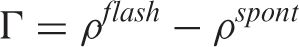Abstract
Objectives:
This research was a case-control study to evaluate functional and effective connectivity patterns in ongoing electroencephalography (EEG) under repetitive photic stimulation in the interictal phase of migraine patients with and without aura compared to nonmigraine controls.
Methods:
EEG was recorded by six scalp electrodes from 19 migraine without aura patients (MO), 19 migraine with aura patients (MA) and 11 healthy subjects (control group (N)). Flash stimuli were presented at 9–27 Hz frequencies. Phase synchronization after Hilbert transform and Granger causality were evaluated filtering the EEG in alpha and beta bands.
Results:
Phase synchronization increased in alpha band in MO, and decreased in beta band in MA, with respect to controls. The intensity of directed interactions in beta band, revealed by Granger causality, increased in MA compared to both MO patients and controls.
Discussion:
There were clear differences in ongoing EEG under visual stimulation, which emerged between the two forms of migraine, probably subtended by increased cortical activation in migraine with aura, and compensatory phenomena of reduced connectivity and functional networks segregation, occurring in patients not experiencing aura symptoms. Further investigation may confirm whether the clinical manifestation of aura symptoms is subtended by a peculiar neuronal connectivity pattern.
Keywords
Introduction
Migraine is an incapacitating disorder of neurovascular origin consisting of episodes of headache, accompanied by autonomic and possibly neurological symptoms. The pathophysiology of migraine episodes is far from being understood, and the occurrence of aura preceding headache seems a complex mechanism related to cortical spreading depression (1,2). Few neurophysiological studies have compared migraine with (MA) and without aura (MO), while most of them described abnormalities of spontaneous and specially evoked brain electrical activity in separate groups of migraine patients (3). An abnormal response to repetitive visual stimulation, consisting of increased amplitude of steady-state visual evoked potentials (SVEPs), was observed in both MA and MO (4,5). Previous studies employing SVEPs at different contrast and frequency of stimulation showed differences between MA vs MO patients, thus suggesting an involvement of the visual associative cortex in patients reporting aura symptoms (6,7).
In order to gain further insight into the interpretation of these phenomena, it may be necessary to investigate the dynamic interactions between brain areas and their modulation in the presence of stimuli. An efficient measure of these interactions is the phase synchronization of electroencephalography (EEG) signals, evaluated by investigating their phase difference. In order to isolate the temporal features of the signal, unlike in coherence or generalized synchronization, the phase is decoupled from the amplitude by means of the Hilbert transform (8). This method allows the consideration of nonlinear characteristics of the signals and the detection of coupling of neuronal networks oscillations. Previously it allowed the identification of intermittent photic stimulation influences and spontaneous EEG activity in healthy subjects by reducing phase synchronization of the posterior dominant alpha rhythm (8–12.5 Hz). Conversely, in MO patients, an opposite pattern of increased alpha rhythm phase synchronization was observed (8,9).
Methods such as correlations, spectral coherence and phase synchronization allow the qualification to show the extent to which two variables are statistically connected, and reveal what in neuroscience is commonly referred to as functional connectivity. It allows the detection of common temporal features of even two distant neural populations due to weak reciprocal interactions or a shared influence of a third variable (10). Another increasingly popular approach, effective connectivity, is based on the flow of connections and information across different brain areas. This allows for the extension of insight provided by functional connectivity by revealing, for example, which is the driver between two temporally correlated time series. These model-based approaches can be purely data driven as Granger causality (GC) (11–14) or biologically inspired such as dynamic causal modeling (15). In order to infer the information flow in nonlinear systems such as the brain, a flexible nonlinear generalization of Granger causality by Kernel methods has been recently developed (16).
These approaches represent a valuable addition to those based on correlation and synchronization analysis (8): The results of functional and effective connectivity represent a significant added value to neuroscience since they allow researchers to pinpoint the temporal pattern of activation and information transfer between cortical areas (17).
This study aimed: 1) to extend the quantification of phase synchronization of ongoing EEG activity during repetitive flash stimulation to MA patients, and 2) to identify Granger causality across brain regions in both types of migraine compared with nonmigraine subjects, thereby obtaining complementary information from functional and effective connectivity patterns.
