Abstract
Aim
The aim of this article is to investigate if children (7–17 years) with frequent episodic tension-type headache (FETTH) or chronic TTH (CTTH) have an altered pain perception compared to healthy controls.
Methods
We applied a pressure of five increasing intensities to m. trapezius and m. temporalis with a Somedic Algometer II. Visual analogue scale-score was rated and area under the curve (AUC) calculated. An average AUC in each person was used as an outcome variable in further univariate multiple linear regression analysis because factor analysis showed that AUC represents only one dimension underlying both muscles.
Results
Participants included 22 children with FETTH, 36 children with CTTH and 57 controls. The CTTH group had a significantly higher AUC compared to the control group (p < 0.001). The FETTH group represented an intermediate state. AUC did not change with increasing age, headache years, headache intensity, headache frequency or sex.
Conclusion
Children with CTTH show significantly increased pain sensitivity in a range of pressures compared to the FETTH group and the controls. Since AUC in m. trapezius and m. temporalis represents only one general latent tenderness, it might indicate that the altered pain perception is mainly due to central sensitisation.
Keywords
Introduction
One of the most frequent primary headache disorders in children is tension-type headache (TTH) (1). In schoolchildren and adolescents the prevalence of TTH is reported to be 10–25%, but 0.1–5.9% of all children suffer from chronic tension-type headache (CTTH) (2,3). Quality of life is often impaired in children with primary headaches (4); chronic pain, which is often associated with mood disorders, lost social relations and decreased school attendance, might have profound consequences for the children in every aspect of their daily lives (5,6). It is believed that episodic headache can convert to chronic headache; however, the mechanisms of the conversion are not clearly established (7). A genetic predisposition to chronification may exist, but environmental factors appear to play a major role (8).
The last two decades have provided substantial knowledge regarding the mechanisms of TTH in adult headache sufferers. Both frequent episodic tension-type headache (FETTH) and CTTH are associated with increased tenderness of the pericranial myofascial tissues (9–11). Where the pain originates is not clearly understood, but it may be the result of nociceptive input from the tender muscles. Stimulus-response functions for pressure versus pain have shown altered qualitative pain perception in patients with CTTH compared with headache-free controls (12). Various studies have contributed to the hypothesis that altered pain sensitivity in FETTH is established through sensitisation of peripheral nociceptors (7) and that prolonged nociceptive input from tender pericranial muscles results in sensitisation at the spinal/trigeminal level and in higher-order neurones converting FETTH to CTTH (7,13–21).
Only very few studies have focussed on the pathophysiological mechanisms of TTH in children and adolescents. The few studies that exist have primarily concentrated on Total Tenderness Score (TTS) in pericranial tissue (22–24) and pressure pain threshold (PPT) in children with FETTH (22,24–26). Results have indicated an increased TTS, while the results comparing PPT in children with FETTH and controls have been conflicting.
In order to prevent headache chronification and to ensure optimal treatment, further investigating of the pathophysiological mechanisms of pain perception in children with TTH is imperative.
We hypothesised that children with TTH, especially those with very frequent headache, have a different pain perception from children without pain, which could represent sensitisation in the pain pathway. The aim of the present study was to conduct a reliable examination of stimulus-response functions for pressure versus pain in order to examine the differences in pain perception in children aged 7–17 years with FETTH or CTTH compared to healthy controls. Secondarily, our goal was also to examine any relationship between stimulus-response functions and headache frequency, headache intensity or headache years.
