Abstract
Objective
We aimed to evaluate associations of human leukocyte antigen variants with migraine or headache in hospital and population-based settings.
Methods
The case-control study population, aged 30–70, included 605 clinic-based migraine patients in a medical center and 8449 population-based participants in Taiwan Biobank (TWB). Clinic-based cases were ascertained by neurologists. Participants in Taiwan Biobank were interviewed by a structured questionnaire including headache and migraine history; among them, 2394 had headache or migraine history while 6055 were free of headache and served as controls. All subjects were genotyped by Axiom Genome-Wide Single Nucleotide Polymorphism Arrays and imputed for eight classical human leukocyte antigen genes. Human leukocyte antigen frequencies were compared between clinic-based and self-reported patients and controls. We utilized likelihood ratio tests to examine human leukocyte antigen-disease associations and logistic regressions to estimate the effect of human leukocyte antigen alleles on migraine.
Results
Human leukocyte antigen-B and C showed significant associations with clinic-based migraine (q-value < 0.05). Human leukocyte antigen-B*39:01, human leukocyte antigen-B*51:01, human leukocyte antigen-B*58:01 and human leukocyte antigen-C*03:02 were significantly associated with migraine, with age and sex-adjusted odds ratios (95% CIs) of 1.80 (1.28–2.53), 1.50 (1.15–1.97), 1.36 (1.14–1.62) and 1.36 (1.14–1.62), correspondingly. Clinic-based migraineurs carrying human leukocyte antigen-B*58:01 or human leukocyte antigen-C*03:02 had 1.63 (1.11–2.39) -fold likelihood to have chronic migraine with medication-overuse headache compared to episodic migraine. However, no human leukocyte antigen genes were associated with self-reported headache or migraine in the community.
Conclusions
Human leukocyte antigen class I genetic variants are positively associated with risk of clinic-based migraine but not self-reported migraine or headache and may contribute to migraine chronification and medication overuse.
Introduction
The immune mechanisms implicated in migraine initiation and disease progression remain elusive (1). Among migraineurs, 8% exhibit chronic migraine (CM) (headaches on ≥15 days per month for >3 months, of which ≥8 days fulfil migraine criteria), which is frequently complicated with medication overuse headache (MOH) (2). This highly disabling sub-form of migraine is more refractory to treatment. There is strong need to better understand migraine disease pathogenesis and identify possible clinical strategies.
Previous studies suggested that migraine is genetically and environmentally similar to inflammatory diseases (3). Headache was also found associated with the immune response-regulating STAT6 gene (4). The human leukocyte antigen (HLA) complex is highly polymorphic and plays a key role in human adaptive immune responses. HLA encodes class I and class II major histocompatibility complexes (MHCs) that present antigens to CD8+ and CD4+ T cells, respectively (5). HLA typing methods have evolved rapidly from serological typing in the 1960s to various deoxyribonucleic acid (DNA)-based techniques (6,7). Past studies investigating HLA-migraine correlations typed HLA with antibody-based microlymphocytotoxicity tests and polymerase chain reaction (PCR) using specific probes and primers (8–12). These previous techniques with low resolution, at only 1-field serotype level, limited the information to examine relationships between variants and migraine. In addition, past studies had small sample sizes (8,10–12), limiting sufficient statistical power to detect associations, and examined limited HLA genes (9,10,12).
High-resolution HLA genotyping can be achieved by next-generation sequencing (NGS) platforms (7). However, high costs and complicated experimental techniques make it non-practical in large-scale studies. In the past decade, the widespread popularity of genome-wide association studies (GWAS) has accumulated a large amount of SNP array data, and algorithms to impute HLA alleles from SNP array have been developed (13). Using population-specific references, a study showed the accuracy of HLA imputation ranged from 95.1% to 99.6% in an independent validation group (14). This demonstrates that HLA imputation based on high-resolution references that reflect the linkage disequilibrium pattern of the target population can be highly accurate.
In this case-control study, we applied a high accuracy HLA imputation technique (15) to examine the associations of eight HLA genes with migraine or headache in hospital and population-based settings. In an exploratory analysis, we further investigated the association of HLA variants with CM and MOH.
