Abstract
Neuroimaging studies have explored cerebral activation patterns in patients with cluster headache (CH) during attacks and have revealed activation of multiple brain areas known to belong to the general pain-processing network. However, it is still unclear which changes in brain metabolism are inherent to the shift from the ‘in bout’ to the ‘out of bout’ period. We measured cerebral glucose metabolism in 11 episodic CH patients during the cluster and again during the remission period with 18F-fluoro-2-deoxy-D-glucose-positron emission tomography (FDG-PET) and compared these data with 11 healthy controls. ‘In bout’ compared with ‘out of bout’ scans were associated with increases of metabolism in the perigenual anterior cingulate cortex (ACC), posterior cingulate cortex, prefrontal cortex, insula, thalamus and temporal cortex. Decreases in metabolism were observed in the cerebellopontine area. Compared with healthy volunteers, hypometabolism in the patient group (‘in bout’ and ‘out of bout’) was found in the perigenual ACC, prefrontal and orbitofrontal cortex. Thus, FDG-PET in CH patients revealed ‘in bout’ activation of brain structures which are involved in descending pain control. Compared with controls, the regional brain metabolism was constitutively decreased in most of these structures, irrespective of the bout. This finding indicates a deficient top-down modulation of antinociceptive circuits in CH patients. We suggest that trigger mechanisms of CH are insufficiently controlled and thus promote the initiation of the bout period and acute attack.
Introduction
Cluster headache (CH) is an excruciating primary headache syndrome with strictly unilateral attacks, which are short in comparison with migraine headache. Most patients are affected by an episodic form and only 10–20% suffer from chronic CH. During the cluster period, patients experience daily attacks with very intense periorbital pain and autonomic symptoms. Neuroimaging studies have explored cerebral activation patterns of primary headache syndromes during attacks and have found posterior hypothalamic activation in CH (1, 2) and brainstem activation in migraine (3–5). The hypothalamic activation is thought to be relatively specific for CH, because the hypothalamus is one mediator of the biological clock and CH typically displays a circadian and seasonal rhythm (6). Furthermore, deep brain stimulation of this area has been proven beneficial in CH patients (7–9) and colocalization of a structural change in the same area has been shown by voxel-based morphometry (10). Magnetic resonance spectroscopic abnormalities have recently been found in the hypothalamus in CH (11, 12). Another brain structure related to the circadian rhythm and probably involved in CH pathophysiology is the pineal gland, for which an alteration in opioidergic receptor status has been shown (13). Although these studies have shed light on changes during the ‘in bout’ period, it is still unclear which changes in brain metabolism are accompanying the shift from the ‘in bout’ to the ‘out of bout’ period.
Impaired pain perception with decreased pain thresholds has been shown in CH (14–16), which has led to the assumption of a dysfunction of pain control systems (14, 15). Preliminary data from a functional imaging case of a spontaneous CH attack have provided some evidence for activation of the periaqueductal grey (PAG) matter, a brain area crucial for endogenous pain control (2).
We investigated episodic CH patients during the cluster and again during the remission period of their headache disorder in comparison with healthy volunteers using fluoro-2-deoxy-D-glucose-positron emission tomography (FDG-PET) to gain insights into associated changes in brain metabolism. We expected metabolic changes in brain regions involved in the generation of the circadian rhythm as well as in structures belonging to the so-called pain control system, i.e. prefrontal cortex, (para-) limbic regions such as the insula and perigenual anterior cingulate cortex (ACC) and brainstem nuclei.
Methods
Population
Patients
Twelve men (mean age 46.3 years, range 33–60 years) with episodic CH but with no other neurological, internal or psychiatric disorder were included in the study. Diagnosis of episodic CH according to the criteria of the International Headache Society was confirmed by two neurologists experienced in the diagnosis and treatment of headache (V.P. and T.R.T.). Clinical characteristics of the headache patients are summarized in Table 1. For the patient group, one of the PET scans was acquired during the ‘bout’ period (but without acute pain during the scanning, last attack >6 h prior to PET) and the other when the patients were ‘out of bout’ (last attack >6 weeks prior to PET). Five patients were first scanned ‘out of bout’ and then ‘in bout’, the other six patients in the opposite order. The average time between the two PET scans in the patient group was 5 months. All patients fasted 4 h before the scan. For the ‘in bout’ scans, the patients were ‘in bout’ for at least 1 week at the time of scanning. All patients answered clinical pain questionnaires [pain adjective list by Geissner and visual analogue scales (VAS)] (17). Due to the severity of the headache syndrome, all patients were allowed to continue the use of their prophylactic medication (Table 1). Clinical magneticresonance imaging of the brain was unremarkable in all but one patient, who had a pituitary tumour and was excluded from the PET study because symptomatic CH was suspected.
Clinical characteristics of the 12 episodic cluster headache patients (all male)

The patient was excluded from the positron emission tomographic study as clinical magnetic resonance imaging showed a pituitary tumour.
