Abstract
Objective
To investigate the role of calcitonin gene-related peptide (CGRP) in persistent post-traumatic headache (PTH) attributed to mild traumatic brain injury (TBI).
Methods
A total of 100 individuals with persistent PTH attributed to mild TBI and 100 age- and gender-matched healthy controls were enrolled between July 2018 and June 2019. Blood was drawn from the antecubital vein and subsequently analyzed using a validated radioimmunoassay for human CGRP. Measurements were performed on coded samples by a board-certified laboratory technician who was blind to clinical information.
Results
CGRP plasma levels were lower in subjects with persistent PTH (mean, 75.8 pmol/L; SD, 26.4 pmol/L), compared with age- and gender-matched healthy controls (mean, 88.0 pmol/L; SD, 34.1 pmol/L) (p = 0.04). No correlation was found of CGRP plasma levels with monthly headache days (r = −0.11; p = 0.27), monthly migraine-like days (r = 0.15; p = 0.13), headache quality (r = −0.14; p = 0.15), or a chronic migraine-like headache phenotype (r = −0.02; p = 0.85).
Conclusions
CGRP plasma measurements are unlikely a feasible blood-based biomarker of persistent PTH. Future studies should assess whether CGRP plasma measurements can be used to predict development of persistent PTH.
Introduction
Post-traumatic headache (PTH) is a secondary headache disorder (1) and a common sequela of mild traumatic brain injury (TBI) (2), also known as concussion. Despite the widespread prevalence and disability associated with PTH, little is known about the underlying disease mechanisms (3). Based on recent data from animal models of PTH (4–6), calcitonin gene-related peptide (CGRP) has emerged as a possible biomarker candidate. In concussed rodents, two studies have demonstrated that CGRP antagonism attenuates cutaneous allodynia (5–6). These animal data suggest that CGRP might mediate mechanisms of peripheral and central sensitization in individuals with PTH. Furthermore, elevated CGRP plasma concentrations have been reported in individuals with migraine, compared with healthy non-headache controls (7), although there is also discordant evidence (8). Interestingly, PTH often mimics a migraine-like headache, but no study has investigated CGRP plasma concentrations in individuals with PTH. Therefore, we conducted a study to assess whether plasma levels of CGRP are elevated in individuals with persistent PTH, compared with age- and gender-matched healthy controls.
Methods
Study population
We enrolled 100 individuals with persistent PTH attributed to mild TBI and 100 age- and gender-matched healthy controls. Study participants were recruited from the outpatient clinic of the Danish Headache Center, the Danish post-concussion syndrome support group website (hjernerystelsesforeningen.dk), and from neurological departments and rehabilitation centers in the Capital Region of Denmark. Healthy non-headache controls were recruited through the Danish research subject website (forsøgsperson.dk) and posters placed at various public institutions in the Capital Region of Denmark.
This article is part of a larger parental study that was approved by the Health Research Ethics Committee of the Capital Region of Denmark (H-18011477). All participants gave written consent after receiving detailed oral and written information. The study was conducted at the Danish Headache Center in accordance with the Declaration of Helsinki (9), with later revisions. The study was registered on ClinicalTrials.gov (identifier: NCT03791515) and approved by the Danish Data Protection Agency.
PTH case definitions
The diagnosis of persistent PTH attributed to mild TBI was made in accordance with the 3rd edition of the International Classification of Headache Disorders (ICHD-3) (1). Other inclusion criteria for subjects with persistent PTH included a) mild TBI to have occurred at least 12 months prior to study participation and b) age between 18–65 years. Exclusion criteria were a) any history of primary headache disorder (except infrequent tension-type headache), b) any history of whiplash injury or more than one TBI, c) pregnant or nursing women, d) cardiovascular or cerebrovascular disease, e) pre-trauma psychiatric disorder (unless well-regulated), and f) medication-overuse headache as defined by the ICHD-3 (1). Furthermore, we required no intake of analgesics within 24 hours prior to study participation.
Healthy controls
Inclusion criteria for healthy controls were a) age between 18–65 years, b) no history of known head trauma or whiplash injury, c) no history of primary headache disorder (except infrequent tension-type headache), d) no first-degree relatives with primary headache disorder, e) no daily intake of medicine other than oral contraceptives, f) no history of neurological or psychological disorders, g) no history of structural heart disease.
