Abstract
This review explores a large series of observations from clinical and experimental studies on the interactions between migraine and the extrapyramidal system (EPS). A critical appraisal of these data suggests that the EPS is somehow involved in migraine. However, primary involvement of the EPS in the pathophysiology of migraine, as hinted at by the apparent concomitance of migraine, extrapyramidal symptoms and diseases, as well as by the common involvement of neurotransmitters and pathways, cannot as yet be proven. On the other hand, the involvement of EPS in migraine may reflect its more general role in the processing of nociceptive information and/or may be part of the complex behavioural adaptive response that characterizes migraine.
Introduction
The extrapyramidal system (EPS) is a complex neural network, encompassing the cerebral cortex, thalamus, basal ganglia, brainstem nuclei and cerebellum, which provides a proper execution of voluntary movements by correct processing of sensory-motor information in the brain. The core of this system is represented by the basal ganglia, consisting of five interconnected nuclei (caudate, putamen, globus pallidus, substantia nigra and subthalamic nucleus), functionally interposed between the cortex and the thalamus, that are involved above all in motor and cognitive functions. Experimental and clinical studies have shown that the basal ganglia are involved in nociception (1). In fact, the basal ganglia process both non-noxious and noxious somatosensory information and contribute to the modulation of nociceptive information and to sensory-discriminative, affective and cognitive aspects of pain.
Interestingly, some data now point to a role of the basal ganglia also in migraine. Functional neuroimaging studies have revealed that the substantia nigra (SN), together with the red nucleus (RN) and occipital cortex, is activated during the attack (2, 3). Furthermore, a relationship between migraine and the extrapyramidal nuclei, pathways and neurotransmitters is suggested by various other clinical and experimental evidence that has accumulated over recent years.
The present review will critically explore suggestions and lines of evidence on the interactions between migraine and the EPS, focusing on the following major issues: (i) the possible role of the extrapyramidal system in migraine; (ii) the relationship between extrapyramidal symptoms or diseases and migraine; and (iii) the interactions between anti-migraine drugs and the EPS.
Has the EPS a role in migraine?
Basal ganglia and migraine
The basal ganglia are involved in the processing of noxious information and probably represent a gating mechanism for the regulation of nociceptive inputs to higher centres (1). In particular, the SN, globus pallidus (GP) and striatum are involved in modulating nociceptive neuronal and behavioural responses. It is unlikely that the basal ganglia play a significant role in the spatial localization of noxious stimuli as nociceptive neurons in the SN, striatum and pallidus have large receptive fields; they are more likely to exert a discrete encoding of stimulus intensity, probably associated with grading motor response to escape from pain (4–6). Ascending nociceptive signals probably provide redundant information to the basal ganglia through multiple parallel pathways. The SN and ventromedial striatum receive afferences from the dorsal raphe nucleus that seem to account for pain modulation, whilst the GP, striatum and SN receive inputs probably engaged in processes related to emotions from the cingulate cortex, amygdala and prefrontal cortex. Efferents from the SN pars reticulata (SNr) and GP project to the ventral anterior/ventral lateral and ventromedial/ventrolateral complex of the thalamus, as well as to the intralaminar nociceptive neurons of the thalamus (7). Moreover, pure nociceptive neurons project from SN to the striatum. This nigrostriatal pathway is involved in the processing and sensory gating of nociceptive information. This has recently been confirmed in vivo by the demonstration of a reduction in striatal fluorodopa uptake (a marker of nigrostriatal function) in patients with burning mouth syndrome (8).
Recent reports point to an involvement of the basal ganglia also in migraine headache.