Healthy volunteers
Eleven healthy male volunteers (average age 37.5 years, range 31–53 years) were scanned once in a resting condition. The healthy volunteers had either no or extremely few headache days per year. None of these volunteers had had a previous diagnosis of a primary headache syndrome or other chronic pain conditions.
All participants gave written informed consent and the local ethics and the radiation protection authorities approved the study protocol.
Data acquisition and analysis
PET scanning
All study participants were imaged using static FDG-PET in 3D mode with septa retracted on a Siemens ECAT EXACT HR+ Scanner (CTI, Knoxville, TN, USA). A neck-shield (NeuroShield; Scanwell Systems, Lavigne St. Montreal, Canada) was used to reduce random count rates. Subjects were positioned with the head parallel to the canthomeatal line within the gantry. An antecubital vein cannula was used to administer the tracer as i.v. bolus (185 MBq 18F-FDG for each scan). During scanning, all subjects rested and had their eyes closed. A sequence of three frames of 10 min was started and afterwards combined into a single frame. After attenuation correction by a standard ellipse-fitting method, the PET data were reconstructed by filtered backprojection with a Hamm filter (cut-off frequency 0.5 cycles/projection element) into 60 image planes and into a 128 × 128 pixel image matrix (pixel size 2.1 × 2.1 mm).
Preprocessing
As CH is a strictly lateralized syndrome (18), the PET scans of the patients with left-sided headache were mirrored in the axial plane to be able to analyse all subjects together. Thereafter, all images were transformed into standard stereotactic space (Talairach space) by linear scaling and non-linear warping using the NEUROSTAT program (19, 20). Normalization was performed with NEUROSTAT instead of the statistical parametric mapping software (SPM2), because the latter program does not provide a FDG template, and normalization of FDG data to the H215O-PET template of SPM has been shown to be inaccurate (21). Spatial smoothing was performed with a Gaussian kernel of 6 mm full width at half maximum (FWHM).
Statistical parametric mapping analysis
Statistical group comparisons (paired t-test) of ‘in bout’ and ‘out of bout’ scans were conducted with SPM2 (Wellcome Department of Imaging Neuroscience, London, UK) and MATLAB 6.5 (Mathworks Inc., Sherborn, MA, USA). For comparison of patients vs. volunteers (‘in bout’ vs. volunteers and ‘out of bout’ vs. volunteers), voxelwise unpaired t-tests were performed. Since the headache and control groups were not exactly age matched, age was included as covariate of no interest in the design matrix to remove possible confounding effects.
The resulting maps of t-statistics were transformed to the unit normal distribution SPM{Z} and thresholded at P < 0.001, uncorrected, for all contrasts. The minimal cluster extension (number of adjacent activated voxels) was set at ≥5 contiguous voxels passing the significance threshold.
Results
Clinical pain ratings
The average VAS pain rating for attacks during the ‘in bout’ periods was 8.3 out of 10 (SD 1.6), indicating severely disabling pain episodes. Similarly, the patients noted high scores of affective (mean 45.2; SD 9.6) and sensory pain (mean 35.1; SD 7.1) components as rated on the adjective lists by Geissner.
PET analysis
Qualitative analysis
The visual qualitative analysis of the FDG-PET scans of patients (‘in bout’ and ‘out of bout’) and healthy volunteers did not show abnormalities in any case.
Analysis of ‘in bout’ vs. ‘out of bout’ scans
In the statistical parametric mapping analysis, significant increases of brain metabolism were detected during the ‘in bout’ compared with the ‘out of bout’ period in the perigenual ACC, posterior cingulate cortex, orbitofrontal cortex including nucleus accumbens, ventrolateral prefrontal cortex, dorsolateral prefrontal cortex (DLPFC) and temporal cortex (middle temporal gyrus) (Fig. 1, Table 2). Decreases were observed in the cerebellopontine area bilaterally (z = 4.04 at 25/−45/−32, cluster size 33 voxel; z = 3.78 at −18/−41/−34, cluster size 11 voxel; not shown). No changes (hypo- or hypermetabolism) were detected in the hypothalamus or pineal gland even when a less conservative threshold of P < 0.01, uncorrected was used.
