Abstract
Background
Medication overuse headache may be associated with widespread alterations along the thalamocortical pathway, a pathway involved in pain perception and disease progression. This study addressed whether brain metabolites in key regions of the thalamocortical pathway differed between chronic migraine patients with medication overuse headache and without medication overuse headache.
Methods
Magnetic resonance spectroscopic imaging was used to map metabolites in the bilateral anterior cingulate cortices, mid cingulate cortices, posterior cingulate cortices, and the thalami. Sixteen patients with medication overuse headache were compared with 16 matched patients without medication overuse headache and 16 matched healthy controls.
Results
Glutamate and glutamine in the right mid cingulate cortex and myo-inositol in the left anterior cingulate cortex were significantly higher in patients with medication overuse headache than patients without medication overuse headache, but similar to healthy controls. Both patient groups exhibited reduced N-acetyl-aspartate and creatine in the thalamus, reduced myo-inositol in the right anterior cingulate cortex, and elevated choline in the right mid cingulate cortex. Finally, a negative association between myo-inositol laterality index in the anterior cingulate cortices and number of days per month with acute medication use was found across all patients.
Conclusions
Patients with medication overuse headache were characterized by a distinct concentration profile of myo-inositol, a glial marker, in the anterior cingulate cortices that may have arisen from medication overuse and could contribute to the development of medication overuse headache.
Keywords
Introduction
Headaches are often treated with acute abortive medication such as simple or complex analgesics, non-steroidal anti-inflammatory drugs (NSAIDs), ergotamine, or triptans. However, excessive intake may lead to worsening of pre-existing primary headaches causing the transformation, for example, from episodic migraine (EM) to chronic migraine (CM). About 1–2% of the general population and up to 50% of patients with chronic daily headaches suffer from medication overuse headache (MOH) (1,2). As medication overuse is one of the leading risk factors for headache chronification (3), it is of great importance to understand how acute medications interact with central headache generating pathways. Evidence suggests that both MOH and CM are associated with widespread functional and structural alterations along the thalamocortical pathway, a pathway involved in pain perception, central sensitization and disease progression (4–8). Nonetheless, the mechanisms by which frequent medication exposure leads to chronic headache remain largely unknown.
Magnetic resonance spectroscopic imaging (MRSI) is a non-invasive imaging technique that allows quantification of several different brain metabolites, with a diverse range of functions, from multiple regions in a relatively short time. In a previous MRSI study of ours, N-acetyl-aspartate (NAA), an index of neuronal integrity, was found to be reduced in the bilateral thalami and the right anterior cingulate cortex (ACC) in CM without MOH, but not in EM (9). Although these results provide further evidence for thalamocortical dysfunction in CM, they are likely non-specific to CM as, for example, thalamic NAA reductions have been found in other chronic pain conditions (10,11). Hence, similar findings may likely also be observed in CM patients with MOH. The additional diagnosis of MOH may, however, also be associated with distinct neurochemical alterations along the thalamocortical pathway. Such alterations may result as a direct consequence of the medication or as an interaction between the medication and headache-generating mechanisms.
To address these issues, we employed MRSI of the bilateral medial walls of the brain in CM patients with and without MOH and in matched healthy controls. By controlling for the underlying primary headache disorder; that is, CM in this study, it becomes possible to examine whether central changes can be ascribed to MOH. A region-of-interest analysis was performed in the bilateral thalami, ACC, mid cingulate cortices (MCC), and posterior cingulate cortices/precuneus (PCC). These regions have previously been implicated in trigeminal pain processing and in the pathophysiology of CM with and without MOH (7,9,12–15). Based on the above considerations, we hypothesized that CM patients with MOH would exhibit similar NAA changes as previously observed in CM patients without MOH; that is, in the bilateral thalami and in the right ACC. An explorative analysis was further performed for all brain metabolites in all of the regions of interest to examine potential differences between the two patient groups.
Materials and methods
Study population
CM patients with and without medication overuse were recruited from the Headache Clinic of Taipei Veterans General Hospital. Both diagnoses were made according to the International Classification of Headache Disorders 3rd edition (ICHD-3) (16). Medication overuse was defined as the intake of ergotamine, triptans, or combination analgesics on ≥10 days/month or simple analgesics on ≥15 days/month for ≥3 months. All patients enrolled in the study fulfilled the following additional criteria: (a) Having had ≥15 days/month of headache in the preceding three months; (b) ≥8 of these days had to be with migrainous headache; that is, moderate-to-severe headache with one or more of the following features: Unilateral pain or pain worse on one side of the head, pulsatile nature, photophobia and phonophobia, nausea or vomiting, or pain made worse by physical activity or responsive to ergotamine or triptans. (c) Patients utilized the same types and amount of acute medication that they had used prior to enrollment throughout the study period; (d) refrained from taking any acute medications 24 hours prior to scanning; (e) had not previously received migraine prophylaxis, and (f) only had migraine without aura. Healthy controls without any history of migraine or other primary headaches were recruited. Infrequent tension-type headache (<1 headache day/month) was, however, allowed. Apart from the migraine diagnosis in the patient groups, both patients and healthy controls were normal in physical and neurological examinations and did not have any systemic diseases, psychiatric disorders or conditions incompatible with MRI. Recruitment and scanning took place between March 2014 and July 2016 after approval of the study by the Institutional Review Board of Taipei Veterans General Hospital (VGH IRB: 2012-04-046B). All participants gave written, informed consent in accordance with the Helsinki Declaration (6th revision).