Autoradiographic studies
Some authors have studied the distribution of specific [3H]sumatriptan-binding sites (5-HT1B, 5-HT1D, 5HT1F receptors) in the human brain (9–11). Interestingly, they observed that, besides the visual cortex (layers II–III), the highest density of specific [3H]sumatriptan-binding sites was found in the SN and medial GP (mGP), the basal ganglia output nuclei. It is worth noting that the density of [3H]sumatriptan-binding sites in the SN and GP is almost threefold that in areas crucially involved in migraine pathogenesis, such as the trigeminal nucleus caudalis, nucleus tractus solitarius, area postrema and periacqueductal grey. The 5-HT1B receptor seems to be the most abundant receptor subtype in the SN and GP (11). In rat, 5-HT1B receptors control the release of neuronal dopamine from the striatal synaptosome as pre-synaptic heteroreceptors (12–14). In man, 5-HT1B receptors are located pre-synaptically on striatonigral and, to a lesser extent, on striatopallidal projecting neurons containing dynophine/SP and enkephalin, respectively. This has been demonstrated by the finding that the density of 5-HT1B receptors is preserved in Parkinson's disease, increased in progressive supranuclear palsy and markedly decreased in striatonigral degeneration and Huntington's chorea (15, 16), in agreement with the results of experimental models of selective lesions of the SN with 6-hydroxydopamine and of the caudate-putamen with kainic acid in rat (17, 18). Finally, 5-HT1B receptors are probably involved in the pre-synaptic regulation of the glutamatergic cortical outflow to subcortical areas. Therefore, within the basal ganglia these receptors seem to be involved in the pre-synaptic regulation of excitatory (glutamate), inhibitory (GABA) and peptidergic neurotransmitters (19, 20) (Fig. 1).

The figure shows a schematic representation of the functional organization of the basal ganglia (adapted from Blandini et al.). SNc = substantia nigra pars compacta; SNr = substantia nigra pars reticulata; STN = subthalamic nucleus; LGP = globus pallidus lateralis; MGP = globus pallidus medialis. Striatal GABAergic neurons containing dynorphin and substance P as co-transmitters and expressing dopamine D1 receptors project to the SN and MGP (direct pathway), thereby inhibiting GABAergic neurons of the output nuclei and leading to a disinhibition of the thalamic nuclei. On the other hand, striatal GABAergic neurons containing enkephalin and expressing dopamine D2 receptors project to the LGP (indirect pathway), which in turn sends projections to the STN. This pathway activates the SN and MGP, thus increasing their inhibitory control over the motor thalamus. 5-HT1B receptors modulate both excitatory corticostriatal afferences and inhibitory striatonigral and striatopallidal projections.
Immunocytochemistry
Peripheral nerves from selected cranial autonomic and sensory ganglia innervate extracranial and intracranial large cerebral arteries. Using light and electron-microscopic immunocytochemistry, it has been shown that the SN and ventral tegmental area actively control the cerebral microcirculation and permeability of the blood–brain barrier through mesocortical dopaminergic neurons that innervate penetrating arterioles and capillaries in the cerebral cortex (21). These fibres are strategically positioned to adjust both microcirculation and neuronal activity in discrete cortical regions.
Functional neuroimaging
Blood oxygen level dependent (BOLD) functional magnetic resonance imaging (fMRI) has provided the strongest finding pointing to a role of the basal ganglia in migraine, with bilateral activation of the SN and RN being demonstrated during spontaneous and visually activated attacks of migraine with and without aura. In addition, a persistent activation of the RN and SN has very recently also been shown in chronic daily headache, using high-resolution MRI techniques (2, 3). Although the mechanisms underlying these observations have yet to be established, both the SN and RN have been implicated in nociception and autonomic dysfunction (22).
Cerebral blood flow (CBF) studies
The study of local CBF using the stable xenon-enhanced computed tomographic (Xe CT-CBF) method during spontaneous attacks of migraine with and without aura has revealed the presence of hyperperfusion in the basal ganglia as well as in the cerebral cortex, thalamus and subcortical white matter of both hemispheres (23, 24). However, this pattern of hyperperfusion does not appear to be peculiar to migraine as it is also observed during spontaneous cluster headache attacks.
Cerebellum and migraine
Genetic studies indicate that migraine may be a central Ca++ channellopathy. The α1 subunit of the P/Q Ca++ channels is highly expressed in the cerebellum (25) and mediates Ca++ influx across pre-synaptic and somatodendritic membranes, thus triggering fast neurotransmitter release and other key neuronal responses. Mutations in its encoding gene, CACNA1A, can provoke familial hemiplegic migraine, an unusual subtype of migraine with aura, and episodic ataxia type 2 (26). In addition, there is an involvement of the CACNA1A gene-containing region on 19p13 in migraine with and without aura (27). Interestingly, an abnormal functioning of the lateral cerebellum in migraine has recently been suggested by the detection of subclinical cerebellar signs, namely hypermetria, in both migraine with and without aura, possibly reflecting a slight dysfunction of genetically abnormal Ca++ channels (28). Furthermore, it is worth noting that mild symptoms of postural instability that may be related to vestibulocerebellar dysfunction, such as dizziness, vertigo and dysequilibrium, are common during and between attacks in migraineurs, especially in those with aura (29).
