Abstract
A global, double-blind, randomized, placebo-controlled, dose-finding, parallel-group trial was conducted to compare three oral doses of RPR100893 (1, 5, 20 mg), a substance-P antagonist, and placebo in 139 patients with migraine attacks with moderate or severe headache intensity. Headache intensity and improvement of headache after 2 h were not different in the four treatment groups. Therefore, it is concluded that this oral substance-P antagonist in the doses used here is not effective.
Introduction
Apart from the new 5-HT1B/
Patients and methods
Patients aged 18–65 years had to fulfil the criteria for migraine with or without aura according to the International Headache Society (5). The usual exclusion criteria for migraine trials were applied. The protocol was approved by all local Ethical Review Boards. Patients were recruited in 21 centres internationally. After giving informed consent they were randomized into one of four groups. Patients treated one migraine attack with moderate or severe headache intensity with 1 mg, 5 mg or 20 mg RPR100893 or placebo. Patients were asked to report to the centre within 48 h after using study medication. Headache severity was measured on a 4-point scale (0 = no pain, 1 = mild pain; 2 = moderate pain; 3 = severe pain) 1, 2, 4 and 8 h after study intake and expressed for the 2-h time point as the mean pain intensity (mean pain index). Additional efficacy measures included functional disability (3-point scale), presence or absence of nausea, vomiting, photo- or phonophobia. Since this was a phase II trial, extensive safety measurements were taken (vital signs, ECG, laboratory assessment, biochemistry, haematology). Rescue medication was allowed 2 h after intake of study medication. Ergots and sumatriptan were not accepted as rescue medication.
Statistical analysis
The primary efficacy analysis was based on the number of patients who obtained complete or almost complete headache relief within 2 h of the onset of treatment. Assuming a response to verum of 70% and to placebo of 30%, a sample size of 144 patients was required. The analysis was performed by Rhone Poulenc Rorer. The first author had no access to the source data, but to the official trial report.
Results
Baseline demographics and migraine characteristics were well balanced across placebo and the three doses of RPR100893 (Table 1). There was no difference between placebo and the three doses of RPR100893 for pain relief or change in mean pain intensity after 1, 2, 4 and 8 h (Table 1). The same was true for functional disability and the absence or presence of nausea, vomiting, photo- or phonophobia and the use of rescue medication after 2 h. In the overall response scale 48% of patients with placebo and 51%, 48% and 29% with 1 mg, 5 mg and 20 mg RPR100893 ranked treatment as good or excellent 2 h after intake of study medication. Asthenia (3–8%) and somnolence (3–10%) were the most frequently reported adverse events (AEs). As for the other AEs, there was no difference between RPR100893 and placebo. In summary, all three doses of RPR100893 were ineffective in the treatment of acute migraine attacks.
Baseline demographics and migraine characteristics
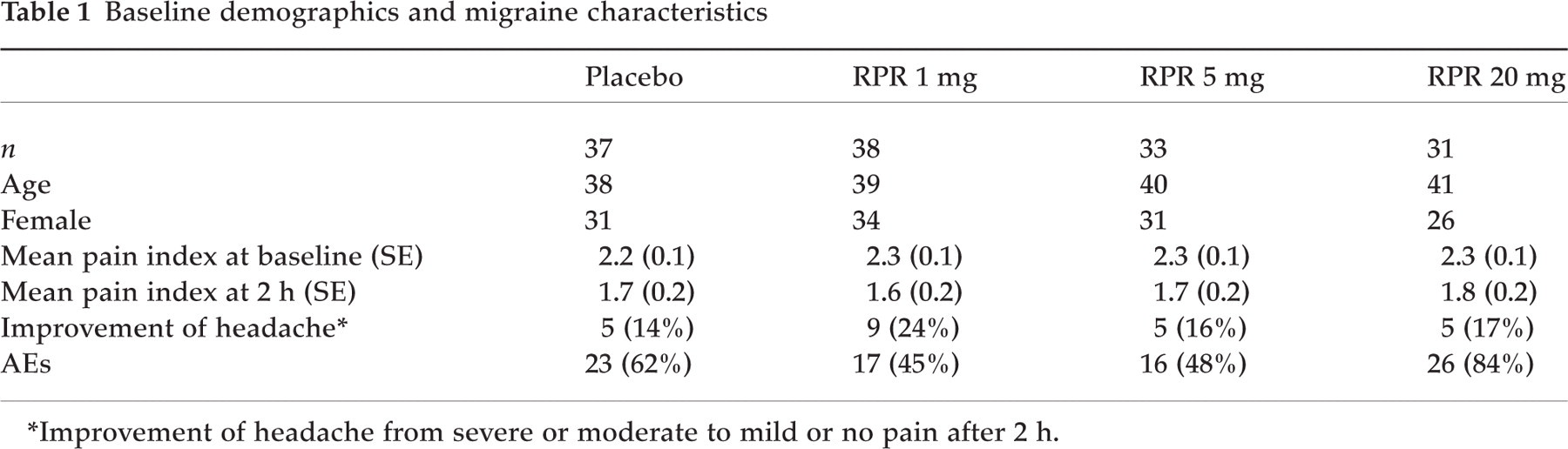
∗Improvement of headache from severe or moderate to mild or no pain after 2 h.
Discussion
This trial showed that three doses of an oral substance-P antagonist are not effective in the treatment of migraine attacks. One major shortcoming of this study is the oral formulation. Without parenteral application there was no way to be sure that the drug reached biologically active plasma levels. The fact that other substance-P antagonists were not effective in migraine attacks, however, indicates that the therapeutic principle might be wrong (6–8). Substance-P antagonists are also ineffective in the prophylaxis of migraine (9). There was, however, never a trial investigating the combination of substance-P antagonists with other drugs for the treatment of migraine attacks in order to find out whether there could be a synergistic effect with non-steroidal anti-inflammatory drugs or triptans. Since the development of substance-P antagonists has been terminated, these trials will probably never be performed.
Footnotes
Acknowledgements
The following investigators participated in the trial and were members of the study group (location at time of trial): Arthur H. Elkind (Mt. Vernon, USA), William T. Ellison (Greer, USA), Frederick G. Freitag (Chicago, USA), Jerome Goldstein (San Francisco, USA), Daniel Henry (Sandy, USA), Frank W. Kilpatrick, Madison (USA), Jack A. Klapper (Denver, USA), David Kudrow (Encino, USA), Ninan N. Mathew (Houston, USA), Robert B. Nett (San Antonio, USA), Joel R. Saper (Ann Arbor, USA), Stephen D. Silberstein (Philadelphia, USA), Jerry R. Tindel (Austin, USA), Thomas M. Walshe III (Boston, USA), Andres Bes (Strasbourg, France), Guy Chazot (Lyon, France), Michel Lhuilier de Cordoze (LeMans, France), Hans-Christoph Diener (Essen, Germany), Ulrich Gerbershagen (Mainz, Germany), Hartmut Goebel (Kiel, Germany), Gunther Haag (Windach, now Elzach, Germany), Erhard Hartung (Duesseldorf, Germany), Hans-Dieter Langohr (Fulda), Anette Beckmann-Reinhold (Königstein, Germany). The trial was supported by a grant from Rhone Poulenc Rorer (RPR) (now Aventis) USA and France. The trial was designed by the principal investigator (H.C.D.) and Larry K. Powe (RPR).
