Abstract
Background
Recent clinical findings suggest that oxytocin could be a novel treatment for migraine. However, little is known about the role of this neuropeptide/hormone and its receptor in the trigeminovascular pathway. Here we determine expression, localization, and function of oxytocin and oxytocin receptors in rat trigeminal ganglia and targets of peripheral (dura mater and cranial arteries) and central (trigeminal nucleus caudalis) afferents.
Methods
The methods include immunohistochemistry, messenger RNA measurements, quantitative PCR, release of calcitonin gene-related peptide and myography of arterial segments.
Results
Oxytocin receptor mRNA was expressed in rat trigeminal ganglia and the receptor protein was localized in numerous small to medium-sized neurons and thick axons characteristic of A∂ sensory fibers. Double immunohistochemistry revealed only a small number of neurons expressing both oxytocin receptors and calcitonin gene-related peptide. In contrast, double immunostaining showed expression of the calcitonin gene-related peptide receptor component receptor activity-modifying protein 1 and oxytocin receptors in 23% of the small cells and in 47% of the medium-sized cells. Oxytocin immunofluorescence was observed only in trigeminal ganglia satellite glial cells. Oxytocin mRNA was below detection limit in the trigeminal ganglia. The trigeminal nucleus caudalis expressed mRNA for both oxytocin and its receptor. K+-evoked calcitonin gene-related peptide release from either isolated trigeminal ganglia or dura mater and it was not significantly affected by oxytocin (10 µM). Oxytocin directly constricted cranial arteries
Conclusion
Oxytocin receptors are extensively expressed throughout the rat trigeminovascular system and in particular in trigeminal ganglia A∂ neurons and fibers, but no functional oxytocin receptors were demonstrated in the dura and cranial arteries. Thus, circulating oxytocin may act on oxytocin receptors in the trigeminal ganglia to affect nociception transmission. These effects may help explain hormonal influences in migraine and offer a novel way for treatment.
Introduction
The recent success of migraine drugs that target calcitonin gene-related peptide (CGRP) (1) has stimulated interest in other regulators of the trigeminovascular pathway as possible targets for migraine therapy (2,3). One novel candidate is oxytocin (OT), a nonapeptide neurotransmitter/hormone that was discovered in 1906 by Sir Henry Dale (4) and was the first peptide hormone to be sequenced and synthesized (5). Recently, it has been reported that intranasal or intravenous administration of OT can relieve migraine headache (6–8). This is an intriguing result for a neurohormone best known for its roles in parturition, lactation and social behaviour (9). OT has been shown to have analgesic actions and to act centrally to suppress incoming sensory nociceptive signals (10,11). However, OT does not cross the blood-brain barrier (BBB), suggesting a peripheral site of action for its anti-migraine effect. As of yet, little is known about the role of OT and OT receptors (OTR) in regulating the trigeminovascular system and how it might be involved in migraine pathophysiology.
The supraoptic and paraventricular nuclei of the hypothalamus are the primary sources of OT for both central and peripheral actions (12). Hypothalamic OT neurons innervate numerous sites in the brain, including areas involved in autonomic and sensory function (13,14). In particular, descending OT projections innervate the medullary and spinal dorsal horn regions where trigeminal sensory afferents terminate (12,15,16). Stimulation of this pathway or local OT application inhibits spinal processing of nociceptive input from A∂- and C-fibers and suppresses pain responses (15).
Magnocellular neurons in the hypothalamus secrete OT into the circulation via axonal projections to the posterior pituitary gland. Circulating OT also has anti-nociceptive effects (10,17), but these actions ought to involve sites outside the BBB. The peripheral mechanisms by which OT suppresses pain responses are not well studied. Several reports indicate the presence of OTR, and possibly OT, in the sensory dorsal root and trigeminal ganglia (18–22). However, there are some discrepancies in these findings, perhaps due to differences in methodology (e.g. antibodies used) and/or model (e.g. inflammation). Because of the key role of the trigeminovascular system in migraine pathophysiology and treatment (1), a more comprehensive understanding of OT-related mechanisms in this system is warranted. In the current study, we used normal animals to investigate OT and OTR gene expression, localization and function.
Materials and methods
All animal procedures in this study followed the guidelines of the European Communities Council (86/609/ECC) and were approved by the Regional Ethical Committee on Animal Research, Malmö/Lund, Sweden (M17-15) and the Danish Animal Experimentation Inspectorate.