Methods
Study subjects and data collection
The case-control study included 2999 cases and 6055 controls with sufficient HLA information for data analyses. The case groups consisted of 605 migraine patients diagnosed in a hospital and 2394 self-reported headache or migraine patients in the community. The clinic-based migraine patients were recruited from Taipei Veterans General Hospital (TVGH), a tertiary medical center in Taiwan. On the other hand, the self-reported headache or migraine cases were participants in the Taiwan Biobank (TWB). The control group consisted of those who were free of headache or migraine selected from TWB. Figure 1 illustrates the selection of study subjects. Detailed information on the clinic-based migraine cases and TWB study subjects is described below.
Flow chart depicting the selection methods of cases and controls.
Clinic-based migraine patients: Patients who sought medical attention for headache at TVGH between 2010–2013 were enrolled. Personal information, medical history and headache history were collected by structured questionnaires. The individuals were diagnosed by board-certified neurologists according to the International Classification of Headache Disorders 2nd edition (ICHD-2) based on the interviews, the questionnaires, and previous medical records. Patients were further sub-classified into episodic migraine and CM with or without MOH based on ICHD-3 criteria. To increase homogeneity of the genetic composition of the study population (16,17), patients with Beck Depression Inventory scores greater than 20, comorbid schizophrenia and comorbid epilepsy were not recruited. Among the 1040 clinic-based migraine cases with both questionnaire and genotyping data, 772 patients aged between 30–70 years old were included in the subsequent analyses (Figure 1(a)).
Self-reported headache and migraine subjects and control group in TWB: The TWB is a general population-based public biobank that recruited cancer-free adults between 30–70 years of age from 2008 to 2016. Participants were interviewed with structured questionnaires including basic demographic information and personal and family medical history. In particular, the questionnaire inquired about a) personal and family history of migraine and b) headache or migraine in the past 3 months. Individuals who reported headache or migraine in the past 3 months were further asked whether the pain was mild, moderate, or severe.
In total, 2823 subjects who reported having a history of migraine or headache in the past 3 months but who were free of depression, bipolar disorder, schizophrenia and epilepsy were defined as the “self-reported headache” case group. “Self-reported migraine” was further defined from self-reported headache cases by excluding subjects without a personal history of migraine who reported unknown or mild degrees of headache pain in the past 3 months (Figure 1(b) left).
The TWB subjects who reported a) no personal history of migraine, b) no family history of migraine, c) no headache or migraine in the past 3 months, and d) no vertigo were the control group. As in the case groups, subjects with depression, bipolar disorder, schizophrenia, or epilepsy were excluded, leaving 7196 subjects included in the subsequent analyses (Figure 1(b) right). TWB subjects who did not answer the questionnaire were excluded.
Ethics or Institutional Review Board approval
The study protocol was approved by the Institutional Review Boards (IRB) of Taipei Veterans General Hospital and National Yang-Ming University. All of the study participants provided informed consent at time of enrollment.
Single nucleotide polymorphism (SNP) genotyping
All study subjects performed whole-genome SNP genotyping at the National Center for Genomics Medicine, Academia Sinica, Taiwan. Clinic-based migraine cases had 642,832 SNPs genotyped by Affymetrix Axiom Genome-Wide CHB 1 Array Plate (CHB Array; Affymetrix, Inc., Santa Clara, CA, USA), which was designed to maximize coverage of common Han Chinese variants (18). SNP genotyping was originally performed on clinic-based migraine patients for the purpose of conducting a GWAS study on migraine without aura (18). TWB subjects had 653,291 SNPs genotyped using the Affymetrix Axiom Genome-Wide TWB Array Plate (TWB Array; Affymetrix, Inc., Santa Clara, CA, USA), which was designed based on the CHB Array and further included SNPs previously reported to have clinical significance (19). The two arrays overlapped in 512,065 SNPs on autosomal DNA and were previously validated to have a high average concordance rate of 99.6% (19).