Dopamine and migraine
Dopamine (DA), the key neurotrasmitter of the EPS, is believed to play a role in migraine pathogenesis (30–32). This hypothesis first arose from purely clinical considerations. In fact, pain excepted, almost all symptoms characterizing an attack, from hypothalamic prodromal (yawning, sleepiness) to brainstem accompanying (nausea, vomiting, dizziness) and limbic postdromal (euphoria) symptoms, point to an activation of the dopaminergic system.
Pharmacological evidence subsequently confirmed this hypothesis. Drugs acting on DA receptors are useful in the acute treatment of migraine. Central (metoclopramide) and peripheral (domperidone) anti-emetics, widely employed for the treatment of accompanying symptoms (33–35), have also displayed migraine-abortive properties. In addition, typical and atypical neuroleptics have been recommended for the acute treatment of migraine in the emergency setting, as well as for the therapy of transformed and/or refractory migraine attacks (36–40).
Drugs that act on the dopaminergic system are also effective as migraine prophylaxis. Both flunarizine, a calcium channel blocker with a clear antidopaminergic effect (41), and DA-agonists, such as bromocriptine, lisuride and dihydroergocryptine, are effective as migraine-preventive therapy (42–44). Finally, continuous subcutaneous infusion of apomorphine (a selective dopamine D1 and D2 receptor agonist) has been proposed as an alternative therapeutic tool for migraine with a high attack frequency resistant to conventional prophylaxis (45).
Dopamine receptors: evidence and doubts
DA receptors are located in sites crucially involved in migraine. The DA D1-like receptor superfamily includes the D1 (DRD1) and D5 (DRD5) receptors, whereas the DA D2-like receptor superfamily includes the D2 (DRD2), D3 (DRD3) and D4 (DRD4) receptors (46). DRD1 are present in the thalamus, supraoptic and suprachiasmatic nuclei. DRD2 are widespread in the hypothalamus, raphe nuclei and periacqueductal grey and, together with DRD3 and DRD4, in the intermediate and medial subnuclei of the solitary nucleus and dorsal motor vagal nucleus. DRD3 are located in the area postrema (47, 48). Moreover, DRD1 are located on the muscular layer of arterial walls, whereas DRD2 are present pre-synaptically on sympathetic perivascular endings that modulate norepinephrine release (49), suggesting a role in haemodynamic modulation and possibly in hypotension during migraine attacks (50).
There are data indicating that DA receptors in migraine patients possess peculiar functional and genetic features:
Activation threshold. Migraine patients are characterized by a condition of dopaminergic hypersensitivity, as demonstrated over the last 30 years by numerous pharmacological studies involving an acute challenge with levodopa, l-deprenyl and DA agonists (30, 51–55). Both pre- and post-synaptic DA receptors in migraineurs are characterized by a lower activation threshold. In fact, using very low apomorphine doses (2 µg/kg), which at most activate pre-synaptic receptors in healthy controls, migraine patients display a higher frequency not only of yawning and somnolence (due to the activation of DA pre-synaptic receptors), but also of nausea, vomiting and other autonomic symptoms (due to the activation of DA post-synaptic receptors) (55). DRD3, DRD4 and DRD5 receptors on peripheral blood lymphocytes are up-regulated in migraine patients (56, 57), probably as peripheral expression of this dopaminergic hypersensitivity.
Genetics. Genetic studies carried out in the late 1990s, though still debated, did raise the possibility that specific alleles of the DRD2 are implicated in the determination of the susceptibility, clinical expression and psychiatric comorbidity of migraine (Table 1, 58–64).
