Abstract
Background
The pathophysiology of reversible cerebral vasoconstriction syndrome is unclear. An unbiased systems-based approach might help to illustrate the metabolite profiling and underlying pathophysiology.
Methods
Urine samples were collected from reversible cerebral vasoconstriction syndrome patients and matched controls recruited in Taipei Veterans General Hospital. 1H-Nuclear magnetic resonance was used to initially explore the metabolic profile, and liquid chromatography tandem mass spectrometry was then used to identify metabolic alterations in reversible cerebral vasoconstriction syndrome. Untargeted metabolite screening was randomly performed on 10 reversible cerebral vasoconstriction syndrome patients and 10 control subjects in the discovery phase. The selected untargeted metabolites were further validated on 47 reversible cerebral vasoconstriction syndrome patients during their ictal stage (with 40 of them having remission samples) and 47 controls in the replication phase.
Results and conclusion
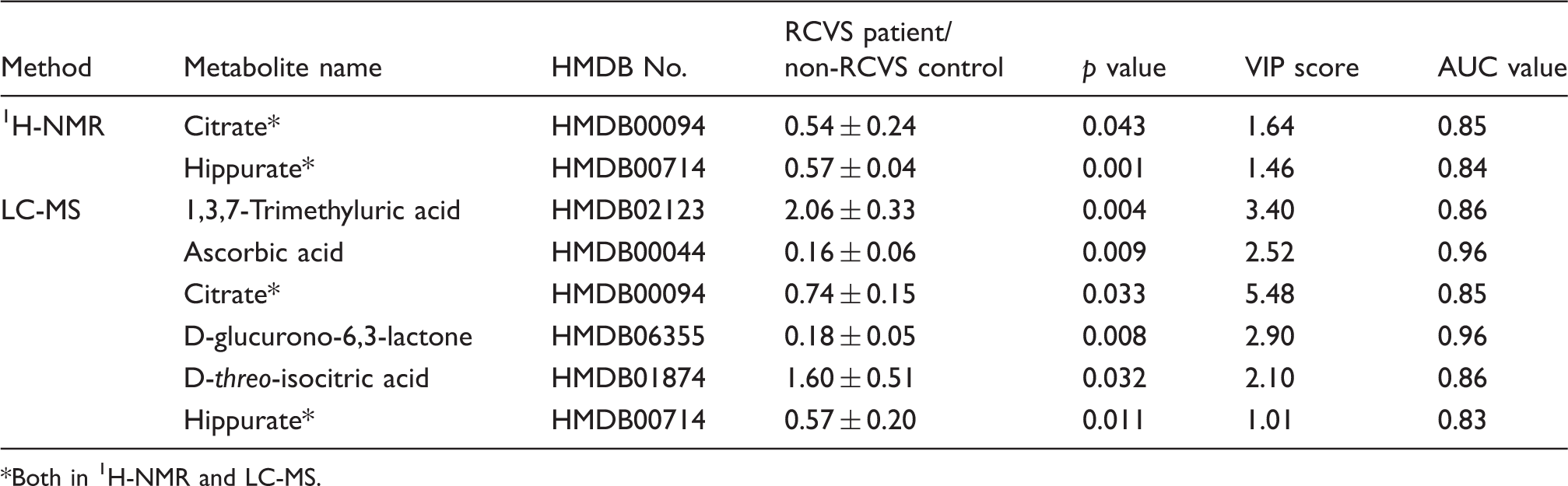
Six metabolites-hippurate, citrate, 1,3,7-trimethyluric acid, ascorbic acid, D-glucurono-6,3-lactone, and D-threo-isocitric acid-with t-test derived p-value < 0.05 and VIP score >1, were identified as potential urine signatures that can well distinguish reversible cerebral vasoconstriction syndrome subjects at ictal stage from controls. Among them, citrate, hippurate, ascorbic acid, and D-glucurono-6,3-lactone were significantly lower, and 1,3,7-trimethyluric acid and D-threo-isocitric acid were higher in reversible cerebral vasoconstriction syndrome patients. Of these, four selected metabolites, citrate, D-glucurono-6,3-lactone, ascorbic acid, and 1,3,7-trimethyluric acid, returned to normal levels in remission. These metabolites are related to pathways associated with free radical scavenging, with the hub molecules being associated with endothelial dysfunction or sympathetic overactivity. Whether these metabolites and their implicated networks play a role in the pathogenesis of reversible cerebral vasoconstriction syndrome remains to be confirmed.