HLA allele imputation
Imputation of HLA alleles based on dense SNP genotypes is an efficient way to fine map association signals with human diseases within the complex MHC region. In this study, HLA imputation was performed using the R package HIBAG (15), which predicted HLA genotypes by averaging posterior probabilities over an ensemble of classifiers. SNP information was extracted from an extended MHC region between 25,759,242 to 33,534,827 base pairs based on hg19 positions. The TWB array included more SNPS in the extended MHC region than the CHB array. Based on a Taiwanese reference, we built two ensembles of classifiers with SNPs extracted from the CHB array and TWB array respectively. HLA imputation was conducted for HLA-A, HLA-C, HLA-B, HLA-DRB1, HLA-DQA1, HLA-DQB1, HLA-DPA1, and HLA-DPB1 genes at 2-field resolution for individuals genotyped with the CHB array and TWB array respectively. Subjects with posterior probabilities ≥0.5 at all eight HLA loci, including 605 clinic-based migraine cases, 2394 self-reported headache cases, 911 self-reported migraine cases and 6055 controls were the study populations included in subsequent analyses (Figure 1).
Assessment of population stratification
Population stratification between clinic-based migraine patients and controls was assessed with the genomic inflation factor (GIF) and principal component analysis (PCA) using PLINK version 1.9. We extracted SNPs common to the CHB and TWB Arrays. Quality control (QC) criteria removed SNPs with call rates <95% and minor allele frequencies <3% in cases and controls combined, as well as SNPs that showed significant (p < 1 × 10−6) deviation from Hardy-Weinberg equilibrium in controls. SNPs were further linkage disequilibrium-based pruned with a variance inflation factor threshold of 2. The GIF was subsequently calculated and PCA was conducted to identify the top principal components (PCs). PC1 and PC2 were plotted to assess systematic differences in the genetic composition between cases and controls.
Statistical analysis
The baseline characteristics of migraine, headache, and controls were compared. Due to violation of the normality assumption (Kolmogorov-Smirnov test p-values < 0.01 and non-linear quantile-quantile plots), the non-parametric Wilcoxon rank-sum test was used to test differences in the ages of case and control groups. The categorical variables, including age groups (in groups of 10), gender, body-mass index (BMI) groups (according to WHO recommendations) (20), and migraine subclass were tested by the Chi-squared tests. In order to evaluate which HLA genes were associated with migraine or headache, we included all alleles of a given HLA gene in the models and examined whether the logistic models improved significantly. We compared two logistic models by a likelihood ratio test (LRT). One model included age and sex as covariates (null model) and the other included all alleles of a given HLA gene plus age and sex (full model). All of the alleles included had population frequency >1%. To account for multiple comparisons, we used false discovery rate (FDR) to obtain q-values.
After identifying specific HLA genes associated with migraine or headache, we further examined which specific HLA alleles were associated with either migraine or headache at a given HLA gene. We compared the various HLA alleles of case and control groups using Fisher’s exact tests and performed FDR adjustments separately at each HLA gene. To assess the magnitude of the associations of HLA alleles on migraine or headache, we performed logistic regressions. The crude and adjusted odds ratios (ORs) with 95% confidence intervals (CIs) were estimated. The multiple logistic regressions included the covariates age and sex, which were previously reported as relevant factors for migraine or headache (21). The population attributable fractions of significant alleles were calculated as previously reported (22,23) by
In addition to the associations of HLA and migraine or headache, we validated four SNPs (rs10803531, rs7565931, rs3781545, rs655484) which have been reported among clinic-based patients in our previous study (18). We validated the associations of the four SNPs with self-reported migraine or headache in the community. We used Cochran-Armitage trend tests to examine the genotypes (AA, AB, and BB) and the associations with migraine or headache. Multiple logistic regressions were performed to obtain ORs and 95% CIs after adjustment for age and sex. All of the p-values and q-values in this study were two-sided and considered as statistically significant when <0.05. The analyses were performed in the SAS statistical software package (version 9.4; SAS Institute Inc., Cary, NC) and PLINK (version 1.9) (24).
Data availability statement
Anonymized data from TWB is available for application by contacting biobank@gate.sinica.edu.tw. In addition, analyzed data related to this study, intended for reasonable use, can be shared by request by contacting the corresponding author.
Results
Baseline characteristics of study subjects
Baseline characteristics of study subjects.