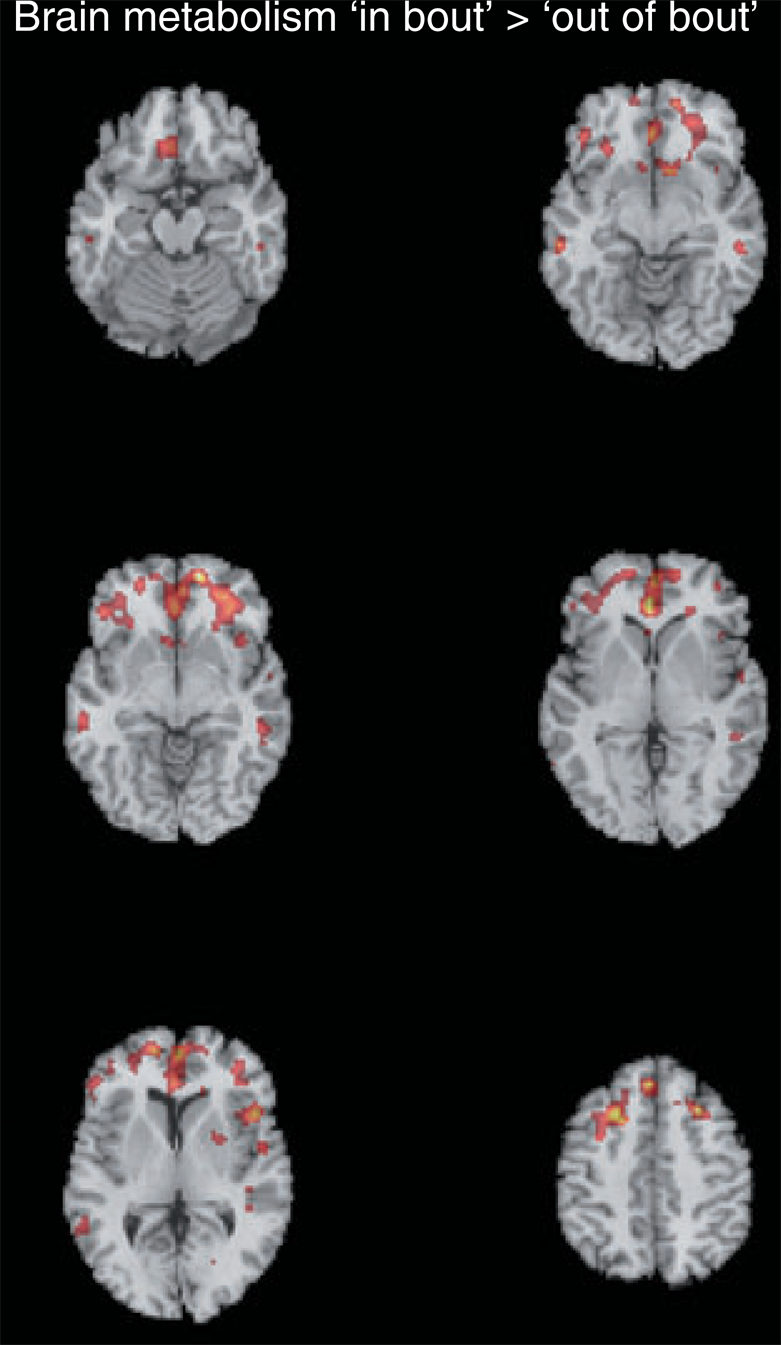
Voxelwise statistical comparison of 18F-fluoro-2-deoxy-D-glucose-positron emission tomography data from cluster headache patients during the cluster period but out of acute pain vs. the identical patients scanned again during the remission period (6 mm full width at half maximum smoothing during preprocessing; ‘in bout’ >‘out of bout’; paired t-test). The statistical parametric map (thresholded at P < 0.005 for descriptive purposes) is overlaid on a skull stripped normalized structural magnetic resonance image (MRI) in axial planes (average of 27 T1-weighted scans of the same individual from the Montreal Neurological Institute) as provided by the MRICRO program package (http://www.sph.sc.edu/comd/rorden/mricro.html). The MRI was stereotactically normalized with NEUROSTAT before overlay. Left side of the images is ipsilateral to the pain.
