Abstract
Introduction
Cephalic autonomic symptoms occur in 27‒73% of migraine patients during attacks. The role of parasympathetic activation in migraine attack initiation remains elusive. Low frequency stimulation of the sphenopalatine ganglion increases parasympathetic outflow. In this study, we hypothesized that low frequency stimulation of the sphenopalatine ganglion would provoke migraine-like attacks in migraine patients.
Methods
In a double-blind randomized sham-controlled crossover study, 12 migraine patients with a sphenopalatine ganglion neurostimulator received low frequency or sham stimulation for 30 min on two separate days. We recorded headache characteristics, cephalic autonomic symptoms, ipsilateral mechanical perception and pain thresholds, mean blood flow velocity in the middle cerebral artery (VMCA) and diameter of the superficial temporal artery during and after stimulation.
Results
Five patients (42%) reported a migraine-like attack after low frequency stimulation compared to six patients (50%) after sham (p = 1.000). We found a significant increase in mechanical detection thresholds during low frequency stimulation compared to baseline (p = 0.007). Occurrence of cephalic autonomic symptoms and changes in mechanical perception thresholds, VMCA and diameter of the superficial temporal artery showed no difference between low frequency stimulation compared to sham (p = 0.533).
Conclusion
Low frequency stimulation of the sphenopalatine ganglion did not induce migraine-like attacks or autonomic symptoms in migraine patients. These data suggest that increased parasympathetic outflow by the sphenopalatine ganglion neurostimulator does not initiate migraine-like attacks.
Keywords
Introduction
The initiating mechanisms of migraine are complex and may involve the cephalic autonomic nervous system. Cephalic autonomic symptoms (CAS) such as lacrimation, conjunctival injection, eyelid edema, rhinorrhea and ptosis occur in 27‒73% of migraine patients during attacks (1–4). The presence of CAS indicates activation of the trigemino-autonomic reflex resulting in increased parasympathetic outflow (5). The sphenopalatine ganglion (SPG) is the major source of parasympathetic innervation to the skin and glands in the facial region as well as cephalic blood vessels (6). Therefore, SPG activation has previously been proposed to contribute to the development of migraine pain (7). In addition, the SPG contains sensory fibers, and SPG stimulation may therefore modulate sensory nerve function (6).
In recent years, emerging studies are targeting electrical stimulation of the SPG as a possible novel therapy in patients suffering from cluster headache (CH) and migraine (8–10). A multicenter clinical trial (NCT01540799) was initiated to investigate the therapeutic response to high frequency (HF) SPG stimulation in high-disability migraine patients. The rationale behind this trial was that HF SPG stimulation (120 Hz) might abort a migraine attack by blocking parasympathetic outflow. Recently, we showed that low frequency (LF) 20 Hz stimulation of the SPG induced CAS, but no CH attacks (11). This study demonstrated that SPG stimulation is a unique model to investigate if the parasympathetic nervous system contributes to the initiation of primary headaches. Whether LF stimulation of the SPG triggers migraine headache is unknown.
In the present study, we hypothesized that LF stimulation of the SPG would induce migraine-like attacks, increase parasympathetic outflow and activate sensory afferents. To test this hypothesis we conducted a double-blind randomized crossover study to investigate migraine induction following LF and sham stimulation in migraine patients with an implanted SPG stimulator.
Methods
We recruited migraine patients in 2016 from the Pathway M-1 study (CIP-003): Sphenopalatine Ganglion Stimulation for the Treatment of High Frequency, High Disability Migraine Headache (ClinicalTrials.gov: NCT01540799). This trial was terminated in 2018 due to insufficient recruitment, power and funding. The patients had medically intractable migraine attacks and reported pain to be unilateral in at least 75% of their migraine attacks. CAS was not an inclusion criterion. Exclusion criteria were medication overuse headache (MOH), somatic and psychiatric diseases among others. All patients were included during the open label phase of the Pathway M-1 study, in which the patients could treat their migraine freely and without limitations. In this study, we enrolled all patients irrespective of their headache response to therapeutic stimulation. The rechargeable, multichannel neurostimulation system consists of a small inductively powered SPG neurostimulator (Pulsante™ SPG Microstimulator System), which is inserted into the sphenopalatine fossa by a maxillofacial surgeon. The neurostimulator is powered and controlled by an external remote control.
The study was approved by the Ethics Committee of Copenhagen (H-1-2011-152) and the Danish Data Protection Agency. The study was conducted according to the latest revision of the Helsinki II declaration of 1964 and was registered at ClinicalTrials.gov (NCT02510742).
The patients were informed that SPG neurostimulation might induce headache or migraine in some individuals, but the timing and headache characteristics were not further explained.
Design and result of the pilot experiment
We conducted three single-blinded pilot experiments to determine the optimal LF SPG stimulation for the main experiment. The patients included in the pilot were chosen randomly because of their availability on the experimental days. Induction of headache, changes in CAS and mean blood flow velocity of middle cerebral artery (VMCA) by transcranial Doppler were used as markers for parasympathetic outflow. One migraine patient was enrolled in each pilot experiment. The patients’ usual amplitude and pulse width were used because this should be the most optimal stimulation for each individual patient and this was configured and tested during the Pathway M-1 trial. Only the frequency parameter was changed in the present pilot and main study.