Imaging protocol
Scanning was performed between 9am to 6pm for all participants. Images were acquired using a 32-channel head coil on a 3T MR scanner (Trio, Siemens Medical Solutions, Erlangen, Germany). To diminish motion artifacts during scanning, the participants’ heads were immobilized with cushions inside the coil. A high-resolution 3D Magnetization Prepared Rapid Acquisition Gradient Echo anatomical scan (repetition time/echo time/flip angle: 2530 ms/3.03 ms/7 degrees; field of view: 224 × 256 × 192; voxel size: 1 × 1 × 1 mm3) was initially acquired for localization of the left and right medial walls for subsequent positioning of MRSI slices. Accelerated MRSI was implemented using a proton echo-planar spectroscopic imaging sequence (17–19). Two 14-mm thick sagittal MRSI slices covering the medial walls in each hemisphere were acquired separately with an in-plane resolution of 8 × 8 mm2, using the following parameters: repetition time =1.5 sec, echo time = 30 ms, 32 × 32 spatial matrix, field of view = 256 × 256 mm2, number of excitations per slice = 16. The acquisition order of left and right slices was balanced across participants. A non-water suppressed MRSI scan was acquired using a single average for automatic phase correction and calibration of metabolite concentrations. For each hemisphere, eight slices of outer-volume lipid suppression were applied along the perimeter of the brain to suppress the lipid signal. Shimming and adjustment for water suppression was carried out automatically. Following each MRSI scan, multi-slice sagittal T1-weighted images were collected using a gradient echo sequence (repetition time/echo time/flip angle: 250 ms/2.61 ms/70 degrees; field of view: 256 × 256; spatial matrix: 256 × 256; slice thickness: 2 mm). Seven slices were collected in each hemisphere yielding 14-mm volumes covering the same spatial locations as the two MRSI scans. These T1 images were used as anatomical references in the following analysis.
Data processing
A detailed description of the data processing procedures can be found in Niddam et al. (9). In short, spectra were quantified with the LCModel software package (20). Metabolic signals were calibrated to the non-water suppressed data using the water scaling method (21) and absolute concentrations were obtained by further adjusting for partial volume and tissue relaxation effects (22). Voxels with a cerebrospinal fluid tissue probability larger than 0.3 were discarded. Finally, adjusted concentration maps were generated for the following five metabolites: Total choline (tCho; including glycerophosphocholine and phosphocholine), total creatine (tCr; including creatine and phosphocreatine), Glx (glutamate and glutamine added together), myo-inositol (mI), and total NAA (tNAA; including N-acetyl-aspartate and N-acetylaspartyl-glutamate). Concentration values in voxels with unsatisfactory LCModel fits (Cramer–Rao Lower Bound >30%) were discarded. A cut-off value of 30% allows for inclusion of the majority of the data and avoid potential biased concentration estimates at group level while maintaining a reasonable quality of fitting (23). Left and right individual T1 images were warped into the corresponding template images chosen from a representative participant (24). The corrected metabolite images were then warped into the template space using the T1 warp parameters.
Statistical analysis
For each participant, mean metabolite concentrations were extracted from regions of interests placed bilaterally in the thalami and in the dorsal/rostral ACC, MCC and PCC/precuneus (Figure 1) (see Supplemental material for the anatomical regions covered by each region). For all regions of interest, differences in metabolite concentrations between the three subject groups were examined by analysis of covariance using age, gender and headache status on the scan day (migrainous or non-migrainous grouped together) as covariates. Results passing a corrected threshold of p < 0.05, controlling for the false discovery rate (40 tests, i.e. five metabolites and eight regions), were considered significant. Pair-wise post-hoc analyses among the three groups were performed using Bonferroni tests. Associations between metabolite concentrations and clinical parameters (maximum headache severity, duration of migraine history, duration of chronic migraine history, headache and migraine frequencies, and number of days per month with acute medication use for headache) were examined for all regions exhibiting significant between-group differences using Pearson correlation coefficients. Prior to statistical analysis, all data series were checked for outliers using Grubb’s test. All statistical tests were two-tailed and were performed using IBM SPSS (version 21.0).