Results of paired t-testing with SPM2 for the comparison of FDG metabolism in cluster headache patients ‘in’ vs. ‘out of bout’ (‘in’ > ‘out of bout’)
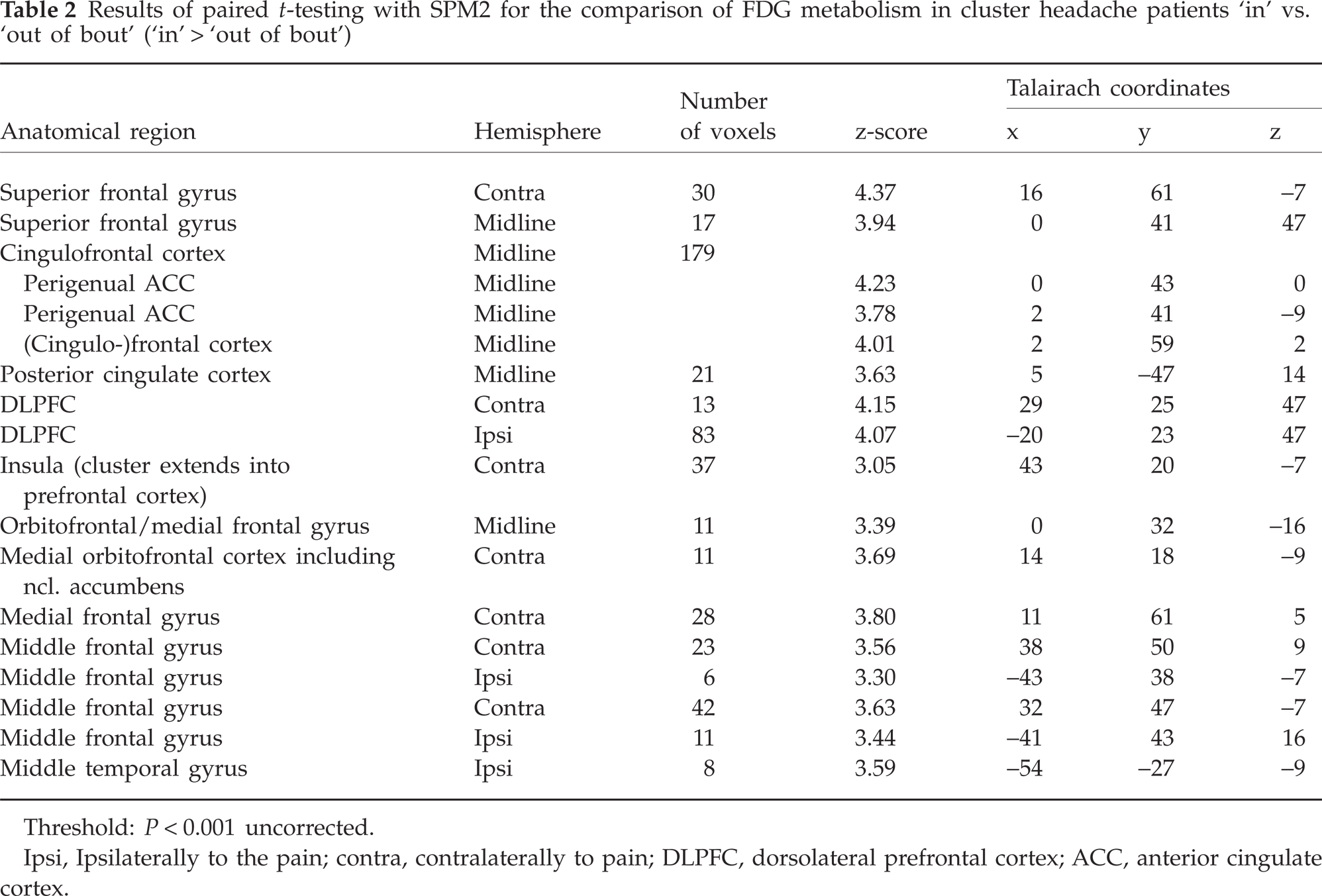
Threshold: P < 0.001 uncorrected.
Ipsi, Ipsilaterally to the pain; contra, contralaterally to pain; DLPFC, dorsolateral prefrontal cortex; ACC, anterior cingulate cortex.
Hypometabolism in CH patients vs. healthy volunteers
Compared with healthy volunteers, a hypometabolism in the patient group (‘in bout’ and ‘out of bout’) was found in the prefrontal cortex, including the orbitofrontal cortex (Fig. 2, Table 3). This reduction was more pronounced ‘out of bout’ than ‘in bout’. The comparison of the ‘out of bout’ scans with the healthy volunteer data (Table 3) evidenced additional decreases of brain metabolism in the patient group in the perigenual ACC, insula, secondary somatosensory cortex (S2), striatum, posterior cingulate cortex and temporal lobe (P < 0.001, uncorrected).