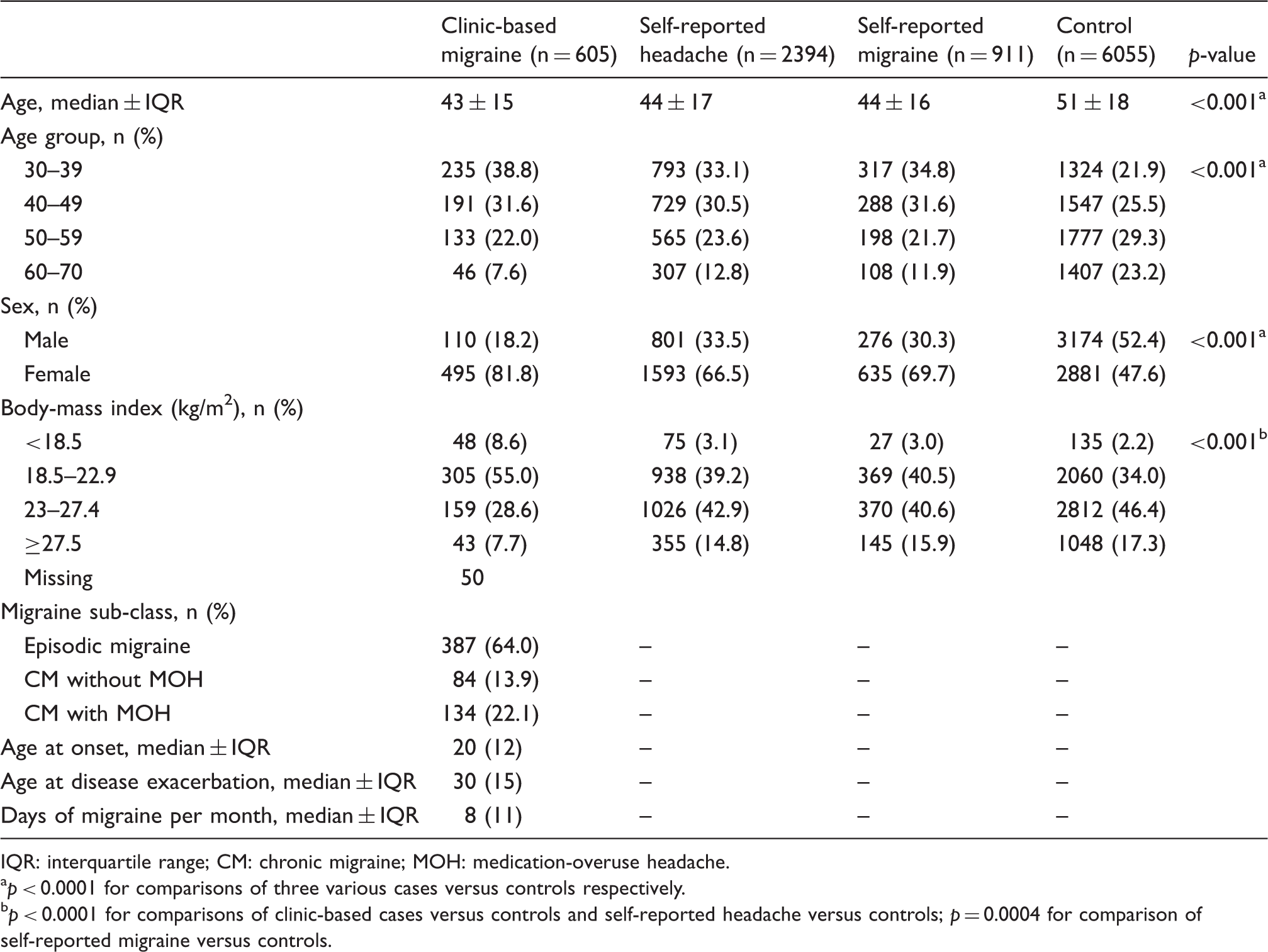
IQR: interquartile range; CM: chronic migraine; MOH: medication-overuse headache.
p < 0.0001 for comparisons of three various cases versus controls respectively.
p < 0.0001 for comparisons of clinic-based cases versus controls and self-reported headache versus controls; p = 0.0004 for comparison of self-reported migraine versus controls.
HLA associated with clinic-based migraine
Q-values of associations between HLA genes and migraine or headache patients in the hospital and the community.

Significant associations between HLA alleles of class I genes and clinic-based migraine.

OR: odds ratio; CI: confidence interval.
