Abstract
Objective
This meta-analysis evaluates pressure pain sensitivity values in symptomatic and distant pain-free areas comparing individuals with tension-type headache to controls.
Databases and data treatment
Electronic databases were searched for cross-sectional or prospective case-control studies comparing pressure pain thresholds in patients with tension-type headache to headache-free controls. Data were extracted by three reviewers. The methodological quality was assessed by the Newcastle-Ottawa Quality Assessment Scale. Meta-analyses of trigeminal, extra-trigeminal (neck) and distant pain-free areas in tension-type headache were compared to headache-free controls. Frequency of tension-type headache and gender were taken into account.
Results
Twenty studies were included. Patients with tension-type headache exhibited lower pressure pain thresholds than headache-free controls: Trigeminal (MD −49.11 kPa, 95% CI −66.05 to −32.17), cervical spine (MD −88.17 kPa, 95% CI −108.43 to −67.92) and distant pain-free areas (MD −98.43 kPa, 95% CI −136.78 to −60.09). Differences were significant for chronic, episodic, and mixed episodic and chronic tension-type headache within the trigeminal and neck (symptomatic areas), but only significant for chronic tension-type headache (MD −102.86, 95% CI −139.47 to −66.25 kPa) for distant pain-free areas. In general, women had lower pressure pain thresholds than men. The methodological quality ranged from fair (45%) to good (40%). The results showed a high heterogeneity and publication bias.
Conclusion
This first meta-analysis addressing pressure pain thresholds differences in symptomatic and distant pain-free areas between patients with tension-type headache and controls found low to moderate evidence supporting the presence of pressure pain hypersensitivity in the trigeminal and neck areas in tension-type headache in comparison with headache-free controls. Sensitivity to pressure pain was widespread only in chronic, not episodic, tension-type headache (moderate evidence).
Introduction
In 2016, neurological disorders were the leading cause of disability-adjusted life-years and the second leading cause of deaths worldwide (1). Headache is the most common condition attended by neurologists in clinical practice. Similarly, in the Global Burden of Disease Study 2017, headache was the second most prevalent of all disorders and the second leading cause of years lived with disability (2). Headache causes substantial pain and social and health costs (3,4). Tension-type headache (TTH) is the most prevalent primary headache affecting around 80% of the population at some time during their lives (5) and with a point prevalence of 42% in the general population (6). A recent longitudinal study found that the 1-year prevalence of TTH increased during the last decade from 16% to 21% (7).
Despite its prevalence, TTH is the most neglected primary headache, probably because its underlying mechanisms are poorly understood (8). However, both peripheral and central sensitization mechanisms have been demonstrated (9). In fact, the most accepted theory is that the episodic form is peripherally related whereas the chronic form is more centrally related (10). One common clinical manifestation of these sensitization processes is the presence of sensitivity to pressure pain (11). Pressure pain threshold (PPT) measurement with a pressure algometer is normally used to measure mechanical sensitivity (12). There is evidence supporting that TTH exhibits pressure pain hypersensitivity, as expressed by lower PPTs compared to headache-free populations (13). Hypersensitivity to pressure pain in patients with TTH has been investigated in many studies and in one systematic review (14) and one meta-analysis (15). Both reviews concluded that PPTs were consistently lower in the trigeminal (14) and cervical (15) areas in individuals with TTH as compared to headache-free people. Neither review compared data between episodic and chronic TTH. Additionally, both reviews investigated only pain sensitivity in symptomatic areas, such as the head or neck. The presence of pressure pain hyperalgesia in trigeminal and cervical symptomatic areas can be related to central but also peripheral mechanisms. To confirm the presence of central sensitization, pressure pain sensitivity in distant extra-trigeminal pain-free areas is required. No meta-analysis has quantitatively investigated the presence of widespread hyperalgesia to pressure pain in distant pain-free areas in patients with TTH. Therefore, the current systematic review and meta-analysis analyzed differences in PPTs between individuals with TTH and controls in symptomatic trigeminal and cervical pain areas and distant (extra-trigeminal) pain-free areas.
Methods
This systematic review and meta-analysis adheres to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (16). The international OPS Registry registration link is https://doi.org/10.17605/OSF.IO/R29HY
Systematic literature search
Electronic literature searches were conducted on MEDLINE, CINAHL, PubMed, SCOPUS and Web of Science databases from their inception to 15 May 2020. We also screened the reference lists of the papers that were identified in database searches and contacted authors, if necessary. Bibliographical database search strategies were conducted with the assistance of an experienced health science librarian.
Population: Adult (age ≥18 years) women and men with a diagnosis of TTH according to the International Headache Society (IHS) Criteria (17–20) and no other primary or secondary headache or concomitant chronic pain condition.
Comparator: A comparative group of headache-free subjects without any musculoskeletal pathology serving as controls.
Outcomes: The primary outcome measure was PPT assessed with a pressure algometer at trigeminal (head), cervical spine, or distant (extra-trigeminal) pain-free area.
The search strategy for each database is available in Supplemental Material 1.