Keywords
Introduction
Reversible cerebral vasoconstriction syndrome (RCVS) is a neurovascular disorder characterized by abrupt, severe (mostly thunderclap) headaches and reversible segmental vasoconstrictions of cerebral arteries (1,2). Several large-scale studies have demonstrated that RCVS is not uncommon and should be treated as a clinical emergency because of its substantial risk of complications (3–5). Up to one-third of RCVS patients might experience posterior reversible encephalopathy syndrome, ischemic stroke, or intracranial hemorrhages, including convexity subarachnoid, intracerebral, and even subdural hemorrhages (3–7). Patients presenting with multiple thunderclap headaches without significant complications have been labeled as “purely cephalalgic” (2), in whom vasoconstrictions (2,7,8), hyperintense vessels (8,9), and white matter hyperintensity lesions (7,9,10) might be the only initial imaging findings. However, it should be noted that even when the thunderclap headaches are remitted, vasoconstriction might still be severe and the patients at risk of vascular complications (6,7). In addition, RCVS is not always monophasic. Our recent long-term follow-up study showed that RCVS could recur in 5–10% of patients (11), suggesting that some patients might be susceptible to this disease.
Despite the fact that its clinical features are largely characterized, studies of the pathophysiology of RCVS are still in their infancy. Altered cerebrovascular autoregulation may play a central role in the pathogenesis of RCVS (2,12), which might be caused by complex interactions between autonomic dysregulation (13), increased oxidative stress (14), impaired endothelial repairing capacity (15), or genetic predisposition (16). Nevertheless, the exact molecular mechanisms are largely unknown. It is likely that RCVS is a complex disease caused by a variety of factors that could not be completely elucidated by a hypothesis-driven approach. Currently, no reliable biomarker is available for RCVS. Urine is a non-invasive and easily collected biofluid, and a urine metabolomic approach for RCVS has not yet been explored. Metabolomics, one of the systems biology technologies (17), might be helpful for exploring potential pathogenic factors to diagnose RCVS and predict treatment response, and pave the way for treatment of RCVS.
In this study, we investigated the perturbed metabolic patterns in urine samples from patients with RCVS and attempted to formulate a possible pathogenic network based on the correlation between these metabolites and clinical parameters. Combined 1H-NMR and LC-MS/MS analyses were applied to find altered metabolites in RCVS.
Materials and methods
Participants, settings, and clinical evaluations
This study prospectively recruited patients with RCVS from the Headache Center of Taipei Veterans General Hospital (TVGH), a 2909-bed national medical center in Taiwan, from 2010 to 2014. The diagnosis of RCVS was based on criteria we reported previously (11). The criteria were adapted from the RCVS diagnostic elements proposed by Calabrese et al. (1) and also in concordance with the proposed criteria for “headaches attributed to RCVS” in the 2018 International Classification of Headache Disorders 3rd edition (ICHD-3) (code 6.7.3) (18). The diagnostic investigations, the demonstration of the reversibility of vasoconstrictions, and therapeutic protocols were detailed previously (6,7,11). Sex and age-matched volunteer controls who did not have major systemic illnesses or past histories of headache disorders except for infrequent episodic tension-type headaches (<1 day/month) were recruited from hospital and university staff, as well as from the general population through advertisements.
The inclusion criteria for all participants included a) being aged between 20–65 years, b) being able to fully understand the objectives of the study, and c) being willing to join the study. The exclusion criteria included a) being a current smoker; b) uncontrolled hypertension (systolic blood pressure >160 mmHg, diastolic blood pressure >100 mmHg) except for the transient blood pressure surge accompanying headache attacks in RCVS patients; c) known cardiovascular or cerebrovascular disease, e) any neovascularization-associated conditions such as cancer or diabetes; and f) a history of using vasoconstrictors, illicit drugs, or taking antioxidants regularly. A matched proportion of control subjects with grade 1 hypertension (systolic blood pressure 140–159 mmHg, diastolic blood pressure 90–99 mmHg) were permitted to enroll since some RCVS patients might have hypertension. For the purpose of replication, we recruited two independent cohorts by the time of subject recruitment. The discovery cohort consisted of patients who entered the study after 2014, whereas the validation cohort was composed of patients recruited from 2010 to 2014. To evaluate the difference between ictal and remission stages, patients with RCVS were also asked to participate in the study at the remission stage.