Immunohistochemistry
Ten Wistar male rats were euthanized by CO2 inhalation followed by decapitation. TG were carefully dissected and placed in 4% paraformaldehyde (PF) in phosphate-buffered saline (PBS) for 2–4 hours (23), followed by incubation overnight in Sörensen’s phosphate buffer (pH 7.2) containing 10% and 25% sucrose in turn. Thereafter, the tissue was embedded in Yazulla embedding medium (30% egg albumin, 3% gelatin) and cryo-sectioned at 10 µm. The sections were stored at −20°C until use.
Sections were washed in PBS containing 0.25% Triton-X (PBS-T) for 15 minutes followed by application of the primary antibody (Table 1) with incubation overnight at +4°C in moisturized incubation chambers. The following day, the sections were washed twice in PBS-T for 15 minutes prior to incubation with secondary antibodies (Table 1) for 1 hour at room temperature. Finally, the sections were washed 2 × 15 minutes and mounted with Vectashield mounting medium containing 4’,6-diamidino-2-phenylindole (DAPI) (Vector Laboratories, Burlingame, CA, USA).
Details of the primary and secondary antibodies.

Three different OT antibodies and four OTR antibodies were tested. Anti-rabbit oxytocin AB911 from Millipore and anti-guinea pig oxytocin PA1-18416 from Thermo Fisher displayed consistent and comparable results and were therefore used in the mapping. As regards to OTR antibodies, anti-goat OTR ab87312 from Abcam and anti-rabbit OTR aa43-129 from LifeSpan Biosciences also showed consistent and comparable results and were therefore used in the present study. Immunohistochemistry was performed three to six times per antibody. For further details of the antibodies used, see Table 1.
Omission of primary antibodies served as negative controls. The sections were examined in a light and epifluorescence microscope (Nikon 80i, Tokyo, Japan) equipped with Nikon DS-2MV camera. Finally, images were processed using Adobe Photoshop CS3 (v0.0 Adobe Systems, Mountain View, CA).
According to Forbes and Welt (24), the mean size of the neurons in adult rat TG is 26.13 µm (see Figure 1, a modification of size-frequency histogram of rat TG). Hence, the majority of cells are currently categorized as “small cells” i.e. <30 µm. Only a few percent of the neurons in rat TG are “large cells” (>60 µm). The remaining cells, approximately 1/3, are classified as “medium-sized cells”; that is, 30–60 µm.
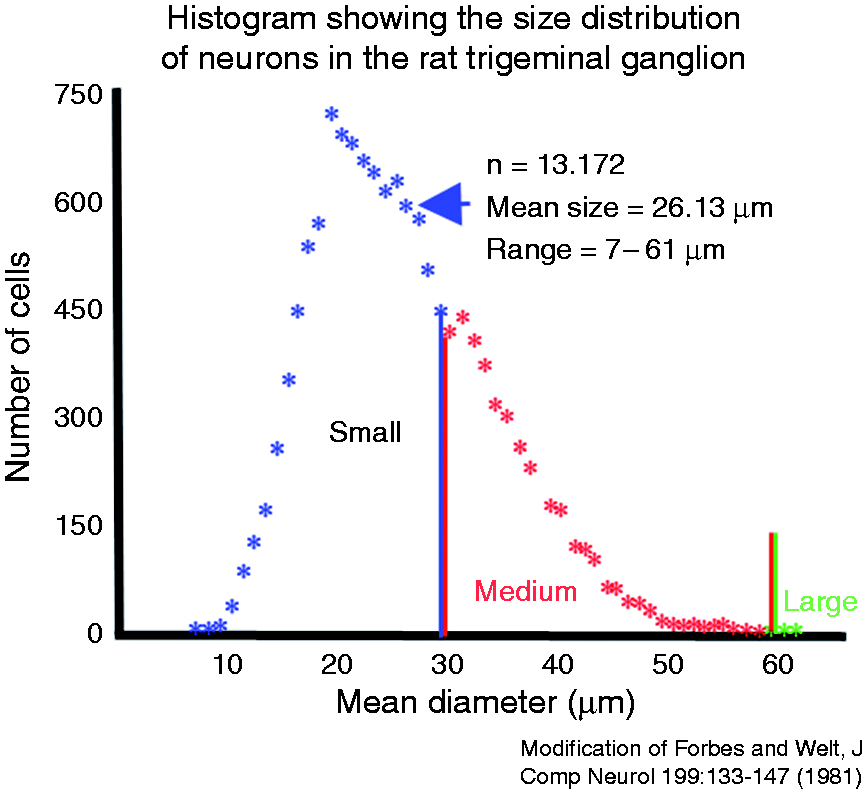
A modification of a size-frequency histogram showing the size distribution of neurons in the trigeminal ganglion. From Forbes and Welt.24
Estimation of the prevalence of OTR, calcitonin gene-related peptide (CGRP), receptor activity-modifying protein 1 (RAMP1) and double-stained cells in the TG were determined by counting intermingled cells in pooled ophthalmic-maxillary and mandibular areas of each ganglion (24). We counted cells only intermingled in fibers, since our experience is that neurons close to the surface may show artefactual fluorescence. All negative and immunoreactive OTR, CGRP, RAMP1 or double-stained cells were counted in each area. The mean percentage of positive neurons (small and medium sized) in three slides/rat from three rats was calculated.