Dopamine receptor genes and migraine
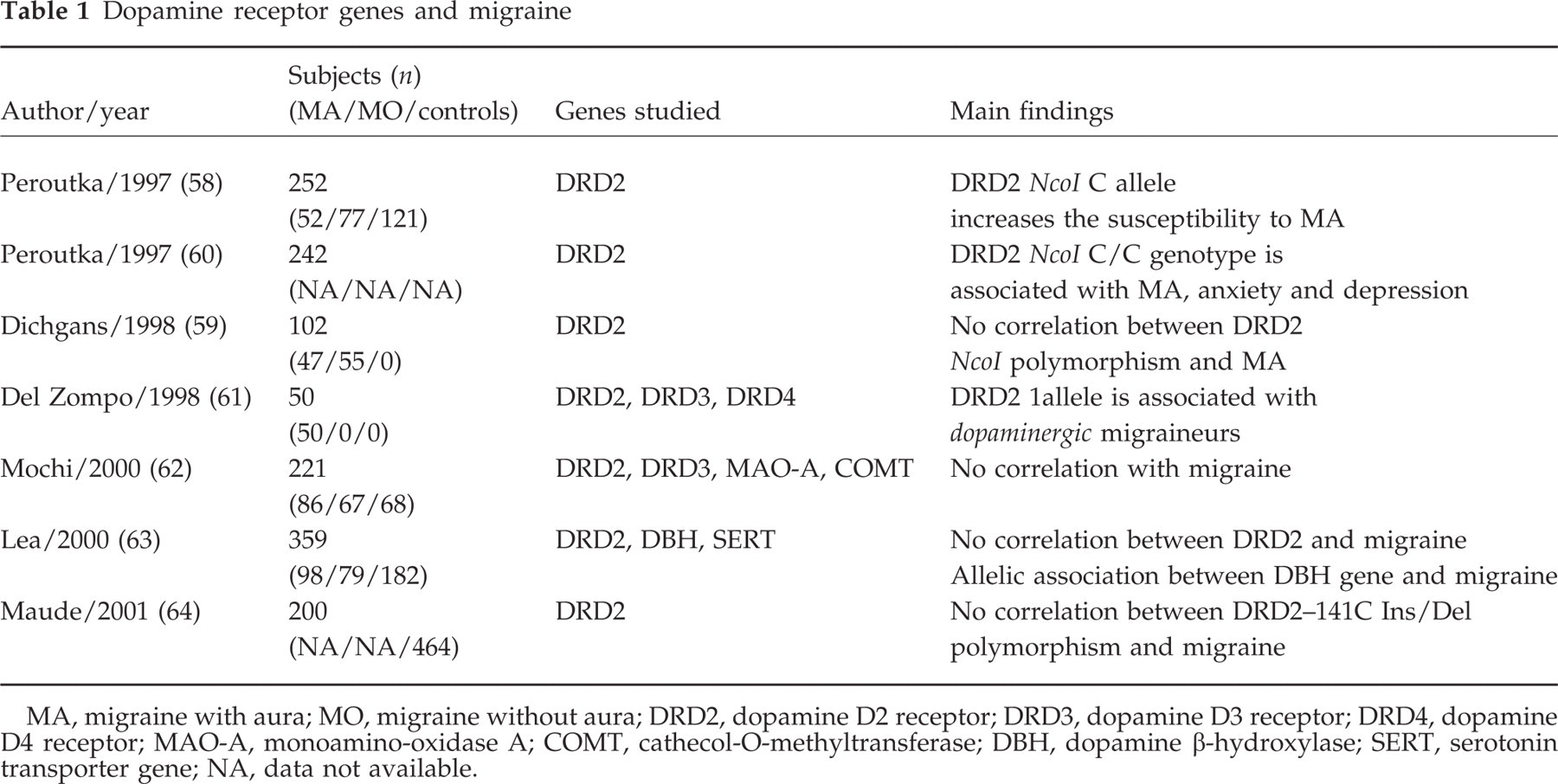
MA, migraine with aura; MO, migraine without aura; DRD2, dopamine D2 receptor; DRD3, dopamine D3 receptor; DRD4, dopamine D4 receptor; MAO-A, monoamino-oxidase A; COMT, cathecol-O-methyltransferase; DBH, dopamine β-hydroxylase; SERT, serotonin transporter gene; NA, data not available.
All the above findings indicate that the dopaminergic system is clearly involved in migraine, but also reveal that the dopaminergic model of migraine, though fascinating, represents a working hypothesis at this stage. The dopaminergic hypersensitivity in migraineurs, demonstrated by numerous clinical and pharmacological studies (30, 51–57), to this day represents the strongest support for this hypothesis. Other issues deserve further investigation. For example, no full agreement exists on genetic studies as recent findings seem to dampen the initial enthusiasm. In particular, the possibility that specific polymorphisms within the DRD2 gene are associated with migraine is controversial (58–64). The possibility of inducing attacks with apomorphine is also debated (55). Finally, the efficacy in migraine of dopamine agonists for prophylaxis needs to be confirmed in further, larger controlled studies.
Is there any link between extrapyramidal symptoms or diseases and migraine?