Urine and plasma sample collection
Each participant provided a 15 ml sample of middle-stream urine and 10 ml of peripheral blood collected from the antecubital vein upon registration. In patients with RCVS, samples were collected prior to administration of any treatment. A remission urine sample was also collected in RCVS patients at least 3 months after the ictal stage and having been off nimodipine for at least 1 month. Urine samples were centrifuged (1500 g for 10 min at 4℃), aliquoted, and stored in −80℃ freezer until analysis. Blood samples were collected with EDTA tubes on ice. Plasma was separated immediately by centrifugation at 2000 rpm for 15 min and was stored at −80℃ until analysis.
Ethics
The study protocol was approved by the Institutional Review Board of TVGH (IRB No. 98-05-04 A). All participants provided written informed consent before entering the study. All clinical investigations were conducted according to the principles expressed in the Declaration of Helsinki. The corresponding authors had full access to all of the data in the study and had final responsibility for the decision to submit the research for publication.
Data availability statement
The data that support the findings of this study are available from the corresponding authors on request.
Metabolome profile for untargeted metabolites in discovery phase
1H-NMR spectroscopy analysis was performed mainly as described previously on a Varian Unity INOVA 500 MHz spectrometer with a regular probe (19). Analysis of metabolites by LC-MS is highly complementary to metabolite profiling by 1H-NMR. Thus, we used LC-MS/MS to identify additional metabolic changes. LC-MS profiling was conducted by Agilent 1290 ultra-high performance liquid chromatography (UHPLC) coupled with 6540 - Quadrupole-Time-of-Flight (QTOF) mass system (Agilent, Santa Clara, CA, USA) and performed as described in Bruce et al. and Zelena et al. (20,21). MS raw data were converted to the mzXML format using Trapper (ISB) and processed by TIPick, an in-house package. After TIPick processing, scaling-based normalization was performed according to the total ion abundances from each UHPLC-MS data set (22).
Validation of untargeted metabolites
Standards of hippurate, D-glucurono-6,3-lactone, 1,3,7-trimethyluric acid, and potassium D-threo-isocitrate were purchased from Sigma (St. Louis, MO, USA). 8-iso-Prostaglandin F2α (8-iso-PGF2α) and d4-PGF2α were purchased from Cayman Chemicals (Ann Arbor, MI, USA). Hippurate, 1,3,7-trimethyluric acid in urine and 8-iso-PGF2α in plasma, were quantified by an UHPLC system (Accela; Thermo, USA) equipped with a C18 column (1.0 × 150 mm, 3 µm, T3; Waters, Milford, MA, USA) with a triple quadrupole mass spectrometer (TSQ QUANTUM ACCESS MAX, Thermo, USA). The mass spectrometer was operated using selected reaction monitoring (SRM) with negative ion mode for detection of hippurate, 1,3,7-trimethyluric acid, and 8-iso-PGF2α. The precursor ion/fragment ion (collision energy) values of hippurate, 1,3,7-trimethyluric acid and 8-iso-PGF2α were 178.06/134.19 (15 eV), 209.06/194.1 (20 eV), and 353.16/193.16 (28 eV), respectively. For D-glucurono-6,3-lactone and D-threo-isocitric acid validation on an UHPLC system (Ultimate 3000; Dionex, Germany) with a C18 column (2.1 × 150 mm, 3 µm, Atlantis T3; Waters, Milford, MA, USA) was coupled with a hybrid QTOF mass spectrometer (maXis impact, Bruker Daltonics, Bremen, Germany) with an orthogonal ESI source in negative ion mode. The fragmented ions (collision energy) of D-glucurono-6,3-lactone and D-threo-isocitric acid were 113.02 m/z (5 eV) and 111.00 m/z (15 eV), respectively. The concentrations of ascorbic acid and citric acid in urine were measured by a commercial ascorbate assay kit (Cayman Inc., USA) and citrate assay kit (BioVision Inc., USA), respectively.