Methods
Study design
Cross-sectional case-control study
Participants
Patients
Consecutive patients (aged 7–17) were diagnosed with TTH according to the International Classification of Headache Disorders, second edition (ICHD-II) (27) by an experienced paediatrician at the Children’s Headache Clinic in Denmark (CHC) and screened for study enrolment from May 2009 to May 2011. All children underwent a detailed interview and neurologic examination carried out by a paediatrician at their first visit to the centre. The age of 7 was assumed to be the limit of cognitive capability to perform the tests correctly. The visual analogue scale (VAS) score has previously been tested as reliable and valid in children over 5–6 years of age (28). Inclusion criteria were: 1) diagnosed with FETTH or CTTH and 2) between 7 and 17 years of age. Exclusion criteria were: 1) children in whom suspicion of medication-overuse headache existed; 2) children who received prophylactic treatment; or 3) children who also had more than one migraine day per month.
Controls
Healthy controls were recruited from two schools in the Copenhagen area. The enrolment and examinations took place from May 2009 to November 2010. The children were told face to face about the study by a paediatrician in the classroom. Children who were interested in participating received a letter with the written information to take home to their parents. The parents were asked to respond by e-mail, and responding parents were subsequently contacted by telephone by the same paediatrician and informed oral consent was given. Written informed consent was provided later.
Children were excluded if they had more than 12 headache days per year or suffered from any kind of chronic illness. The exclusion criteria were based on the assumption that infrequent TTH frequently occurs among children (1,3), making it difficult to recruit children with no headache at all. Furthermore, assuming the presence of infrequent headache (<one day per month) would not affect pain perception measured by the test being applied.
Setting
Within the space of three weeks from their first visit at the CHC and before any interventions were initiated, patients were tested at the CHC. Healthy controls were tested in their own schools during the school day.The participating children were not permitted to take pain medication 12 hours before the test to prevent interference with any pain perception in the myofascial tissues. During the test all children were examined by the same physiotherapist with paediatric experience, and each child was assisted by the same paediatrician. The physiotherapist was blinded for the child’s answers, but not for disease/control status. She had no information about each patient’s headache history and the children were unknown to her. Besides stimulus-response functions the children were also examined with TTS, PPT and individually PPT-adjusted suprathreshold stimuli. The results will be published in another paper.
Lasting in total 30 minutes, the tests was conducted in a quiet, comfortable room. The FETTH and CTTH children were accompanied by a parent. Parents of the healthy school children were invited to participate, but refrained from attending in the belief that their children felt secure. All procedures were carefully explained before each test.
The child was placed in a vertical position in an adjustable chair with head and armrests. The chair was individually adjusted to obtain relaxed muscles during the test. The child was examined dressed but was asked to uncover the shoulder and neck area.
Variables
Variables included VAS score, area under the curve in m. trapezius (AUCtrap), AUC in m. temporalis (AUCtemp), the average AUC (T-AUC), age, sex, headache frequency, headache intensity and headache years.
Self-reported measurements at the first visit to the clinic
The self-reported measurements at the first visit to the clinic involved the following: 1. Headache frequency = headache days per month; 2. Headache intensity = the most dominating visual VAS score during each headache attack; and 3. Headache years = total number of years with headache.
Data were obtained by interviewing the parents and child and from a headache diary covering the month immediately prior to the interview.
Apparatus
The same Somedic Algometer II was used for all subjects and calibrated with a 100 kPa weight before each test day (29).
Test
Algometry has previously shown to be reliable (intra and inter-rater) in measuring PPT in children (30).
The Somedic Algometer II was used to apply five different pressure intensities to the test sites: S1 (m. trapezius – the point halfway between C7 and Acromion) and S2 (the anterior temporal region – where palpation was used to find the muscle belly during contraction. The instrument was placed perpendicular to the skin surface. Only the non-dominant side was tested to avoid any difference in tissue composition and pain perception according to hand dominance. The intensities were: 23 kPa, 45 kPa, 67 kPa, 89 kPa and 111 kPa.
Alternating between the two muscles, each type of pressure was applied in random order for a total of four seconds, using the first two seconds to reach the intended pressure. The child was asked to score the pain level with a pencil on a printed VAS showing a happy face on one end and a crying face on the other end. The child was told that the happy face represented no pain and the crying face the worst pain they could imagine. The VAS score was later measured 0–10.0 cm. The time interval between stimuli at the same muscle was a minimum of one minute.