Selection criteria
The systematic review included cross-sectional or prospective case-control studies where at least one group included adults with any form of TTH and one group included headache-free controls. The specific inclusion criteria were: i) Adults diagnosed with TTH according to IHS criteria (any edition); ii) full text report published in English (not abstract, letter, or editorial); iii) a control group including headache-free subjects; and iv) the study should include PPTs assessed with a pressure algometer as an outcome. In studies that evaluated numerous types of sensory thresholds with quantitative sensory tests, such as thermal or electrical pain thresholds, only PPT data measured with an algometer was extracted. We excluded studies: a) not published as a full-text journal article; b) assessing pressure pain sensitivity with other modalities (e.g. Von-Frey monofilament); or c) experimental pain model of headache.
Screening, selection process and data extraction
Articles identified from the different databases were independently reviewed by three authors (CFdlP, GPM, MJSN). First, the duplicates were removed. Second, the title and abstract of the articles were screened for potential eligibility. Third, a full-text read of potentially eligible studies was conducted. Authors were required to achieve a consensus on the included trials. In case of discrepancy between the reviewers, a fourth author (LB) participated in the process to reach the consensus for inclusion of the study.
Data from each trial, including population, diagnosis, sample size, outcome and type of algometer, were extracted independently by three authors (CFdlP, GPM, MJSN). They had to achieve a consensus on each item on the data extraction. If disagreement occurred, a fourth author participated in the determination (JO). No differences were observed in data extraction among authors.
Assessment of risk of bias/methodological quality
Risk of bias of the studies was independently assessed by three authors using the Newcastle-Ottawa Scale. The Newcastle-Ottawa Scale is a star rating system (a total of nine stars) to evaluate the risk of bias/methodological quality of case-control/cohort studies (21). It includes three main fields: Case selection, comparability and exposure. Case selection is formed by four items: Case definition, representativeness of cases, and definition and selection of controls. Comparability evaluates the analysis of comparison (e.g. controlled for age, gender, or other factors) between groups. Exposure assesses the ascertainment for case and control groups (blinding assessment) and non-response rate (refusing to participate in the study). A maximum of nine stars can be awarded. Studies scoring ≥7 are considered of good quality; those scoring 5 or 6 are of fair quality and studies scoring 0–4 are of poor quality (22).
Risk of bias was also determined by the same three authors (CFdlP, GPM, MJSN), and differences were discussed. In case of disagreement, a fourth researcher arbitrated a consensus decision (RJ).
Level of evidence
To evaluate the level of evidence, the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach was used (23). The level of evidence was classified as high, moderate, low or very low according to downgrading factors such as i) risk of bias (Newcastle-Ottawa Scale), defined as several studies’ limitations that are a substantial risk of bias across the evidence; ii) inconsistency of the results/unexplained heterogeneity when the results presented statistically significant (p < 0.05) moderate-high heterogeneity (I2 > 60%); iii) indirectness of evidence when the evidence was not directly comparable to the question of interest (e.g. population, exposure, comparator, outcome); iv) imprecision of the results when the studies had few participants and few events (wide confidence intervals as judged by reviewers); and v) probability of publication bias if the funnel plot presented substantial asymmetry according to studies missing from body of evidence and statistically significant Egger Test, resulting in an over or underestimate of true effects from exposure (24). The level of evidence was classified as high when all items were negative; moderate when one item included serious risk or yes; low when two items showed serious risk or one item showed very serious risk; or very low when all items had serious risk or two or more items showed very serious risk. Again, this process was independently performed by three authors, with the participation of a fourth if needed.
Data synthesis and analysis
The meta-analysis was conducted using the Review Manager statistical software (RevMan version 5.3) and R software 4.0.0. Data synthesis was categorized by diagnosis (chronic tension-type headache, CTTH; frequent episodic tension-type headache, FETTH or TTH, if frequency distinction was not provided) and by gender (men, women).
We extracted sample size, means and standard deviations for each variable. When the study reported only standard errors, they were converted to standard deviations. When necessary, the mean scores and standard deviations were estimated from graphs with the GetData Graph Digitizer v.2.26.0.20 software. If the study presented non-parametric values, they were converted to means and standard deviations (25,26). Eventually, if a study reported mean differences with their interval confidence or standard error of the means, we also estimated standard deviations, as needed.
Mean and standard deviation values of every point (head, neck, and distant pain-free) were obtained using the dmetar package for R software and pool.groups function. Also, when the data of different groups (men/women) was presented separately, the pool. groups function was also used to obtain the mean and standard deviation of the total sample for avoiding repetition of the same control group in the quantitative analysis. The between-groups mean differences, with their 95% confidence intervals (CI) of each study, were obtained. A random-effect model was used to determine the overall effect size (MD). Pressure pain thresholds were expressed in kPa, when another unit was presented in the studies (e.g. kg) they were converted to kPa (1 kg/cm2 = 98.066 kPa).
Data extracted from the articles are summarized in tables (see Results section). If multiple sites were analyzed in the same area, for example, temporalis, masseter, or frontalis for the trigeminal area, the average of all PPTs on each area (trigeminal, neck, or distant pain-free) was calculated for each meta-analysis. In addition, absolute mean PPT values for each area (trigeminal, neck, distant pain-free) were also pooled regardless of the location and compared between patients with TTH and headache-free controls. For those studies reporting results by subgroups (CTTH/FETTH, men/women) each subgroup was treated as a separate study. For studies with a single headache-free control group and different patient subtypes, we pooled data from each group of patients versus the same control group.
