Abstract
Background
The characteristics of the hypersensitivity to auditory stimuli during the interictal period in episodic migraine are discussed. The combined use of event-related potentials, time-frequency power and phase-synchronization can provide relevant information about the time-course of sensory-attentional processing in migraine and its underlying mechanisms.
Objective
The aim of this nested case-control study was to examine these processes in young, female, episodic migraine patients interictally and compare them to controls using an active auditory oddball task.
Method
We recorded, using 20 channels, the electrophysiological brain activity of 21 women with episodic migraine without aura and 21 healthy matched controls without family history of migraine, during a novelty oddball paradigm. We collected sociodemographic and clinical data as well as scores related to disability, quality of life, anxiety and depression. We calculated behavioural measures including reaction times, hit rates and false alarms. Spectral power and phase-synchronization of oscillatory activity as well as event-related potentials were obtained for standard stimuli. For target and novel stimuli, event-related potentials were acquired.
Results
There were no significant differences at the behavioural level. In migraine patients, we found an increased phase-synchronization at the theta frequency range and a higher N1 response to standard trials. No differences were observed in spectral power. No evidence for a lack of habituation in any of the measures was seen between migraine patients and controls. The Reorienting Negativity was reduced in migraine patients as compared to controls on novel but not on target trials.
Conclusion
Our findings suggest that migraine patients process stimuli as more salient, seem to allocate more of their attentional resources to their surrounding environment, and have less available resources to reorient attention back to the main task.
Keywords
Introduction
Migraine is a neurosensory brain disorder involving altered sensory processing, which has been associated with thalamocortical dysrhythmia (1). Previous studies have reported abnormal neurophysiological responses in migraine, which can be summarized as enhanced sensory processing and a lack of habituation (1) during the interictal period. Nonetheless, while these abnormalities have been exhaustively analyzed in the visual modality, results obtained in audition remain inconsistent (2).
Auditory oddball tasks combined with electrophysiology (EEG) are one way of better understanding auditory processing, also allowing the study of attention and behavioural readjustments to environmental demands (3). In its active form, repetitive sounds, standard stimuli, and two types of less frequent, differential sounds, target and novel stimuli are presented to the participant, who is instructed to respond to targets only (4,5).
N1 and P2 Event-Related Potentials (ERPs) have been linked to early allocation of attention and to evaluative processing of repetitive standard stimuli. N1 is a negative electrophysiological component (peak 75–150 ms post-stimulus), which is affected by stimulus salience and the degree of attention (6,7). P2 (150–250 ms), a positive component, has been related to higher order perceptual processing (8,9), early allocation of attention and initial awareness (6). Most previous studies reported no group differences in either the N1 or P2 amplitude or habituation in migraine patients as compared to controls (3,10–13). However, other studies did observe an increased N1-P2 amplitude in migraine (14,15).
Target and novel sounds during active oddball tasks allow us to evaluate aspects of sensory-perceptual and attentional processing, which are essential to better comprehend the relationship between the evaluation of incoming information, the management of attentional resources, and the behavioural response to external stimuli (see Table 1 for a detailed description of the specific cascade of sensory and attentional processes, from the MMN to the RON, involved in the processing of novel and target stimuli) (4,16–19). Novel stimuli generate a cascade of electrophysiological components including the mismatch negativity (MMN), early P3a, late P3a and reorienting negativity (RON), which are related to detection and processing of unexpected events as well as subsequent attentional reorientation. Similarly, processing of target stimuli involves the MMN, P3b and RON, which are linked to contextual memory comparisons necessary for the behavioral response and the reorientation of attention back to the main task. Within the migraine literature, some studies have reported equal MMN amplitude (10,19) and similar P3a amplitude in patients as compared to controls (3,13,20). Furthermore, some studies have reported a reduced P3b amplitude in response to deviant or target stimuli (3,11,21,22), whereas others did not encounter group differences (19,23,24).
Cascade of effects of novel and target stimuli on the sensory-attentional system.

Note: Novel stimuli lead to a cascade of event-related potentials, which include, in order of appearance, the Mismatch Negativity (MMN), the early and late P3a, and finally the Reorienting Negativity (RON) (4,17,18). ERPs associated with target stimuli include the MMN, the P3b and the RON (4,16–19).
While ERPs can provide us with relevant information about the time-course of sensory-attentional processing, the transformation of data into time-frequency (TF) measures of power and phase-synchronization (or inter-trial phase coherence) can give information about more general underlying processes. Power is related to the amount of simultaneously active neurons measured at any given point in time, whereas phase-synchronization gives an index of phase consistency across trials. For example, after the presentation of standard, target or novel stimuli, there is a rise in theta activity, which coincides with the period in which the N1 and P3a components have their maximum amplitude. Similarly, an increase in low-beta activity has been observed after the presentation of novel stimuli (5,25). Phase-synchronization, together with measures of power, could partially contribute to ERP activity (26), especially during the presentation of repetitive stimuli. However, despite these measures offering a more direct link to thalamocortical activity when compared to ERPs, to the best of our knowledge, neither power nor phase-synchronization have been studied in auditory processing, in migraine patients interictally.