Statistics
LC-MS and 1H-NMR spectrum datasets were processed by SIMCA-P + (version 12.0, Umetrics, Umeå, Sweden). For multivariate statistical analysis, principal component analysis (PCA) and partial least squares discriminant analysis (PLS-DA) were used for regression in MS and NMR spectra. The loading plot and S-plot were then used to find all metabolites that contributed to the separation between the studied groups. Selected peaks were assigned by comparison with a spectrum of standard compounds (www.bml-nmr.org) (23) and Chenomx NMR software suite (Version 7.1, Chenomx, Inc.). To determine which metabolites contribute most to the discrimination between RCVS patients and controls, we performed a variable importance in the projection (VIP) analysis. Metabolites were discriminated as potential RCVS-relevant signatures by a VIP cut-off value of 1. Random forest (RF) classification and mean decrease accuracy were used to further refine the features that could discriminate the metabolic changes, and out of bag (OOB) error of RF is 0.1. To increase the reliability of RCVS prediction, we calculated the receiver operating characteristic (ROC) curve analysis on the basis of a logistic regression model to determine the area under the curve (AUC) as a measure for the predictive ability of metabolites in RCVS. Analyses involving the correlations between clinical parameters and the urine metabolites were performed with the IBM SPSS Statistics software package, version 23.0. Descriptive statistics are presented as the mean ± standard deviation, median (range), or as the number (percentage). Student’s t, Fisher’s exact, or chi-square tests were used for comparisons between groups when appropriate. Paired t-test or Wilcoxon signed-rank test was used to compare paired data. The correlations between clinical parameters and the urine metabolites were evaluated with multivariable linear or logistic regression. All calculated p-values were two-tailed. Statistical significance was defined as a p < 0.05.
Bioinformatics analyses
Biological pathway and functional annotation of metabolomics data were further analyzed by Ingenuity Pathway Analysis (IPA) Software (Ingenuity Systems, Mountain View, CA) and MetaboAnalyst (24).
Results
Participants
The characteristics of the current cohort.

Only in females; bEvaluated by transcranial and carotid color-coded sonography.
ICH: intracerebral hemorrhage; PRES: posterior reversible encephalopathy syndrome; TCH: thunderclap headache; VMCA, VACA, VPCA, VBA: mean flow velocity of the middle cerebral artery, anterior cerebral artery, posterior cerebral artery and basilar artery, respectively.
Metabolite profiling in urine of RCVS patients using NMR- and LC-MS-based metabolomics
Principal component analysis (PCA) of 1H-NMR data showed a trend of inter-group separation, as shown on the scores plot in Figure 1(a). The plot of PC1 versus PC2 scores shows separation between the control and RCVS patients clearly in PC2 but only slightly in PC1 (Figure 1(a)). Scores plot obtained from partial least squares discriminant analysis (PLS-DA) showed clear clustering of samples into two groups (Figure 1(b)), suggesting that urine metabolomes differed between RCVS patients and controls. In addition, PCA and PLS-DA of LC-MS data both indicated a trend of inter-group separation (Figure 1(c) and 1(d)).
Multivariate analysis for RCVS patients and controls. Untargeted metabolomics of RCVS patients and non-RCVS controls were analyzed by 1H-NMR and LC-MS/MS. PCA and PLS-DA score plots were based on 1H-NMR data (a) and (b), and LC-MS/MS data (c) and (d) of urine samples from control group (red) and RCVS group (green).
Identification of potential metabolites with discriminative features
Five metabolites identified by 1H-NMR spectroscopy showed remarkable differences between the two groups (Supplemental Table 1): citrate, guanidinoacetate, hippurate, alanine, and valine. In addition, the LC-MS/MS metabolomic study showed D-xylose, D-ribose, DL-arabinose, 1,3,7-trimethyluric acid, uridine, tehalose, lactulose, kojibiose, 3-methoxy-4-hydroxymandelate, and glycylproline were significantly increased in RCVS patients. In contrast, ascorbic acid, D-glucurono-6,3-lactone, alanine, carnitine, 3-hydroxytetradecanolycarnitine, hippurate, mannitol, D-sorbitol, dulcitol, and 4-methoxyphenylacetic acid were decreased in RCVS patients (Supplemental Table 1).
From the results of 1H-NMR data, citrate and hippurate were identified as important contributors that contribute most to the discrimination between RCVS patients and controls (Figure 2(a)). From LC-MS results, citrate, betaine, valine, 1,3,7-trimethyluric acid, D-glucurono-6,3-lactone, acetylcarnitine, 3-pyridylacetic acid, ascorbic acid, isobutyrylcarnitine, butyrylcarnitine, D-threo-isocitric acid, D-xylose, D-ribose, DL-arabinose, paraxanthine, pyrrolidonecarboxylic acid, and hippurate were identified (Figure 2(b)). Combining the above data, we selected 13 metabolites as candidates that simultaneously fulfilled the criteria of having VIP > 1 and p < 0.05 (1,3,7-Trimethyluric acid, ascorbic acid, butyrylcarnitine, citrate, D-glucurono-6,3-lactone, D-threo-isocitric acid, D-xylose, D-ribose, DL-arabinose, hippurate, isobutyrylcarnitine, and pyrrolidonecarboxylic acid) (Supplemental Table 1). ROC curve analysis was then used to evaluate the strongly predictive AUC (>0.8) between RCVS patients and controls.