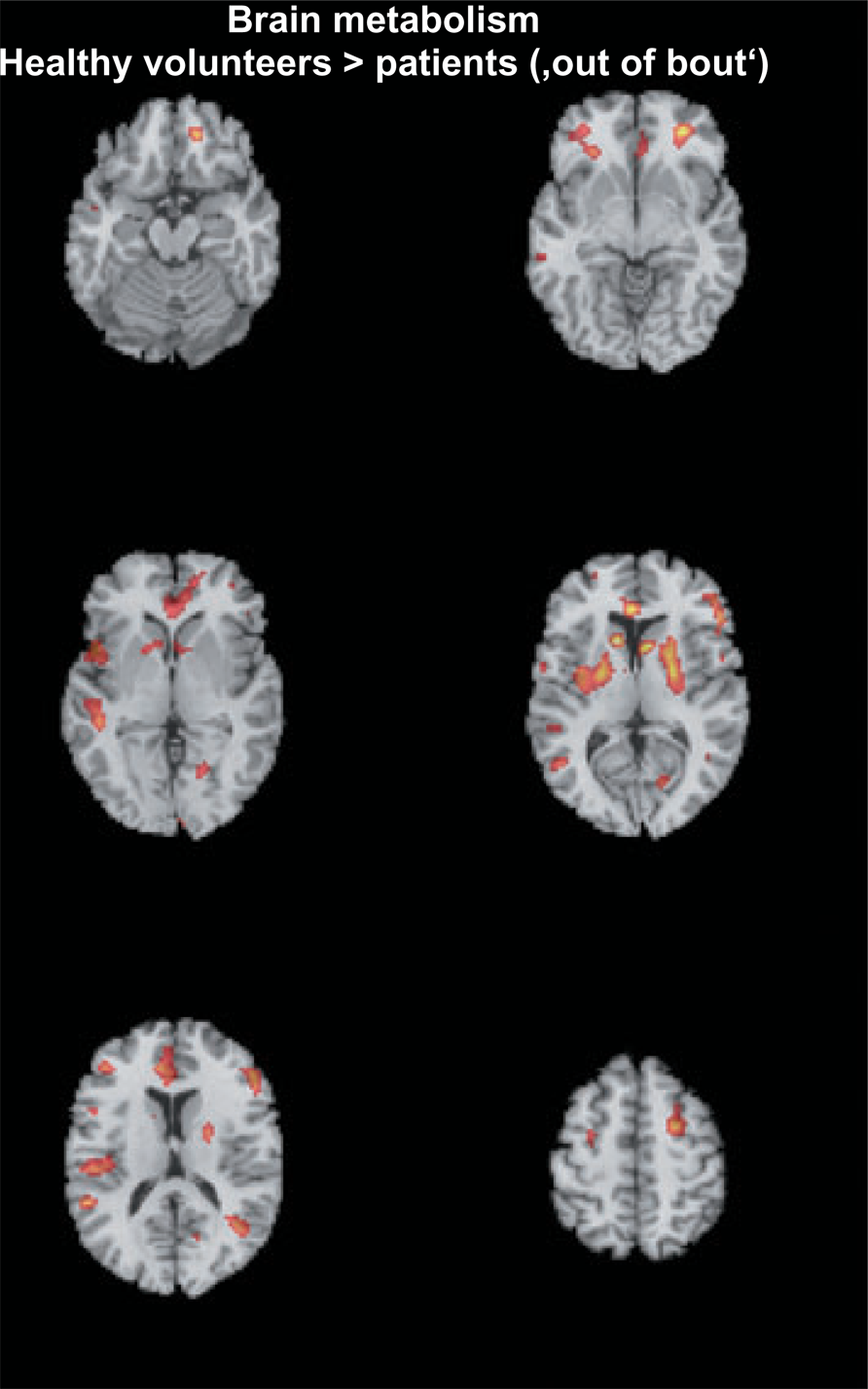
Voxelwise statistical comparison of 18F-fluoro-2-deoxy-D-glucose-positron emission tomography data from cluster headache patients out of the cluster period and out of acute pain vs. healthy volunteers. The statistical parametric map (thresholded at P < 0.005 for descriptive purposes; volunteers > patients ‘out of bout’; unpaired t-test) is overlaid on a skull stripped normalized structural magnetic resonance image (MRI) (average of 27 T1-weighted scans of the same individual from the Montreal Neurological Institute) as provided by the MRICRO program package. The MRI was stereotactically normalized with NEUROSTAT before overlay.
Results of unpaired t-tests with SPM2 for the comparisons of FDG metabolism in healthy volunteers vs. cluster headache patients (hypometabolism in patients)
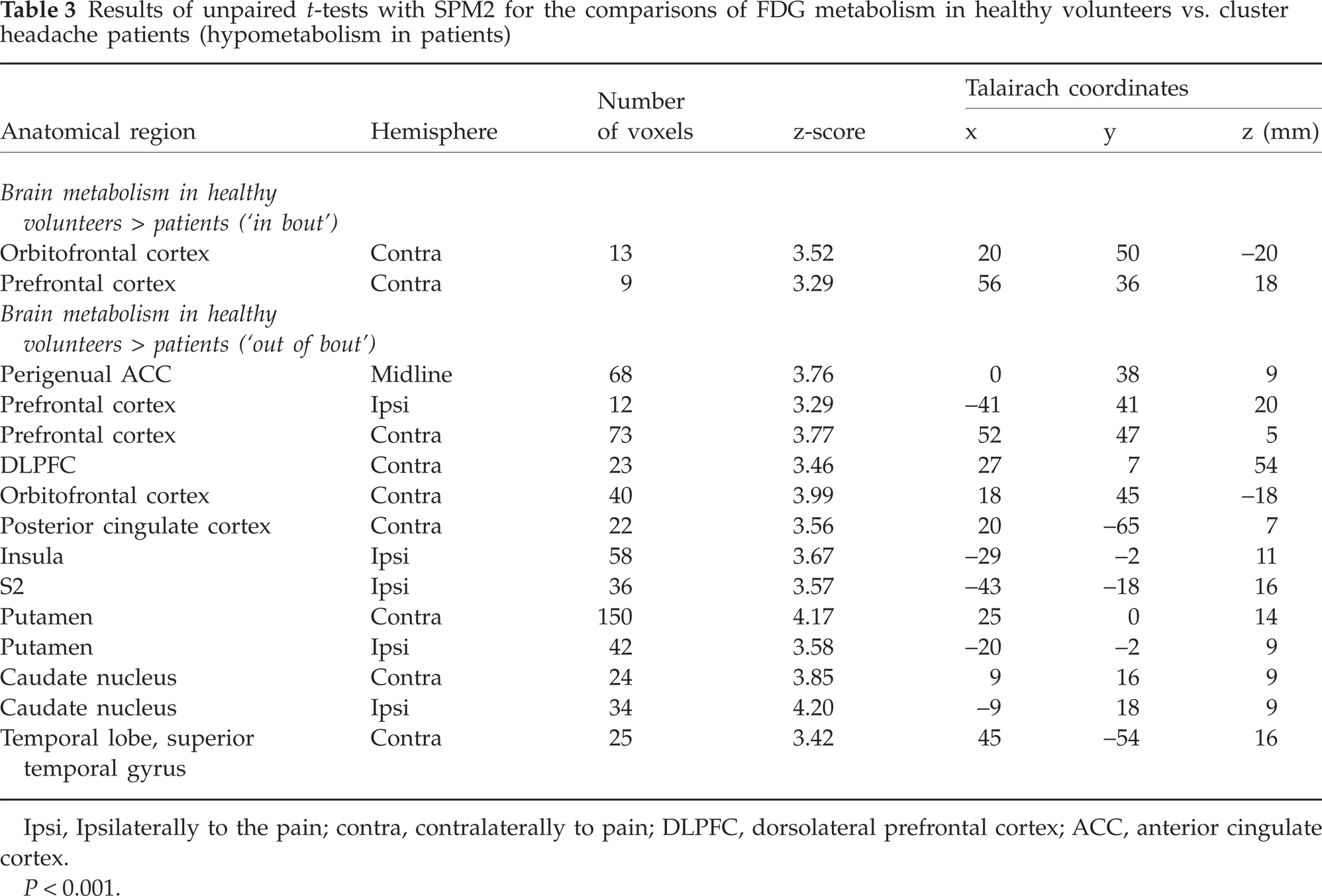
Ipsi, Ipsilaterally to the pain; contra, contralaterally to pain; DLPFC, dorsolateral prefrontal cortex; ACC, anterior cingulate cortex.
P < 0.001.
Hypermetabolism in CH patiens vs. healthy volunteers
Increases in FDG metabolism in patients compared with healthy controls were detected in the parietal lobe and postcentral gyrus (Table 4, not shown as figure). The comparison of brain metabolism in patients ‘out of bout’ vs. healthy volunteers evidenced an additional metabolic increase in the temporal lobe of the patient group (P < 0.001, uncorrected).
Results of unpaired t-tests with SPM2 for the comparisons of FDG metabolism in healthy volunteers vs. cluster headache patients (hypermetabolism in patients)
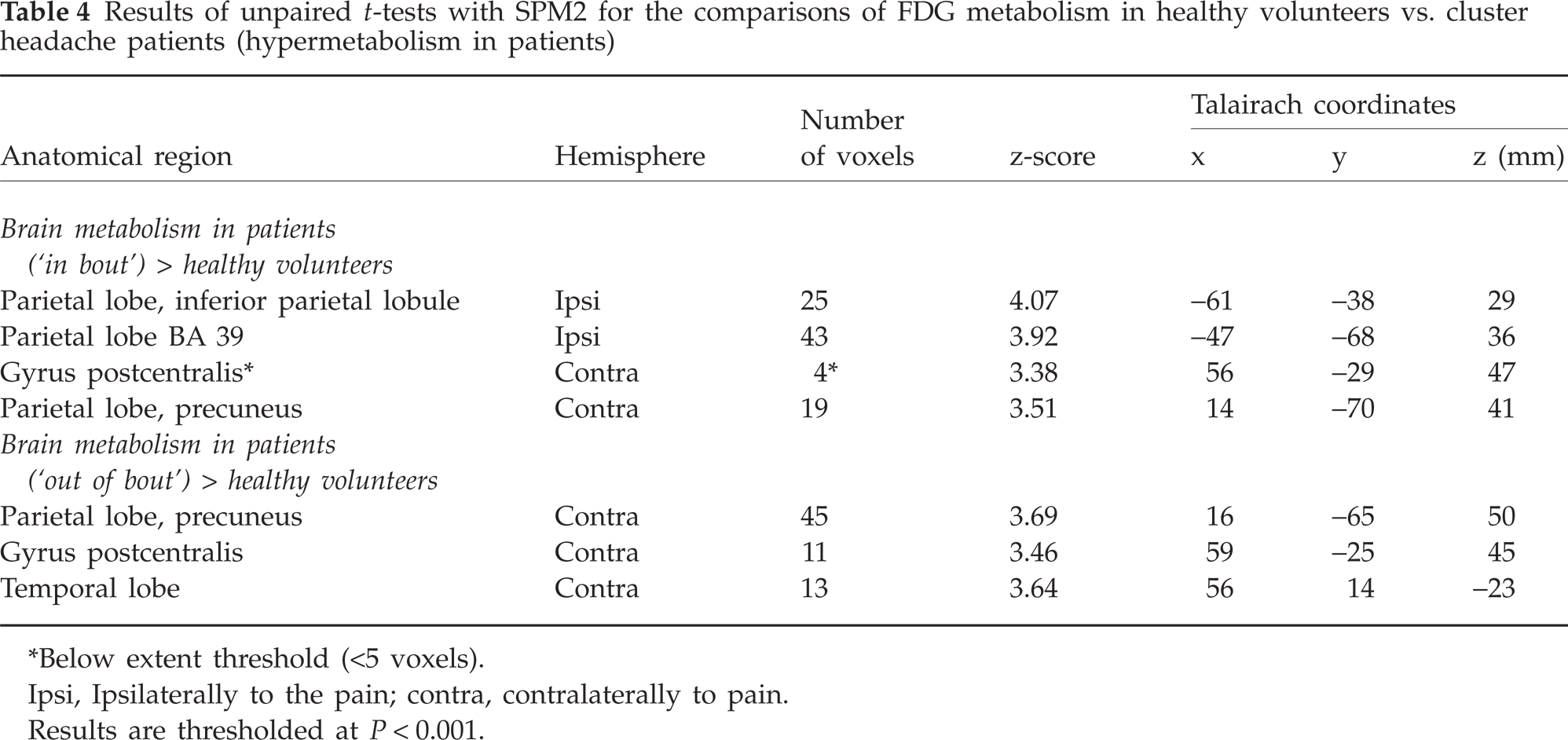
Below extent threshold (<5 voxels).
Ipsi, Ipsilaterally to the pain; contra, contralaterally to pain.
Results are thresholded at P < 0.001.
Analysis of results for the hypothalamus and pineal gland