Clinical signs suggestive of EPS involvement may spontaneously occur in migraine patients. Subclinical hypermetria has been detected in migraineurs with and without aura (see point 1.2). In addition, ‘hypokinetic behaviour’ is seen in the vast majority of patients during the attack, whereas ‘akathisic behaviour’ is very rare. This is in sharp contrast to other pain syndromes originating from the head, such as cluster headache or dental pain, during which the patient always appears restless. However, given the concomitance of sensitivity to movements, sounds and light in migraine, it is very unlikely that these behaviours derive from a primary involvement of the EPS.
Migraine has been investigated in some extrapyramidal diseases such as Parkinson's disease, essential tremor and Tourette's syndrome (Table 2).
Migraine frequency in movement disorders
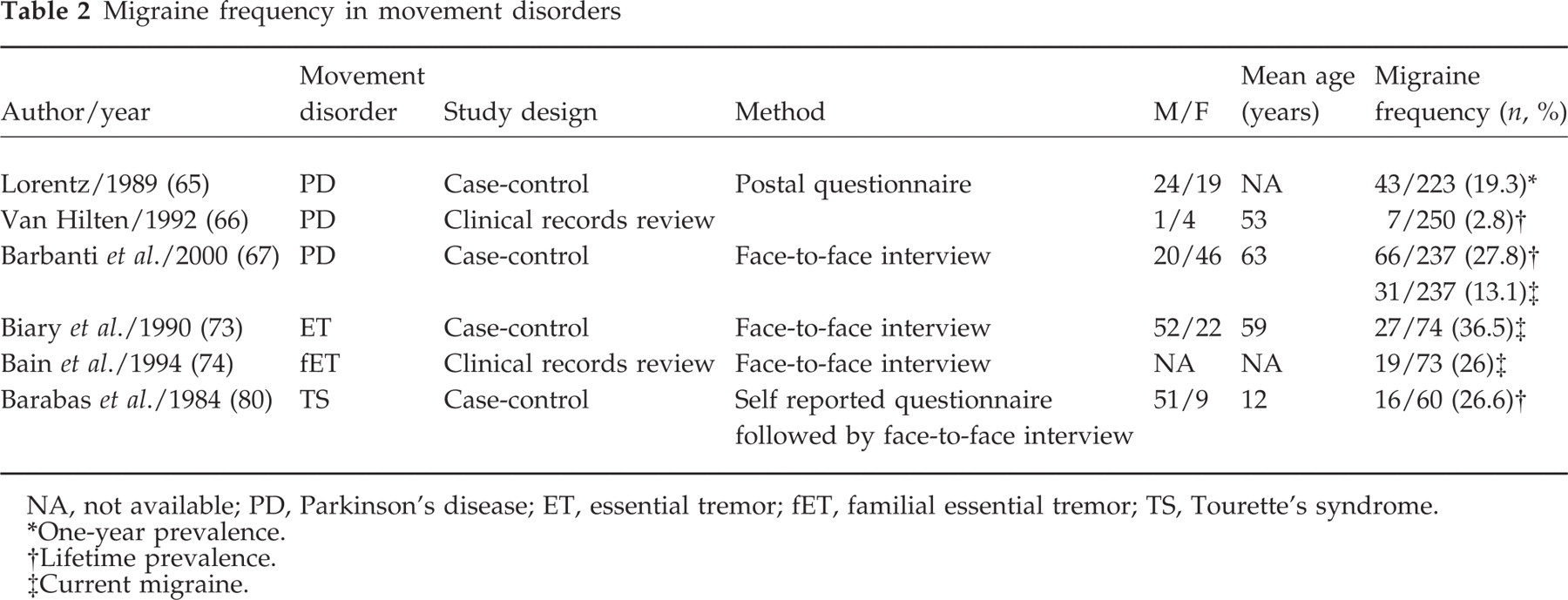
NA, not available; PD, Parkinson's disease; ET, essential tremor; fET, familial essential tremor; TS, Tourette's syndrome.
∗One-year prevalence.
†Lifetime prevalence.
‡Current migraine.