The heterogeneity of the studies was assessed using the I2 statistic. The Cochrane group has established the following interpretation of the I2 statistic: 0–40% may not be relevant/important heterogeneity; 30–60% suggests moderate heterogeneity, 50–90% represents substantial heterogeneity, and 75–100% represents considerable heterogeneity (27). Publication bias was assessed using a funnel plot; if there was asymmetry and suspicion of publication bias, Egger’s Test was realized to detect publication bias. The assessment of publication bias was only evaluated if 10 different studies were included within the same meta-analysis.
Results
Study selection
The electronic search initially identified 1376 potential studies for review. After removing duplicates, 611 studies remained. Five hundred and eighty-seven (n = 587) were excluded based on examination of their title and/or abstract, leaving 24 articles (27–50), for full-text analysis. Three studies were excluded because they did not include a control group (40–42) and one because it had an experimental design (43). Finally, 20 studies (27–39,44–50), were included in the systematic review and meta-analysis (Figure 1).
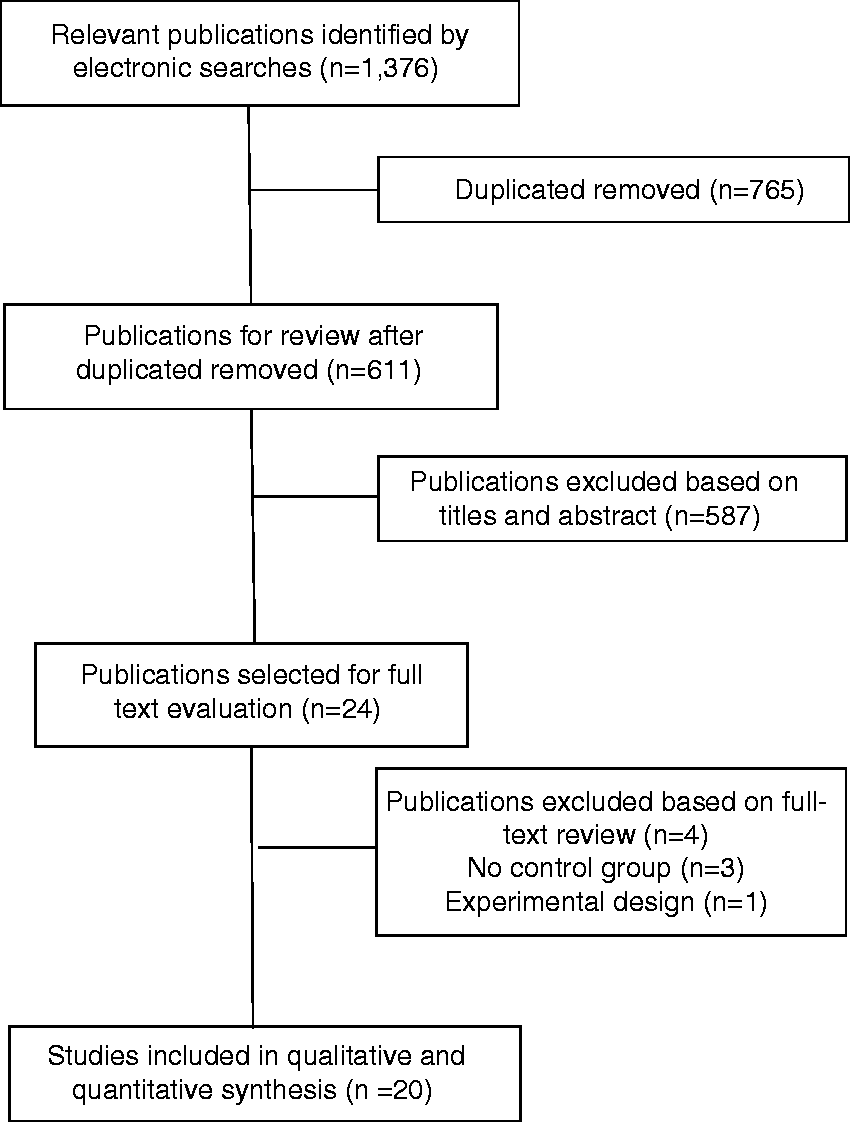
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram.
Study characteristics
The characteristics of the populations of the included studies are shown in Table 1. All articles diagnosed the group of patients with TTH according to the IHS criteria (n = 9, first edition 1988; n = 8 second edition 2004; n = 2 third edition, beta version 2013, n = 1 third edition 2018). Seven articles included patients only with CTTH, four articles patients with just FETTH, four included both patients with CTTH and FETTH, whereas the last four did not differentiate between the episodic or chronic form (TTH). Five studies included only women and the remaining 15 included men and women. The total sample included 1003 patients (284 men, 719 women) and 1066 headache-free controls (504 men, 562 women). Three hundred and twenty-three (n = 323) were patients with CTTH (66 men, 257 women), 422 were patients with FETTH (130 men, 292 women) and 258 (88 men, 170 women) were patients with TTH without differentiation by frequency. All studies included a comparable headache-free control group, but only in five studies was the control group mentioned to be age-matched. In the remaining studies, the control group was comparable in gender and age to the patient group. In 10 studies (50%), patients were asked to not take analgesic medication 24 h or longer before the assessment. Few articles mentioned prophylactic treatment, but overall there was insufficient presentation of medication information. Most studies evaluated patients in headache-free periods or when headache intensity was less than 3/10.