Methods
Subjects
EEG was recorded from 19 patients (seven males, 20–44 years old) experiencing aura, for whom a diagnosis of typical aura with migraine headache (18) (International Classification of Headache Disorders II (ICHD-II) cod. 1.2.1,-n = 17 patients) and nonmigraine headache (ICHD-II cod. 1.2.2, n = two patients) were performed. The mean time of patients’ migraine history was respectively eight ± 5.5 years; the mean headache frequency was 2.1 ± 1.2 days in a month in the last three months. Nineteen MA patients (four males, age 21–45 years. ICHD-II cod 1.1.) (18) were also included in the study. They reported migraine episodes for 10 ± 4.4 years, their mean headache occurrence was 2.5 days ± 1.1/headache/months, computed in the last three months. All patients were in the interictal state, the time from the end of the last attack being at least 72 hours, while an interval of at least 48 hours from the next attack was ascertained by a telephonic interview. This was performed in order to exclude patients in the phases preceding migraine. Females were recorded 14.45 ± 4.5 days after menses. No patient was under preventive treatment nor had used symptomatic drugs in the 72 hours preceding the recording session. Eleven healthy subjects, selected among hospital staff and medical school participants, matched for sex (three males, chi square 0.544 not significant (NS)) and age (20–46 years); analysis of variance (ANOVA) was used with age as variable and diagnosis as factor: F 0.98 NS) with the patient groups, not reporting migraine in first-degree relatives, were also included as controls. In the control group, females were recorded 13.32 ± 2.9 days after menses. None of the subjects involved in the study were affected by general medical, neurological or psychiatric diseases.
Recording and stimulation procedure
Flashing stimuli were presented each time with a different rate, namely 9–18–21–24–27 Hz, according to previous studies (9,19).
For each stimulus frequency, a 40-second (s) stimulus interval was followed by a 20-s rest period. The subjects were instructed to relax during the experiment and keep their eyes closed. To avoid drowsiness, they were asked to open their eyes and converse with the experimenter after any session of stimulation at different frequencies. Stimulus frequencies were presented in random order. Each frequency of stimulation was delivered by a flash with 0.29 J energy. The distance from the stroboscope was 20 cm. EEG data were recorded by six scalp electrodes: two occipital channels (O1 and O2), two parietal ones (P3 and P4), a central electrode (Cz) and a frontal one (Fz), referred to the nasion. We used a Micromed Brain Quick apparatus with System Plus Evolution software for EEG data acquisition, analysis and storage, and the strobe lamp was integrated into the apparatus (http://www.micromed.eu/prodsel.asp?cat = 2&prod = 5). The ground electrode was positioned on Fpz. One electrode was placed above the right eyebrow for electro-oculogram (EOG) recording. Impedance was kept at 10 kΩ or less. The sampling rate was 256 Hz.
EEG analysis
The EEG records were first inspected by the first author, who was not aware of the subjects’ identity and diagnosis. A digital filter in the range 0.1–70 Hz was applied first, as well as a notch filter around the line frequency (50 Hz). Records or portion of records that contained drowsiness, sleep or persistent ocular artifacts were deleted. Artifact-free epochs of one s each were analyzed by Fast Fourier Transform (FFT) at 256 Hz frequency resolution, according to the FFT analysis reported in ASA version 4.8.1 by ANT software (http://www.ant-neuro.com): We averaged at least 20 epochs for each frequency (range 21–28 epochs; mean 24.28 ± 2.71). Among SVEP components, the absolute power (uV2) of the EEG bandwidth ±0.15 Hz the value of stimulation frequency was computed according to previous studies (10) and interpolation provided by ASA software. Absolute power in the frequencies of stimulation (fundamental component, F1) was compared across groups by means of Student’s t test, corrected for multiple comparisons.
We further investigated the hyper-synchronization phenomenon previously described in the presence of flash stimuli (9) in the three groups, filtering the EEG signals in alpha (8–12 Hz) and beta (12.5–30 Hz) bands.