Given the discrepancies seen within the auditory evoked potential literature in migraine and the lack of literature related to time-frequency measures in response to auditory stimuli, the objective of this study was to examine whether or not there were significant group differences in ERPs, power or phase-synchronization on an active auditory oddball task between young episodic migraineurs during the interictal period and controls.
Method
This is a double-blind study, using an observational, nested case-control study design.
Participants
We decided to focus on young patients with low-frequency episodic migraine (EM) interictally as compared to healthy controls. From a sample of 45 participants that underwent the EEG recording, 42 participants remained in the study after the EEG artifact rejection (see EEG recordings section and Supplemental material for more details). The final sample consisted of 21 right-handed females with EM without aura (age: 22.99 ± 1.99, attack frequency: 4.38 ± 2.91 days/month), according to the International Classification of Headache Disorders 3rd edition, beta version (ICHD-3 beta) (27), and 21 age- and gender-matched healthy controls (HC) (age: 21.95 ± 2.20, age: t(40) = −1.598, p = 0.118). The inclusion criteria for all participants was to be between 18–30 years old, right-handed, and with normal or corrected-to-normal vision and hearing. Specifically, migraine patients chosen to take part in this study were all low-frequency, episodic migraineurs. There was a clear objective of having a very homogeneous and clinically-similar sample of participants to effectively compare brain responses and activity as well as to reduce possible biases. All participants were individuals who were unsure or unaware of their diagnosis. The exclusion criteria included the presence of other headache, neurological or psychiatric disorders as well as the use of specific medications/recreational drugs. Controls could not match the criteria for any headache (according to ICHD-3 beta), and they could not have any first-degree relatives with migraine (see Supplemental material for details about the recruitment process and exclusion criteria). The neurologist did not inform the participants of their diagnosis until the end of the EEG session to ensure that both the researcher and the participant remained double-blind.
Ethics approval: All participants gave their informed consent prior to participation and received 15 euros compensation at the end of the experimental session. This research study was approved by the Ethics Committee at the Vall d’Hebron Hospital (PR(AG)376/2017).
Procedure
A neurologist confirmed the diagnosis of EM and the absence of migraine (or other headache) and familial antecedents of migraine in HCs. Participants that were designated as fit to continue completed a digital migraine diary during 30 days prior to the experimental session. All participants, including controls, completed the daily calendar to reduce possible biases such as the selection of controls who were not truly headache-free. The diary specifically collected headache presence, duration, intensity and the use of acute treatment, menses and the participant’s sleep-wake cycle. Participants continued to fill in the diary 72 hours after the experiment. The experiment was done on a headache-free day preceded by a 72-hour headache-free window. During the session, participants completed patient-reported outcome (PRO) surveys (see Supplemental material) and underwent an EEG recording while performing an auditory oddball task. The entire session was 2.5 h long.
EEG recordings
We acquired continuous EEGs (digitized, sampling rate 500 Hz, no online filters, Neuroelectrics Enobio, Barcelona). Data was recorded using a 20-electrode EEG cap located over the scalp at standard positions (Fp1/Fp2, F3/F4, F7/F8, Fz, Cz, Pz, C3/C4, T7/T8, P3/P4, P7/P8, O1/O2), together with a common mode sense (CMS), and a driven right leg electrode (DRL) placed on the right mastoid. Electrode impedances were kept below 15 kΩ. EEG activity was re-referenced offline to the mean activity of all electrodes. Participants were instructed to keep their eyes open throughout the entire EEG session. Eye movements were monitored by an electrode placed at the infraorbital ridge of the right eye. From a sample of 45 participants, two EMs and one HC were not included in the final analyses based on EEG muscular and movement-related artifacts.
Auditory oddball paradigm
A variant of the active auditory oddball paradigm (5,25), in which an infrequent target tone (1620 Hz, 60 ms duration, 5 ms rise/fall times, probability of p = 0.2) occurred within a stream of standard tones (1500 Hz, 60 ms duration, 5 ms rise/fall times, probability of p = 0.6), was used. In addition to the standard and infrequent target tones, novel sounds (short excerpts of environmental sounds, such as the barking of a dog or the honking of a car) were also presented (average duration: 60.95 ± 7.61 ms, probability of p = 0.2). The stimuli were presented binaurally with the Presentation Software 18.1 (NeuroBehavioral Systems, San Francisco, CA, USA), through Sony MDR-ZX310APB headphones at 75 dB sound pressure level (SPL) in pseudo-random order with a stimulus onset asynchrony set to 1200 ms (±100 ms). A total of 500 trials (five blocks of 100 trials, 60/100 standard stimuli) were presented (duration ≅ 25min). A central black fixation cross (height, width: 13 pixels) was presented on a black background throughout the entire duration of the experiment. Participants were instructed to keep their eyes open, avoid blinking, and remain fixated on the central cross, at all times, throughout the experiment. They were also instructed to respond as quickly and accurately as possible to the target tones while ignoring the standard and novel tones.