Characteristics of the population and algometer of the included studies
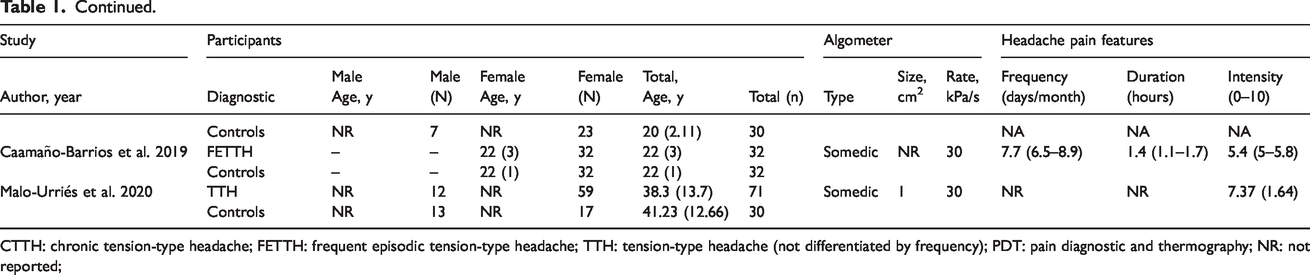
CTTH: chronic tension-type headache; FETTH: frequent episodic tension-type headache; TTH: tension-type headache (not differentiated by frequency); PDT: pain diagnostic and thermography; NR: not reported;
The pressure algometer used in most of the studies (n = 14, 70%) was from the company Somedic (Sösdala, Sweden). The size of the algometer probe surface was reported in 14 (70%) studies and ranged from 0.5 (n = 6) to 1 cm2 (n = 9). Pressure pain thresholds were assessed in trigeminal areas in 17 studies (temporalis n = 13, frontalis n = 4, parietal bone n = 2, masseter n = 1), in the neck in 11 studies (upper trapezius n = 7, cervical spine n = 5), and in distant (extra-trigeminal) pain-free areas in nine studies (tibialis anterior/Achilles tendon n = 6, hand/finger n = 6). Table 2 summarizes PPT findings of the included studies on each point.
Pressure pain thresholds (KPa) of patients with tension-type headache and controls.

CTTH: chronic tension-type headache; FETTH: frequent episodic tension-type headache; TTH: tension-type headache.
The pooled data of all PPTs revealed, in general, that patients with TTH exhibited lower values (mean ± SD, kPa) than headache-free controls at trigeminal, neck and distant pain-free areas (all, p < 0.001) (Table 3). Women showed lower PPTs than men (p < 0.01).
Pressure pain thresholds (pooled means ± SD, KPa) in the trigeminal, extra-trigeminal and distant pain-free areas in adults with tension-type headache and headache-free controls.

CTTH: chronic tension-type headache; FETTH: frequent episodic tension-type headache.
Risk of bias/methodological quality
The methodological quality scores ranged from 4 to 8 (mean: 6.1, SD: 1.1) out of a maximum of 9. Three studies were considered of poor quality (4 stars) (27,36,39), nine were of fair methodological quality (5–6 stars) (29,32,35,37,44,45,47,48,50), and eight were of good quality (7 stars) (30,31,33,34,38,46,49,51). The most frequent biases were lack of control for additional factors as psychological aspects (e.g. anxiety/depression) or ascertainment of exposure (blinding assessment). Differences in scores between reviewers were discussed and solved properly, and arbitration was not needed. The score on risk of bias between reviewers showed an overall agreement of 94%. Figure 2 and Table 4 show the Newcastle-Ottawa Scale on each study and a summary of every item.

Plots of risk of bias (the Newcastle-Ottawa Scale) of the included studies.
Newcastle-Ottawa Quality Assessment Scale evaluating methodological quality/risk of bias.

Pressure pain thresholds according to headache diagnosis
The meta-analysis found that patients with TTH exhibited significant reduced PPT at the head (MD −49.11, 95% CI −66.05 to −32.17, n = 2347, Z = 5.68, p < 0.001, Figure 3), the neck (MD −88.17, 95% CI −108.43 to −67.92, Z = 8.53, p < 0.001, Figure 4) and distant pain-free (MD: −98.43, 95% CI −136.78 to −60.09, Z = 5.03, p < 0.001, Figure 5) areas with high heterogeneity (I2 > 75%) between studies in all the meta-analyses. The heterogeneity was related to the same study (38) on TTH classification in all analyses, and to just one in CTTH (head (27), neck (30), and distant pain-free (44) analyses) and FEETH (head (39), neck (37), and distant pain-free (47) analyses) classifications. The funnel plot showed asymmetry in overall PPTs at the trigeminal area (Supplemental Figure 1) with a significant Egger’s test (p = 0.0028); therefore, high risk of publication bias was possible, but mostly due to just one study (38). No evidence of publication bias for the results in the neck area was found (Supplemental Figure 2).

Summary of forest plot results for meta-analyses of PPTs in the trigeminal area by chronicity. Negative values indicate that patients < controls. Lower PPTs were found in patients with TTH compared with headache-free controls independently of the frequency of the headaches.