Phase synchronization
For all stimulus frequencies, we evaluated the alpha and beta rhythm phase synchronization, according to our previous study (9). We can model the brain rhythms as signals oscillating at several frequencies (e.g. sine waves), localized at the electrodes’ positions. If two signals from different locations maintain more or less the same distance between the same phase of the oscillation (for example, the maximum) throughout the recording, then we say that the two signals are synchronized in phase. In order to isolate the temporal features of the signal, unlike in coherence or generalized synchronization, the phase is decoupled from the amplitude by means of the Hilbert transform (8). The procedure is the following: The signals are filtered in the relevant frequency band. Then for each pair of signals, the difference between the phases is computed. This difference, module 360 degrees, is then displayed in a histogram. If the signals are synchronized in phase, this angle will be more or less constant and the histogram will display a peak; if on the other hand the signals are not synchronized in phase, this angle will change each time and the histogram will be flat. The phase synchronization is defined in terms of the Shannon entropy (“peakedness”) of the histogram (Figure 1, top) (9). To quantify the phase synchronization, the index proposed by Tass et al. (20) was used. The artifact-free EEG signals were filtered in each band with a second order, double-sided Butterworth filter. The phase synchronization index previously described (Figure 1, top) (9) was evaluated for all pairs of electrodes, for all subjects and for all frequencies of the flash stimuli. These indexes were subsequently averaged over all the possible pairs of electrodes for each subject both in the presence of stimuli and in spontaneous conditions. Given that the entire analysis was automatically performed, the blind design was not requested, according to Angelini et al. (9). For each stimulation frequency, we then calculated the difference
Top: Method for computing phase synchronization. Signals are filtered in the relevant frequency band (a), decupling the phase from the amplitude by means of Hilbert transform (b). Then for each pair of signals, the difference between the phases is computed. This difference, module 360 degrees, is then displayed in a histogram (c). If the signals are synchronized in phase, this angle will be more or less constant and the histogram will display a peak; if on the other hand the signals are not synchronized in phase, this angle will change each time and the histogram will be flat. The phase synchronization is defined in terms of the Shannon entropy (“peakedness”) of the histogram (c). Bottom: Method for computing Granger causality. An autoregressive model of a time series X predicts its future from its past, with a certain accuracy measured by the error εX. If now we add to the model information from the past of another time series Y, the accuracy for this new model will be given by the error εX,Y. If εX,Y is significantly smaller than εX then we can say that Y Granger causes X. This model is a nonlinear generalization of Granger causality, by Kernel methods, presented in Marinazzo et al. (16), which allows us to infer the directional information flow in nonlinear and multivariate systems.
Granger causality
In this study we evaluated effective connectivity by means of Granger causality, whose basic idea is the following: We can build an autoregressive model of a time series X to predict its future from its past, with a certain accuracy measured by the error εX. If now we add to the model information from the past of another time series Y, the accuracy for this new model will be given by the error εX,Y. If εX,Y is significantly smaller than εX then we can say that Y Granger causes X. Here we use the nonlinear generalization of Granger causality, by Kernel methods, presented in Marinazzo et al. (16), which allows us to infer the directional information flow in nonlinear and multivariate systems (Figure 1, bottom).
In order to distinguish between direct and conditional influence, a multivariate approach has to be employed (21). This framework has been used in many fields of science, including neural systems (22,23), but limited to the linear case. Kernel Granger causality (KGC) is based on the theory of reproducing Kernel Hilbert spaces for measuring the transfer of information between brain areas (16). In this work we evaluated the global flow averaging across all the pairs, rather than concentrating on the single links for which there can be inter-subject variability.
The statistical analysis was performed considering phase synchronization and Granger causality (average values across electrodes) as variables and frequencies of stimulation and diagnosis as factors, after a Kolmogorov Smirnoff test application to confirm the parametric distribution of data. The Tukey Kramer test was employed as a post-hoc test.
Phase synchronization and Granger causality (average values across electrodes) were also correlated with main clinical features, such as age, age of illness and migraine headache frequency, by means of the Spearman correlation test.