VIP scores from PLS scores of urine samples from RCVS patients and controls. VIP analysis based on the weighted coefficients of the PLS-DA model from 1H-NMR (a) and LC-MS/MS (b) were used to rank the contribution of metabolites to discriminate RCVS patients and controls.
Random forests (RFs) classification and feature selection
Figures 1 and 2 show that all data yielded good classification accuracy. RF models coped well with high dimensional data sets and, more importantly, also provided an insight into the structure of the data under study by quantifying the confidence in classification voting and indicating the importance of each variable for the classification task. To determine which metabolites were more important for differentiating RCVS patients and controls, the top ranked differential metabolites in the respective models were selected according to the percent decrease in a mean of decrease accuracy, when the trial was performed in the absence of the metabolites (Figure 3). The 10 most important NMR-derived metabolites for the classification of RCVS are depicted in Figure 3(a). The top 15 LC-MS/MS-derived metabolites, which contribute most to the classification of RCVS, were shown in Figure 3(b). Alanine and D-glucurono-6,3-lactone were the highest-ranked metabolites by 1H-NMR and LC-MS, respectively. Finally, we integrated the results of Figures 1–3 and Supplemental Table 1 to narrow down the candidate metabolites. We identified hippurate, citrate, 1,3,7-trimethyluric acid, ascorbic acid, D-glucurono-6,3-lactone, and D-threo-isocitric acid as potential RCVS-relevant signature (Table 2). Notably, p values of 1,3,7-trimethyluric acid, ascorbic acid, and D-glucurono-6,3-lactone, were lower than 0.01.
Random forest (RF) assay for RCVS patients and non-RCVS controls. An RF importance measure was used to rank metabolites according to their prognostic importance for RCVS status by 1H-NMR (a) and LC-MS/MS (b). The plot shows each variable on the y-axis, and their importance on the x-axis. They are ordered top to bottom as more to less important. The colored boxes on the right indicate the relative levels of the corresponding metabolite between RCVS patients and controls. Potential biomarker candidates in RCVS. Both in 1H-NMR and LC-MS.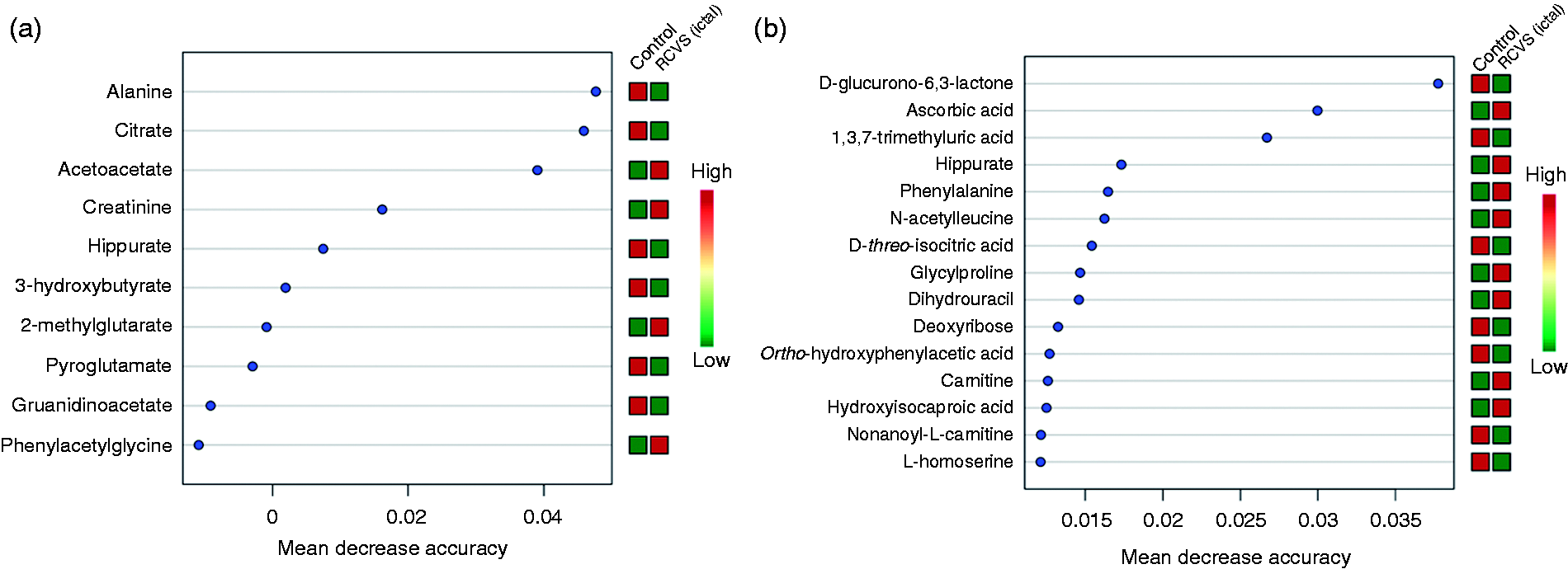
Metabolic pathway analyses of RCVS-related metabolite changes
Pathway analysis using MetPA revealed that six major metabolic pathways; that is, synthesis and degradation of ketone bodies, glycine, serine and threonine metabolism, galactose metabolism, phenylalanine metabolism, ascorbate and aldarate metabolism, and tyrosine metabolism, might be disturbed in RCVS (impact >0.1, p < 0.05) (Figure 4(a)). Furthermore, based on metabolite set enrichment analysis and IPA-curated disease ontology, these pathways are primarily correlated to organismal injury and abnormalities, tyrosine hydroxylase deficiency, and dopamine beta-hydroxylase deficiency, seizure, schizophrenia, neuroblastoma-related disease, glomangioma, neurological disease, and immunological disease (data not shown). Of these, only seizures have been clinically shown to have a close relationship with RCVS (4).