Behavioral analyses
Reaction times (RTs) for target stimuli were obtained. For each participant, RTs that were ±3 standard deviations from their mean were excluded. The percentage of no-response (‘miss’) trials was also obtained by dividing the number of misses by the number of hits. Finally, the percentage of false alarms on standard and novel trials was calculated by dividing the number of standard/novel trials with a recorded response by the total number of standard/novel trials. Two-sample t-tests were used to compare these measures between groups. In addition, for standard stimuli, separate values were obtained for each block and submitted to repeated-measures analyses of variance (ANOVAs) with Time (Blocks 1–5) as a within-subject factor and Group (EM/HC) as a between-subject factor.
EEG pre-processing
First, the EEG was offline-filtered with a 50 Hz notch filter (type Parks-McClellan, order 180). Then, EEG data from ERP and TF analyses was pre-processed differently from ITC EEG data to avoid nonlinear phase distortions in ITC data (28). For ERP and TF, a Butterworth infinite impulse response (IIR) band-pass filter from 0.1 to 100 Hz was applied (high-pass: Frequency 0.1 Hz, order 2, cutoff −6 dB; low-pass: frequency 100 Hz, order 4, cutoff −6 dB). For ITC analyses, a Hamming windowed sinc finite impulse response (FIR) band-pass filter (zero-phase), from 0.1 to 100 Hz, was applied (high-pass: Frequency 0.1 Hz, order 16500, cutoff −6 dB; low-pass: frequency 100 Hz, order 66, cutoff −6 dB). Finally, epochs were separately obtained for standard, target and novel tones (ERPs: 100–1000 ms; power/phase-synchronization: −2000–2000 ms post-stimulus onset) and were baseline-corrected from −100 ms until 0 ms post-stimulus onset, as done in previous studies (5,25). Only correct trials were used to obtain the epochs. Correct trials were considered to be: Correct detections in response to target stimuli and no response to standard and novel tones. No participant had to be excluded due to an insufficient number of correct trials. Epochs exceeding ±100 µV in the electro-oculogram (EOG) or EEG were automatically detected and removed from further analysis, then a visual inspection of the data was performed to confirm that artifacts were correctly detected.
Single trial convolution was performed via frequency-domain multiplication, in which the Fourier-derived spectrum of the ERP data was multiplied by the spectrum of the wavelet, and the inverse Fourier transform was taken. A six-cycle complex Morlet was used. A separate time series of complex wavelet coefficients was obtained for each frequency from 1 Hz to 40 Hz (linear increase). These complex coefficients, containing both real and imaginary components, were used to derive the power and phase-synchronization (i.e. inter-trial coherence). Power was computed with respect to baseline. Both measures were obtained for each trial and averaged for each participant before performing the grand-average.
Electrophysiological analyses
The EEG analyses were divided as a function of standard trials and target/novel trials. For standard trials, the individual amplitude was separately obtained for each component and for each of the five blocks. The ERP mean amplitudes were set-centered on the peak activity of each component (N1: 75–125 ms; P2: 175–225 ms; N2: 250–350 ms). For spectral power and phase-synchronization, theta (3–8 Hz, 0–400 ms), alpha (8–12 Hz, 0–200 ms), and beta-gamma (12–40 Hz, 0–200 ms) frequency ranges were defined based on maximum activity and previous literature (5,25).
In the case of novel and target trials, the difference waveform ERPs were obtained between novel/target and standard trials. The following components were obtained, for novel: MMN (175–225 ms), Early P3a (225–275 ms), Late P3a (275–325 ms), and RON (350–450 ms); and for target stimuli: MMN (175–225 ms), P3b (450–550 ms), and RON (400–600 ms). For all ERP components, the mean amplitudes were set-centered on the peak activity. See Supplemental material for spectral power of target and novel trials.
Sample size was determined based on previous literature, which corroborated that 10–25 participants, with at least 20 artifact-free trials per condition, are enough for this kind of nested case-control EEG study (29,30). Consequently, participants with <20 correct responses or artifact-free trials of any condition were excluded from the analyses (30).
Standard trials were submitted to repeated-measures ANOVAs with Electrode location (Fz, Cz, Pz) and Block (1–5) as within-subject factors and Group (EM/HC) as a between-subject factor. A similar procedure was used for novel and target trials; however, the repeated-measures ANOVAs only had one within-subject factor: Electrode location (Fz, Cz, Pz) and one between-subject factor: Group (EM/HC). In addition, we used post-hoc t-tests comparisons when necessary.
For all analyses, p-values were corrected using the Greenhouse-Geisser correction for nonsphericity when appropriate.