No increases or decreases in FDG metabolism (‘in bout’ and ‘out of bout’) were detected in the hypothalamus or pineal gland in patients compared with healthy volunteers (even when using a less conservative threshold of P < 0.01, uncorrected).
Discussion
We present FDG-PET data of episodic CH patients ‘in bout’ and ‘out of bout’. Glucose is the major substrate for brain metabolism and FDG is therefore suitable for measuring the glucose metabolic pattern (hexokinase activity) and detecting pathological changes in local synaptic activity (22).
In our study, patients were allowed to continue the use of their prophylactic medication (Table 1). Because of the incapacitating nature of CH, it seemed unethical to ask the patients to stop their medication with the risk of increasing their headache frequency. Theoretically, the medication might impact the FDG metabolism during the ‘bout’ period; however, all patients still had attacks although on medication, and no such changes have been reported for verapamil thus far. As regards lithium therapy, a decrease in temporoparietal FDG metabolism has been reported in a single case of lithium intoxication with a lithium serum level of 2.1 mmol/l (23), but only two of the CH patients were on lithium at the time of the scanning ‘in bout’ without any signs of intoxication. In any case, as we observed hypermetabolism during the ‘bout’ period in most brain structures, the medication might have led to underestimation of the differences in brain metabolism between the ‘in bout’ and ‘out of bout’ periods.
For the comparisons of CH patients vs. healthy volunteers, a slight difference in mean age was present between the groups. This problem was handled by including age as a covariate of no interest in the SPM analysis, thereby minimizing the confounding effect.