The first pilot experiment was a stimulation-response study, in which one patient received 30 min SPG stimulation of 10, 20, 40, 80 and 120 Hz successively separated by a 30 min rest period. We observed rhinorrhea and a decrease in VMCA of 8% on the ipsilateral side immediately after 20 Hz stimulation. We observed no decrease in VMCA during the other frequencies of stimulation. Only at 80 Hz stimulation, a transient headache was immediately induced with a maximal intensity of four on a 0–10 verbal rating scale (VRS) lasting 20 min.
In the second pilot experiment, using the same patient as in the first pilot experiment, the patient received 20 Hz SPG stimulation for 30 min on three different study days. On each day, rhinorrhea was reproduced and a decrease in VMCA of 5%, 11% and 18% was detected. However, the patient did not develop headache on any of the three study days.
In the third pilot experiment, another migraine patient was allocated to receive 20 Hz SPG stimulation or sham stimulation (amplitude = 0) on two experimental days. The patient developed an ipsilateral migraine-like attack with a maximal intensity of seven on VRS after 10 min of SPG stimulation with CAS (conjunctival injection and nasal congestion) on the active day. The patient treated these symptoms with HF stimulation for 20 min and reported a headache improvement from seven to three on the VRS and resolution of CAS within 1 hour. On sham day, the patient developed a headache after 20 min localized on the right side of the neck with no autonomic or accompanying symptoms. The patient described it as a tension-type headache, which differed from her migraine headache. There was a decrease in VMCA on the ipsilateral side of 10% after 20 min on the active day and an increase of 4% on the sham day.
Based on these pilot experiments, the 20 Hz LF SPG stimulation in 30 min was chosen for the main study.
Design of the main experiment
We included 12 migraine patients in a double-blind, sham-controlled crossover study. The patients were randomly allocated to receive LF (20 Hz and amplitude: 0.25–2.2 mA) or sham (20 Hz and amplitude = 0) stimulation on two experimental days separated by at least 5 days. Two of the patients also participated in the three pilot experiments.
All patients arrived non-fasting at the clinic between 8:30–10:30 a.m. The patients were migraine free for at least 24 h before the start of the study but were allowed to have mild to moderate non-migraine headache because most of them had daily headache. If patients reported that the headache was developing into a usual migraine attack, the experiment was postponed. Patients were placed in a supine position in a quiet room with a constant temperature. After 15 min of rest, baseline recordings were performed. Immediately after baseline recordings, SPG stimulation (LF or sham) was applied for 30 min. Each patient’s usual pulse width and electrode configuration were kept constant. All patients were informed that the two kinds of stimulation would be different from their usual therapeutic HF stimulation. An investigator (EKH) programmed the stimulator according to a balanced randomization code generated by online randomization software (12). The randomization code was kept in the laboratory for safety reasons and was available only to EKH until the study was completed. Both patients and lab technicians recording headache quality, CAS responses and vascular changes were blinded to the neurostimulator setting.
Headache characteristics, CAS, diameter of the superficial temporal artery (STA) and vital parameters were recorded at baseline, 10, 20, 30 and 40 min after stimulation. Due to signal interference, it was not possible to measure VMCA in real time during SPG stimulation. Therefore, VMCA was recorded at baseline, 30 min, 33 min, 37 min and 40 min after start of stimulation. Von Frey hair and pinprick tests were recorded ipsilaterally before, during and after stimulation. If the stimulation induced a migraine-like attack, the patients were allowed to treat with the SPG stimulator, and/or with their usual acute migraine medication. After stimulation, the patients were observed for a minimum of 20 min before being discharged from the hospital.
Headache intensity and questionnaire
Headache intensity was recorded at baseline and every 10 min up to 40 min on a VRS from zero to 10. Zero is no headache, one represents a very mild headache (including a sensation of pressing or throbbing or otherwise altered sensation in the head not associated with pain), five is headache of moderate intensity and 10 is the worst headache imaginable (13). Headache characteristics, associated symptoms, CAS, whether headache mimicked usual migraine, adverse events, rescue therapy (SPG stimulation or triptan) and its efficacy were also assessed. After hospital discharge, the patients were carefully instructed to continue recording their headache by a self-administered headache questionnaire every hour until 11 h post-stimulation or until they had gone to bed. The questionnaire included headache characteristics and associated symptoms according to the ICHD-3 beta criteria (14) as well as adverse events, premonitory symptoms (unusual fatigue, yawning, neck stiffness, mood swings) and whether the reported headache mimicked the spontaneous migraine attacks.
Migraine-like attack criteria
We used the previously described definition for migraine-like attacks (Table 1) (15–17).
The following criteria defined a migraine-like attack.