Regions of interest in the left and right medial walls. The eight regions covered the anterior cingulate (red), mid cingulate (green), posterior cingulate (blue), and the thalamus (yellow) in both hemispheres. Please refer to Supplemental Materials for information on how these regions were defined and which anatomical sub-regions they covered.
Results
Demographics and clinical profiles
Demographics of healthy controls and patients (mean ± SD).
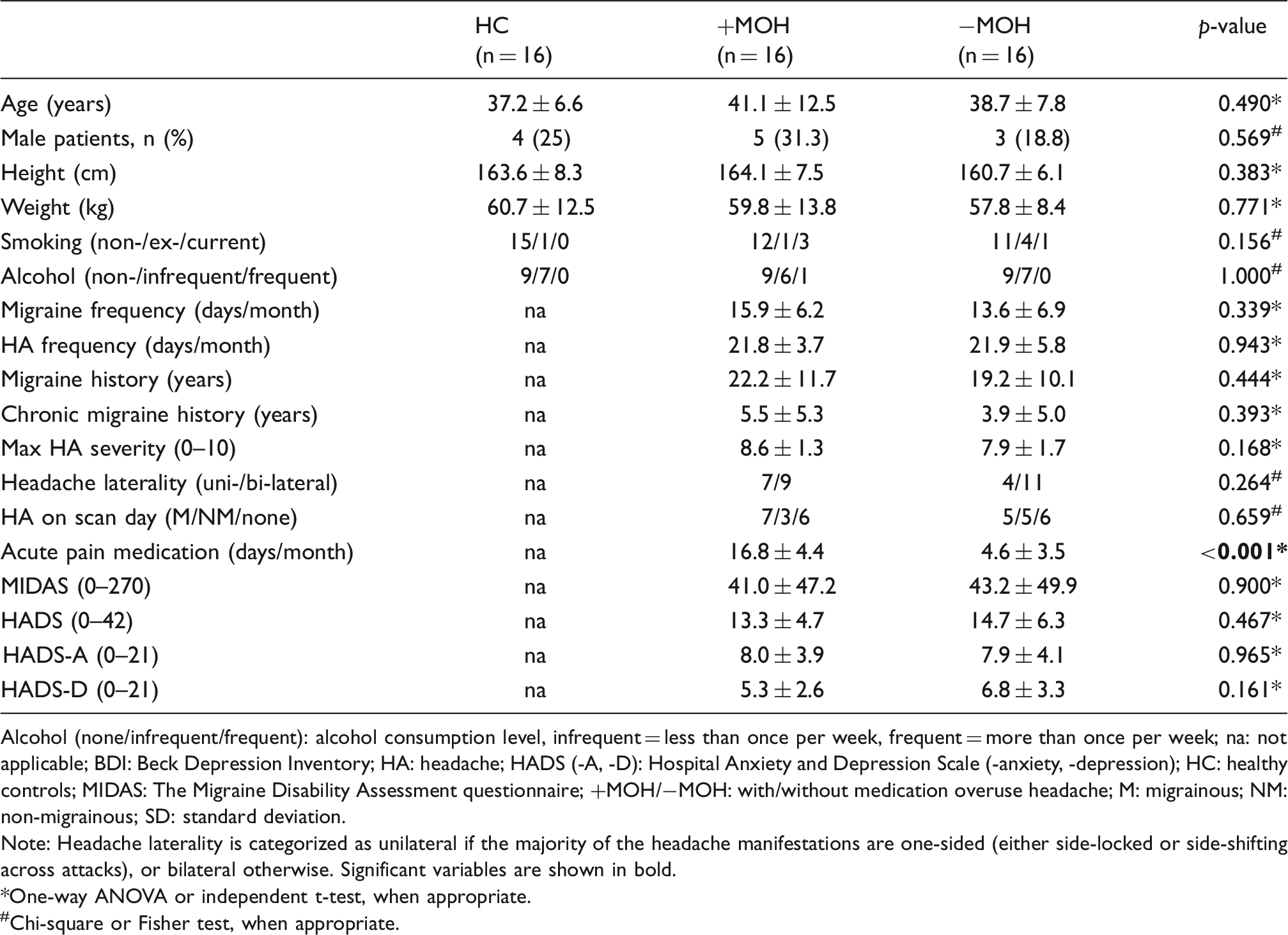
Alcohol (none/infrequent/frequent): alcohol consumption level, infrequent = less than once per week, frequent = more than once per week; na: not applicable; BDI: Beck Depression Inventory; HA: headache; HADS (-A, -D): Hospital Anxiety and Depression Scale (-anxiety, -depression); HC: healthy controls; MIDAS: The Migraine Disability Assessment questionnaire; +MOH/−MOH: with/without medication overuse headache; M: migrainous; NM: non-migrainous; SD: standard deviation.