Parkinson's disease (PD)
In cross-sectional studies, the lifetime prevalence of migraine in PD ranges from 19.3% to 27.8% in case-control studies (65–67) (Table 2). PD seems to modify the course of migraine. In a retrospective study of five parkinsonian migraineurs, van Hjlten (66) found that after the onset of PD migraine disappeared in one patient, clearly decreased in two and was unchanged in the remainder. He concluded that PD clearly changed the course of migraine in patients without a family history of migraine, hypothesizing a different role for the dopaminergic system in migraineurs with and without a family history of migraine. In a larger cross-sectional study (67), we confirmed that PD seems to shorten the course of migraine: approximately two-thirds of PD patients had an improvement in or remission of migraine after PD onset, showing a significantly lower frequency of current migraine than in otherwise healthy age- and sex-matched controls, regardless of migraine characteristics and menopause.
The modification of migraine course might be due to the anti-migraine properties of drugs used in PD therapy. This may be the case for dopamine agonists, but is less likely to be so for levodopa, the most frequently used anti-parkinsonian drug. In fact, reports of a dramatic improvement in migraine with levodopa in a PD patient (68) and in a similar unpublished case by Dr Melvin Yahr were not confirmed in a subsequent pilot study (69). Similarly, amantadine, a dopaminergic and antiglutamatergic drug, at a dose of 200 mg/day, was ineffective as migraine prophylaxis in a 4-week, double-blind, placebo-controlled crossover study (70).
Alternatively, migraine may be modified by PD pathophysiology. In animals, intracerebral administration of 6-OHDA, which significantly depletes dopamine within the nigrostriatal pathways, results in decreased pain sensitivity as measured by the formalin test but not the tail flick test (71). Moreover, parkinsonian animal models display a degeneration of nigrostriatal collaterals to the thalamus (mesothalamic system) (72). In man, the degeneration of dopaminergic neurons in the SN pars compacta (SNc), the hallmark of PD, triggers a cascade of functional changes leading to a hyperactivation of GABAergic output nuclei, SNr and mGP, which in turn increase inhibitory control over the thalamus (19, 20). Despite the complexity of the problem of pain in PD, it could be speculated that this increased inhibitory activity of the SNr on the thalamus in PD enhances the gating properties of the basal ganglia on nociception.
Essential tremor (ET)
ET and migraine seem to be related. In fact, migraine has a higher prevalence in ET patients than in controls (36.5% vs. 17.7%), whereas ET is much more common in migrainers than controls (22% vs. 1%) (73). In addition, migraine co-segregates with tremor in hereditary ET (74). Interestingly, ET has been described in a parent–child pair affected by hemiplegic migraine, in whom there was an improvement in both migraine and tremor following treatment with propranolol and pizotyline (75). Baloh (76) described a family with migraine, vertigo and ET who showed an improvement in all symptoms following acetazolamide intake and hypothesized a common genetic defect for both migraine and ET. The cerebellum, nucleus dentatus and RN are involved in ET pathophysiology, as has been confirmed by H2O15 PET and fMRI studies that demonstrated a bilateral activation of these structures (77–79). It is noteworthy that both migraine and essential tremor have a strong genetic background, share identical therapeutic options (β-blockers) and show similar bilateral activation of the RN at BOLD fMRI.
Other movement disorders
Migraine is also very common among patients with Tourette's syndrome (TS), a complex, multifaceted condition with multiple motor and vocal tics, characterized by a dysfunction in the basal ganglia and related corticothalamic circuits. One study has shown that migraine prevalence is substantially higher (26.6%) in TS patients than in the general population of school-aged children, with a higher incidence in boys than in girls (80). The authors of the study hypothesized that the coexistence of TS and migraine may reflect a similar abnormality underlying both conditions, such as neurohumoral dysfunction involving the serotonin and/or dopamine metabolism.
Migraine without aura has been described in association with paroxysmal exercise-induced dystonia. No linkage has been found to the loci of paroxysmal non-kinesigenic dyskinesia, familial hemiplegic migraine or familial infantile convulsions and paroxysmal choreoathetosis syndrome (81). Finally, a favourable change in the biological pattern of migraine with aura has been reported in one patient, affected by generalized dystonia secondary to perinatal hypoxia, after the use of tetrabenazine (50 mg), a DA depletor (82).
Do anti-migraine drugs affect EPS?
Some of the most common agents used in migraine therapy also have a clear effect on the basal ganglia, with potential implications on the EPS.
Sumatriptan
Like all triptans, sumatriptan, is a selective 5-HT1B/1D receptor agonist with a negligible action, if any, on dopamine receptors. Nevertheless, symptoms that are essentially dopaminergic, such as nausea and somnolence, are among its most frequent adverse events. The potential occurrence of extrapyramidal symptoms associated with sumatriptan sheds light on the possibility of a direct action of this drug also on the basal ganglia.