Results
Behavioral results
In regards to behavioral measures, no significant differences between HC and EM were found on measures of RTs (HC: 512.53 ±73.29 ms, EM: 486.13 ±41.19 ms; t(40) = 1.602, p = 0.117), percentage of misses (HC 4.86 ±3.79%, EM 5.61 ±5.63%; t(40) = −0.506, p = 0.616), and false alarm rates on novel trials (HC 3.29 ±1.65%, EM:3.91 ±2.24%; t(40) = −1.023, p = 0.312). On standard trials, no modulation of the false alarm rate was observed across blocks (main effect of Block: F(4,160) = 2.466, p = 0.106), or as a function of group (main effect of Group: F(1,40) = 0.001, p = 0.978; Block × Group interaction: F(4,160) = 0.102, p = 0.854).
Electrophysiological results
Repetitive stimuli ERPs
See Figure 1 and Table 2 for repeated-measures ANOVAs. Typical N1, P2 and N2 ERP components were observed across blocks. A significant main effect of Electrode indicated the presence of a central distribution for N1 and P2, and a fronto-central distribution for N2. The lack of a significant main effect of Block together with the absence of a significant interaction of Block × Electrode for the N1 and N2 components, indicated that the amplitude of these components did not habituate across blocks. On the other hand, the amplitude of the P2 component was significantly reduced in the last block as compared to the first one, as seen by the significant main effect of Block and a significant Block × Electrode interaction.

Grand mean ERP waveforms for standard trials, including the 1st (solid black line), 2nd (pointed dark grey line), 3rd (pointed medium grey line), 4th (pointed light grey line), 5th (solid blue line), at midline electrodes (Fz, Cz, and Pz), from −100 to 800 ms, −3/+3 µV, for both the EM (a) and HC (b) groups. Scalp distribution (−3/+3 µV) for 1st minus 5th block of standard trials involves the N1, P2 and N2.
Repeated-measures ANOVA on the ERP components related to standard trials.

Note: The following components and time-windows were studied: N1 (175–225 ms), P2 (350–450 ms), and N2 (250–350 ms). The repeated measures ANOVA included B: Block (1st, 2nd, 3rd, 4th, 5th) and E: Electrode location (Fz, Cz, Pz) as within-subject factors and G: Group (Control, Migraine) as the between-subject factor. The degrees of freedom were as follow: B (4,160), B × G (4,160), E (2,80), E × G (2,80), B × E (8,320), B × E × G (8,320), and G (1,40).Bold values represent P<0.05.
In terms of group comparisons, a trend for increased N1 amplitude in the migraine group as compared to the control group was observed (see Table 2, N1, Group analyses, G). No differences were reported for P2 and N2 components between groups as reflected by a lack of a main effect of group and significant interactions. The lack of significant Block × Group interactions and Block × Electrode × Group interactions suggested that both groups presented a similar pattern of habituation (habituation of P2 and no habituation of N1 and N2).
Spectral power of repetitive stimuli
See Figure 2 and Table 3 for repeated-measures ANOVAs. As can be observed in Figure 2 and corroborated by statistical analyses, typical theta, alpha and beta-gamma activities were elicited in both groups, with certain habituation across blocks. The significant main effect of Electrode indicated the presence of a central distribution for all three frequencies. Habituation across blocks was confirmed for theta and alpha, but not for beta-gamma (see significant and non-significant main effects of Block in Table 3). No significant main effect of Group was obtained in theta, alpha, and beta-gamma power. Furthermore, a lack of significant Group × Block interactions implied that there were no differences between HC and EM in respect to the three frequencies. There were no significant group differences in habituation either.

Grand mean event-related spectral perturbation representing changes in power with respect to baseline of standard trials, separated by blocks, at midline electrodes (Fz, Cz, Pz), for the migraine (a) and the control group (b). The increase/decrease of power is represented in percentage of change (−100/+100%), from −100 to 800 ms. Topographical maps of the spectral power difference between the 1st block and the 5th block, are depicted for each frequency of interest (theta: 3–8 Hz, 0–400 ms; alpha: 8–12 Hz, 0–200 ms; and Beta-Gamma: 12–40 Hz, 0–200 ms) and group.
Repeated-measures ANOVA on the time-frequency spectral power of standard trials.

Note: The following frequency ranges and time-windows were studied: theta (3–8 Hz, 0–400 ms), alpha (8–12 Hz, 0–200 ms), and beta-gamma (12–40 Hz, 0–200 ms). The repeated measures ANOVA included B: Block (1st, 2nd, 3rd, 4th, 5th) and E: Electrode location (Fz, Cz, Pz) as within-subject factors and G: Group (Control, Migraine) as the between-subject factor. The degrees of freedom were: B (4,160), B × G (4,160), E (2,80), E × G (2,80), B × E (8,320), B × E × G (8,320), and G (1,40).Bold value represent P<0.05.