Summary of pathway related to RCVS and metabolite-metabolite interaction network analysis. (a) Network pathways were identified by MetaboAnalyst software. Network analysis of differentially expressed metabolites was performed by ingenuity pathway tools (www.ingenuity.com) on metabolites annotated in the Ingenuity database. Significant changes in (b) vitamin and mineral metabolism and the free radical scavenging network were identified. The red nodes represent upregulated metabolites and green nodes downregulated metabolites. The red words are hot spots of the network.
Metabolic network differences between RCVS patients and controls
To further investigate the comprehensive relationship of candidate metabolites to RCVS, the IPA network algorithm tool was used to build a connectivity network. Pathway analysis was applied to the data contained in Supplemental Table 1 to highlight possible molecular mechanisms underlying the differential expression of metabolites in RCVS. Known mutual interactions among differentially expressed metabolites were used to construct metabolite networks, including direct and indirect relationships. This network analysis exhibited significant perturbation on the metabolic network, vitamin and mineral metabolism and free radical scavenging-related network (Figure 4(b)).
Validation of selected urine metabolite for RCVS
We validated the six urine metabolites with the highest discriminative utility (p < 0.05, VIP > 1, and AUC > 0.8) in the replication cohort (i.e. 47 RCVS patients with ictal samples (with 40 with matched remission samples) and 47 controls) (Table 2). In the ictal stage, the urine levels of 1,3,7-trimethyluric acid and D-threo-isocitric acid in RCVS patients were higher than in controls; hippurate, citrate, D-glucurono-6,3-lactone, and ascorbic acid levels were lower in RCVS patients than in controls (Figure 5). Moreover, we found that levels of four culled metabolites (1,3,7-trimethyluric acid, D-glucurono-6,3-lactone, ascorbic acid, and citrate) recovered in remitted RCVS patients compared to their ictal stage (paired t-test). However, levels of D-threo-isocitric acid and hippurate did not recover but ran a reversed trend in the remission stage. In a smaller subset including only patients with purely cephalalgic RCVS (i.e. excluding patients with complications), we found that most of the urine levels of the metabolites were significantly different in patients than those in controls, although the difference in hippurate and D-glucurono-6,3-lactone became borderline (Supplemental Table 2). Thus, we deemed that 1,3,7-trimethyluric acid, D-glucurono-6,3-lactone, ascorbic acid, and citrate were promising as an RCVS-relevant signature.