For double immunohistochemistry, the procedure was repeated two consecutive times. The first primary antibody was matched with its appropriate secondary antibody before the second round of primary antibodies was applied and finally mounted. Negative controls were performed for each set by omitting the primary antibody. Any resulting immunofluorescence would suggest unspecific binding of the secondary antibodies.
Cryo-sections of TGs were stained using Hematoxylin and Eosin (Htx 4 min, Eosin 1 min). The staining was done in order to examine the morphology and condition of the tissue.
Analysis of mRNA with qPCR
Rat TG, TNC and hypothalamus were dissected out for RNA extraction from six rats. The hypothalamus was gently “spooned” out using curved forceps. Both left and right TG were dissected and used for one sample. Brain stem tissues containing the TNC were carefully dissected and both left and right TNC were used for one sample. After dissection, all three types of tissue were cut into smaller pieces with a scalpel and immediately frozen on dry ice.
Tissue was homogenized on dry ice 6 × 20 sec in lysing matrix D tubes containing 1.4 mm ceramic spheres (MP Biomedicals, USA) and lysis buffer (ML buffer) from the NucleoSpin miRNA isolation kit (Macherey-Nagel, Germany) using a FastPrep-24™ 5G instrument (MP Biomedicals, CA, USA). The NucleoSpin miRNA isolation kit was used to extract total RNA according to the manufacturer’s protocol. Total RNA concentration was measured by the NanoDrop 2000 UV-Vis spectrophotometer (ThermoFisher Scientific, MA, USA). A ratio of sample absorbance at 260 nm and 280 nm in the range of 1.9 to 2.2 was acceptable.
2000 ng RNA from TG, TNC and hypothalamus was synthesized to cDNA using the RT2 First Strand Kit (Qiagen, MD, USA) according to the manufacturer’s protocol. qPCR was performed in a 10 µl reaction volume containing RNAase free water, 20 × TaqMan gene expression assay (ThermoFisher Scientific, USA), 2 × TaqMan universal PCR master mix (ThermoFisher Scientific, MA, USA) and 2 µl cDNA using the QuantStudio 12K Flex real-time PCR system (ThermoFisher Scientific, MA, USA) with ROX as a passive reference. The thermal cycling condition included an initial denaturation step at 50°C for 2 min and 95°C for 10 min followed by 45 PCR cycles at 95°C for 15 sec and 60°C for 1 min. A no-template was used as negative control for all TaqMan gene expression assays where RNAse-free water was added instead of cDNA. All TaqMan gene expression assays were pipetted in triplicate for each sample: OT (
Calculations and statistics for qPCR
Tissue from six male Sprague-Dawley rats (250–350 g, Taconic, Ejby, Denmark) was divided into three groups: TG, TNC and hypothalamus (n = 6/group). The threshold cycle (Ct) is the intersection between the amplification curve and the threshold line, and it was determined using QuantStudio 12K Flex software (ThermoFisher Scientific, MA, USA). Technical triplicates of Ct values were averaged for each TaqMan gene expression assay. Ct values are plotted on the y-axis using GraphPad Prism 8. The y-axis is log and reversed. Data is presented as mean ± standard error of the mean (SEM), and
CGRP release studies
Rats (18 male Sprague-Dawley rats, ∼ 300 g) were anaesthetized by CO2 inhalation and decapitated. TGs were dissected out and immersed in 10 mL synthetic interstitial fluid (SIF, composition: 108 mM NaCl, 3.5 mM KCl, 3.5 mM MgSO4, 26 mM NaHCO3, NaH2PO4, 1.5 mM CaCl2, 9.6 mM NaGluconate, 5.6 mM glucose and 7.6 mM sucrose; pH 7.4.) at 37°C for 30 min. TGs were randomized, and placed in Eppendorf tubes in a heating block at 37°C. TGs were again washed five times (each wash 10 minutes), with 300 µL SIF.