Mean blood flow velocity of middle cerebral artery (VMCA)
VMCA was recorded bilaterally using hand-held 2-MHz probes and transcranial Doppler (TCD; Multidop X, DWL, Sipplingen, Germany). Marks were drawn on the skin to ensure accurate repositioning in each acquisition. Four recordings were sampled and averaged at each time point. One recording was a time-averaged mean over 4 seconds or approximately four cardiac cycles. We avoided fixed probes, which may cause discomfort and headache. Identification of the middle cerebral artery (MCA) and marking reproducible fix points were done as previously described (18,19). TCD recordings of all patients on both days were performed by the same skilled examiner (LE).
Diameter of superficial arteries
The diameter of the frontal branch of the STA on the headache side ipsilateral to the stimulation side was measured using high-resolution ultra-sonography, 20 MHz, bandwidth 15 MHz (Dermascan C, Hadsund, Denmark) (20,21). Marks were drawn on the skin and the coordinates of these were measured and recorded to ensure reproducibility of the measuring points from day to day. The same skilled examiner (WG) performed all the measurements.
Mechanical perception and pain thresholds
Mechanical perception and pain thresholds were measured on the ipsilateral cheek corresponding to the second branch of the trigeminal nerve (V2) at baseline, 15 min (during stimulation) and 45 min (after stimulation). All measurements were assessed by the same investigator (EKH) to avoid interpersonal bias.
The mechanical detection threshold was determined by using a set of 12 calibrated von Frey hairs (VFH; Marstock, Marburg, Germany; diameter 0.4–0.65 mm, force 0.25–512 mN). Beginning with a force of 16 mN, which each subject felt, the next lower hair was applied until the subject no longer felt the stimulus. Then the next higher hair was applied until the subject noticed the stimulus. Using the method of limits, five infra- and five suprathreshold values were obtained and the geometrical mean was taken as the individual mechanical detection threshold.
The mechanical pain threshold was tested with a series of seven standardized pinprick probes (diameter 0.2 mm, force 8–512 mN). The lowest bucking force (8 mN) was applied first and was followed by a filament with a higher bucking force until the subject signaled a pricking pain. According to the method of limits, five infra- and five suprathreshold values were obtained and the geometric mean was considered as the individual mechanical pain threshold.
Vital parameters
Heart rate (HR) and mean arterial blood pressure (MAP) were measured every 10 min using an auto-inflatable cuff and finger pulse oximeter (ProPac Encore®; Welch Allyn Protocol).
Statistical analysis
All values are presented as mean values ± 95% confidence interval (CI), except headache scores and mechanical perception and pain threshold measurements, which are presented as median values. Baseline was defined as T0 before stimulation start. Immediate phase is defined as 0–60 min and delayed phase as > 60 min–12 h after the beginning of stimulation.
Sample size was chosen on the basis of previous provocation studies performed by our group showing induction of migraine-like attacks in 65% of migraine patients after administration of a pharmacological trigger (16,22,23) compared to 10% after placebo. We assumed that LF compared to sham stimulation would have the same incidences of migraine-like attacks as our previous studies and estimated that inclusion of 12 subjects would be sufficient in a crossover study.
Primary endpoints were the difference in incidence of migraine-like attacks, non-migraine headache and the difference in area under the curve (AUC) for headache intensity scores (0–11 h) between LF stimulation and sham. The secondary endpoints were a) differences in AUC (0–40 min) for VMCA, STA diameter, HR and MAP between LF stimulation and sham, and b) differences in mechanical and pain thresholds before, during and after LF and sham stimulation.
Incidence of migraine-like attacks, non-migraine headache, associated symptoms, premonitory symptoms and adverse events were analyzed as binary categorical data with McNemar’s test. We calculated AUC according to the trapezium rule (24) to obtain a summary measure and to analyze the differences in response after LF and sham stimulation. Baseline was subtracted before calculating AUC to reduce variation between sessions within each subject. Analyses of AUC values were performed with a paired two-way t-test, except headache scores where data were tested with Wilcoxon signed rank test. Differences in mechanical detection and mechanical pain thresholds before (0 min), during (15 min) and after stimulation (45 min) for LF and sham were analyzed with Friedman test. If overall differences were found, post hoc Wilcoxon signed rank test was applied to characterize which time points differed from each other. We tested for period and carry-over effects for all baseline variables with Mann-Whitney test and independent t-test.
All analyses were performed with SPSS Statistics version 22 for Windows (Chicago, IL, USA) and GraphPad Prism 5 (San Diego, CA, USA) for the figures. p-value <0.05 was considered the level of significance.
Results
Twelve female migraine patients completed the study. The mean age was 54 (range 44–68 years) and mean weight was 69 kg (range 50–110 kg). The migraine attack frequency of the patients ranged between 2 and 25 per month at the timepoint of the examination. Eleven (92%) of the patients reported CAS, such as nasal congestion, rhinorrhea, lacrimation, conjunctival injection, eyelid edema and/or ptosis, during their usual migraine attacks. The clinical characteristics of all the patients are described in Table 2. The average time between implantation of the neurostimulator system and the first study day for the patients was 871 days (range: 418–1171 days).