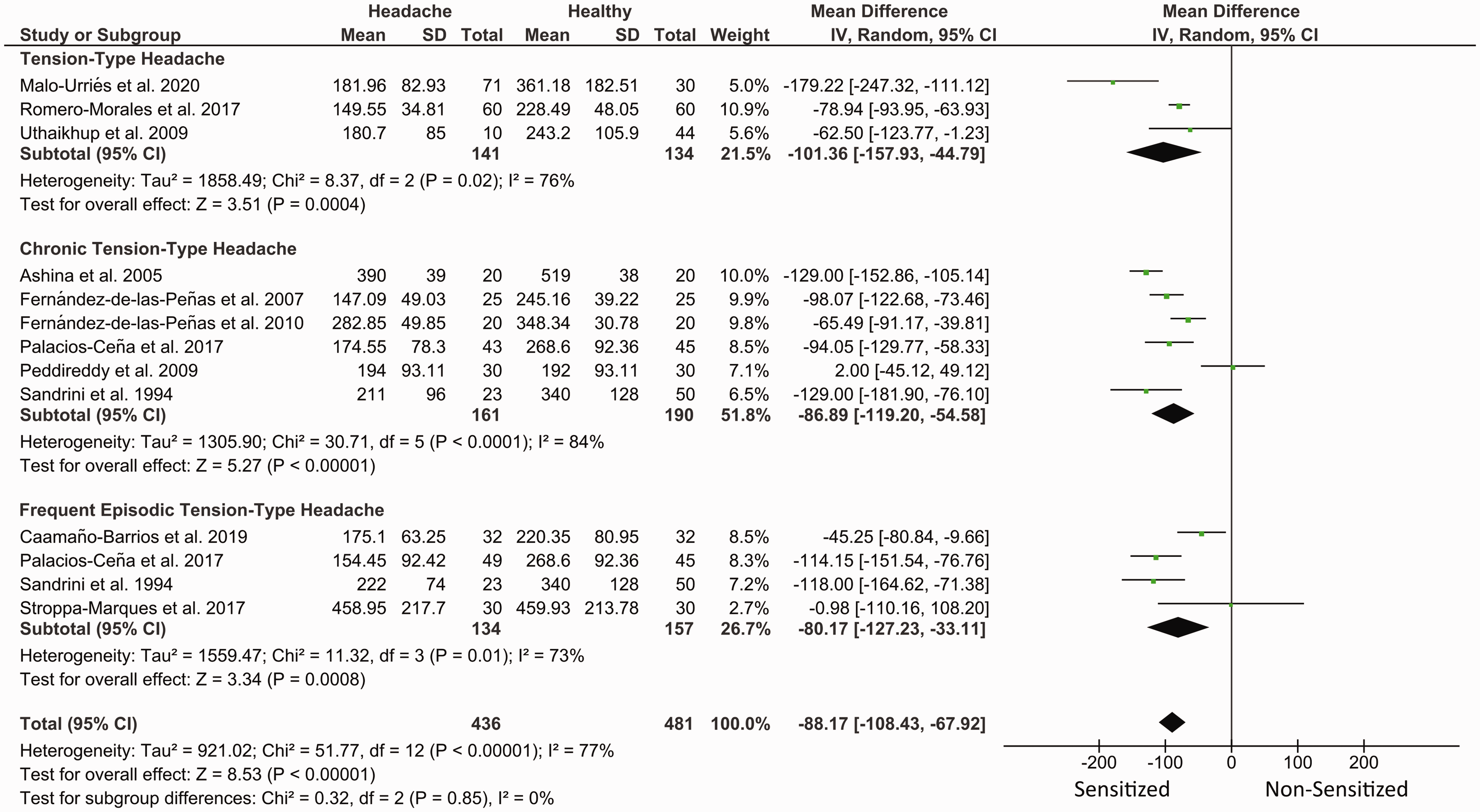
Summary of forest plot results for meta-analyses of PPTs in the neck area by chronicity. Negative values indicate that patients < controls. Lower PPTs were found in patients with TTH compared with headache-free controls independently of the frequency of the headaches.

Summary of forest plot results for meta-analyses of PPTs in distant pain-free areas by chronicity. Negative values indicate that patients < controls. Lower PPTs were found in patients with CTTH compared with headache-free controls.
Pressure pain thresholds by gender at the trigeminal area
Women with TTH exhibited lower PPT at the trigeminal area (MD −48.18, 95% CI −0.80 to −15.56, n = 568, Z = 2.89, p = 0.004, I2 = 91%) when compared with headache-free women (Figure 6). Differences between males with TTH and headache-free controls were not significant (MD −10.94, 95% CI −24.55 to 2.67, n = 282, Z = 1.57, p = 0.12, I2 = 0%). Test for subgroups differences showed significant differences (p = 0.04, I2 = 76.6%).