In haplotype analysis, we found that HLA-A*33:03, HLA-B*58:01, and HLA-C*03:02 construct a common HLA haplotype with a frequency of 9.7% in controls and 13.2% in cases (Supplemental material, Appendix 1). Individuals who carried the haplotype of HLA-A*33:03- HLA-B*58:01- HLA-C*03:02 were associated with clinic-based migraine, and the age-sex adjusted OR was 1.44 (1.20–1.73). The population attributable fraction of the haplotype on clinic-based migraine was 8.1% (Supplemental material, Appendix 2).
Significant associations of HLA alleles with migraine subtypes among clinic-based migraine patients.

Multinomial logistic regression adjusted for age and sex.
OR: odds ratio; CI: confidence interval; CM: chronic migraine; MOH: medication-overuse headache.
A few sensitivity analyses were conducted. First, we adjusted HLA allele imputation QC criteria from posterior probabilities at all HLA genes ≥0.5 to ≥0.6 and ≥0.4, which respectively increased or decreased imputation accuracy while decreasing or increasing call rate. Using different CT values did not affect the significance of the associations discovered in the main analyses (Supplemental material, Appendices 4 and 5).
We also evaluated population stratification. Although clinic-based migraine patients were not recruited from TWB, the GIF was 1.04 and plotting of PC1 versus PC2 did not show differential clustering of cases and controls (Supplemental material, Appendix 6). Therefore, principal components were not adjusted for in the main analyses. To ensure this did not cause bias, we conducted additional analyses where the top five PCs were included as covariates in logistic regressions. The age, sex, PC5-adjusted odds ratios of HLA-A*33:03, HLA-B*39:01, HLA-B*51:01, HLA-B*58:01, and HLA-C*03:02 remained significant, and were separately 1.29 (1.08–1.54), 1.91 (1.35–2.69), 1.47 (1.12–1.93), 1.35 (1.13–1.62) and 1.35 (1.13–1.62).
Finally, to ensure imputing cases and controls with separate sets of classifiers did not result in bias, we imputed clinic-based cases with the TWB array-specific set of classifiers. Due to the lower imputation quality, only 334 cases remained after quality control, resulting in a 44.8% decrease in case sample size. We re-analyzed HLA associations and found the common alleles HLA-A*33:03, HLA-B*58:01 and HLA-C*03:02 remained significantly positively associated with clinic-based migraine. HLA-B*39:01 and HLA-B*51:01 had positive associations, but associations became insignificant likely due to the much smaller sample size (Supplemental material, Appendix 7).
HLA and self-reported migraine or headache in the community
In HLA gene-based LRTs, no HLA genes showed significant associations with self-reported headache or migraine (Table 2). In addition, the four SNPs (rs655484, rs3781545, rs7565931 and rs10803531) found in our previous clinic-based case-control study (18) were not validated in the self-reported migraine or headache cases in the community (Supplemental material, Appendix 8).
Discussion
In this case-control study, we found HLA-A*33:03, HLA-B*39:01, HLA-B*51:01, HLA-B*58:01 and HLA-C*03:02 were positively associated with clinic-based migraine. Among clinic-based migraine patients, HLA-A*33:03, HLA-B*58:01 and HLA-C*03:02 increased the probability of having CM with MOH. On the other hand, self-reported migraine or headache in the community was not associated with HLA class I or class II variants.
Past studies investigating HLA-migraine relationships had small sample sizes, low genotyping resolution and investigated a limited number of genes (8–12). In contrast, our study including thousands of subjects utilized an imputation reference of 1012 Taiwanese individuals who were genotyped with both whole-genome SNP array and NGS (whole genome sequencing) to derive high resolution HLA alleles for eight classical HLA genes from SNP data, allowing cost-efficient and accurate assessment of how the immune response-regulating HLA variants affect migraine phenotypes. To the best of our knowledge, this is the first comprehensive study to examine migraine-related HLA genes, including HLA class I and class II. In addition, it is the largest study to investigate genetic variants and migraine or headache in Asians.