Stimulus-response functions have been conducted in adult TTH-sufferers using a palpometer (12). The original palpometer was specially designed and not commercially buyable, therefore, we chose to use the Somedic Algometer II instead. However, in order to compare results, we standardised the stimuli on the algometer to the palpometer pressures (80–200 arbitrary units (AU)). The two highest stimuli equivalents (180 and 200 AU) were withdrawn, as a pilot study showed that stimuli above 111 kPa were too painful for some children and thus ethically unacceptable because of the participants’ age. Furthermore, to replicate the area of second finger pulpa, to produce results comparable to that previous study, a 2 cm2 tip was used on the algometer.
Study size
The present pain measurement test has never been used in children before, thus making it impossible to conduct sample size estimations a priori. In order to have enough power in the study to show a significant difference between groups, even though it might be lower than in adults (12), our goal was to include 60 patients and 60 controls. The distribution of children in the FETTH and CTTH groups were, however, not determined a priori; they were selected solely on consecutive basis when diagnosed with TTH by CHC.
Approval
Written informed consent was obtained in all patients and controls. The study was approved by the local biomedical research ethics committee H-D-2009-019 and the Danish Data Protection Agency 2009-41-3172.
Statistics
SPSS 20 computer software was used for data analysis. A statistician was consulted for statistical assistance. VAS scores in tests and AUC are characterised by median and interquartile range (IQR). AUC was calculated for the two muscles, m. trapezius and m. temporalis, using the trapezoidal method (31).
The two measurements of tenderness were assumed to be correlated to a degree where a factor analysis in which AUC measurements were assumed to depend on a latent general tenderness variable, θ, might be realistic. According to this model, AUCtrap = α + βtrapθ + εtrap and AUCtemp = α + βtempθ + εtemp, where the βtrap and βtemp parameters are called factor loadings. Loadings are not required to be identical, but if they are, it encourages using the average AUC (T-AUC) as an outcome variable in further multiple regression analyses by general linear models (GLM).
We used a paired-samples t test to test significant differences between AUC in m. trapezius and m. temporalis.
We conducted univariate multiple linear regression on the following dependent variable: T-AUC. The following explanatory variables were used: FIXED FACTORS: group (FETTH/CTTH/control), sex (female/male); COVARIATES: age, headache frequency, headache intensity, headache years. We also included interactions between factors in the models.
The assumptions of linearity, independence, normal distribution of response and homogeneity of variance (Levene’s test insignificant) were satisfied. We used backward elimination of the explanatory variables.
A p value less than 5% was considered statistically significant.
Results
Descriptive data
Patients
Around 400 children attended the clinic in the study period. They were all screened for the inclusion and exclusion criteria by the paediatrician at their first visit. Sixty-four children were confirmed eligible and present on days when the research team also were present and could provide oral information face to face. Six children declined to participate, but 58 children agreed to participate and performed the test. No records of the six children who declined were registered. We included 22 children with FETTH, mean age 10.6 (SD 2.5), 55% females and 36 children with CTTH, mean age 12.6 (SD 2.2) and 78% females.
Controls
Twenty-eight classes with around 20 pupils each (aged 7–15) were told about the study in person by a paediatrician in the classroom. Sixty healthy pupils were included because they expressed interest in participating, but others were later rejected. Fifty-seven children performed the test as three children were absent on the day of examination. The control group had a mean age of 10.7 (SD 2.3) and 63% were female.
Missing values
One control child was clearly distracted at the 67 kPa stimuli in both test sites. Because the stimulus was intermediate, AUC was calculated from VAS scores at the other four stimuli in that child.
Twenty-seven percent of the FETTH group and 86% of the CTTH group had headache at examination.