Clinical characteristics of the 12 patients.
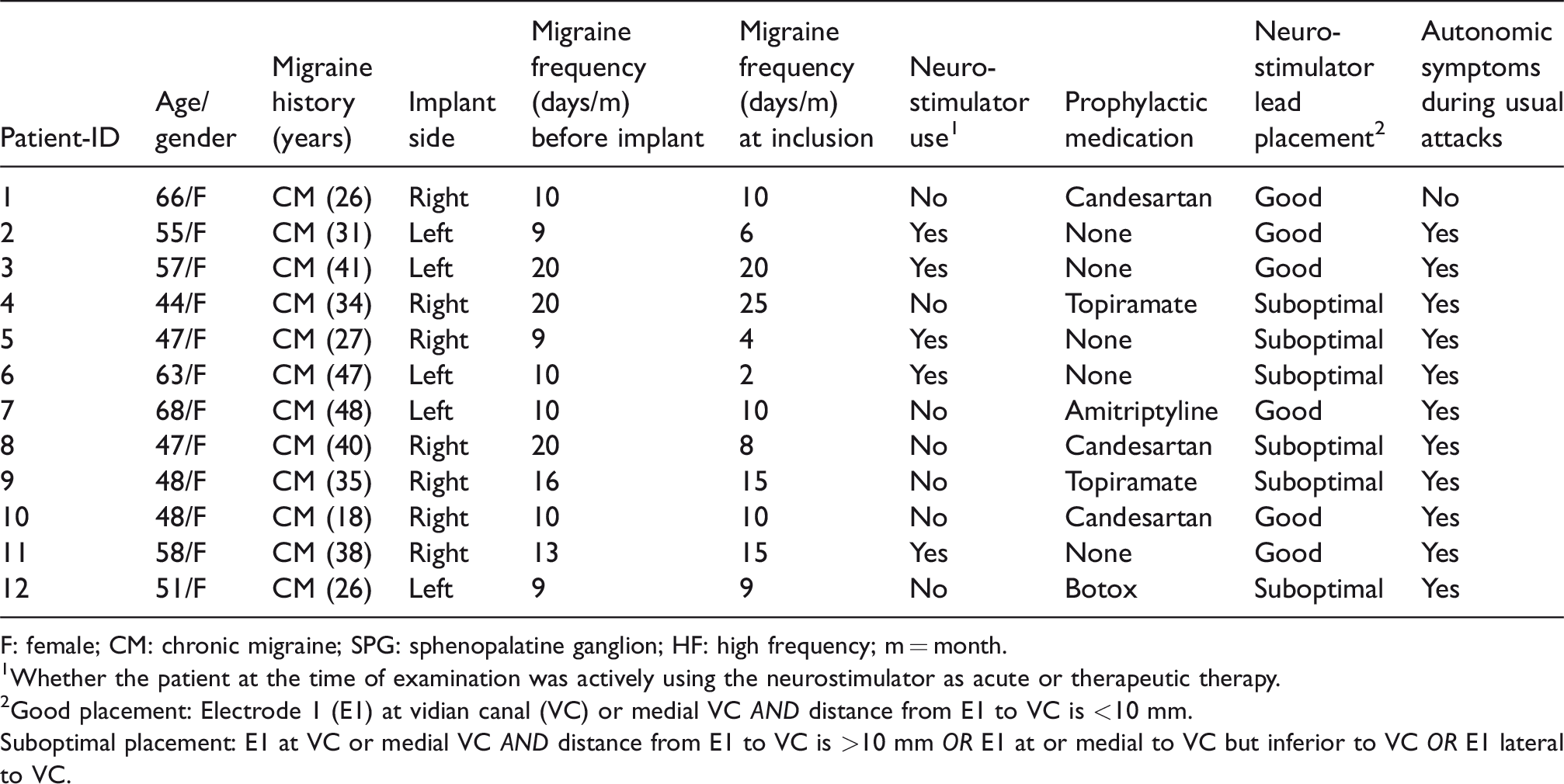
F: female; CM: chronic migraine; SPG: sphenopalatine ganglion; HF: high frequency; m = month.
1Whether the patient at the time of examination was actively using the neurostimulator as acute or therapeutic therapy.
2Good placement: Electrode 1 (E1) at vidian canal (VC) or medial VC AND distance from E1 to VC is <10 mm.
Suboptimal placement: E1 at VC or medial VC AND distance from E1 to VC is >10 mm OR E1 at or medial to VC but inferior to VC OR E1 lateral to VC.
Eight patients reported mild to moderate non-migraine headache, while four patients were headache free at baseline on both experimental days. The median headache intensity at baseline for patients with headache was one on both active and sham day (range 0–6). We could not measure VMCA using TCD in two patients on both study days due to technical issues. Therefore, TCD data for these patients were excluded from the VMCA AUC analysis. There was no carry-over or period effect for baseline values of headache intensity, mean VMCA, STA diameter, heart rate and mean arterial blood pressure.