Study procedures
A semi-structured interview was used to record data on demographics and clinical characteristics. Furthermore, the 12-item Allodynia Symptom Checklist (ASC-12) was used to determine severity of allodynia. Blood samples were collected in EDTA tubes (4 mL) by intravenous cannulation of the antecubital fossa on the non-dominant side. After storage at room temperature for 30 min, blood samples were centrifuged for 10 min at approximately 2000g. Subsequently, aliquots were immediately stored at −80°C until assayed. CGRP plasma concentrations were measured using an in-house developed and validated radioimmunoassay for human CGRP, as previously described (10). A summarized description can be found in previous publications from our research group (11,12). The intraassay coefficient of variation was 2% while the interassay coefficient of variation was 7%. The non-specific binding range was 3% to 4% while the assay detection limit was less than 1 pmol/L. A protease inhibitor was not used, as the assay validation study had shown no effect of enzymatic degradation on plasma levels of CGRP (10). Prior to measurements of CGRP concentrations, plasma samples were randomly arranged to ensure that all assays were conducted without knowledge of group assignment. Measurements were performed on coded samples by a board-certified laboratory technician who was blind to clinical information.
Statistical analysis
Data are presented as means and standard deviations (SD) or frequency counts with percentages. Shapiro-Wilk test was used to determine the normality of continuous data (CGRP plasma concentrations). A two-sample t-test was used to compare mean plasma levels of CGRP between subjects with persistent PTH and healthy controls. The Spearman rank correlation coefficient (rs) was used for analyses of correlation between CGRP plasma levels and various demographic and clinical characteristics. The chi-squared test was used for comparison of frequencies. All tests were two-sided, and statistical significance was determined at p less than .05. All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 25.0, Armonk, NY, USA.
Results
Study sample
One hundred subjects with persistent PTH and 100 age- and gender-matched healthy controls participated in the study and were enrolled between July 2018 and June 2019. The mean age (SD) was 36.0 (11.6) years in subjects with persistent PTH and 35.8 (11.3) years in healthy controls. In addition, the mean BMI (SD) was 24.5 (4.1) kg/m2 in subjects with persistent PTH and 24.3 (3.7) kg/m2 in healthy controls. Selected characteristics of the subjects with persistent PTH are presented in Table 1. For a more detailed presentation of clinical characteristics and treatment patterns in the present study population, readers are referred to a recently published article (13).
Characteristics of the study population.

In 100 subjects with persistent PTH, 61% had a chronic migraine-like headache phenotype, 29% had a combined migraine-like and tension-type-like headache (TTH-like) phenotype, 1% had a “pure” episodic migraine-like phenotype and lastly, 9% had a chronic TTH-like phenotype (Table 1). The mean (SD) number of headache days per month was 25.4 (7.1), with 55% of the subjects currently using preventive medications (Table 1). None of the subjects used anti-CGRP monoclonal antibodies as a preventive medication.
Plasma CGRP
CGRP plasma levels were lower in subjects with persistent PTH (mean ± SD, 75.8 ± 26.4 pmol/L; 95% CI, 70.6–81.0 pmol/L), compared with age- and gender-matched healthy controls (mean ± SD, 88.0 ± 34.1 pmol/L; 95% CI, 81.2–94.8 pmol/L) (p = 0.04) (Figure 1). CGRP plasma concentrations in subjects with persistent PTH are presented in Table 2.
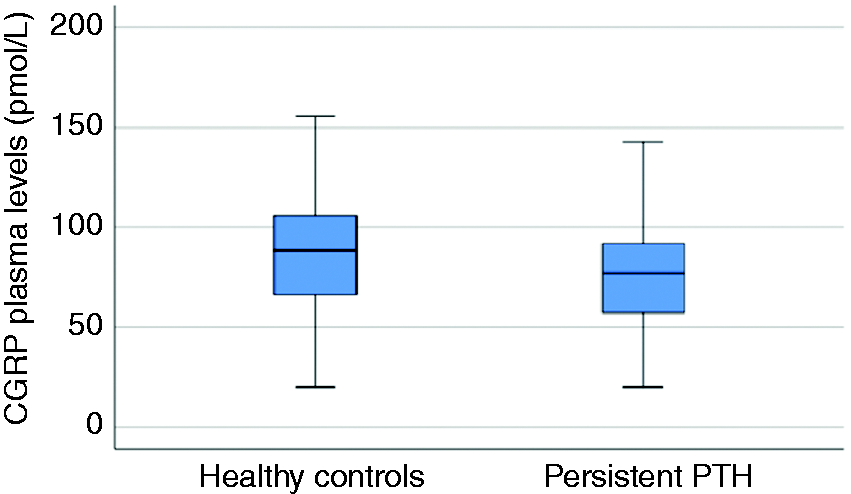
Plasma levels of CGRP in subjects with persistent PTH (n = 100) and healthy non-headache controls (n = 100). In the peripheral circulation (antecubital vein), CGRP plasma levels were lower in subjects with persistent PTH, compared with age- and gender-matched healthy controls (p = 0.04).