Time and phase-synchronization analyses of repetitive stimuli
See Figure 3 and Table 4 for repeated-measures ANOVAs. In Figure 3, both groups had increased phase-synchronization of theta, alpha and beta-gamma on standard trials, with a clear habituation across blocks. Upon visual inspection, the phase-synchronization appeared higher in EM compared to HC. A central topographical distribution was supported by a significant main effect of electrode. Habituation across blocks was also corroborated for theta and alpha but not for beta-gamma. Importantly, the significant main effect of Group confirmed the higher phase-synchronization in theta for the migraine group as compared to the control group (see Table 4, Theta, Group, G). No significant group differences were observed for the other frequencies (neither main effect of group, nor Group interactions). Similarly, no significant group differences were observed regarding the habituation of phase-synchronization of any of the three frequencies (see Table 4).

Grand mean inter-trial coherence (ITC, phase-synchronization) standard trials, separated by blocks, at midline electrodes (Fz, Cz, Pz), for the migraine (a) and the control group (b). The ITC (0/0.35) is depicted from −100 to 800 ms. Topographical maps of the ITC difference (−0.03/0.03) between the 1st block and the 5th block, are depicted for each frequency of interest (theta: 3–8 Hz, 0–400 ms; alpha: 8–12 Hz, 0–200 ms; and Beta-Gamma: 12–40 Hz, 0–200 ms) and group.
Repeated-measures ANOVA on the phase-synchronization of standard trials.
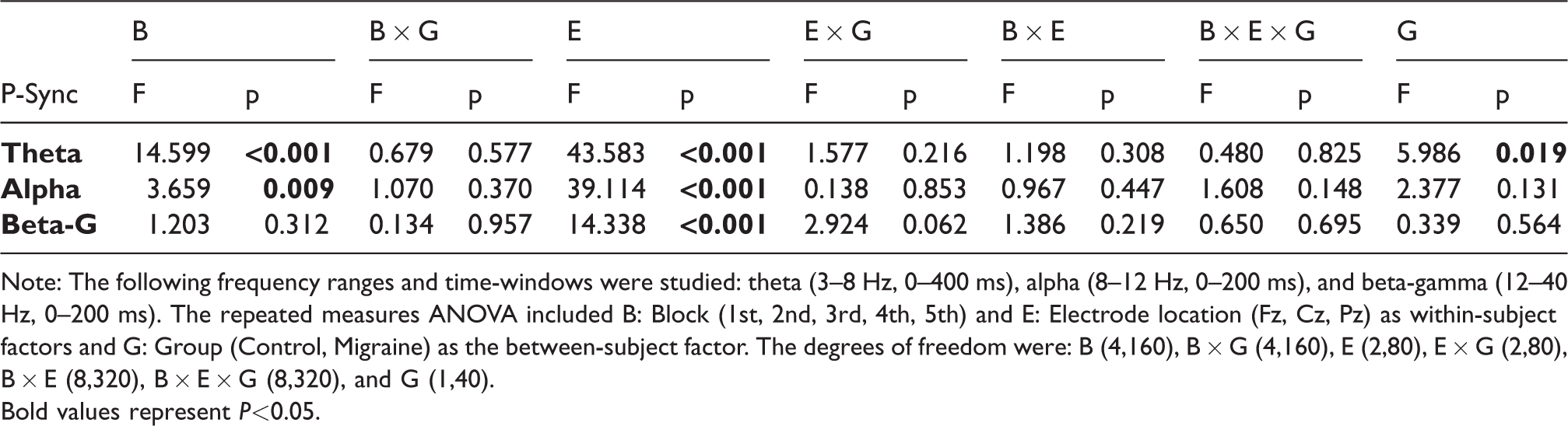
Note: The following frequency ranges and time-windows were studied: theta (3–8 Hz, 0–400 ms), alpha (8–12 Hz, 0–200 ms), and beta-gamma (12–40 Hz, 0–200 ms). The repeated measures ANOVA included B: Block (1st, 2nd, 3rd, 4th, 5th) and E: Electrode location (Fz, Cz, Pz) as within-subject factors and G: Group (Control, Migraine) as the between-subject factor. The degrees of freedom were: B (4,160), B × G (4,160), E (2,80), E × G (2,80), B × E (8,320), B × E × G (8,320), and G (1,40).Bold values represent P<0.05.
Target/Novel ERPs
See Figure 4 and Table 5 for repeated-measures ANOVAs. A cascade of ERP events was observed in response to target stimuli in both groups (see Figure 4) beginning with the appearance of a frontocentral MMN, a centro-parietal P3b, and a frontocentral RON. Topographical distributions were corroborated by the significant main effects of electrode. No significant group differences were observed (see Table 5).