Migraine-like attacks and headache
Five out of 12 (42%) patients reported a migraine-like attack after LF stimulation compared to six (50%) after sham (p = 1.000) (Table 3). Of these attacks, three were reported in the immediate phase (0–60 min) after LF stimulation and two after sham stimulation (p = 0.567). In the delayed phase (>1–12 h), two were reported after LF stimulation and four after sham stimulation (p = 0.567). The median time to onset of migraine-like attacks was 1 h (range 0.33–8 h) after LF and 3 h (range 0.16–10 h) after sham.
Headache characteristics in 12 migraine patients of their spontaneous migraine attacks, after 30 min of SPG stimulation using LF and sham (0–11 h observation period). Patients (n = 4) who developed no headache on both study days are marked with *.
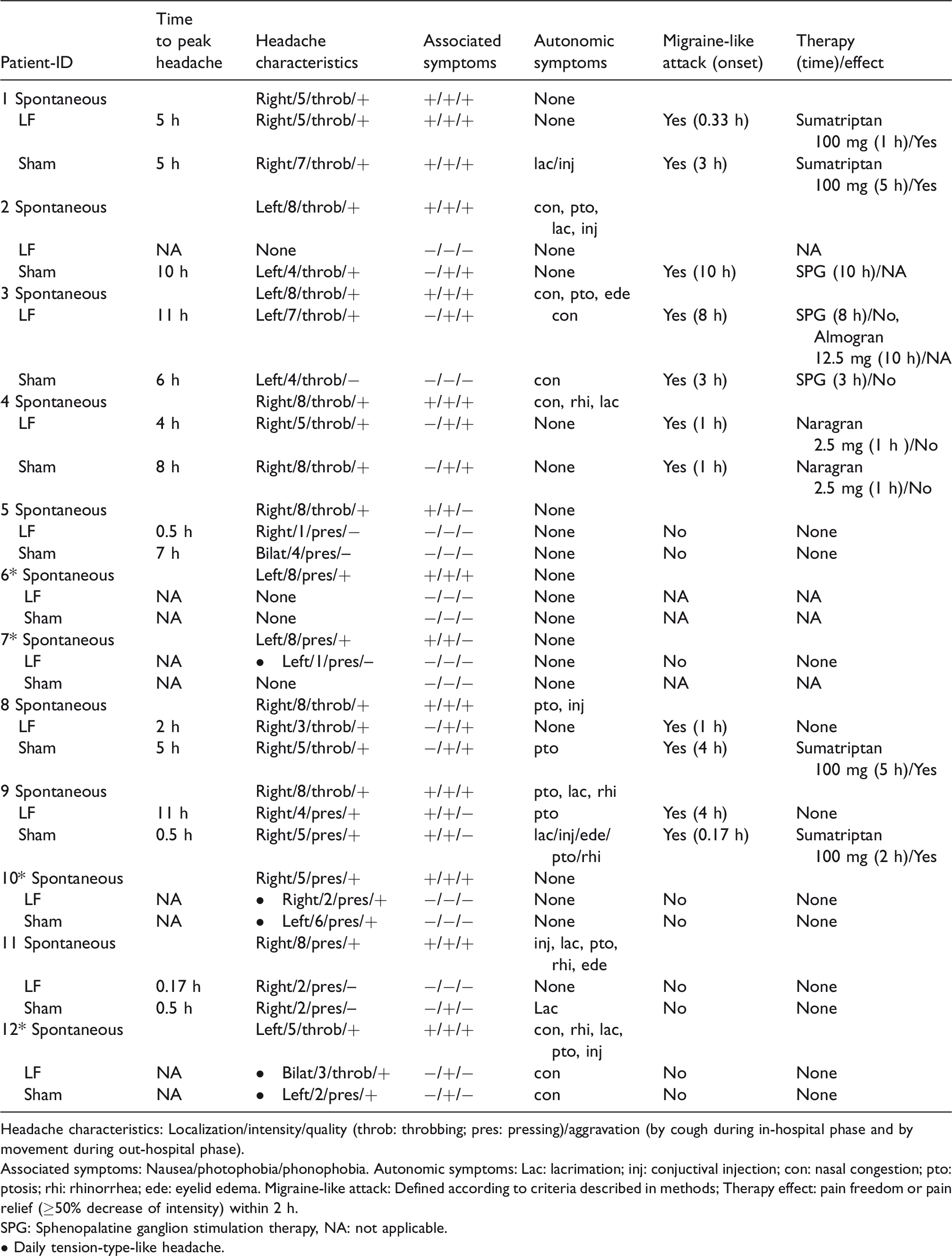
Headache characteristics: Localization/intensity/quality (throb: throbbing; pres: pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
Associated symptoms: Nausea/photophobia/phonophobia. Autonomic symptoms: Lac: lacrimation; inj: conjuctival injection; con: nasal congestion; pto: ptosis; rhi: rhinorrhea; ede: eyelid edema. Migraine-like attack: Defined according to criteria described in methods; Therapy effect: pain freedom or pain relief (≥50% decrease of intensity) within 2 h.