CGRP plasma levels (pmol/L) in subjects with persistent PTH (n = 100).

Correlations
CGRP plasma levels did not correlate with age (r = 0.16; p = 0.12), gender (r = −0.18; p = 0.08), or months with headache attributed to mild TBI (r = 0.12; p = 0.24) in subjects with persistent PTH. There was also no correlation between CGRP plasma concentrations and the following headache characteristics: Monthly headache days (r = −0.11; p = 0.27; Figure 2), monthly migraine days (r = 0.15; p = 0.13), chronic migraine-like headache phenotype (r = −0.02; p = 0.85), throbbing headache (r = −0.09; p = 0.35), headache intensity on the experimental day (r = −0.09; p = 0.39), and aura (r = −0.05; p = 0.64). In addition, we observed no relationship of CGRP plasma concentrations with current use of preventive medication (r = −0.01; p = 0.95) or severity of allodynia (r = −0.08; p = 0.64). Results from the correlation analyses performed are summarized in Table 3. In 75 female subjects of childbearing potential, CGRP plasma levels did not correlate with use of hormonal contraceptives (r = 0.20; p = 0.08) or number of days since day 1 of the last menstrual period (r = −0.06; p = 0.61).

Scatter plot of CGRP plasma levels and monthly headache days in subjects with persistent PTH (n = 100). CGRP plasma levels did not correlate with monthly headache days in subjects with persistent PTH (r = −0.11; p = 0.27).
Correlation of CGRP plasma levels (pmol/L) with demographic and clinical characteristics in subjects with persistent PTH (n = 100).

ASC-12: 12-item Allodynia Symptom Checklist.
Discussion
In this study, we found that CGRP plasma levels were lower in subjects with persistent PTH (mean, 75.8 pmol/L; SD, 26.4 pmol/L), compared with the age- and gender-matched healthy controls (mean, 88.0 pmol/L; SD, 34.1 pmol/L). No correlation was found of CGRP plasma levels with any demographic or clinical variable, including monthly headache days, monthly migraine days, chronic migraine-like headache phenotype, headache quality, and headache intensity on the experimental day.
CGRP is a key signaling molecule in migraine pathogenesis (14) and has recently been implicated in disease mechanisms underlying PTH as well (3–6). CGRP is widely distributed in the trigeminovascular system (14) and its release from trigeminal primary afferents has been well established (15). In rodent models of PTH, three studies have investigated the role of CGRP in the underlying disease mechanisms (4–6). Data from these studies indicate that CGRP plays a modulatory role in nociceptive transmission following head trauma. These findings are interesting for two reasons: a) PTH often mimics a chronic migraine-like headache phenotype (16) and b) previous studies have investigated CGRP in the peripheral circulation as a potential biomarker for chronic migraine (7,8,17). Initial data were promising, showing elevated interictal levels of CGRP in peripheral blood of chronic migraine patients, compared with healthy controls (7). However, a subsequent study failed to reproduce these findings using similar methodology and procedures (8). Thus, it remains unclear whether peripheral blood-based assessments of CGRP levels have clinical utility as a biomarker for chronic migraine.
In the present study, CGRP plasma levels were lower in subjects with PTH, compared with the age- and gender-matched healthy controls. We hypothesize that the daily – often continuously ongoing – headache experienced by those with persistent PTH results in depletion of CGRP in trigeminal afferents. This would explain the low CGRP plasma concentrations found in the present study. In rats, CGRP levels were depleted without recovery for more than a week following capsaicin injection in the paw skin and sciatic nerve (18,19). Based on these data, it could be speculated that capsaicin might result in depletion of CGRP within trigeminovascular structures. Indeed, CGRP expression was decreased in the trigeminal ganglion and trigeminal nucleus caudalis after capsaicin injection into periodontal tissues (20). Interestingly, one recent study reported that CGRP plasma levels were lower in chronic cluster headache patients compared with cluster headache patients in remission (12). Thus, lack of replenishment with newly synthesized CGRP molecules might explain the low plasma concentrations of CGRP in subjects with persistent PTH. This relies on the assumption that capsaicin-mediated pain mechanisms are a reliable measure for continuous headache and its effect on the trigeminovascular system. Taken together, we would, therefore, conclude that there is currently not enough evidence to support that the constant and unremitting headache experienced by individuals with persistent PTH is associated with depletion of CGRP in trigeminal afferents. Thus, more studies are needed to explore this hypothesis.