Sumatriptan may induce extrapyramidal symptoms
Seven cases of extrapyramidal reaction following sumatriptan, namely dystonia and akathisia, have been described in patients exposed to this drug for the first time (83–85) (Table 3). Dystonia has been reported to be mainly axial, involving the neck, the oral musculature and the trunk. In one case, the patient was concomitantly taking loxapine and had a personal history of dystonic reactions to haloperidol (83). Only one of these patients was re-challenged with sumatriptan and showed the recurrence of the same extrapyramidal symptom (akathisia) (85).
Extrapyramidal symptoms with sumatriptan
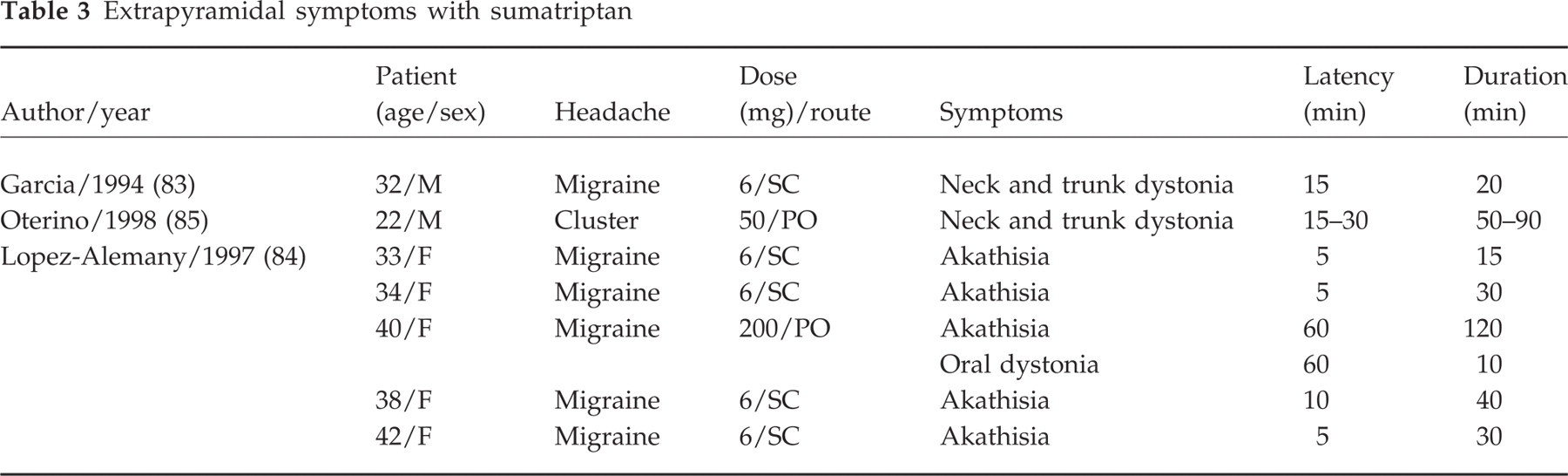
These data are in keeping with a central effect of sumatriptan and suggest that this drug can cross the blood–brain barrier in man, at least in some circumstances. The extrapyramidal effects are probably mediated by 5-HT1B receptors in basal ganglia, in particular by those present on the striatonigral projections whose stimulation might negatively affect motor dopaminergic function, thereby inhibiting nigrostriatal neurons. On these bases, it cannot be excluded that neck pain and stiffness following triptan intake, a typical class-related adverse event whose origin is unclear, might be an expression of focal dystonia, at least in some patients. However, it should be borne in mind that the patients described above are case reports and that nothing is known, except in one case, of the effects produced by further exposure to the drug. Moreover, akathisia may also spontaneously occur, though very rarely, in the early phases of the attack in some patients (86).
Sumatriptan may abolish extrapyramidal symptoms
Essential palatal myoclonus (EPM) is a rare movement disorder with an involvement of the dentato-rubro-olivary triangle and a hyperactivity of the inferior olivary nuclei. Subcutaneos sumatriptan (6 mg) is reported to have completely abolished (EPM) in one patient (87, 88) and partially relieved palatal myoclonus secondary to stroke in another (89), while intranasal sumatriptan (20 mg) was ineffective (90). The beneficial effect of sumatriptan in EPM has been ascribed to an interaction with 5-HT1D receptors in the trigeminal nuclei (with consequent inhibition of involuntary contractions of the trigeminally innervated tensor veli palatini) and/or stimulation of 5-HT1B/1D receptors in the SN and basal ganglia.