Results
The F1 amplitude was significantly increased in MA and MO groups in the 9–27 Hz range of stimulation, with respect to controls, on O1-O2-P3 and P4 electrodes (Figure 2(a) and 2(b)).
Mean values and standard deviations of fundamental components of the steady-state visual evoked potentials (SVEPs), obtained by means of Fast Fourier analysis in the main frequency of stimulation ((a) 17–20; (b) 23–26) in migraine with aura (MA), migraine without aura (MO) and controls (N). The results of t test (threshold for multiple comparison p ≤ 0.01) are shown: N vs MA and N vs MO: *p ≤ 0.01.
Phase synchronization
The pattern of alpha band hyper-synchronization was confirmed for MA, while MA patients did not show alpha band hyper-synchronization in presence of light stimuli (Figure 3, top). The ANOVA test with post-hoc analysis showed that the frequency of stimulation displayed a different effect on alpha synchronization, although no significant differences across frequencies were detected by the post-hoc test. This effect was significantly different in MO patients with respect to MA and N subjects, while the interaction between frequency of stimulation and diagnosis was not significant (Figure 3, top).
Mean values and standard deviations of synchronization index Γ of ongoing electroencephalography (EEG) filtered in alpha (top) and beta (bottom) bands, averaged over subjects and across channels, during photic stimulation at different frequencies in migraine with aura (MA), without aura (MO) and controls (N). The results of analysis of variance (ANOVA) test are reported. Frequency: frequency of stimulation (9–18–21–24–27 Hz). D.F.: degree of freedom; NS: not significant; diagnosis: diagnosis MA vs MO vs N. Frequ*diagn: interaction; significant post-hoc tests are reported.
Regarding the beta band, in MA patients a desynchronizing effect of repetitive light stimulation was observed. This effect was also slightly present in controls (Figure 3, bottom). Clear differences were detectable between the two migraine groups: Phase synchronization between beta rhythms decreased during light stimulation in MA more than in MO patients (Figure 3, bottom)
Granger causality
The stimulus-related modulation of effective connectivity in alpha band, evaluated by means of nonlinear Granger causality, was significantly different across groups: Smaller values were observed in MO patients than with MA patients or the control group (Figure 4, top). Concerning the statistical effect of the interaction diagnosis × frequency of stimulation, a stronger separation between the three groups was observed for stimulation at 24 Hz (Figure 4, top). MA patients exhibited higher Granger causality values in the beta band in the presence of stimuli, while the same values remained virtually constant in the control group and MO patients. In this case, stimulation at 24 Hz, as well as at 9 Hz, resulted in a stronger separation of the three groups (Figure 4, bottom).
The nonlinear Granger causality in alpha (a) and beta band (b) obtained using a Gaussian kernel and averaged over subjects and across channels in the three classes (migraine with aura (MA),without aura (MO) and controls (N)), is depicted as a function of the frequency of stimulation. The results of analysis of variance (ANOVA) test are reported. Frequency: frequency of stimulation (9–18–21–24–27 Hz). D.F.: degree of freedom; NS: not significant; diagnosis: diagnosis MA vs MO vs N. Frequ*diagn: interaction; significant post-hoc tests are reported.
No correlation was found between phase synchronization and Granger causality individual values and the considered clinical features for any frequency of stimulation.
Discussion
The results of our study showed that the patterns of functional and effective connectivity in ongoing EEG rhythms in the presence of repetitive light stimulation were different in MA and MO patients. Moreover, SVEP patterns were found to be similar in the two groups, according to previous reports (4,5), though the comparison of our results to those of other groups may be limited by the lack of photometric luminance measurement and the uncertain homogeneity of visual stimulation conditions.
The incremented SVEPs amplitude confirmed in both migraine types might be linked to a change in neuronal excitability and balance between inhibitory and excitatory circuits in the primary visual cortex (6,7). Important differences between the forms of migraine were found in regard to the effects of visual stimulation on the ongoing EEG, which represents the entity of recordable brain activity outside that specifically induced by intermittent visual stimulation. The evidence of a different pattern of phase synchronization of alpha and beta rhythms between the two migraine groups, in the presence of a similar SVEP behavior (4,5), could suggest that the basal EEG rhythm and the oscillations in the frequencies of photic stimulation may have different resonant properties. This makes sense in view of the complexity of brain functions with different modalities of activation of neuronal network not specifically involved in photic driving response.