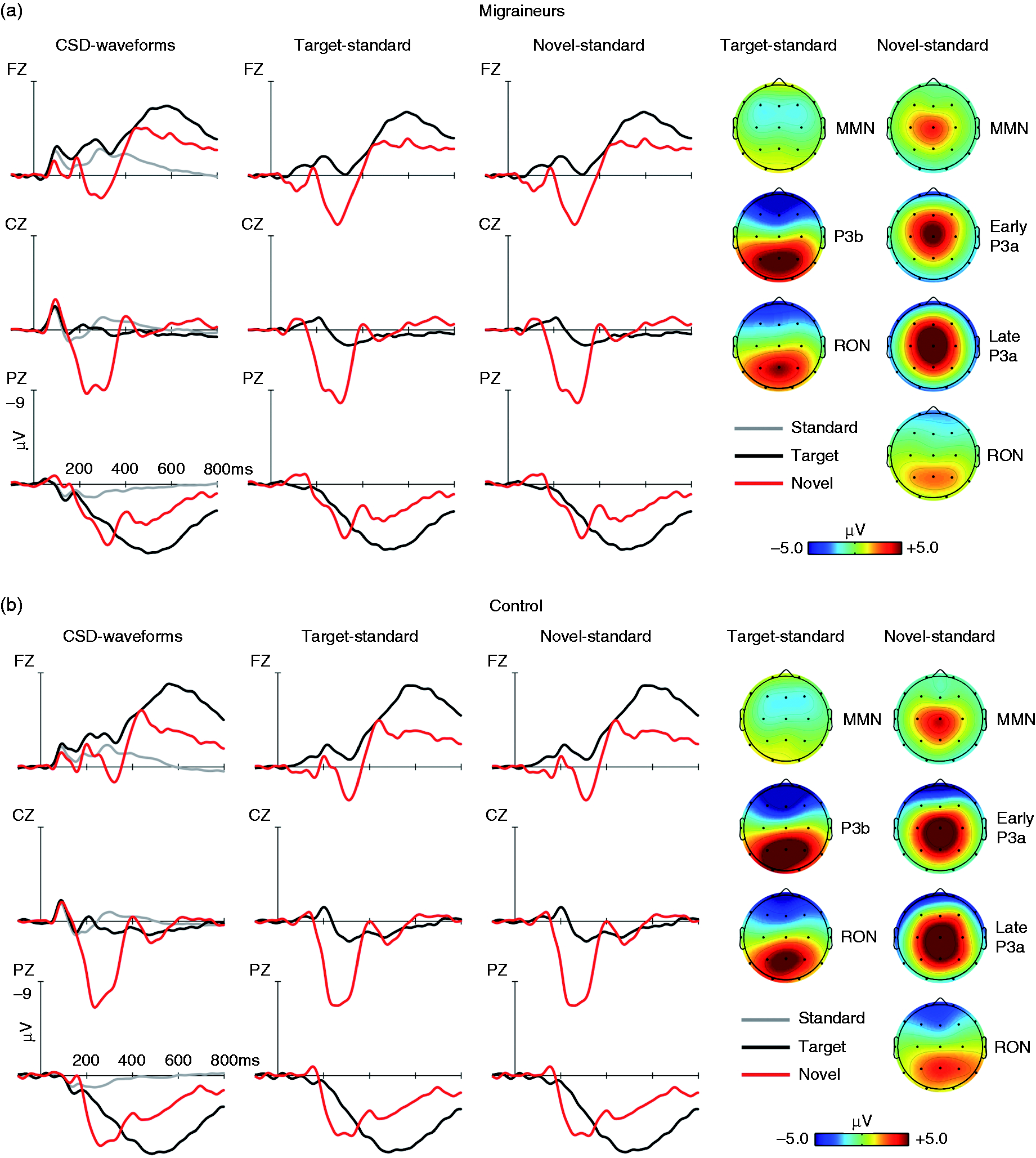
Grand mean ERP waveforms for standard (grey line), target (black line) and novel (red line), at midline electrodes (Fz, Cz, and Pz), from −100 to 800 ms, −9/+9 μV, for both the EM (a) and HC (b) groups. Difference waveforms associated to the target minus standard (black line) and novel minus standard (red line) are showed. Scalp distribution (−5/+5 μV) for target minus standard trials involve the MMN, P3b and RON. Scalp distribution (−5/+5 μV) for novel minus standard involve the MMN, early P3a, late P3a, and RON.
Repeated-measures ANOVA for the ERP difference waveforms related to target and novel stimuli.

Note: The following components and time-windows were included for target stimuli: MMN (175–225 ms), P3b (450–550 ms), and RON (350–450 ms); and for novel stimuli: MMN (175–225 ms), early P3a (225–275 ms), late P3a (275–325 ms), and RON (400–600 ms). The repeated measures ANOVA included E: Electrode location (Fz, Cz, Pz) as the within-subject factor and G: Group (Control, Migraine) as the between-subject factor. The degrees of freedom were as follow: E (2,80), E × G (2,80), and G (1,40).Bold values represent P<0.05.
Similarly, the time-course for responses to novel stimuli included the frontocentral MMN, a central early P3a, a frontal and parietal late P3b, and a frontocentral RON (see Figure 4). Topographical distributions were upheld by significant main effects of electrode. Despite a lack of main effects between groups, a significant Electrode × Group interaction was observed for the early P3a, late P3a, and RON. The post-hoc t-test analyses revealed that the early P3a trends to be reduced in EM as compared to HC at Cz (t(40) = 1.967, p = 0.056) and attains significance at Pz (t(40) = 3.747, p = 0.001). On the other hand, an increased late P3a was observed at frontal sites in EM as compared to HC (Fz: t(40) = −2.087, p = 0.043). A post-hoc analysis of the RON also indicated an amplitude reduction in EM at Fz (t(40) = −2.801, p = 0.008). For time-frequency see Supplemental results.