Validation of selected urine metabolites for RCVS. Selected metabolites from the discovery cohort, including (a) 1,3,7-trimethyluric acid, (b) ascorbic acid, (c) citrate, (d) D-glucurono-6,3-lactone, (e) D-threo-isocitric acid, and (f) hippurate were validated in non-RCVS controls, RCVS patients, and remitted RCVS patients.
8-iso-prostaglandin F2 (8-Iso-PGF2α) level in plasma
Previously, we found that the level of 8-iso-PGF2α, one of the oxidative stress markers and a potent vasoconstrictor, was higher in urine samples in patients with RCVS and highly correlated with disease severity of RCVS (14). Therefore, in addition to urine metabolites, we also checked the plasma level of 8-iso-PGF2α for comparison. The result revealed that the level of 8-iso-PGF2α in the RCVS ictal stage was higher than in controls (Supplemental Figure 1).
Clinical correlates
Although the levels of the metabolites could well differentiate patients from controls and had ictal-interictal differences, none of these metabolites were associated with clinical parameters, including age, gender, headache triggers, blood pressure surge, numbers and durations of thunderclap headaches prior to sample collection, flow velocities of individual cerebral blood vessels, and presence of complications (see Supplemental Table 3 for the correlation between metabolites and major clinical parameters). There was significant difference between the percentages of migraine history in patients with RCVS and controls; however, there was no significant differences in metabolite levels between RCVS patients with or without migraine (Supplemental Table 4).
Discussion
This study combined 1H-NMR and LC-MS/MS to discover potential candidates for urine metabolomic signature in RCVS patients and then further validated these metabolites. In the discovery phase, six metabolites, hippurate, citrate, 1,3,7-trimethyluric acid, ascorbic acid, D-glucurono-6,3-lactone, and D-threo-isocitric acid, were identified as the most discriminative metabolites for RCVS. Of them, hippurate, citrate, ascorbic acid, and D-glucurono-6,3-lactone were significantly lower, and 1,3,7-trimethyluric acid and D-threo-isocitric acid were higher in RCVS patients. These metabolites are related to two metabolic networks including free radical scavenging and vitamin/mineral metabolism. Finally, those metabolites were validated in RCVS patients with their remitted samples. Of note, four of the six metabolites, 1,3,7-trimethyluric acid, D-glucurono-6,3-lactone, ascorbic acid, and citrate, were recovered during the remission stage. Those metabolites provide insights to identify the pathogenesis of RCVS and might be used as potential disease-relevant signatures and therapeutic targets if further validated in different patient populations.
The implicated free radical scavenging and vitamin/mineral metabolism signaling pathways are consistent with our previous findings showing that the level of 8-iso-prostaglandin F2 (8-iso-PGF2α) in urine was higher in patients with RCVS during the ictal stage than that in their remission stage and in controls (14). The non-enzymatic free radical peroxidation product of arachidonic acid, 8-iso-PGF2α, is not only a reliable marker for oxidative stress (14) but also a potent vasoconstrictor that correlated with the vasoconstriction in patients with RCVS and aneurismal subarachnoid hemorrhage (25,26). Consistent with the current study in urine, we also found that RCVS patients during the ictal stage had higher 8-iso-PGF2α in plasma. Interestingly, ascorbic acid has been reported to reduce 8-iso-PGF2α by quenching free radicals (27), and our metabolomic analysis showed decreased ascorbic acid in patients with RCVS during the ictal stage, returning to normal range during the remission stage. Furthermore, D-glucurono-6,3-lactone is an important precursor in ascorbic acid biosynthesis (28,29). Meanwhile, L-arginine could undergo decarboxylation to L-agmatine and metabolism to guanidinoacetate, which have also been shown to inhibit NO synthases (30). Notably, levels of guanidinoacetate and D-glucurono-6,3-lactone were lower in RCVS patients. In addition, as we have demonstrated, levels of endogenous 1,3,7-trimethyluric acid, a methylated analogue of uric acid, were elevated in RCVS patients during the ictal stage and returned to normal level in the remission stage. 1,3,7-trimethyluric acid is a potent antioxidant (31) and its analogues could efficiently scavenge hydroxyl radicals and inhibit lipid peroxidation (32). We speculate that the upregulation of 1,3,7-trimethyluric acid during the ictal stage is an endogenous protective mechanism to counteract increased oxidative stress.