Self-reported measurements
In the FETTH group the median headache frequency was 6.5 days/month (IQR 4–8.1), headache intensity was 5 cm (IQR 4–5.8) on a VAS scale and the children had suffered from headache for a median duration of two years (IQR 1–5.3) prior to the examination. The CTTH group had a median headache frequency of 30 days/month (IQR 21.5–30), a VAS score of five (IQR 4–6) and had suffered from headache for a median duration of two years (IQR 1–3.8) prior to the examination.
Data
Outcome data – VAS score for pressure at stimulus intensities: 23, 45, 67, 89 and 111 kPa in m. trapezius, m. temporalis and the two muscles combined.

Average muscles response: (VAStrap + VAStemp)/2; VAS: visual analogue scale; IQR: interquartile range; FETTH: frequent episodic tension–type headache; CTTH: chronic TTH.
Main results
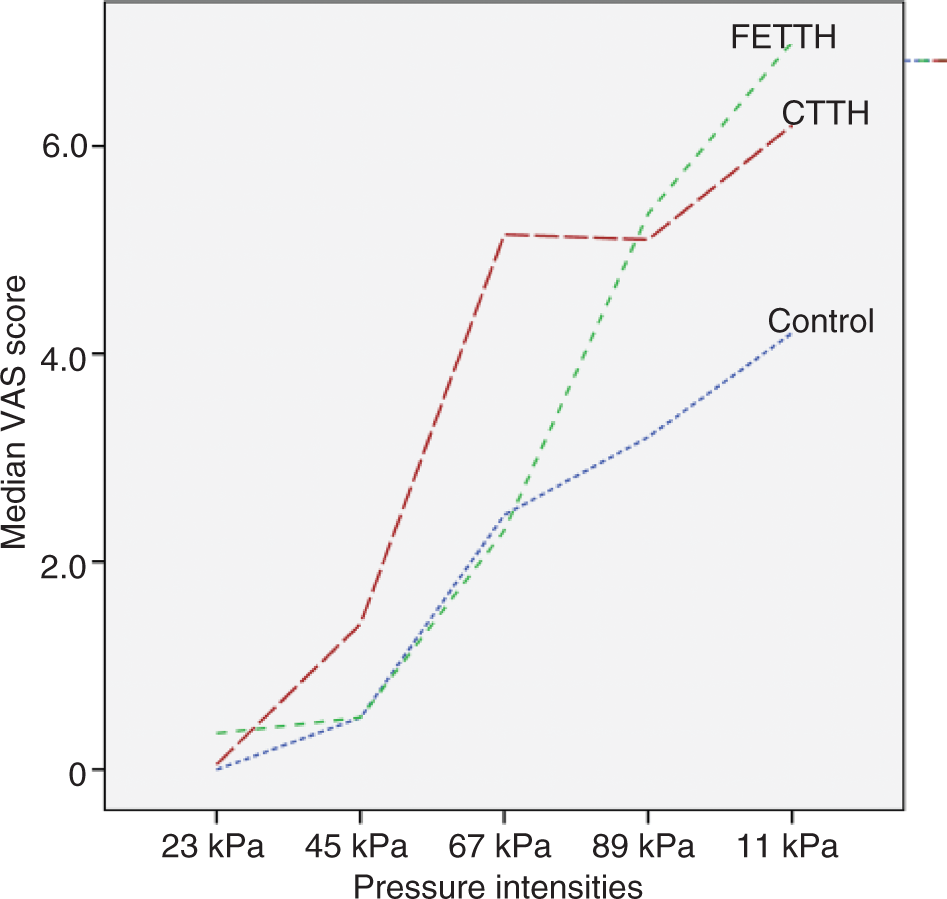
Figure 1 (m. trapezius) and Figure 2 (m. temporalis) present the stimulus-response functions for pressure versus pain. The stimulus-response functions clearly illustrate that the biggest difference in pain perception between the control and the CTTH group are found in the mid-pressure intensities. The two groups appear to approximate each other again in the low and high end of the stimulus range. The CTTH group has a higher VAS score in the whole range of pressure intensities in both muscles.