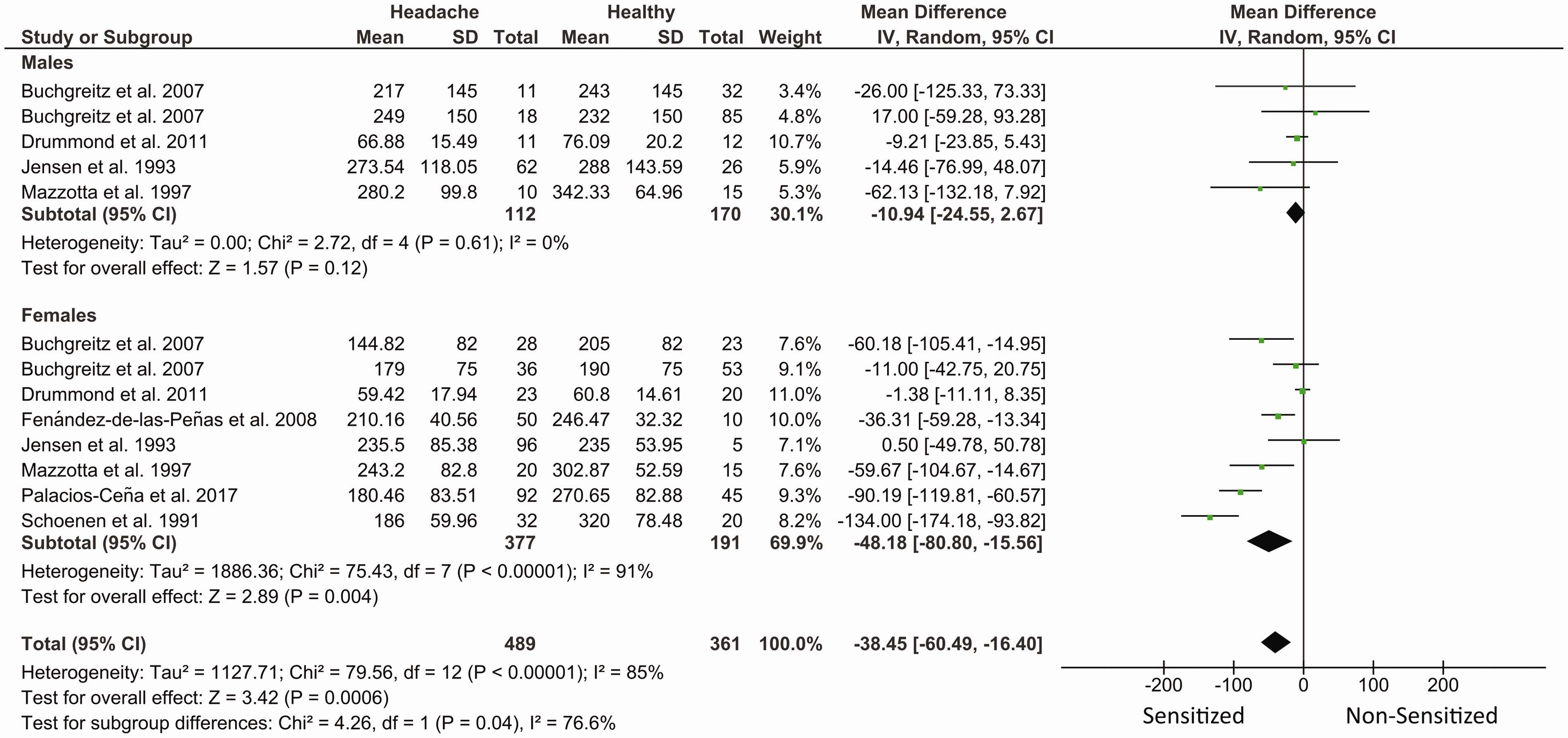
Summary of forest plot results for meta-analyses of PPTs in the trigeminal area by gender. Negative values indicate that patients < controls, whereas positive values indicate that patients > controls. Lower PPTs were found in women with TTH compared with headache-free women.
Level of evidence
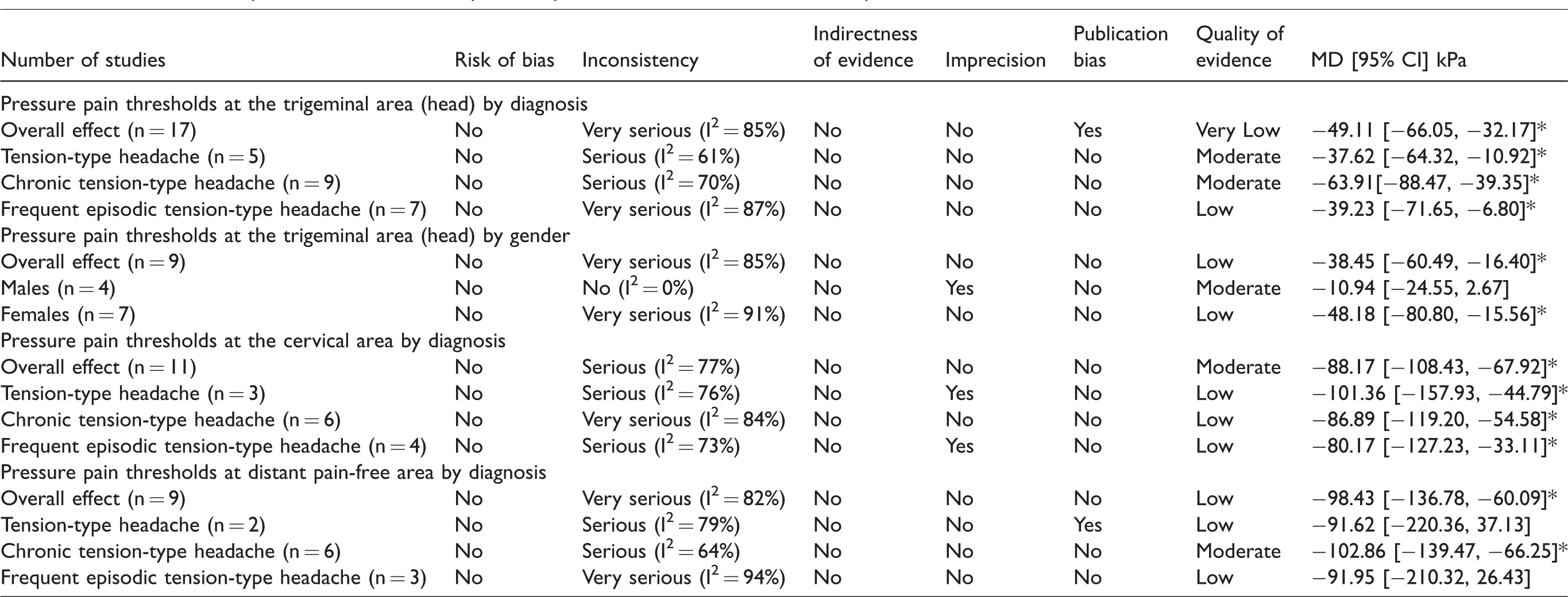
Table 5 displays the details of GRADE assessment showing Risk of Bias (RoB), inconsistency of the results, indirectness of evidence, imprecision of results, and high probability of publication bias. In general, the inconsistency of the results downgraded the evidence assessment, leading to low to moderate evidence in most pooled data.