Note: Headache laterality is categorized as unilateral if the majority of the headache manifestations are one-sided (either side-locked or side-shifting across attacks), or bilateral otherwise. Significant variables are shown in bold.
One-way ANOVA or independent t-test, when appropriate.
#Chi-square or Fisher test, when appropriate.
MRSI findings
Spectra with overlaid LCModel fit from voxels at four locations in each hemisphere are shown in Figure 2 for a representative subject. Metabolite peaks were well defined in all of the selected regions. The spatial distributions of the mean concentrations of the five metabolites were similar across the three groups and in the two hemispheres and resembled those from our previous reports (9,25) (Figure 3). Mean metabolite concentrations were extracted from the four regions of interest in each hemisphere (Figure 1). The corresponding estimated mean standard deviations (Cramer–Rao Lower Bound) are provided in Supplemental Table 2. Significant differences in tNAA concentrations were found in the left and right thalamus (left: F = 7.246, padjusted = 0.005; right: F = 6.447, padjusted = 0.008) and for tCr in the left thalamus (F = 8.016, padjusted = 0.003). Post-hoc analyses revealed significantly lower concentrations in both patient groups compared with healthy controls (Figure 4; left tCr: HC vs. +MOH, p = 0.005; HC vs. −MOH, p = 0.001; left tNAA: HC vs. +MOH, p = 0.008; HC vs. −MOH, p = 0.002; right tNAA: HC vs. +MOH, p = 0.011; HC vs. −MOH, p = 0.004) but no significant differences between the two patient groups (p = 0.999 for all). Significant correlations with clinical parameters were not found for the two metabolites in the thalamic regions of interest.
Spectra (black) with overlaid LCModel fit (red) from voxels at four locations in left and right hemispheres in a representative subject. Voxel locations are marked on the individual anatomical images (in native space) and corresponds to anterior (ant.), middle (mid.), posterior (post.), and inferior (inf.) portions of the medial wall. Metabolite peaks are labeled in the upper right spectrum and ppm denotes parts per million. Interpolated mean concentration maps for the five metabolites (tCho, tCr, Glx, mI, tNAA) in healthy controls (HC), chronic migraine patients without medication overuse headache (−MOH), and chronic migraine patients with medication overuse headache (+MOH). The color-coded maps refer to the absolute concentration levels and are given in mM. Red indicates high concentrations and blue/violet indicates low concentrations. Absolute tNAA and tCr concentrations in the thalamus in the three subject groups. (a) tNAA concentrations were significantly reduced in the both the left and right thalami in chronic migraine patients with (+MOH) and without (−MOH) medication overuse headache relative to healthy controls (HC). (b) tCr concentrations were also significantly reduced in the two patient groups but only in the left thalamus. Error bars indicate standard deviations. Summary of the significant changes in metabolite concentrations in the two patient groups relative to healthy controls. ACC: anterior cingulate cortex; HC: healthy controls; MCC: mid cingulate cortex; +MOH: chronic migraine with medication overuse headache; −MOH: chronic migraine without medication overuse headache; PCC: posterior cingulate cortex; Thal: thalamus. ↓ denotes decreased concentration relative to healthy controls; ↑ denotes increased concentration relative to healthy controls; * denotes −MOH differed from +MOH in the same direction as the arrow.



In the MCC, significant differences were found for tCho (F = 5.230, padjusted = 0.048) and Glx (F = 6.903, padjusted = 0.006) in the right hemisphere. For tCho (Figure 5(a)), post-hoc analysis revealed an increase in CM patients with (p = 0.008) and without (p = 0.049) MOH relative to healthy controls. A significant difference was not found between the two patient groups (p = 0.780). Furthermore, tCho concentrations correlated positively with the duration of CM history across the entire patient population (Figure 5(b); n = 31, r = 0.497, p = 0.005). For Glx (Figure 5(c)), post-hoc analysis revealed a decrease in CM patients without MOH relative to healthy controls (p = 0.020) and CM patients with MOH (p = 0.005). No difference was found between CM patients with MOH and healthy controls (p = 0.999). A significant positive correlation was found across the entire patient population between Glx levels and the number of days per month with acute medication use (Figure 5(d); n = 32, r = 0.402, p = 0.023).
Metabolites in the right mid cingulate cortex (MCC). (a) Both patient groups exhibited elevated tCho concentrations in the right MCC. (b) tCho concentrations were positively correlated with chronic migraine (Mx) history across the entire patient population (n = 31, r = 0.497, p = 0.005). (c) Glx concentrations were significantly reduced in chronic migraine patients without medication overuse headache (−MOH) relative to chronic migraine patients with medication overuse headache (+MOH) and healthy controls (HC). (d) Glx concentrations in the right MCC correlated positively with the number of days per month with acute medication use across the entire patient population (n = 32, r = 0.402, p = 0.023).