SPG: Sphenopalatine ganglion stimulation therapy, NA: not applicable.
• Daily tension-type-like headache.
On LF stimulation day, three out of 12 patients treated their attack with a triptan or SPG therapy and six out of 12 did so on sham day (p = 0.480). The median time to treatment was 1 h (range 1–8) after LF and 3 h (range 1–11) after sham. One attack after LF stimulation responded to triptan treatment, whereas three attacks after sham stimulation responded to triptan. SPG therapy was applied by the patients for one attack after LF stimulation and two attacks after sham stimulation but showed no therapeutic effect for any of these attacks.
We found no difference in the AUC0–11 h for headache intensity between LF and sham stimulation (p = 0.102) (Figure 1). The median peak headache intensity was three (range 1–7) after LF and four (range 2–8) after sham. The median time to peak headache occurred four hrs (0.5–11 h) after LF stimulation and five hrs (0.5–10 h) after sham.

Median (thick line) and individual (thin lines) headache intensity on a 0–10 VRS for 12 migraine patients after LF and sham stimulation. We found no difference between LF and sham stimulation in the immediate (0–1 h), delayed phase (1–11 h) or entire registration period (0–11 h). Median peak headache score after LF stimulation was 4 (range 0–7) and 5 (range 1–9) after sham stimulation.
Mean blood flow velocity of middle cerebral artery (VMCA)
During the in-hospital phase (0–40 min), we found no difference in AUC for VMCA between LF and sham stimulation on both the ipsilateral (p = 0.533) and contralateral side (p = 0.998) (Figure 2). In addition, there was no difference between the ipsilateral and contralateral side during LF stimulation (p = 0.730).
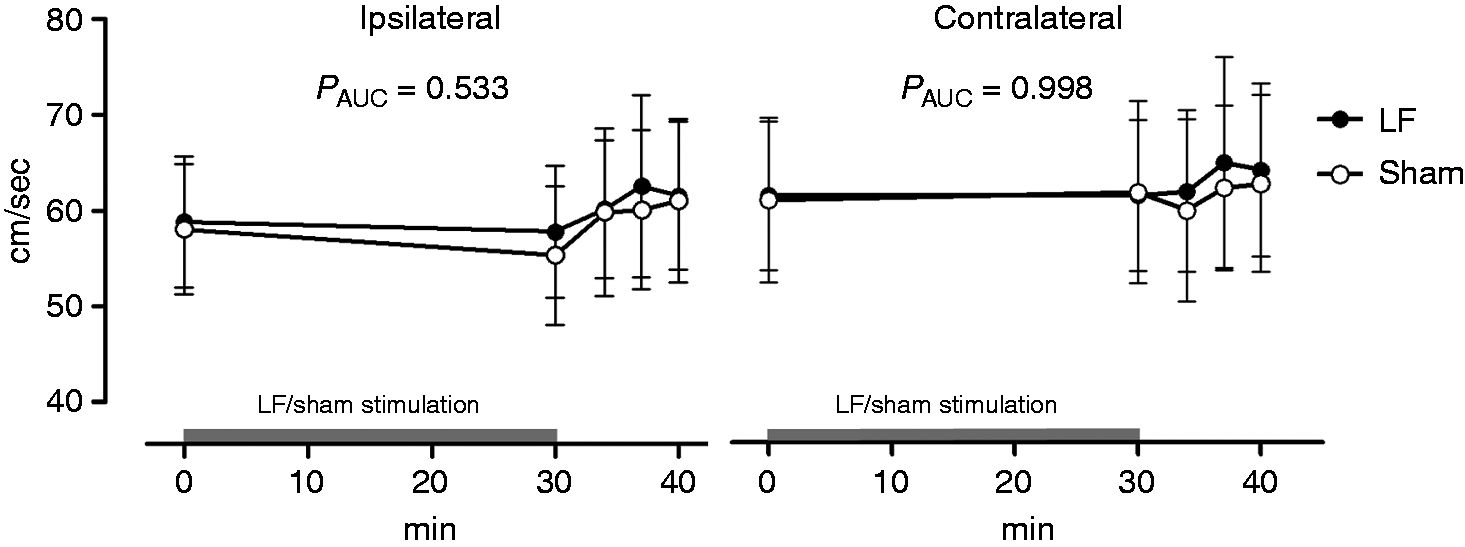
VMCA with 95% CI after 30 min of low frequency (LF) or sham stimulation on the ipsilateral and contralateral side. We found no difference in AUC 0-40 min for VMCA between LF and sham stimulation on the ipsilateral and the contralateral side.
Diameter of superficial temporal artery (STA)
We found no difference in diameter of STA between LF stimulation and sham during the in-hospital phase on the ipsilateral side (AUC0-40 min, p = 0.614).