During the ‘in bout’ period, significant increases in metabolism were revealed in prefrontal brain areas and the perigenual ACC in comparison with the scans in the remission period of the disease. These regions are well known to be involved in supraspinal pain processing (24, 25). More specifically, these brain areas are thought either to be part of descending pain inhibitory circuits, or to modulate the activity of the latter (26, 27). Although many of these putatively pain-modulating (i.e. ACC, prefrontal/orbitofrontal cortex) brain structures showed increased metabolism during the ‘in bout’ period in comparison with the remission period, the metabolism in patients was still decreased compared with healthy volunteers. An insufficient pain-modulating capacity might therefore be one key factor in a complex pathophysiology, which makes the patients more susceptible to the generation of the whole clinical picture of CH. We suggest, therefore, that CH pathology with cortical (frontal) dysfunction of top–down pain control is in contrast with migraine pathology, where possibly a brainstem pathology, i.e. PAG dysfunction, is more critical (3, 4, 28). The specific contribution of the identified brain areas to metabolic changes in CH patients is discussed below.
Perigenual ACC activation has been shown during a spontaneous attack of CH (2). Other studies, investigating nitroglycerin-triggered CH attacks, have shown activation of the ACC, which was located more caudally in direction to the mid-cingulate area of the ACC (1, 29). The perigenual portion itself was activated in studies evaluating emotional processing, which has been shown with multiple experimental paradigms (30). In experimental pain studies, activation of the perigenual ACC has been seen when pain modulation paradigms, stress and anxiety are evaluated (26, 31) and the perigenual ACC has been proposed to be one key region in (opioidergic) pain modulation (32, 33).