Finally, significant differences were found for mI in the left and right ACC (Figure 6(a) and (b); Left: F = 8.806, padjusted = 0.004; Right: F = 9.102, padjusted =0.001). For both regions, post-hoc analyses revealed an mI decrease in CM patients without MOH relative to healthy controls (Left: p = 0.001; Right: p = 0.001). Comparing MOH patients with healthy controls, a significant mI reduction was found in the right ACC for MOH patients (p = 0.004) while no difference was found in the left ACC (p = 0.278). Comparing the two patient groups, mI was significantly lower in patients without MOH than in patients with MOH in the left ACC (p = 0.028) but not in the right ACC (p = 0.999). To further investigate the hemispheric asymmetry in mI levels, the laterality index ([Right-Left]/[Right + Left]) was calculated (Figure 6(c)). An ANCOVA with age, gender and headache status as covariates resulted in a significant difference (F = 4.797, p = 0.014) and post-hoc analysis further revealed a significant difference between the two patient groups (p = 0.011) but not between healthy controls and any of the two patient groups ( + MOH: p = 0.964; −MOH: p = 0.364). In addition, a negative association was found across the entire patient population between the laterality index and the number of days per month with acute medication use (n = 29, r = −0.419, p = 0.024) (Figure 6(d)). Because an association between mI in the ACC and depression scores was reported in a previous study (9), a correlation analysis was also performed for mI in the two ACC regions and the depression sub-scores from the Hospital Anxiety and Depression Scale. However, significant associations were neither found across the entire patient population (left ACC: n = 30, r = −0.067, p = 0.812; right ACC: n = 30, r = −0.218, p = 0.434) nor within each patient group (left ACC: +MOH: n = 15, r = 0.500, p = 0.058; −MOH: n = 15, r = −0.385, p = 0.157; right ACC: +MOH: n = 15, r = 0.374, p = 0.170; −MOH: n = 15, r = −0.190, p = 0.498). However, comparing the correlation coefficients from the two patient groups resulted in a significant difference for the left (z = 2.34, p = 0.019) but not the right ACC (z = 1.43, p = 0.153). Significant between-group differences were not found for any of the metabolites in the PCC. None of the metabolites in any of the regions were found to correlate with mean headache severity, duration of migraine history, or headache or migraine frequencies. A summary of the between-group results is provided in Table 2. Finally, to examine the effect of headache status on the scan day, two groups were generated from the pooled patient cohort (Supplemental Table 1) and ANCOVAs were performed, with age, gender and headache frequency as covariates, for all of the above metabolites and loci exhibiting significant differences. This analysis, however, did not result in any significant findings.
mI concentrations in the left and right anterior cingulate cortex in the three subject groups. (a) In the left anterior cingulate, mI concentrations were significantly reduced in chronic migraine patients without medication overuse headache (−MOH) relative to chronic migraine patients with medication overuse headache (+MOH) and healthy controls (HC). (b) In the right anterior cingulate, mI concentrations were significantly reduced in −MOH and +MOH relative to healthy controls (HC). (c) The laterality index for mI, extracted from the left and right ACC ([Right-Left]/[Right + Left]), was significantly reduced in patients with MOH relative to patients without MOH. (d) The laterality index correlated negatively with the number of days per month with acute medication use across the entire patient population (n = 29, r = −0.419, p = 0.024).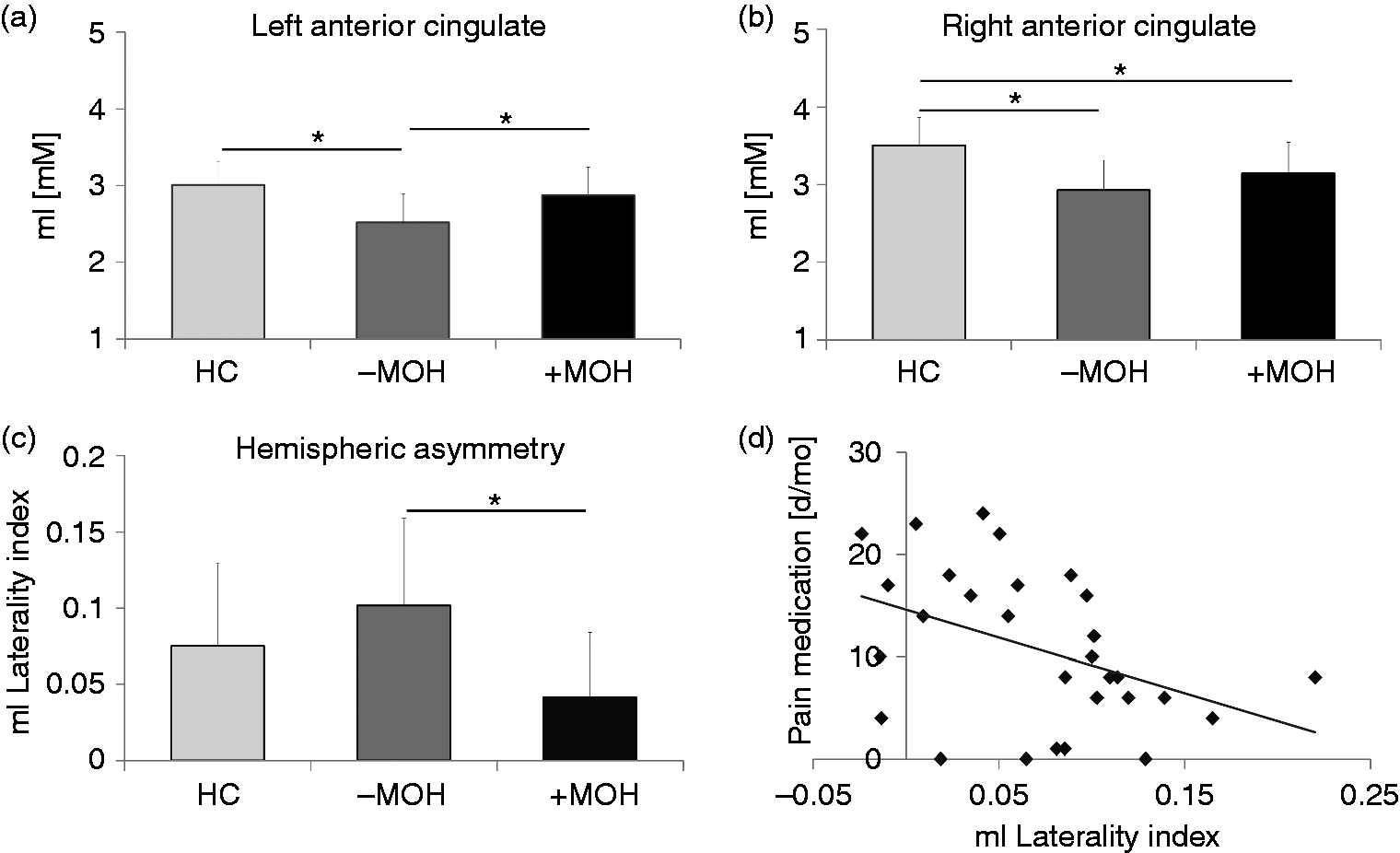