Stimulus-response function for pressure versus pain – m. trapezius. Stimulus-response function for pressure versus pain – m. temporalis.
Sensitivity in m. trapezius and m. temporalis
AUC in the two test muscles was significantly different in a paired-samples t test (p = 0.003). M. temporalis was more sensitive than m. trapezius in the same individual, although the difference was minimal (Figure 3, Table 2).
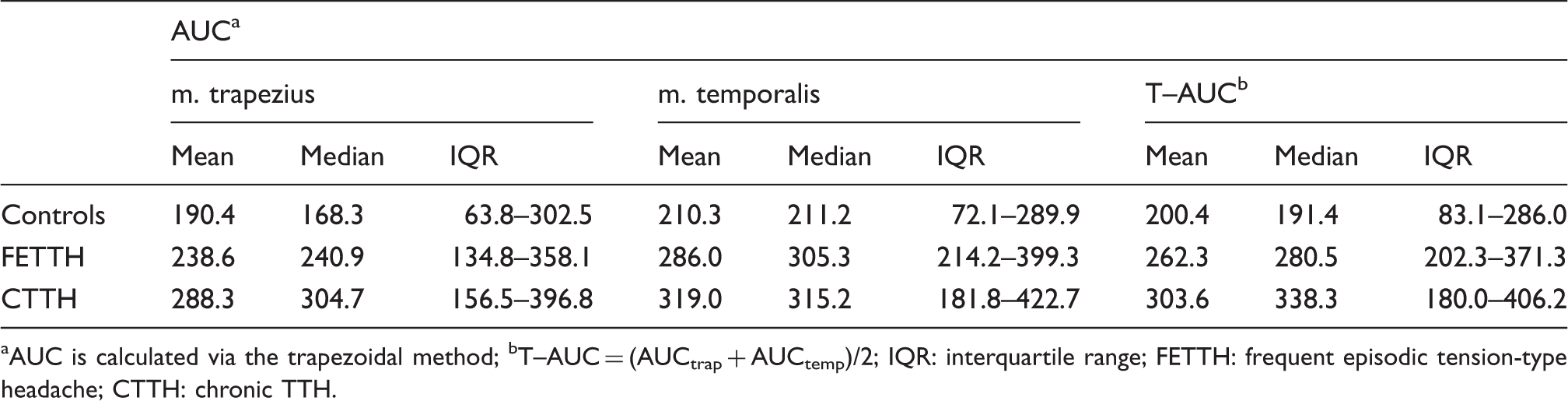
Correlation of AUC in m. trapezius and m. temporalis. Area under the curve for stimulus–response functions in m. trapezius, m. temporalis and the average of the two muscles (T–AUC). AUC is calculated via the trapezoidal method; bT–AUC = (AUCtrap + AUCtemp)/2; IQR: interquartile range; FETTH: frequent episodic tension-type headache; CTTH: chronic TTH.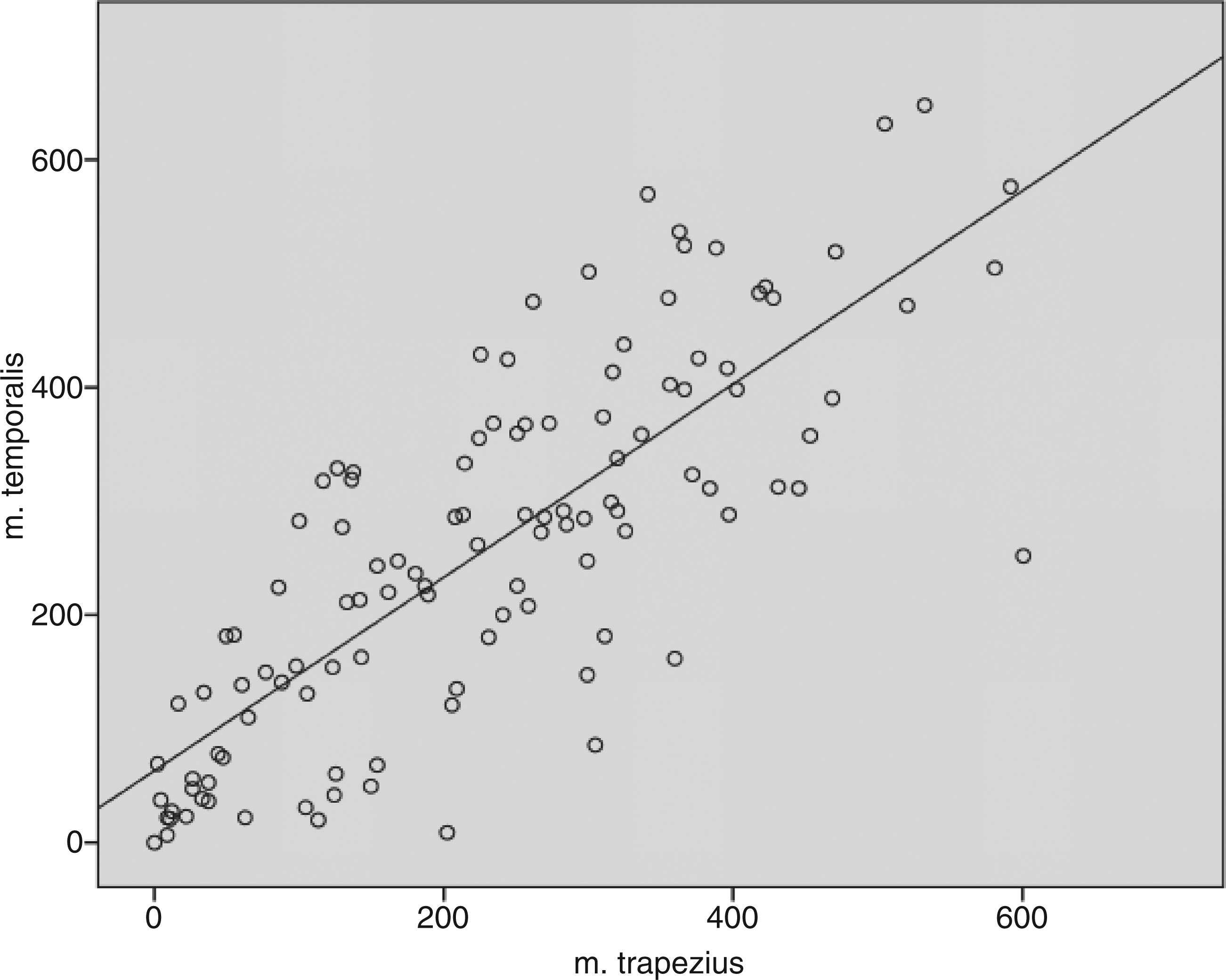
The strong correlation of the two AUC measurements (r = 0.79) and the highly linear association between the two measurements (Figure 3) justified conducting a factor analysis. This analysis confirmed the model and showed that the latent tenderness variable θ explained 89.7% of the variation of the AUC measurements. The analysis also showed that the loadings of the two measurements were almost the same (βtrap = 1 and βtemp = 1.028), therefore the average AUC (T-AUC) was used as the outcome variable in further GLM analyses.
GLM analysis
According to the multiple GLM analyses, sensitivity (T-AUC) did not change with increasing age, sex, headache years, headache intensity or headache frequency, even though the relation between T-AUC and headache frequency is nearly significant (p = 0.067). For this reason, the analysis of the difference between the control group, the FETTH group and the CTTH group reduces to a simple analysis of variance. Of these, the CTTH group had the highest T-AUC level (median 338, IQR 180–406), the control group had the lowest T-AUC level (median 191, IQR 83–286), and the difference between these two groups was highly significant (p < 0.001). The T-AUC level in the FETTH group (median 281, IQR 202–371) was higher than in the control group and lower than in the CTTH group, but the difference between the FETTH level and the level of the two other groups was not significant (p = 0.084 and 0.283, respectively).