Discussion
The aim of this study was to investigate auditory sensory processing during an active oddball task in a group of young, female, low-frequency EM patients in the interictal phase as compared to healthy controls. Our research study proposed a novel approach by pairing classic ERP analyses with spectral power and phase-synchronization, which offer a more holistic approach to understanding the sensory alterations reported in EM. Patients presented: i) an increased response to auditory stimuli, as indicated by increased N1 amplitude and theta phase-synchronization, and ii) an abnormal response pattern to novel stimuli characterized by a reduced early P3a, an increased late P3a, and reduced RON.
Our results suggest that auditory sensory processing is increased in EM patients interictally, which supports previous findings indicating increased sensitivity to sound in patients between attacks (31). An increased N1 suggests that EM individuals process environmental sounds as more salient, and consequently allocate more attentional resources to their processing (6,7), affecting the representation of sound at the neural level, within the sensory auditory cortex (32). A lack of group differences in P2 and N2 components would suggest that higher order perceptual processes and their modulation by attentional top-down mechanisms remain preserved (8). Although some studies have failed to report differences in N1 or P2 amplitude (3,10–13,33), these results are compatible with data from other studies (14,15), which encountered higher N1-P2 amplitudes in families with migraine.
The main finding of the present study was the increased theta phase-synchronization in response to repetitive stimuli in EM. The theta wave is the main contributor to the brain’s response to standard stimuli during active auditory oddball tasks (5). Notably, both power and phase-synchronization may be instrumental in generating and modulating ERPs. However, phase-synchronization may be also involved in maintenance (with progressive habituation) of ERP amplitudes across repetitions (26). Phase-synchronization of theta activity plays a relevant and active role in early sensory processing. It has been suggested to be an internal form of information coding in the sensory cortex (34), which acts by continuously tracking information related to external sensory stimuli (35). In the auditory modality, this process is closely linked to the N1 component (26). Therefore, an increase in phase-synchronization, as seen in our data, could underlie the increased sensory auditory processing seen in EM, confirmed by an increased amplitude of the N1 component. Our results are compatible with studies on visual processing, which also show increased phase-synchronization in EM without aura in particular within the alpha-band during visual stimulation (1,36,37) and with resting-state functional magnetic resonance imaging (rfMRI) studies indicating alterations in connectivity in salience- and auditory-related structures (38). Finally, our results are also congruent with the thalamocortical dysrhythmia hypothesis, suggesting the presence of increased low frequency oscillations in migraine in thalamic structures, which may lead to a hyperresponsiveness of the sensory cortices.
In the current study, we observed habituation of ERPs, spectral power, and phase-synchronization processes in both EM and control groups across blocks. These results support previous studies, which did not find any differences in the habituation of N1 and P2 ERP components to auditory stimuli between migraine patients and healthy controls (3,10–13). Although the thalamocortical dysrhythmia hypothesis suggests that migraine hyperresponsiveness is due to a lack of habituation to sensory stimuli (1), current research findings have opened this up for debate. It remains unclear whether the deficit of habituation, frequently reported in migraine studies, is related to disease itself, or if it could be related with the subtype of migraine (such as episodic or chronic, with or without aura), or to a confounding variable (such as treatment, psychiatric comorbidity, age or methodological differences) (39).
In regards to the cascade of ERPs related to novel and target stimuli, we observed abnormalities in EM. Similarly to previous studies (10,19), but in contrast to others (12,13), no group differences were observed in MMN in response to novel stimuli, which may reflect that early sensory detection of unexpected change or stimuli, is preserved in migraine. Regarding P3a, previous studies reported no differences (13,20), while other studies showed an increased P3a in migraine compared to controls (12). Based on previous basic research studies indicating the presence of two P3a peaks (early and late) with different brain generators, we decided to divide the P3a in two. We observed an abnormal pattern of reduced early P3a and increased late P3a in patients as compared to controls. These results suggested that in EM there was a reduced post-sensory response to novel stimuli (reduced early P3a), which was quickly compensated by the heightened allocation of attentional resources (increased late P3a). A reduced RON in both novel and target stimuli would indicate that patients had difficulty disengaging from distracting novel stimuli and reorienting attention back to the task, which could indicate decreased cognitive flexibility (16). Finally, consistent with one line of evidence, no group differences were observed between migraine patients and healthy controls with respect to the MMN (10,19) and P3b (3,11,22), despite findings from other studies (19,23,24).