Discussion
This study addressed whether CM patients with MOH exhibited similar brain metabolic changes in the thalamocortical pathway as previously observed in CM patients without MOH (9) and whether additional medication-overuse related changes could be observed along this pathway. We showed, in accord with our hypothesis, that CM patients with and without MOH had reduced tNAA in the bilateral thalami. We further showed that mI in the left ACC and Glx in the right MCC significantly differed between the two patient groups but not between MOH patients and healthy controls. Finally, we showed that additional commonalities exist between CM patients with and without MOH for tCr in the left thalamus, for tCho in the right MCC, and for mI in the right ACC. The significance of these findings and their relationship with clinical parameters are discussed below.
Thalamic tNAA levels
We reproduced our previous finding of reduced tNAA concentrations in the bilateral thalami in CM patients without MOH (9). We furthermore extended this finding to CM patients with MOH. This result is in accord with our hypothesis of thalamic tNAA reductions being related to chronic pain in general as such a reduction has also been found in other chronic pain conditions (10,11). The bilateral reduction suggests the presence of compromised neuronal health of the thalami (26) and is congruent with the mainly bilateral or side-shifting headache distributions in both patient groups. Although the thalamus has been implicated in pain processing and disease progression (4–8), and is activated during migraine attacks (27), it is not possible to conclude whether the observed changes in the thalami are a consequence of the headaches or contributes directly to generating or maintaining the headaches; for example, through central sensitization. Moreover, as our region of interest covered a large area of the thalamus (see Supplemental materials), the observed reduction in tNAA cannot be assigned to any specific nuclei. Nevertheless, our results suggest that medication overuse results in similar neuronal dysfunction in the thalamus as observed in other chronic pain conditions.
mI levels in the ACC
As in our previous study (9), CM patients without MOH had reduced mI in the left ACC. This reduction was, however, not found in CM patients with MOH. Additionally, both patient groups had reduced mI in the right ACC. The lower mI concentrations may represent glial dysfunction, as mI is mainly found in glial cells (26). Such dysfunction could have ramifications for neurotransmitter clearance from the synaptic cleft, synaptic plasticity, or synaptogenesis, among others. Since the ACC is engaged during spontaneous migraine attacks (27), it is possible that lower mI levels, or glial dysfunction, could arise from the repetitive attacks and perhaps contribute to headache chronification (28). In contrast, the normal levels of mI in the left ACC in MOH patients suggest the absence of glial dysfunction in this region. A potential explanation for this is provided below. It is noteworthy that the constellation of higher mI in the left ACC and lower mI in the right ACC distinctly characterized MOH patients. This finding could have clinical implications, if confirmed in a larger patient cohort, as it may serve as a potential marker of medication overuse.