Discussion
Quantitative changes in pain perception
The present study shows that children with CTTH have an increased sensitivity (T-AUC) for mechanical pressure compared to the control group (p < 0.001).
Measured by AUC in m. trapezius, an altered pain perception has also been shown in adult patients with CTTH in a previous study (12). Our results are also consistent with previous findings of increased sensitivity in adults suffering from CTTH, when measured by other pain perception assessments such as electrical or thermal thresholds or PPT (11,13,14,32–36); and suprathreshold stimuli (21,37).
T-AUC in the FETTH group was not significantly different from either the control group (p = 0.084) or the CTTH group (p = 0.283), which is paradoxical since the control and CTTH groups were significantly different. The only logical explanation is that the FETTH group must represent an intermediate state between the two groups in the extreme.
To our knowledge no study has previously been conducted on stimulus-response functions for pressure versus pain in adults suffering from FETTH. Our finding of FETTH as an intermediate state combined with the finding of a nearly significant linear positive relation between headache days per month and T-AUC supports Schmidt-Hansen et al.’s hypothesis that FETTH and CTTH seem to represent a continuum of changes in the peripheral and central mechanisms of pain pathways rather than two separate entities (38).
Qualitative changes in pain perception
In the present study m. temporalis has a higher AUC than m. trapezius (p = 0.003), which leads to the conclusion that m. temporalis is the most sensitive muscle in a child with TTH, measured by AUC. Because of this difference between muscles, we evaluate the quality of the graphs separately for the two muscles. If sensitivity quantitatively increases in children with CTTH, the primary expectation is a parallel-displaced function for pressure versus pain for the various headache groups. Looking at the CTTH group and the control group, however, our groups appear to differentiate most in the mid-pressure intensities but approximate each other again in the lower and higher end of the stimulus range, i.e. there are qualitative changes in pain perception (Figures 1 and 2). This pattern is most clearly visible in m. trapezius (Figure 1).
The same qualitative changes were previously found in the stimulus-response study of adults with CTTH (12). Bendtsen et al. concluded that the qualitatively altered response to nociceptor stimulation in tender muscles (linear function) was probably caused by activation and central sensitisation of low-threshold mechanosensitive neurones (LTM) at the level of the spinal dorsal horn/trigeminal nucleus (7).
Peripheral or central sensitisation
A measure of the individual child’s stimulus-response functions for pressure versus pain, AUC represents the perceived pain when tenderness of the myofascial tissue is tested in a broad range of stimulus intensities. The nociceptive inputs can be affected, however, by different segments in the sensory pathway before they are perceived as pain in the cortex cerebri.
It has been proposed that increased TTS in TTH patients reflects the peripheral sensitisation of nociceptors and their neurons, whereas altered PPT and AUC primarily, or in part, reflect central sensitisation at the level of second-order neurons in the spinal dorsal horn/trigeminal nucleus or supra-spinal neurons (7,12).
Even though the two muscles in our study had a significantly different tenderness in the same individual, factor analysis revealed that AUC represents a dimension (latent general tenderness variable θ) common to the two muscles. In other words, the same child will have a different level of tenderness in different muscles, but a latent general tenderness exists, which determines this individual’s “overall” sensitivity level compared to others’. The altered pain perception found in CTTH must therefore represent changes in pain pathways shared by both muscles. The two muscles are sensory innervated by different nerves, which means the changes must be at the level of the central nervous system. Given the combination of AUC as one aspect of factor analysis and the qualitative changes seen, our conclusion is that the observed changes in AUC between groups might represent central sensitisation. However, chronic peripheral sensitisation might also contribute to the changes observed.