GRADE evidence profile for differences in pressure pain thresholds between headache patients and controls.
Discussion
This is the most comprehensive review and meta-analysis conducted to date analyzing differences in PPTs between patients with TTH and headache-free controls. The results found low to moderate evidence supporting the presence of pressure pain hypersensitivity in the trigeminal and cervical areas in TTH in comparison with headache-free controls. Sensitivity to pressure pain was higher and more widespread in people with the chronic form of TTH (moderate evidence). In general, women exhibited lowers PPTs than men.
Findings
We were able to include 20 studies of fair to good methodological quality. Pooled data found lower PPTs in the trigeminal (head), cervical spine and distant extra-trigeminal pain-free areas in patients with TTH compared to headache-free controls. Current results are in line with the systematic review of Andersen et al. (14) and the meta-analysis of Castien et al. (15), which also found that patients with TTH had reduced PPTs in the craniofacial and cervical muscles compared to healthy subjects. Andersen et al. (14) reported PPTs in the temporalis muscle ranging from 248.4 to 264.2 kPa in patients with TTH, values similar to those pooled for the head in the current meta-analysis (mean: 214.9 kPa, n = 931). Similarly, Castien et al. reported a pooled mean difference of −109.57 kPa (95% CI −129.25 to −89.8, n = 5) in people with CTTH (15); again, similar to our data (CTTH mean: −86.89 kPa, 95% CI −119.20 to −54.58). We should note that Castien et al. (15) included in their analysis the study by Tüzün et al (52), which investigated PPTs in adolescents and not adults, and therefore was excluded from our analysis. The current meta-analysis is the first providing data of PPT differences in distant (extra-trigeminal) pain-free areas in people with TTH. We confirm that patients with CTTH have lower PPT values in distant pain-free areas compared to headache-free controls.
It is important to determine if differences between patients with TTH and controls are clinically relevant and determine the presence of pressure pain hyperalgesia. Romero-Morales et al. (35) determined that a difference of 16.18 kPa in the temporalis muscle (head) and of 78.94 kPa within the upper trapezius (neck) could be considered as the minimal clinically relevant difference in individuals with TTH. Walton et al. found a minimal detectable change of 42.7 kPa for the cervical spine and of 86.3 kPa for the tibialis anterior (distant pain-free area) in asymptomatic subjects (53). Our pooled estimates were larger than all these values, supporting the presence of both real differences (larger than test error) and clinically important differences between patients and controls at symptomatic (head and neck) and distant pain-free areas. Additionally, Andersen et al. (14) determined a cut-off value of 230 kPa for PPTs applied at sites on the head, suggesting that lower PPT values could be considered as a sign of sensitization and a risk for development or promotion of headaches.
The pooled data reported for the head and the neck in the current meta-analysis support the presence of trigemino-cervical sensitization in individuals with TTH in both subtypes, FETTH and CTTH. Additionally, the presence of hypersensitivity to pressure pain in distant extra-trigeminal pain-free areas also supports the presence of widespread central sensitization, but only in CTTH. These results support the pathophysiological model of sensitization in TTH where the episodic form seems to be more peripherally dominant and the chronic form more centrally dominant (10). In fact, evidence supports that development of central sensitization is a continuous process related to an increase in the frequency of headaches (47). The current meta-analysis would support that widespread hypersensitivity to pressure pain is present in CTTH but to a lesser extent or not at all in FETTH. Nevertheless, the lack of information related to headache clinical features in several studies does not permit determination of the exact frequency of the headaches and proper classification of patients with frequent or infrequent episodic forms. In addition, the small number of studies analyzing distal pain-free area PPTs in patients with episodic TTH means we cannot exclude that lack of findings in the FETTH population were merely due to low power.
Interestingly, the presence of widespread pressure pain hyperalgesia has not been consistently reported in patients with migraine, since lower PPTs were found in local, but not in remote pain-free, areas (54). It is possible that the widespread sensitivity to pressure pain is a common manifestation of TTH, but is not as common in migraine. Although rarely specified, methodological issues may also play a role here as some migraine patients also suffer from coexisting TTH. The findings may also be related to the fact that pathogenesis of TTH is most associated with musculoskeletal disorders, mainly featuring pressure pain hyperalgesic responses, whereas migraine pathogenesis involves more activation of sub-cortical structures and the trigemino-vascular system. This hypothesis would explain that impairments in nociceptive gain processing in migraine patients may be more modality, measure, and location specific (54) than in TTH.