To study release from the dura, the skull was cut mid-sagittal and the brain halves were carefully removed while the cranial dura was left attached to the skull. Skull halves were transferred to a beaker containing 250 mL of SIF and washed two times (each wash 15 min). Both skull halves were randomized and placed in a humid chamber above a water bath to maintain temperature at 37°C and washed five times (each wash 10 min) with 300 µL SIF.
After 10 min incubation with 300 µL SIF, 200 µL samples for measuring the basal CGRP release were collected from all the tissues, mixed with 50 µL enzyme immunoassay buffer (containing protease inhibitors) and stored at −20°C until analysis, within a week after the experiment was performed. Previous studies have shown that there is no significant difference between the basal CGRP release from the left and the right side of the tissues; thus, one side was used to test OT and the other served as a vehicle control using a paired design shown to reduce experimental and biological variations in this assay (25,26).
CGRP release from the TG samples and the skull halves was first measured under basal conditions and then in the presence of different concentrations of OT or vehicle (after a 20-min incubation period) in order to test for release stimulated by OT
The samples were processed using commercial EIA kits (SPIbio, Paris, France) to measure CGRP release. From both tissues, 200 µL of sample was mixed with 50 µL of EIA buffer, which was also added to the CGRP standard. Determination of CGRP content was calculated based on this standard curve. Antibody in the CGRP EIA kit is directed against human-CGRP α/β, but it has 100% cross-reactivity with rat and mouse CGRP (26,27). The protocol was performed following the manufacturer’s instructions. Briefly, samples were incubated in at 4°C for 16–20 h, washed, and incubated with Ellman’s reagent. Following 60 min of incubation, the optical density was measured at 410 nm using a micro-plate photometer (Tecan, Infinite M200, software SW Magellan v.6.3, Männedorf, Switzerland). Statistical analysis was performed using paired two-sided Student’s t-test in Graph Pad 8.0.
Contractile responses of cranial arteries ex vivo
Contractile responses of rat (male Sprague Dawley ∼ 300 g) basilar, middle cerebral and middle meningeal arteries (commonly named cranial arteries) were measured
Results
Immunohistochemical localization of OTR and OT in TG
To validate our findings, four different anti-OTR antibodies were tested. Both the Abcam (ab87312) and LifeSpan (aa43-129) antibodies displayed consistent and comparable results and were therefore used for localization mapping in this study.
In the present study, immunoreactivity for OTR was observed in numerous small and medium-sized neurons, and in thick axonal fibers within the rat TG (Figure 2(a)). The OTR immunoreactivity in cell bodies was observed mainly in the cytoplasm in a mesh-like pattern, characteristic of the endoplasmic reticulum (Figure 2(a)). Counting of the cells in the ophthalmic-maxillary and mandibular divisions together showed that 35% of the small neurons and 80% of the medium-sized neurons expressed OTR (Figure 3(c)).