FETTH represents an intermediate group. If AUC reflects central sensitisation, we must conclude that the FETTH group is also partly centrally sensitised. This finding is supported by two recent studies by Fernández-de-las-Peñas et al., who documented significantly decreased PPTs in both the muscles and nerves of children with FETTH (mean 12.7 and 14 days/month) (24,25). Another study by Anttila et al., however, could not detect any difference in PPT between the FETTH group and the controls, but in this case the headache frequency was very low (mean three days/month) (22).
Limitations
Blinding the physiotherapist would have been the optimal study design. The set-up, however, made it impossible as the controls were examined at school to obtain enough healthy volunteers. No information about the patient’s headache history was available to the physiotherapist, who did not know any of the children. Bias would create differences between headache groups as our physiotherapist would expect the sick children to be more sensitised.
Although we tried to establish the same confident atmosphere, the children were examined in two different surroundings (CHC/school) with the risk of environmental nocebo. The expectation of pain and the unknown setting in the sick children might increase their sensitivity compared to the controls.
We did not check for psychiatric co-morbidity or fibromyalgia in a standardised manner; however, it was not our impression that the children had serious co-morbidity.
Distinguishing between children with or without headache on the day of examination is another limitation. One could argue that the current headache status could interfere with pain perception and tenderness. Whether this is true for stimulus-response functions for pressure versus pain is unknown; however, this was not the case in a study of PPT in adults (38). Pericranial tenderness assessed by TTS seems to be influenced by the actual headache status, although increased TTS is found in TTH both on days with and without headache (39).
Another limitation could be that only children suffering from headache were accompanied by a parent at the examination. If the controls were more anxious about participating alone it would have induced bias in terms of a higher AUC in the control group, thus counteracting the real differences visible between sick children and the controls. Despite this limitation, we still found a significant difference in AUC between the groups.
Conclusion
In children suffering from CTTH for an average duration of two years, pain perception for pressure versus pain is quantitatively and qualitatively altered. These changes seem to be a continuum of changes, with the FETTH representing an intermediate state between controls and children with CTTH. Whereas AUC in m. trapezius and m. temporalis represents only one dimension common for both muscles, it might indicate that the altered pain perception mainly is due to central sensitisation.
Our results need to be confirmed in future studies. However, our results might have several implications for children with TTH in the future. There has been a tendency among the general population not to recognise TTH in children and adolescence because of a consensus that headache in children is not a disease but just a way to excuse psychogenic problems. Our study may contribute to changing this opinion. The finding of progressing central sensitisation emphasises that TTH in children is a neurological disorder and that early recognition and treatment by health personnel is necessary to avoid progression along the spectrum from infrequent to CTTH. Furthermore, the finding of central sensitisation establishes a “set of” for new drug trials in order to find efficient prophylactic medication for children with CTTH. Future research in children with TTH should aim at ways of reducing sensitisation and identifying the sources of peripheral nociception.
Clinical implications
Children with chronic tension-type headache (TTH) have a quantitatively and qualitatively altered pain perception for pressure versus pain. These changes seem to be a continuum, with frequent episodic TTH representing an intermediate state. Since the area under the curve in m. trapezius and m. temporalis represents only one general latent tenderness common for both muscles, it might indicate that the altered pain perception is due to central sensitisation
Footnotes
Acknowledgement
We would like to thank the healthy children from Sengeløse School and Søndervangs School, as well as the patients at the CHC who agreed to participate.
Funding
The work was supported by The Lundbeck Foundation (grant number R19-A2040, R34-A3618); the Dagmar Marshalls Foundation; the Tømrermester Jørgen Holm and wife Elisa F. Hansen’s Memorial Trust; the Capital Region of Denmark (grant number R120-A3256); the Danish Headache Society; and the Professor Torben Iversen Travel Fund for Young Paediatricians.
Conflict of interest
None declared.