Migraine is recognized as a disease mainly involving neuronal factors (25). Interestingly, strong genetic and environmental links between immune diseases and migraine were recently discovered (3). Furthermore, elevation of inflammatory mediators including histamine, IL-6, and TNF was observed in migraine patients (1,26). The associations of HLA class I variants and clinic-based migraine found in this study paralleled previous findings and suggested immune mechanisms of migraine. Calcitonin gene-related peptide (CGRP) is a neuropeptide postulated to modulate pain signals in the trigeminovascular system (27). Although mechanisms of action are still unclear, clinical trials of CGRP antagonists in migraine patients showed promising results (1,25). CGRP is hypothesized to induce peripheral neurogenic neuroinflammation, which may play a role in migraine chronification (1). In addition to CGRP, pituitary adenylate cyclase activating peptide (PACAP) is another neuropeptide implicated in migraine (26). IgE-crosslinking and various neuropeptides induced human mast cell degranulation and release of PACAP (28). This possible link between mast cells and migraine is interesting given that mast cells were reported to present antigens to CD8+ T cells through MHC class I, altering CD8+ T cell effector functions (29). Our findings, coupled with past studies, suggest HLA class I presentation of antigens and CD8+ T cell responses may play a role in migraine onset and chronification, illustrating the need for further investigations of immune mechanisms in migraine.
Among clinic-based patients, we identified episodic migraine and CM with or without MOH based on detailed clinical evaluations. HLA-A*33:03, HLA-B*58:01 and HLA-C*03:02 increased the risk of CM with MOH, but not CM without MOH, compared with episodic migraine. We speculate that drug molecules may alter migraine-related immune responses, which might contribute to the exacerbation or chronification of headaches especially upon excessive exposure of painkillers; that is, medication overuse. HLA-B*58:01 was previously reported to be associated with allopurinol-induced adverse drug reactions (30). HLA-related drug hypersensitivities were hypothesized to be the result of alloreactive T cell responses due to drug molecules altering HLA binding groove structure (31). Whether specific or non-specific migraine medications mediate MOH in the same manner remains unknown. In the future, identifying specific drug use associated with CM with MOH may be interesting. Determination of the drug molecules involved, along with peptide-HLA binding assays (31), may help provide insight into the mechanisms of MOH in migraine.
No associations between HLA variants and self-reported migraine or headache were found in our study. The population-based cases were defined by self-reported migraine or headache. The prevalence of tension-type headache in Asia was 16.2% (32). Therefore, the self-reported migraine or headache may be more heterogeneous than those diagnosed in hospitals, resulting in loss of study power. Additionally, population-based cases were possibly milder, with genetic components contributing less than those in clinic-based patients. Despite higher heterogeneity in self-reported cases, a large-scale GWAS in UK Biobank discovered 28 loci associated with self-reported headache (4). The study included over 200,000 subjects with 74,461 cases and 149,312 controls and thus may be more sensitive to detect the associations. The loci reported in the UK Biobank study partly overlapped with a previous large-scale migraine GWAS (33), which included self-reported cases as well as rigorously defined clinic-based migraine patients. The studies reported associated loci with diverse functions, from regulating immune responses (STAT6), forming ion channels (TRPM8), to intracellular signaling (LRP1) (4,33). The findings implied complex mechanisms, including immune responses, are involved in headache and migraine. However, whether the variants found in Caucasians could be validated in Asian populations remains to be investigated. In addition, the homogeneity of the case population should be considered when aiming to identify genetic associations that reflect mechanisms of a specific disease. Regarding this, our carefully ascertained clinic-based migraine cases provide an advantage.
There were several limitations in our study. First, it is not possible to rule out the true causal gene being in linkage disequilibrium with HLA. Tumor necrosis factor alpha (TNFα)-308 G/A (rs1800629) A allele was previously reported to be positively associated with migraine in Asians (34). This allele, located on chromosome 6, is also linked with the haplotype HLA-A*33- HLA-B*58 in Chinese (35,36). It is possible that the rs1800629 risk allele, instead of HLA-A*33:03- HLA-B*58:01- HLA-C*03:02, is the causal variant. However, previous studies on associations between TNFα and migraine were conflicting (37). The HLA region includes extensive linkage disequilibrium (38). Fine-mapping and functional studies are needed to confirm whether the five HLA alleles discovered in our study mechanistically affect migraine. Second, migraine is most prevalent in young and middle-aged adults (32). The memory of previous migraine history among individuals with advanced age may not be correct. The non-differential misclassification may result in estimates toward the null when we examined the HLA associations with self-reported migraine or headache. Third, the generalizability of our findings to migraine patients of all ages and ethnic groups requires caution. The study participants were 30 to 70 years old. However, disease that manifests in younger or older patients may have different genetic components. Additionally, this study included Taiwanese and should be validated in other ethnic groups. Allele frequencies and linkage disequilibrium patterns differ between ethnicities, resulting in differed genetic associations with disease. HLA-A*33:03- HLA-B*58:01- HLA-C*03:02 is a common haplotype in Han Chinese but the frequency was less than 1% in European Caucasians (39). However, HLA-B*39:01 and HLA-B*51:01 were similarly prevalent in our study population and in Caucasians (39).