mI levels within the ACC showed a right hemisphere preponderance (positive laterality index) in all subject groups, albeit with a significantly reduced asymmetry in patients with MOH relative to patients without MOH. Hemispheric lateralization in the ACC has previously been observed functionally, structurally and neurochemically and may be related to processes involved in physiological arousal and attention reorientation (9,29–31). The leftward hemispheric shift in MOH patients may be linked to the role of the ACC in the generation of the subjective affective experience. According to the prevailing model of emotion processing, the left frontal brain system is mainly involved with behavioral activation and responds to appetitive and rewarding stimuli (32). It has furthermore been implicated in substance abuse (32,33). Acute abortive medication may thus act as a rewarding stimulus, perhaps through psychotropic properties, which predominantly targets the left frontal brain system and at the same time alters the interhemispheric balance. The resulting effect could potentially counter the effect of reduced mI observed in CM in the left ACC and perhaps contribute to the development of MOH. This notion is further supported by the association between hemispheric laterality and the number of days with acute medication use as well as the widespread changes observed in the reward system of the brain in MOH (34).
Glx levels in the MCC
Glx, glutamate (Glu) and glutamine (Gln), quantified together, were reduced in the right MCC in CM patients without MOH but not in CM patients with MOH. Furthermore, Glx concentrations were positively correlated with the number of days per month with acute medication use across the entire patient population. Glu is the most abundant excitatory neurotransmitter in the brain and Gln is a precursor of Glu (26). The Glu–Gln cycle is considered a marker for regulation of synaptic activity and a decrease in Glx, as observed in patients without MOH, may not only represent reduced excitatory synaptic activity but also a general downregulation of the Glu–Gln cycle. An alternative explanation could be that Glu is diverted away from the energy-demanding Glu-Gln cycle towards energy producing oxidative metabolism (35). Such a shift would occur during high energy demand, as observed during repetitive neuronal depolarization, and is contingent upon high Glu levels in the synaptic cleft. It is, however, unclear if such a shift can be sustained over a longer period of time and how this would affect the overall Glx concentration. Nevertheless, decreased Glx suggests the presence of altered synaptic regulation perhaps as a compensatory mechanism in response to neuronal stress resulting from ongoing nociceptive input. In support of this, the MCC is involved in trigeminal nociceptive transmission (14) and contributes to cognitive and affective aspects of pain processing (36). Furthermore, functional and structural alterations in the MCC have been found in migraine patients (37). It is noteworthy that other MRS studies in migraine found increased Glu or Glx in the occipital cortex or decreased Glu in the cerebellum (38). Together with the current study, this suggests that the direction of Glu or Glx change may depend on the brain region.
The absence of a difference between patients with MOH and healthy controls and the concomitant positive association between Glx and number of days per month with acute medication use suggest the medication to reverse or prevent the glutamatergic dysfunction observed in CM patients without MOH. Interestingly, a single NSAID dose alters the cerebral blood flow in the MCC (36,39). Moreover, NSAIDs exert a neuroprotective effect and have been shown to specifically protect against glutamate decreases in animal models of Alzheimer’s disease (40). Overuse of NSAIDs also appears to reduce the high incidence of white matter lesions observed in CM without MOH (41). Taken together, the effect of medication on Glx levels may not directly contribute to MOH symptomatology but may instead represent a non-specific neuroprotective effect of hitherto unknown functional significance.
Additional commonalities between the two patient groups
Reduced tCr concentrations were found in the left thalamus in both patient groups. This finding confirms our previous explorative result obtained in CM patients without MOH (9). Total creatine mainly consists of the compound creatine and its phosphorylation product phosphocreatine, two essential constituents of brain energy metabolism (26). The lower levels of tCr suggest an altered energy homeostasis in the left thalamus in both patient groups. The concomitant finding of lower tNAA concentration further suggests that the reduced energy demand or supply in the left thalamus is associated with a state of compromised neuronal health that either could result from or contribute to the headaches. It is unclear why the tCr reduction was left lateralized, but it suggests that the two sides of the thalamus are afflicted in a different manner, perhaps due to different underlying modulating mechanisms.