The current observations with FDG-PET regarding the perigenual ACC are in line with a recent correlation analysis between opioidergic receptor binding in CH and disease duration, which revealed decreasing receptor availability depending on disease duration in the rostral ACC (13).
The prefrontal cortex, and particularly the DLPFC, are also thought to participate in pain modulation by inhibiting the neuronal coupling along the ascending midbrain–thalamic–cingulate pathway via descending fibre systems. This has been pointed out in the context of a PET study comparing heat pain with experimentally induced allodynia (34). Moreover, there is electrophysiological evidence from animal experiments, where stimulation of the prefrontal cortex was able to depress the response of midbrain neurons to painful stimulation of the hind-paw (35).
The orbitofrontal cortex has also been linked to pain modulation (36). This view is supported by the fact of orbitofrontal activation in thalamic deep brain stimulation and motor cortex stimulation for pain relief, as well as by opioid administration (31). Recently, a similar change in orbitofrontal FDG metabolism, as seen here in CH, has been reported in chronic medication-overuse headache (MOH). In this study, the orbitofrontal decrease in metabolism was present during the abuse of analgesics in comparison with a control population, but persisted after withdrawal of analgesics (37). The authors suggest that this hypoactivity favours ongoing medication overuse and predisposes the patient to a relapse of MOH. Such a relation of orbitofrontal hypoactivity to substance abuse in MOH is supported by orbitofrontal hypometabolism in addiction (38). However, there is no clear evidence for a ‘cluster headache personality’ (39) and only limited evidence for addictive behaviour in CH (40). Therefore, the orbitofrontal hypoactivity in our study might be related rather to pain-modulation circuits, although addictive and pain-modulation pathways are likely to share similar networks.
The insula is of exceptional importance in pain processing, as it is very robustly activated by virtually any somatic or visceral pain stimulus (31), including facial pain and CH attacks (1, 2, 29). In particular, the anterior insula has been implicated in the determination of stimulus intensity (41), anticipation of pain (42), distraction from pain (43), coding of pain unpleasantness (44), interoceptive pain (45) as well as in the development of asymbolia for pain, i.e. absence or decrease in reaction to nociceptive stimuli (46). Pain-induced insular activity can be modulated by opioids (47) and placebo (48). Moreover, the insular cortex has been shown to modulate pain itself and either produce pro- or antinociception (49). Considering these complex functions of the insula and the multiple connections to other brain areas, including the PAG (50), pain perception might be either altered directly by changes at the cortical insular level or indirectly by insular influences on the descending inhibitory system, i.e. PAG/rostral ventral medulla.
In addition to the changes in pain-modulating brain structures, the comparison of patient data with healthy volunteers evidenced increases of metabolism in the contralateral postcentral gyrus in patients. This might be taken as a direct consequence of the frequent pain attacks during the ‘bout’. However, it is surprising that the hypermetabolism continues ‘out of bout’, which is possibly a sign of a longlasting reorganization in the trigeminal sensory system.
Structures known to be mediators of the biological clock and proposed to be involved in CH pathogenesis, i.e. the hypothalamus and pineal gland (1, 13), showed no significant changes in FDG metabolism, even when lowering the statistical threshold. Possible changes might be relatively subtle in amplitude and therefore not detectable by means of FDG-PET. However, it is also possible that hypothalamic activation is restricted to initiation or maintenance of the acute attacks and therefore no change is evident when investigating patients out of acute pain. Future studies will need to resolve these questions.
We also observed striatal hypometabolism in patients compared with healthy volunteers. Generally, striatal function in pain processing has not been extensively and specifically studied, although striatal activation has been repeatedly reported in pain stimulation paradigms (51, 52) and also in CH (1, 29, 53). The basal ganglia have been implicated in the motor response to pain (31), but recently, striatal D2 receptors have also been related to the modulation of pain (54). Taking these aspects into account, the metabolic changes in the basal ganglia have to be interpreted with caution, but an association with deficient pain-modulating circuits in the CH patients seems worthy of consideration.
Conclusion
We have shown changes in the brain metabolism of CH patients in cortical and subcortical structures known to be involved in pain modulation. As hypometabolism in these brain regions was present during the ‘bout’ as well as ‘out of the bout’ period, we suggest that defective pain control mechanisms are one important component in a more complex pathophysiology, which makes the patients more susceptible to the generation of the whole clinical picture of cluster headache.
Acknowledgements
We thank Brigitte Dzewas and Coletta Kruschke for their technical assistance and the radiochemistry group for their reliable supply of radiopharmaceuticals. We thank Torsten Wüstenberg for helpful input. The study was supported by a grant from the ‘Deutsche Migräne und Kopfschmerzgesellschaft’ (DMKG).