In the present study, we were able to confirm reduced phase synchronization of alpha rhythm during repetitive light stimulation in the control group, as opposed to the increased synchronization in MO patients (9,24). In the control group, the reduced phase synchronization of alpha rhythm under intermittent visual stimulation, together with a slight increase of causal connection across electrodes, may suggest that in this condition a reduced resonance of thalamic activity may occur (25) with a disruption of basal alpha and an activation of different cortical networks (26). Conversely, in MO patients the concurrence of increased synchronization and the reduced information flow in the alpha band may be due to the weak interactions between cortical regions and the resonance of rhythmic activity generated at the sub-cortical and probably thalamic level (27,28). The reduction of causal links across scalp regions coexisting with the temporal synchrony of oscillating rhythms could indicate a synchronous activation due to a unique pacemaker probably located in the thalamus. Accordingly, many studies account for a thalamocortical dysrhythmia in migraine (29–32), which may work in different ways during intermittent visual stimulation in MA and MO. The alpha hyper-synchronization, which may be ascribed to a compensatory phenomenon against generalized cortical involvement under intermittent photic stimulation, was absent in MA, which resembled controls in alpha rhythm phase synchronization and causality. A pattern of reduced phase synchronization and increased information flow across channels in beta band was evident in MA and clearly differentiated the two forms of migraine. The beta rhythm is generally associated with a state of cortical activation, and up to now few reports have described its changes under intermittent visual stimulation (26,33). In this study Granger causality measured the total amount of information transfer across electrodes in any direction, which may be interpreted as a sign of activation of different interacting functional networks (34). In the case of MO, compensatory phenomena of reduced connectivity and functional networks segregation may limit the extension of cortical recruitment. The lack of correlation between the alpha and beta phase synchronization and Granger causality and clinical features such as frequency of headache may indicate that it was the presence of aura that formed these neurophysiological patterns. Previous studies outlined neurophysiological differences between the two forms of migraine. Conte et al. (35) found an increased facilitation of primary motor cortex in the interictal phase of MA patients compared with both MO and the control group. Other studies suggested more pronounced and diffused cortical recruitment under visual stimulation in MA (6,7,30,36,37). In particular, increased early high-frequency oscillations in the gamma band range during visual stimulation were attributed to a peculiar pattern of thalamocortical dysrhythmia leading to decreased intracortical inhibition in migraine patients reporting aura symptoms (30). Bjørk et al. (38) employed brief trains of intermittent flash stimulation and found attenuation of driving responses to 18 Hz and 24 Hz only in MO migraineurs in the inter-ictal phase, confirming a dysregulation of cortical excitability with different fluctuating properties in the two forms of migraine. So far, our study may add to our existing knowledge about phenotypical differences in the migraine brain, pending several limits. The biological basis of changes in phase synchronization and causal connection of brain networks under visual stimulation are complex and not completely explained (33,39–41). In addition, the employment of additional recording channels and possibly more complex patterns of visual stimulation may reinforce present results (6). Differences in functional abnormalities of migraine brain may indicate the expression of phenotypic traits subtending different clinical features (42,43). A challenge for future studies would be the search for mechanisms underlying different migraine syndromes in view of more specific and customized therapeutic approaches. Our results outline clear differences in EEG activity under visual stimulation between MA and MO patients and may presently indicate the potential utility of functional and effective connectivity patterns in understanding the mechanism subtending aura symptom perception.
Clinical implications
Migraine with (MA) and without (MO) aura are associated with different patterns of functional and effective connectivity in ongoing EEG under repetitive photic stimulation. In MA we observed an increase of effective connectivity in the beta band, whereas increased functional connectivity in alpha band was reported in MO patients. Patterns of effective and functional connectivity across brain areas may subtend the different phenotypic expression of the two forms of migraine.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.