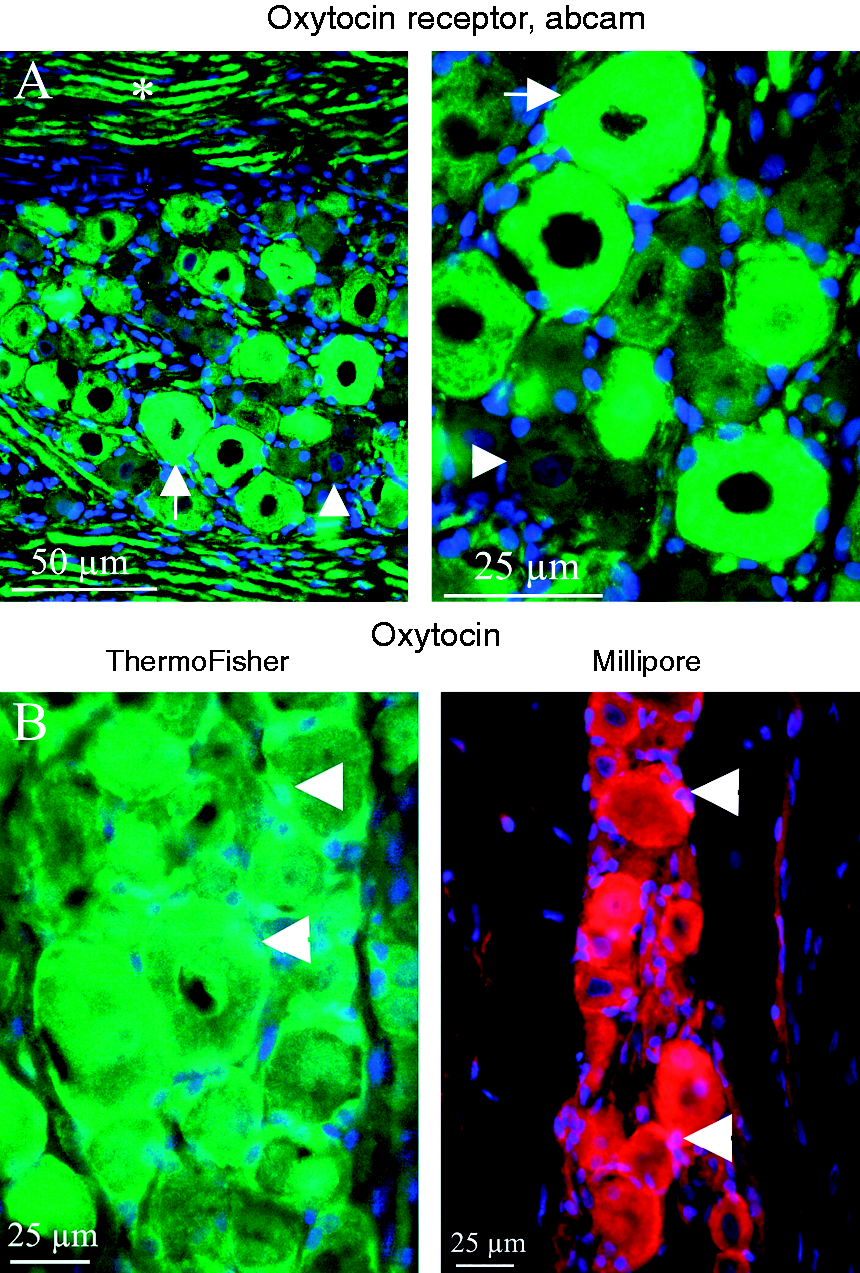
OTR and OT immunoreactivity in rat TG. (a) Numerous neurons were immunopositive for OTR (Abcam antibody, green), in particular the medium-sized neurons (arrow). Staining was cytoplasmic, with a mesh-like appearance. Not all neurons were positive, in particular smaller-sized neurons (arrowhead). OTR immunoreactivity was also evident in the thick axonal fibers (asterisk). Nuclei were stained with DAPI (blue). (b) Two different antibodies against the neuropeptide OT consistently stained satellite glial cells (arrowheads) surrounding neuronal cell bodies; ThermoFisher PA1-18416 (green) and Millipore AB911 (red). With the Millipore antibody, some neurons also appeared immunopositive. Nuclei were stained with DAPI (blue).

Co-localization of OTR and CGRP or RAMP1 in rat TG. (a) Double immunohistochemistry using antibodies to OTR (red, LifeSpan Ab) and CGRP (green) was used to determine possible co-localization in rat TG. Most CGRP-positive cells (thin arrows) were small- sized neurons that did not show co-localization with OTR. In contrast, OTR-labelled cells were primarily medium-sized neurons (arrow head) and fibers. A small number of neurons were positive for both antibodies (thick arrow). In these cells, OTR and CGRP appear to be located in distinct intracellular compartments (merged image, lower panel). (b) Double immunohistochemistry using antibodies to OTR (red, LifeSpan Ab) and RAMP1 was used to demonstrate double staining of the two receptors. Many of the medium-sized neurons (47%) and small neurons (23%) were positive for both antibodies. The mesh-like staining of OTR coincided with RAMP1 staining. (c) Size-frequency histogram showing percentage of TG neurons expressing OTR, CGRP, Ramp1, and double staining OTR/CGRP and OTR/RAMP1.
Positive immunofluorescence for the OT peptide was also detected in the rat TG (Figure 2(b)). Three different antibodies were tested (anti-rabbit OT AB911 from Millipore, anti-guinea pig OT PA1-18416 from Thermo Fisher, anti-rabbit OT orb185729 from Biorbyt LLC). AB911 and PA1-18416 were found to give consistent and comparable results. With these antibodies, positive immunoreactivity for OT was found in satellite glial cells (SGC) that surround trigeminal neurons. The antibody from Millipore appeared to stain some neurons, but this was not seen using PA1-18416. No immunoreactivity was observed in nerve fibers. No counting was made since the OT expression was mainly found in SGC.