Limitations: Our study has some limitations in relation to the features of our sample, which consisted of young adult women (19–28 years) with low-frequency EM without aura. It is possible that our sample can only partially explain migraine symptomology and might not be generalized to the usual population of migraine patients seen in clinical practice, which report higher frequency of attacks and more associated symptomology. Another factor that might have an impact on the results is the age of migraine onset, but unfortunately this variable was not collected in this research study, given that many of the participants that received a migraine diagnosis were unaware or unsure of whether or not they had migraine. Therefore, it was difficult to pinpoint a specific onset time. Future studies might evaluate how disease evolution and associated symptomatology may impact auditory sensory processing and consequently ERPs, spectral power, and phase-synchronization. There is also an important methodological aspect to consider, which is a basic principle of habituation: The presentation of other (usually strong or salient) stimuli results in a recovery or in a disruption of habituation to a standard repetitive stimulus (40). In this experiment, habituation was examined using an active Oddball task, comprised of standard, novel and target stimuli. Thus, the results regarding habituation should be interpreted with caution, as the presence of target and novel stimuli could break the habituation chain and bias data.
Conclusions
In conclusion, the presence of an increased theta phase-synchronization and a larger N1 in EM could indicate the presence of a hypersensitivity to auditory stimuli in low-frequency EM patients between attacks. Patients appear to process repetitive stimuli as though they were more salient and seem to allocate more of their attentional resources to their surrounding environment. Finally, in the presence of particularly salient stimuli (such as novel and target), which could be considered biologically relevant, patients show a reduced RON, or a difficulty in reorienting their attention from the distracting stimulus back to the task at hand. This would suggest decreased cognitive flexibility in patients with EM as compared to controls. Ultimately, patients show a hypersensitivity to auditory stimuli and maladaptive allocation of attentional resources, which could explain the auditory alterations reported interictally in EM.
Article highlights
The perceived salience of auditory stimulus is increased in episodic migraine as seen in greater theta phase-synchronization and higher N1 amplitude. The reduced amplitude of the reorienting negativity suggested that episodic migraine patients have less available resources to reorient attention back to the main task.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420951509 - Supplemental material for Neurophysiological correlates of abnormal auditory processing in episodic migraine during the interictal period
Supplemental material, sj-pdf-1-cep-10.1177_0333102420951509 for Neurophysiological correlates of abnormal auditory processing in episodic migraine during the interictal period by Adrià Vilà-Balló, Angela Marti-Marca, Marta Torres-Ferrús, Alicia Alpuente, Victor José Gallardo and Patricia Pozo-Rosich in Cephalalgia
Supplemental Material
sj-pdf-2-cep-10.1177_0333102420951509 - Supplemental material for Neurophysiological correlates of abnormal auditory processing in episodic migraine during the interictal period
Supplemental material, sj-pdf-2-cep-10.1177_0333102420951509 for Neurophysiological correlates of abnormal auditory processing in episodic migraine during the interictal period by Adrià Vilà-Balló, Angela Marti-Marca, Marta Torres-Ferrús, Alicia Alpuente, Victor José Gallardo and Patricia Pozo-Rosich in Cephalalgia
Supplemental Material
sj-pdf-3-cep-10.1177_0333102420951509 - Supplemental material for Neurophysiological correlates of abnormal auditory processing in episodic migraine during the interictal period
Supplemental material, sj-pdf-3-cep-10.1177_0333102420951509 for Neurophysiological correlates of abnormal auditory processing in episodic migraine during the interictal period by Adrià Vilà-Balló, Angela Marti-Marca, Marta Torres-Ferrús, Alicia Alpuente, Victor José Gallardo and Patricia Pozo-Rosich in Cephalalgia
Footnotes
Acknowledgements
We especially wish to thank all the participants for their great collaboration in the present project. Statistical analysis was conducted by Adrià Vilà-Balló.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AVB, AMM, VJG report no conflicts of interest. AVB has received a postdoctoral contract courtesy of “La Caixa” Foundation, and was supported by the Spanish MICINN Juan de la Cierva postdoctoral grant (FJC2018-036804-I). AA has received honoraria as speaker for Allergan. MTF has received honoraria as a speaker for Allergan, Chiesi, Eli Lilly and Novartis. PPR has received honoraria as a consultant and speaker for Allergan, Almirall, Biohaven, Chiesi, Eli Lilly, Medscape, Neurodiem, Novartis and Teva. Her research group has received research grants from Allergan, AGAUR, la Caixa foundation, Migraine Research Foundation, Instituto Investigación Carlos III, MICINN, PERIS; and has received funding for clinical trials from Alder, Electrocore, Eli Lilly, Novartis and Teva. She is a trustee member of the board of the International Headache Society and a member of the Council of the European Headache Federation. She is on the editorial board of Revista de Neurologia. She is an editor for Frontiers of Neurology and the Journal of Headache and Pain. She is a member of the Clinical Trials Guidelines Committee of the International Headache Society. She has edited the Guidelines for the Diagnosis and Treatment of Headache of the Spanish Neurological Society. She is the founder of ![]() . PPR does not own stocks from any pharmaceutical company.
. PPR does not own stocks from any pharmaceutical company.
The authors report no disclosures relevant to the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project leading to these results has received funding from “La Caixa” Foundation under the project code LCF/PR/PR16/51110005”.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