Valproate
Valproate, one the most effective prophylactic drugs for migraine, can induce tremor and parkinsonism (91). Its mechanism of action in migraine is believed to be due to an increase in brain GABA levels and to a suppression of migraine-related events in the cortex, perivascular parasympathetics or trigeminal nucleus caudalis (92). However, there is substantial evidence that a direct action on the SN is a key effect of valproate. In fact, valproate induces a reduction in the firing rate of SNr neurons (93), while its metabolite trans-2-en-valproate accumulates in the SN during prolonged treatment in rats (94). These effects on the SN are thought to represent an important mechanism through which this drug exerts its anticonvulsant properties, and are likely to account for the extrapyramidal symptoms.
Caffeine
Caffeine, a non-selective adenosine A1 and A2A receptor antagonist, is in some patients a useful migraine-abortive agent, currently used as an adjunctive analgesic in combination with acetaminophen and non-steroidal anti-inflammatory drugs, which also plays a relevant role in the regulation of basal ganglia motor functions. Although the exact mechanisms of its analgesic effect are still largely unknown, they are likely to involve both peripheral and central sites (95, 96). Besides its analgesic properties, caffeine modulates locomotor activity and striatal neuropeptide expression through interactions with the dopaminergic system by means of mechanisms only partially understood. Striatum is highly rich in adenosine A2A receptors, which are thought to play a pivotal role in basal ganglia functions (97). Caffeine stimulates motor behaviour through the blockade of adenosine A2A receptors in the GABAergic striatopallidal neurons, where they are co-localized with dopamine D2 receptors (98). For these reasons, caffeine and other adenosine A2A antagonists are being viewed as promising tools in the treatment of Parkinson's disease (99).
Conclusions
In the present review, we have explored a large series of observations derived from basic science, neurophysiological, neuroimaging and clinical studies on the interactions between the EPS and migraine. A critical appraisal of the data reviewed raises some noteworthy points of discussion. Firstly, some areas of the EPS may be implicated in the pathophysiology of migraine. The SN, which actively controls cerebral microcirculation and the permeability of the blood–brain barrier, is activated, together with RN, during migraine attacks and shows, like the mGP, a very high density of 5-HT1B receptors, the target of triptans. Furthermore, the cerebellum reveals a high expression of the α1 subunit of the P/Q Ca++ channels, involved in the pathogenesis of familial hemiplegic migraine and, probably, of more common forms of migraine. Secondly, extrapyramidal symptoms are not uncommon in migraine patients. Migraineurs manifest subclinical cerebellar signs such as hypermetria that reflect slight dysfunctioning of genetically abnormal Ca++ channels. In addition, postural and kinetic tremor seems to be more frequent in migraineurs than in controls. Thirdly, there seems to be a peculiar relationship between migraine and some extrapyramidal diseases. ET and TS are characterized by a high prevalence of lifetime migraine, whereas the prevalence of current migraine is lower in PD patients than in controls. Fourthly, some anti-migraine drugs may affect basal ganglia motor functions. Sumatriptan may induce, though infrequently, acathisia and dystonia, probably by acting on 5-HT1B receptors located on the SN and mGP. Valproate can cause tremor and parkinsonism, possibly exerting a direct action on the SN, whereas caffeine stimulates motor behaviour through the blockade of adenosine A2A receptors in the GABAergic striatopallidal neurons.
The interpretation of all these findings is debatable. A primary involvement of the EPS in the pathopysiology of migraine cannot be demonstrated as yet, in spite of the apparent concomitance of migraine, extrapyramidal symptoms and diseases, as well as of the common involvement of extrapyramidal neurotransmitters and pathways, which seems to suggest common potential pathogenetic factors. Nevertheless, the EPS, which provides the proper execution of voluntary movements by processing of sensory-motor information in the brain, actively participates in modulation of pain and nociceptive sensorimotor integration, thereby providing a co-ordinated response to damaging or potentially damaging stimuli (escape behaviour). Therefore, the involvement of the EPS in migraine may reflect its more general role in the processing of nociceptive information and/or may be part of the complex behavioural adaptive response that characterizes migraine.