Immunohistochemical localization of OTR relative to CGRP and its receptor
Double immunohistochemistry was performed with OTR and CGRP or the CGRP receptor component receptor activity-modifying protein 1 (RAMP1) antibodies to determine possible OT regulation of CGRP-containing neurons in the ganglion (Figure 3).
It has been shown that the mean diameter of CGRP-positive neurons is approximately 20 µm (30), which we confirm in the present study by showing that 55% of the small neurons and 29% of the medium-sized neurons expressed CGRP (Figure 3(c)). In the majority of these trigeminal neurons there was no co-localization, indicating OTR is not generally present in CGRP neurons. In a small number of medium-sized neurons in which OTR and CGRP were found together (10%) intracellular co-localization was not observed, indicating CGRP and OTR are expressed in different cytoplasmic compartments (Figure 3(a) and (c)). CGRP staining showed a cytoplasmic pattern resembling that for the Golgi apparatus, whereas OTR staining appeared similar to that of the endoplasmic reticulum. No small neurons expressed OTR and CGRP together. We also used OTR and RAMP1, a CGRP receptor component, double immunohistochemistry (31); 23% of the small cells and 47% of the medium-sized neurons expressed OTR and RAMP1 in the same cell (Figure 3(b) and (c)). The RAMP1-positive cells also expressed the other component of the CGRP receptor, CLR (calcitonin receptor-like receptor), indicating that receptors for OT and CGRP co-localize in the TG.
The mRNA expression of OTR and OT in the trigeminal pathway
The immunohistochemical findings indicated the presence of OT peptide and OTR protein within the trigeminal system pathway. To determine whether the OT and the OTR genes (

Expression of OT and OTR mRNA in the trigeminal pathway. qPCR was used to determine expression of mRNA for (a) oxytocin and (b) oxytocin receptor in rat TG, brainstem TNC and hypothalamus (HT, positive control tissue). Relative amounts of mRNA transcript were determined as Ct (threshold cycle) values. Data are presented as median and interquartile range, n = 6. The threshold line indicates the limit of detection for the assay.
Effect of OT on CGRP release in TG and dura mater
Since OTR was expressed in some CGRP-containing trigeminal neurons, we investigated if exogenously applied OT could affect CGRP release, either from the TG or from trigeminal afferents innervating the dura mater (Figure 5). Isolated preparations of dura and TG were used to measure CGRP release under both basal conditions and following stimulation with 60 mM K+. In both tissues, K+-evoked a substantial release of CGRP. We did not observe any significant effects of OT (10 µM) on basal release of CGRP from the dura mater preparation (Figure 5(a)) or the TG (Figure 5(b)), indicating OT does not trigger CGRP release

Effect of OT on CGRP release from the dura or TG in situ. Release of CGRP from isolated preparations of rat (a) dura mater and (b) TG are shown. Using matched tissue samples, release was first measured under basal conditions in the absence of treatment (“Dura/TG baseline”). Measurements of CGRP release were then made following addition of OT (10 µM) or vehicle (“Dura/TG veh/ago”). Finally, depolarization-evoked CGRP release was measured after addition of 60 mM K+ in the presence of OT (10 µM) or vehicle (“Dura/TG K+”). Data are shown as mean ± SEM, with n = 6 for the dura and n = 9 for the TG. (C) K+-evoked CGRP release from individual pairs of TG treated with OT and vehicle are shown, illustrating the variability in responses between pairs with no OT effect (non-inhibitory) and OT inhibitory effects (inhibitory).
Vasomotor effects of OT on cranial arteries
Cranial arteries are another important target of peripheral trigeminal afferents. We investigated potential contractile effects of OT on isolated segments of two cerebral arteries (middle cerebral artery, MCA, and basilar artery, BA) and an extracerebral artery (middle meningeal artery, MMA) of the rat (Figure 6). OT elicited concentration-dependent contractions in each of the three arteries. The pEC50 values for OT were similar in the MCA, 7.34 (7.53 to 7.14, 95% CI) and the BA, 7.19 (7.30 to 7.08, 95% CI). OT was somewhat less potent in the MMA with a pEC50 of 6.93 (7.02 to 6.81, 95% CI) in the MMA. We initially tested a known OTR antagonist, L368899 (10 µM) and found that it significantly inhibited OT-evoked contractions in all three arteries (Figure 6).