Higher tCho concentrations were found in both patient groups in the right MCC compared with healthy controls. Elevated tCho may reflect increased membrane synthesis or turnover as choline-containing compounds play an important role in membrane synthesis (26). Alternatively, since choline-using reactions require energy, the elevated choline levels in the patient groups may also indicate an impaired energy supply in the MCC (26). Such changes could arise as a consequence of migraine attacks occurring at high frequency over many years as suggested by the positive association with CM history found in the present study. The observed right lateralization may be driven by interactions with the right lateralized ventral attention system (29), which subserves attention reorientation in response to salient and behaviorally relevant stimuli such as repetitive migraine attacks.
Limitations and methodological considerations
We were unable to reproduce our previous finding of reduced tNAA concentrations in the right ACC. This can in part be explained by the different approaches to defining the regions of interest. In the present study, a larger ACC region was used in order to improve the reproducibility (25), consequently resulting in reduced specificity. Our previous finding of an association between right thalamic tNAA and the duration of migraine history in CM patients without MOH was also not reproduced in the present study (9). However, sub-significant negative correlations were found in both of our patient populations. The smaller study populations in the present study likely account for this difference. Also, we did not find a negative correlation between mI in the left ACC and depression scores, as previously reported in migraine patients without MOH (9). Explanations for the absence of a relationship across the entire patient population could be the significantly different coupling between mI and depression scores in the two patient groups of the current study as well as the different depression scales used. Study limitations of our study include that excitatory glutamatergic neurotransmission was not assessed directly due to unreliable estimates. Furthermore, orbitofrontal dysfunction known to exist in MOH was also not assessed due to poor signal quality in this area (see Figure 3). Finally, we did not specifically account for the potential effect of head movement during scanning. However, the relatively large ROIs; that is, integration over numerous voxels, in combination with signal averaging using a high number of acquisitions (NEX = 16) reduces the sensitivity of our results to head movements (42). Hence, we consider it unlikely that head movements have systematically biased our results.
Conclusions
CM with MOH was associated with a distinct concentration profile of mI, a glial marker, in the bilateral ACC that may have arisen from medication overuse and could contribute to the development of MOH. Furthermore, excessive use of acute abortive medication may prevent or reverse the glutamatergic dysfunction observed in the MCC of CM patients without MOH. Finally, similar neuronal- and glial-related abnormalities were found in the two patient groups in regions involved in pain processing suggesting common headache mechanisms.
Article highlights
Magnetic resonance spectroscopic imaging was used to measure neurochemicals in key areas of the thalamocortical pathway in CM patients with and without MOH. CM with MOH was associated with a distinct concentration profile of mI, a glial marker, that may have arisen from medication overuse and could contribute to the development of MOH. Excessive use of acute abortive medication may prevent or reverse glutamatergic dysfunction observed in CM without MOH. Shared neuronal- and glial-related abnormalities were found in CM patients with and without MOH in regions involved in pain processing, suggesting common headache mechanisms.
Supplemental Material
CEP908579 Supplemental Material - Supplemental material for Brain metabolites in chronic migraine patients with medication overuse headache
Supplemental material, CEP908579 Supplemental Material for Brain metabolites in chronic migraine patients with medication overuse headache by David M Niddam, Kuan-Lin Lai, Shang-Yueh Tsai, Yi-Ru Lin, Wei-Ta Chen, Jong-Ling Fuh and Shuu-Jiun Wang in Cephalalgia
Supplemental Material
CEP908579 Supplemental Table 1 - Supplemental material for Brain metabolites in chronic migraine patients with medication overuse headache
Supplemental material, CEP908579 Supplemental Table 1 for Brain metabolites in chronic migraine patients with medication overuse headache by David M Niddam, Kuan-Lin Lai, Shang-Yueh Tsai, Yi-Ru Lin, Wei-Ta Chen, Jong-Ling Fuh and Shuu-Jiun Wang in Cephalalgia
Supplemental Material
CEP908579 Supplemental Table 2 - Supplemental material for Brain metabolites in chronic migraine patients with medication overuse headache
Supplemental material, CEP908579 Supplemental Table 2 for Brain metabolites in chronic migraine patients with medication overuse headache by David M Niddam, Kuan-Lin Lai, Shang-Yueh Tsai, Yi-Ru Lin, Wei-Ta Chen, Jong-Ling Fuh and Shuu-Jiun Wang in Cephalalgia
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DMN, KLL, SYT, YRL and WTC report no disclosures. SJW has served on the advisory boards of Allergan, and Eli Lilly Taiwan. He has received speaking honoraria from the Taiwan branches of Pfizer, Elli Lilly and GSK. He has received research grants from the Taiwan National Science Council, Taipei-Veterans General Hospital, and Taiwan Headache Society. JLF is a member of a scientific advisory board of Novartis and has received research support from the Taiwan National Science Council and Taipei-Veterans General Hospital.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Ministry of Science and Technology (105-2628-B-010-011-MY2) and the Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education in Taiwan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