Another possible explanation for the present findings is that local CGRP release can only be accurately detected in the cranial circulation. We collected blood from the antecubital vein and the transport of CGRP from cerebral structures to peripheral blood might hamper reliable plasma measurements given the short half-life of CGRP in venous blood (21). It has been argued that the external jugular vein represents a more suitable target to accurately assess cerebral outflow. However, only 22% of external jugular vein blood flow originates from the cerebral circulation (22). Also, elevated peripheral CGRP levels have been reported in several migraine studies (17). Thus, it seems feasible to detect differences in CGRP levels based on blood drawn from the antecubital vein.
Pre-analytical confounders might also impact our findings. Although we used a validated radioimmunoassay, the analytical assay sensitivity and specificity to detect cranial changes in CGRP plasma concentrations remains unknown. In addition, longitudinal follow-up studies are needed to determine inter-subject fluctuations in CGRP plasma concentrations over time. In the present study, we found that CGRP plasma levels ranged from 20–143 pmol/L in subjects with persistent PTH and from 20–181 pmol/L in healthy controls. Thus, future studies should emphasize large samples to avoid over- or misinterpretation of CGRP plasma measurements. Furthermore, it should be noted that some of the preventive medications used might influence plasma levels of CGRP, although we did not find any correlation of CGRP plasma levels with current use of preventive medications. It would have been ideal to have a larger sample, enabling correlation analyses of CGRP plasma concentrations with each drug class of preventive medications used. Lastly, the present study was not designed to assess possible effects related to gender differences and use of hormonal contraceptives. Indeed, some evidence suggests that such factors might influence plasma measurements of CGRP (23,24).
Conclusions
CGRP involvement has been reported in preclinical models of PTH (4–6). However, our data shows that CGRP plasma concentration is unlikely a feasible biomarker of persistent PTH. As such, there are still no valid biomarker of persistent PTH. Nevertheless, more studies are needed to replicate our findings and future studies should also assess whether CGRP plasma measurements can be used to predict development of persistent PTH.
Clinical implications
CGRP plasma levels were lower in individuals with persistent PTH, compared with age- and gender-matched healthy controls. Plasma CGRP concentration measurements are unlikely a feasible biomarker of persistent PTH. Future studies should assess whether CGRP plasma measurements can be used to predict development of persistent PTH.
Footnotes
Author contributions
HA: Study concept and design, acquisition of data, analysis (including statistical analyses) and interpretation, drafting the manuscript.
HMA-K: Acquisition of data and critical revision of the manuscript for important intellectual content.
AI: Acquisition of data and critical revision of the manuscript for important intellectual content.
SA: Analysis (including statistical analyses) and interpretation, critical revision of the manuscript for important intellectual content.
NRJ: Critical revision of the manuscript for important intellectual content.
FMA: Study concept and design, critical revision of the manuscript for important intellectual content.
MA: Study concept and design, critical revision of the manuscript for important intellectual content.
HWS: Study concept and design, critical revision of the manuscript for important intellectual content, supervision.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: SA has received honoraria from Allergan and Teva and consultant fees from Amgen and Allergan; honoraria for consulting for Allergan, Amgen, Eli Lilly, Novartis, Promius, Satsuma, Supernus, and Theranica. FMA is a lecturer or scientific advisor for Teva and Novartis. MA has received personal fees from Alder BioPharmaceuticals, Allergan, Amgen, Eli Lilly, Novartis, and Teva, and also participated in clinical trials as the principal investigator for Alder, Amgen, electroCore, Novartis, and Teva. MA also serves as an associate editor of Cephalalgia, associate editor of Headache and co-editor of the Journal of Headache and Pain. MA is President of the International Headache Society, and General Secretary of the European Headache Federation. MA also reports research grants from Lundbeck Foundation, Research Foundation of the Capital Region of Copenhagen, and Novo Nordisk Foundation. HWS received speaking fees from Novartis and Teva. The other authors declare no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a grant from the Rigshospitalet Research Foundation (F-23340-02).