Effect of oxytocin on vascular tone of intra- and extracerebral arteries
However, because OT and OTR show structural similarities, respectively, with the peptide vasopressin and its receptors, OT agonists and antagonists may act at vasopressin receptors, especially at higher concentrations (32). To test if OT constricts cranial arteries via OTR or a vasopressin receptor, we also analysed the effects of vasopressin and the selective vasopressin V1A receptor antagonist (SR49059) in the BA (Figure 6(c)). SR49059 (100 nM) significantly inhibited the vasomotor effects of OT. Moreover, vasopressin, itself, evoked contraction of the BA with a much higher potency (pEC50 = 9.82; 10.04 to 9.62, 95% CI) than that found for OT. This order of potency (vasopressin > OT) is indicative of vasopressin receptors. In addition, the vasopressin response was also significantly inhibited by 10 µM L368899, indicating a lack of OTR specificity at this concentration in the artery. Thus we conclude that, at higher concentrations, OT can constrict cranial arteries by acting on vasopressin V1A receptors, but there is no evidence supporting OTR-mediated contractions in the cranial arteries.
Discussion
Although OT was among the first neuropeptides to be discovered, its effect on trigeminal signal transmission has been largely ignored. The present study demonstrates a significant role of OT and OTR throughout the trigeminovascular pathway, within ganglionic neurons as well as central targets of trigeminal afferents. From this perspective, it appears OT may influence both central migraine mechanisms, in part via descending hypothalamic pathways (10,16), as well as peripheral processing of headache pain via circulating OT. OTRs in the TG are outside the BBB (33) and readily accessible as potential targets for migraine therapy. Indeed, it has been reported that intranasal and intravenous administration of OT provides migraine relief (6−8).
Our results underscore a major role for OTR within the TG. The
In this study, we showed that the CGRP receptor, verified by positive staining with RAMP1 (and in a few experiments verified that also the other component CLR co-stained), and the OTR co-localized in the mid-sized neurons. From our point of view, this is representative for Aδ-fibers and this was verified by examination that in myelinated fibers contained both RAMP1 and OTR positivity (37). However, in contrast to the dense distribution of CGRP in the sensory unmyelinated C-fibers, these were devoid of OT, thus a similar relation at peripheral sites of the trigeminal nerves is unlikely. The way OT might modify peripheral signaling would be via blood-borne hormone distribution.
Similar to what was reported for dorsal root ganglia (DRG) (19), the majority of OTR-positive neurons were negative for CGRP immunostaining; no double staining was seen in the small cells and only approximately 10% of the medium-sized neurons expressed both OTR and CGRP. We have shown previously that CGRP is expressed primarily in small cells and thin fibers characteristic of sensory C-fibers (31). In the small number of trigeminal neurons that did co-express OTR with CGRP, the intracellular pattern of immunoreactivity was distinct but different. CGRP staining was characteristic of the Golgi apparatus, consistent with a peptide being packaged vesicles for transport and secretion, whereas the receptor staining resembled that for the continous membrane system of endoplasmic reticulum. Tzabazis et al. (20) also found numerous OTR-positive neurons in rat TG. However, in contrast to the present study, they observed OTR immunoreactivity extensively co-localized with CGRP. They used a rat inflammation model and found that inflammation significantly upregulated levels of OTR protein in the ganglion. It is possible that under these extreme conditions, there may be a shift in neuronal expression. Discrepancies among studies of OTR localization in sensory ganglia (18–20) may also reflect the use of different antibodies. We tested four different antibodies and found only two that gave consistent staining results. Further studies need to take into account the antibodies used as well as the possibility that conditions such as inflammation or culture may affect OTR expression.
Because we did find OTR in a small number of CGRP-positive neurons, we tested whether OT affects depolarization-evoked CGRP release from either isolated TG or dural afferents in a cranial cup model. No significant effect was found in either tissue with 10 µM OT. This is consistent with the relative lack of OTR we observed in CGRP-containing neurons. Our findings, however, differ from the study by Tzabazis et al. that found significant effects of 10 and 100 µM OT (but not 1 µM) on capsaicin-stimulated CGRP release from rat dura mater after induced inflammation (20). The relevance of the latter findings is unclear as the effective concentrations were much higher than physiological OT levels.