The burden of MOH among migraine is high, with 22% of our clinic-based migraine cases having CM with MOH. Individuals with MOH who tried to decrease medication intake experienced withdrawal symptoms and had a high relapse rate of 20–40% within the first year of drug withdrawal (40). Therefore, screening for patients at high risk of MOH, coupled with better communication of the risks of medication overuse, may be an efficient prophylactic strategy to reduce disease incidence. Screening for HLA-B*58:01 is recommended in clinical guidelines before the use of allopurinol (41); our findings demonstrate it may have potential to alleviate the considerable burden of MOH in migraine as well.
In conclusion, we found that HLA class I variants increased susceptibility to clinically diagnosed migraine in the hospital but not self-reported migraine in the community. Among clinic-based patients, HLA-A*33:03, HLA-B*58:01 and HLA-C*03:02 conferred risk of CM with MOH. These findings implicate immune mechanisms in migraine pathogenesis.
Clinical implications
This large case-control study found HLA class I variants are associated with clinic-based migraine and contribute to medication overuse-related disease chronification. Screening for HLA-B*58:01 has potential to alleviate the burden of medication overuse headache in migraine. Independent validation in different populations is needed to assess the clinical utility of the results in other ethnic groups.
Supplemental Material
CEP902228 Supplemental material - Supplemental material for HLA class I alleles are associated with clinic-based migraine and increased risks of chronic migraine and medication overuse
Supplemental material, CEP902228 Supplemental material for HLA class I alleles are associated with clinic-based migraine and increased risks of chronic migraine and medication overuse by Claire Huang, Shih-Pin Chen, Yu-Han Huang, Hsuan-Yu Chen, Yen-Feng Wang, Mei-Hsuan Lee and Shuu-Jiun Wang in Cephalalgia
Footnotes
Acknowledgements
We thank all study participants for their generous contribution and the TWB for making the data available. We thank the National Center for Genome Medicine for the technical support in the genotyping.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CH, SPC, YHH, HYC, and MHL have nothing to disclose; YFW reports grants from Taiwan Ministry of Science and Technology, grants from Taipei Veterans General Hospital, personal fees from Eli Lilly, Taiwan, personal fees from Pfizer, Taiwan, personal fees from Novartis, Taiwan, personal fees from Chugai, Taiwan, personal fees from Orient EuroPharma, Taiwan, outside the submitted work; SJW reports personal fees from Eli-Lilly and Company, personal fees from Pfizer Taiwan, personal fees from Daiichi-Sankyo, personal fees from Norvatis Taiwan, grants from Ministry of Science of Technology, grants from Ministry of Education, grants from Taipei Veterans General Hospital, grants from National Yang-Ming University, outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Ministry of Science and Technology, Taiwan (grant number: MOST-107-2314-B-010-021 (to SPC) and MOST 105-2628-B-010-003 MY4 and MOST 107-2314-B-010-004 MY2 (to MHL) and MOST 108-2321-B-010-014-MY2, MOST 108-2321-B-010-001-, and MOST 108-2314-B-010-023-MY3 (to SJW)), Ministry of Education of Taiwan, Higher Education Sprout Project (107BRC- B201, B202, B203 and B204 (to SJW and SPC)), Ministry of Health and Welfare, Taiwan (MOHW107-TDU-B-211-123001 and MOHW 108-TDU-B-211-133001 (to SJW)), and Taipei Veterans General Hospital, Taiwan (VGH-106-D9-001-MY2-2 (to SJW)). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