Mechanical perception and pain thresholds
We found a significant increase in mechanical detection thresholds during LF stimulation compared to baseline (p = 0.007), which returned to normal 15 min after simulation (Figure 3). However, we found no changes in mechanical pain thresholds during LF stimulation. Mechanical detection and pain thresholds did not change during sham stimulation.

Box-and-whisker plot representing medians and quartiles of the mechanical detection thresholds (mN) measured by Von Frey hairs before (0 min), during (15 min) and after LF or sham stimulation (45 min) in 12 patients. We found a significant change in detection threshold for LF stimulation (Friedman’s test, p = 0.031) but not for sham (p = 0.098). Post hoc test showed a significant increase in detection thresholds during LF stimulation compared to baseline (Wilxocon signed rank test, p = 0.007).
Heart rate and mean arterial blood pressure
No difference in heart rate and mean arterial blood pressure was found between LF stimulation and sham during the in-hospital phase (HR AUC0–40 min, p = 0.351 and MAP AUC0–40 min, p = 0.419).
Autonomic symptoms and non-headache symptoms
Six out of 12 patients reported CAS after both LF stimulation and sham (p = 1.000) (Table 4). In the immediate phase, all six patients reported CAS (0–60 min) after LF stimulation and five patients after sham stimulation. An explorative sub-analysis of the six patients who reported CAS after LF stimulation showed no difference in headache response compared to sham day.
Cephalic autonomic symptoms (CAS) and non-headache symptoms reported by patients after LF and sham stimulation (0–11 h).

Data are shown as (n): number of patients.
*p-value: McNemar’s test.
Non-headache symptoms reported after LF such as unusual fatigue, neck stiffness, yawning, mood swing, poor concentration and hunger were quite few and not different from the sham day (Table 4).
Sub-analysis of patients who are using the SPG neurostimulator
Of the 12 patients, five patients were actively using the SPG therapy as acute or prophylactic therapy at the time of examination and claimed efficacy of the therapy. The seven patients who did not use the SPG neurostimulator may be due to lack of efficacy or because they prefer triptans as their treatment, since these were the most frequent causes of not using the SPG neurostimulator from the previous SPG trial (8–10). We did a sub-analysis of the five patients who were using the SPG therapy to see whether they had a clear response in headache intensity, VMCA, STA or mechanical perception and pain thresholds compared to patients that do not use the SPG therapy and sham stimulation. We found no associations between usage of SPG therapy and any of the parameters.
Sub-analysis of lead placement
Of the 12 patients, six had a good neurostimulator lead placement, six had a suboptimal placement, whereas none had bad placement (Table 2). All lead placements were assessed by CT scans. We did a sub-analysis to investigate whether patients with a good lead placement had a clear response in headache intensity, VMCA, STA or mechanical perception and pain thresholds compared to patients with suboptimal placement and sham stimulation. We found no associations between lead placement and any of the parameters.
Discussion
The major outcome of the present study is that LF stimulation of SPG did not induce migraine-like attacks or CAS in migraine patients. LF stimulation caused a significant increase in mechanical detection thresholds, but not in mechanical pain threshold. Additionally, we found no differences in VMCA, STA diameter, HR and MAP between LF stimulation and sham. None of the migraine patients in the present study reported pain from the maxillary distribution, which suggests that LF stimulation of SPG (similar to HF stimulation) with the applied amplitudes does not activate nociceptive fibers.
Involvement of the SPG in migraine pathophysiology is plausible given that a) parasympathetic efferent fibers of the SPG can modulate and activate meningeal and dural nociceptors (25–27); b) activation of SPG can lead to dural plasma protein extravasation (28) and neurogenic inflammation (29,30) causing dilation of intra- and extracranial arteries (20,31,32); and c) intranasal lidocaine, which is presumed to reach the SPG, is able to abort migraine attacks in migraine patients suggesting a role for the SPG in the development of migraine pain (33,34). Among migraine patients, CAS are common (1–4,35), but it is unknown whether parasympathetic activation triggers migraine headache or whether activation of trigeminal afferents leads to altered parasympathetic outflow.
The lack of CASs (markers for parasympathetic activation) after LF in migraine patients is in contrast to our previous findings in CH patients who reported CASs after LF 20 Hz stimulation of the SPG (11). Suboptimal lead placement definitely appears linked to stimulation and CAS response (36). However, difference in lead placement of the SPG neurostimulator is unlikely to explain the difference in our studies because implantation of the neurostimulator was performed by the same surgeon using the same technique with the same variance in sub-optimal lead placements. In support, we also found no association in sub-analysis between lead placement and any of the measured variables. Regarding the stimulation frequency, several animal studies have shown that LF stimulation of the SPG (10–20 Hz) for 1.5 to 45 min in dilation of intra- and extracranial arteries, increased cerebral blood flow (CBF), and plasma protein extravasation in the dura mater (28,37–41). Therefore, we expected LF stimulation of 20 Hz to induce migraine-like attacks with CAS, increase STA diameter and decrease VMCA (a surrogate measure for vasodilatation) (18) due to parasympathetic activation. It is possible that LF stimulation of SPG does not cause sufficient parasympathetic outflow to induce migraine-like attacks in migraineurs because they are less susceptible to the initiation of CAS than CH patients. Five of the migraine patients in the present study did not experience CAS during their spontaneous attacks and explorative sub-analysis of the six patients who reported CAS after LF stimulation showed no difference in headache response compared to sham day. Thus, migraine patients may only experience CAS at some of their most severe attacks, in contrast to CH where CAS is a much more prominent symptom.