In the trigeminovascular pathway, the intracranial and cerebral arteries are key targets for the peripheral afferent nerves from the TG. In the present study, we demonstrated
In a recent publication, we demonstrated the distribution of OT and OTR in the rat brain (14). We demonstrated the presence of OTR and OT in the trigeminovascular pathway, and also examined the key target of the central trigeminal afferents, the TNC in the medulla (also referred to as the spinal trigeminal nucleus or Sp5) (1). In addition, we summarized that cerebral cortex, cerebellum, hypothalamus, pons and Sp5 were regions where CGRP and OT expressions matched (14).
Detailed neuronal tracing studies from the temporal artery, the middle meningeal artery, the superior sagittal sinus (40–42) and cerebral arteries showed that these regions project to the most caudal part of the TNC and the spinal C1-C3 regions (43). Thus, these projection regions represent V1 and V2 of the TG, and the distribution areas they showed were somatotopically different and likely convey nociceptive information from the craniofacial area to second order neurons that project to supraspinal sites in the pain pathway (44). We found that mRNA for both OT and OTR was expressed in this region (present data and Warfvinge 2020 (14)). Immunohistochemistry confirmed the presence of OT in some of the TNC neuronal cell bodies. This localization is somewhat unique in the CNS, in that our recent survey of OT immunoreactivity in rat brain revealed primarily OT-positive fibers but few labeled cell bodies outside of the hypothalamus (14). With regards to OTR, we were unable to detect immunoreactivity in the TNC, in spite of positive mRNA results in tissue containing the TNC and our findings of OTR in A∂ fibers in the TG. This result may be due to technical issues, such as difficulties in visualizing receptors that may be diffusely localized on neuronal membranes. Previous studies using receptor autoradiography have reported OTR binding sites in the TNC region of humans and monkeys (45,46). Moreover, local application of OT has been shown to inhibit firing of TNC neurons stimulated by peripheral trigeminal nerves and this effect is blocked by OTR antagonists, indicating the involvement of functional OTRs in this region (16).
Within the TG, we observed OT immunoreactivity in satellite glial cells and in few neurons. This agrees with early reports of the presence of chromatographically identified OT peptide in human and rat TG (21,22). The latter study also showed the number of neurons exhibiting OT immunoreactivity was greatly increased following colchicine treatment to block axonal transport (22). In the current study, however, we were unable to definitively detect mRNA for OT in the ganglion. Whether the presence of OT reflects a low level of synthesis or cellular uptake of the peptide remains to be determined. The TG lacks a BBB (33) and is therefore accessible to circulating OT that might accumulate in the satellite glia.
It is becoming clear that OT functions as an endogenous modulator of nociceptive transmission in the trigeminal nociceptive pathway. This realization opens up a new perspective on trigeminal physiology and pathophysiology and, in particular, the understanding and treatment of migraine. While there is much to be learned, OT likely influences both central and peripheral aspects of migraine. There is a growing appreciation of the key role of the hypothalamus in initiating and maintaining migraine attacks (47,48). Thus, it is particularly intriguing that OT is an output of hypothalamic neurons that influences trigeminal, spinal and central brain areas associated with migraine (14,49). Moreover, the involvement of OT may help explain the well-known, but little understood, hormonal influences in migraine (50). OT, with its direct anti-migraine effects, may provide the link between estrogen levels and migraine susceptibility in women (51); OT and OTR expression is regulated by estrogen (52,53) and fluctuations in circulating OT levels are similar to those for estrogen (51,54,55). This aspect needs to be explored further. The encouraging clinical results (6–8) point to the potential of OT and other OTR agonists as novel treatments for migraine in both men and women.
Key findings
The neurohormone oxytocin is an important regulator in the trigeminal pathway. Oxytocin receptor mRNA was expressed in rat trigeminal ganglia and receptor protein was extensively localized in myelinated A∂ sensory neurons and fibers. Immunoreactivity of OT was observed in trigeminal satellite glial cells. OTR were not found to play a role in peripheral trigeminal targets. Cranial arteries showed no oxytocin receptor-mediated vasomotor responses The central target of trigeminal afferents, the trigeminal nucleus caudalis, expressed mRNA for OT and OTR. Circulating OT, either released from the neurohypophysis or administered for migraine treatment, does not cross the blood-brain barrier and likely acts on receptors within the trigeminal system to modify nociceptive mechanisms.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Lundbeck foundation, Lundbeck Grant of excellence [no. R59-A5404], the Swedish Medical Research Council, and a research grant from the International Headache Society (KAH). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