We also observed that women had lower PPTs than men in all locations regardless of the presence or absence of headache. The presence of higher hyperalgesic mechanical responses in women is consistent in the literature and is in line with higher responsiveness to nociceptive stimuli associated with biological, hormonal or biopsychosocial factors (55,56). One interesting finding was that differences between patients with TTH and headache-free controls in the trigeminal area were more consistent in women than in men. Future studies including large sample sizes and differentiating by gender are needed to further confirm these results since we were not able to pool PPTs by gender in other locations.
Strengths and limitations
One of the strengths of the current review was the rigorous methodology applied for literature search, study selection, screening for eligibility, assessment of risk of bias, and pooling analysis of data by three authors. We included a total of 20 studies, a greater number than those included in previous reviews on TTH (14,15). In addition, several studies fulfilled several recommendations provided by Andersen et al. for assessment of pressure algometry (14): An algometer probe size of 1 cm2, using the mean value of three consecutive measures and control groups comparable by age.
Nevertheless, some limitations are also recognized. First, we could not conduct a meta-regression due to the high heterogeneity between the studies. The lack of data on several clinical outcomes (e.g. gender, or headache features) could influence the results. Although all studies applied ICHD for the inclusion of the participants, not all studies reported on the severity/frequency of headaches and consecutiveness of patients. A limited number of studies reported PPTs separately in women and men and most studies did not control for the menstrual phase in women, which may influence the pain thresholds. Therefore, gender differences in PPTs were only able to be pooled in the trigeminal area, but not in the cervical spine and distant extra-trigeminal pain-free areas. The present PPT differences by gender should be considered with caution. We pooled data for PPT values but there was a great variety of sites (e.g. frontalis, temporalis, parietal) and tissues (e.g. muscle, joint or bone) measured, mostly in the trigemino-cervical region. Only articles published in English were included, but because the majority of available articles are in English, this has probably not had a major impact on the overall findings. Finally, publication bias was possible in the meta-analysis of PPTs in the trigeminal area, but this was mostly related to a single study (38).
Clinical and research implications
The present review and meta-analysis highlights the importance of pressure pain hypersensitivity in TTH as manifestation of peripheral and central sensitization and opens several questions for future research. First, standardized protocols for PPT assessment would permit better characterization of differences between individuals with TTH and controls. Second, PPTs should be provided separately by gender, age, and frequency of headaches. This will lead to more solid and robust conclusions and better differentiation between episodic and chronic forms of TTH. Third, studies investigating quantitative sensory testing in TTH have mainly focused on pressure thresholds; however, thresholds and tolerances to other stimuli; for example, thermal, vibration, or electrical, are needed. In fact, the German Research Network for Neuropathic Pain protocol includes 13 measures to assess sensory profiles in patients with neuropathic pain by measuring loss of sensory function, presence of allodynia, and hyperalgesia (57). The application of the entire protocol in individuals with TTH could permit better understanding of the altered nociceptive pain processing in this population.
Conclusions
This meta-analysis found low to moderate evidence supporting lower PPT values in trigeminal and cervical symptomatic pain areas in patients with TTH when compared with headache-free controls. In addition, sensitivity to pressure pain was widespread only in chronic, not episodic, TTH (moderate evidence). Hypersensitivity to pressure pain was localized in the episodic form and widespread in the chronic form. In addition, women were more sensitive than men. Current results support the neurophysiological model of peripheral sensitization in patients with episodic TTH and central sensitization in chronic TTH.
Key findings
This systematic review and meta-analysis found low to moderate evidence supporting
pressure pain hypersensitivity in tension-type headache in comparison with
controls. Patients with tension-type headache, either episodic or chronic, exhibited pressure
pain hyperalgesia within the trigeminal and cervical regions. Sensitivity to pressure pain was widespread only in chronic tension-type
headache.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420958384 - Supplemental material for Evidence of localized and widespread pressure pain hypersensitivity in patients with tension-type headache: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-cep-10.1177_0333102420958384 for Evidence of localized and widespread pressure pain hypersensitivity in patients with tension-type headache: A systematic review and meta-analysis by César Fernández-de-las-Peñas, Gustavo Plaza-Manzano, Marcos J Navarro-Santana, Jes Olesen, Rigmor H Jensen and Lars Bendtsen in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_0333102420958384 - Supplemental material for Evidence of localized and widespread pressure pain hypersensitivity in patients with tension-type headache: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-cep-10.1177_0333102420958384 for Evidence of localized and widespread pressure pain hypersensitivity in patients with tension-type headache: A systematic review and meta-analysis by César Fernández-de-las-Peñas, Gustavo Plaza-Manzano, Marcos J Navarro-Santana, Jes Olesen, Rigmor H Jensen and Lars Bendtsen in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_0333102420958384 - Supplemental material for Evidence of localized and widespread pressure pain hypersensitivity in patients with tension-type headache: A systematic review and meta-analysis
Supplemental material, sj-pdf-3-cep-10.1177_0333102420958384 for Evidence of localized and widespread pressure pain hypersensitivity in patients with tension-type headache: A systematic review and meta-analysis by César Fernández-de-las-Peñas, Gustavo Plaza-Manzano, Marcos J Navarro-Santana, Jes Olesen, Rigmor H Jensen and Lars Bendtsen in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