Furthermore, since the patients were recruited from a terminated trial, we were not able to differentiate between patients who had long-term efficacy of the SPG therapy or not. A sub-analysis of the five patients who were actively using the SPG therapy and had self-reported efficacy did not reveal that LF stimulation had a stronger physiologic effect on them compared to sham.
Interestingly, we observed in migraine patients an increase in mechanical detection threshold, but not in mechanical pain threshold, during LF stimulation, suggesting a selective depression of sensory Aβ-fiber function but not C-fiber function. This may be possible because different fiber types need different electric field strengths to be activated (42,43), but it may also suggest different neurophysiologic mechanisms underlying migraine and CH. Notably, the mean increase in mechanical detection threshold of 0.41 mN caused by LF stimulation is similar to the response seen during transcutaneous electrical nerve stimulation (TENS). It is used as preventive therapy for migraine (44) and had shown to cause an increase in mechanical detection threshold of 0.45 mN when comparing pre-stimulation with during-stimulation on the thenar eminence (45). TENS is employed in two forms: High frequency (>50 Hz) with low intensity or low frequency (<10 Hz) with high intensity (44). Thus, LF stimulation of the SPG and TENS may possibly share some similar mechanisms. However, in contrast to LF SPG stimulation, TENS also increases mechanical pain threshold (46).
We acknowledge the limitation that blinding might be compromised since the patients could feel a difference between the two stimulations applied, as sham does not cause any sensation. However, the patients were not told that one of the stimulations was sham, but only that we would stimulate using frequencies that differed from what they were used to. Another limitation could be that chronic migraine patients may develop many spontaneous attacks, which could blur the results. Notably, we had an unusual high sham provocation of migraine-like attacks (50%) compared to the normal placebo reaction in experimental provocation studies, which lies between 0–30% (15,47–49). It is possible that the high frequency of migraine attacks in our severely affected patients is responsible for the high sham response.
Conclusion
Even though neurostimulation of SPG using HF has become accepted as a promising therapy system for chronic CH, it is still unclear how this stimulation physiologically exerts its effects. The present study involving a unique group of migraine patients with SPG implants gave us an excellent opportunity to investigate the physiological effects of LF stimulation of the SPG and activation of the parasympathetic nervous system. We found that LF stimulation of SPG in 12 migraine patients did not induce migraine-like attacks, CAS or changes in vascular responses. Our findings suggest that LF stimulation of SPG does not cause sufficient parasympathetic outflow to induce migraine-like attacks, CAS or changes in cerebral hemodynamics in migraine patients. The physiological mechanisms and role of SPG stimulation in migraine therefore remain to be elucidated.
Clinical implications
LF stimulation of the SPG does not induce migraine-like attacks or autonomic symptoms in migraine patients. LF stimulation cause a significant increase in mechanical detection thresholds, but not in mechanical pain threshold. Our results suggest that parasympathetic outflow activated by the SPG neurostimulator is not sufficient to induce migraine-like attacks.
Footnotes
Acknowledgements
The authors thank all the patients who participated in this study, lab technicians Winnie Grønning Nielsen and Lene Elkjær for their excellent and dedicated assistance and to Professor Jes Olesen for valuable input to the paper. Further thanks to funding from Novo Nordisk Foundation (NNF11OC1014333), Independent Research-Medical Sciences (FSS) (DFF-1331-00210A), Lundbeck Foundation (R155-2014-171), FP7-EUROHEADPAIN (602633) and Autonomic Technologies, Inc.
Author contributions
SG: Study concept and design, acquisition of data, analysis (including statistical analyses) and interpretation, drafting the manuscript. KF: Acquisition of data and critical revision of the manuscript for important intellectual content. HWS: Study concept and design, critical revision of the manuscript for important intellectual content. AC: Study concept and design, critical revision of the manuscript for important intellectual content. RHJ: Study concept and design, interpretation of study result, supervision and critical revision of the manuscript. MA: Study concept and design, interpretation of study result, critical revision of the manuscript for important intellectual content and supervision.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RHJ is an advisory board member for Autonomic Technologies Inc., Allergan, Electrocore and Medotech, and has received honoraria and lectured for MSD, ATI, Pfizer, Allergan, Berlin-Chemie, and Norpharma. MA is a consultant and/or scientific adviser/speaker for Allergan, Alder, Amgen, ATI and Eli Lilly. SG has received travel grant from the ATI. HWS is a consultant and/or scientific adviser/speaker for Novartis, TEVA, BalancAir and Eli Lilly. AC was previously employed by ATI as research/chief scientist. KF has nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Autonomic Technologies Inc.