A number of the “hub” molecules in our network analysis, including Ca2+, cyclic AMP, nitric oxide, prostaglandin E2, and ascorbic acid, are associated with endothelial dysfunction or sympathetic overactivity that has been demonstrated to be important in the pathogenesis of RCVS (13,15) . Of note, calcium channel blockers have been demonstrated to be effective in aborting thunderclap headaches in RCVS patients (3,33), which is consistent with the finding of Ca2+ to be in the hub of putative networks. In addition, our network analysis showed dopamine and its precursor, L-dopa, to be involved in vitamin and mineral metabolism. We speculate that it might be associated with blood pressure surge or pain modulation in RCVS patients, but no clinical or basic reports have yet described how dopamine levels fluctuate during RCVS. The association between these metabolites and RCVS were indirectly derived from pathway analysis, which remains to be validated in future.
In this study, RCVS-relevant metabolomic signatures were obtained by combining two different techniques with successful replication in independent cohorts. Urine accommodates many bio-reactions and a final by-product of metabolism, which may reflect the status of the human body, such as pregnancy and aging (34); the identified metabolites might be highly relevant to the pathogenic mechanism of RCVS. However, the lack of correlation between the metabolites and velocities suggests that although these metabolites were elevated during the ictal stage, they might not be directly involved in vasomotor control. Rather, these metabolites are likely the downstream products of certain (yet-to-be-explored) biological pathways that were involved in the pathogenesis. In addition, our study also has limitations. First, obtaining an accurate and sensitive peak extraction for target compounds is a crucial step in urine metabolite screening, and commonly creatinine is used to normalize the urine data. However, the measured creatinine levels varied a lot in our RCVS individuals. This normalization may lead to under- or over-estimation of the measured metabolites. Therefore, we used TIPick, an in-house package (35), instead of creatinine for sample normalization. Second, the exploratory study is for hypothesis generating. Hence, the results were not corrected by multiple comparison. In addition, all patients recruited in this study were Taiwanese and the sample size was limited; thus, the results may not be generalizable to patients of other ethnicities. Further studies from independent samples of large scale will be needed to validate the utility of these signatures. Third, we could not completely exclude the possibility that some patients with RCVS or controls had taken vitamin or antioxidant supplements prior to entering the study, thus confounding the results. Nevertheless, the finding that these metabolites could normalize in RCVS patients during the remission stage is a strong proof that these results are highly relevant to RCVS. Fourth, the urine samples were collected prior to the initiation of nimodipine treatment. We could not completely exclude the possibility that some patients with RCVS had taken painkillers prior to entering the study and these drugs could have confounded the results, although direct metabolites of painkillers were not detected in our samples. Furthermore, as far as we know, current evidence supporting the interaction between these metabolites and acetaminophen or non-steroidal anti-inflammatory drugs is scarce. Finally, it is possible that migraine might contribute to the differences between patients and controls; however, the lack of differences between RCVS patients with migraine and RCVS patients without migraine suggested that migraine might not be an important contributor to the production of these metabolites.
In summary, we have performed, for the first time, a comprehensive analysis of urine metabolites of RCVS patients. Using discovery, validation, and remission cohorts, we have identified multiple urine metabolites associated with the disease process. Our study demonstrated that the urine metabolomic approach provides a non-invasive and promising tool to discover potential metabolomic signature in RCVS patients and new insight into the pathophysiology of RCVS.
Clinical implications
Six metabolites, hippurate, citrate, 1,3,7-trimethyluric acid, ascorbic acid, D-glucurono-6,3-lactone, and D-threo-isocitric acid, were identified as potential urine biomarkers for RCVS. These selected metabolites are related to six pathways that are responding to two metabolic networks including vitamin and mineral metabolism and the free radical scavenging-related network, which were disturbed in RCVS patients.
Supplemental Material
CEP897621 Supplemental Material1 - Supplemental material for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome
Supplemental material, CEP897621 Supplemental Material1 for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome by Wei-Hsiang Hsu, Shuu-Jiun Wang, Yen-Ming Chao, Chao-Jung Chen, Yen-Feng Wang, Jong-Ling Fuh, Shih-Pin Chen and Yun-Lian Lin in Cephalalgia
Supplemental Material
CEP897621 Supplemental Material2 - Supplemental material for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome
Supplemental material, CEP897621 Supplemental Material2 for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome by Wei-Hsiang Hsu, Shuu-Jiun Wang, Yen-Ming Chao, Chao-Jung Chen, Yen-Feng Wang, Jong-Ling Fuh, Shih-Pin Chen and Yun-Lian Lin in Cephalalgia
Supplemental Material
CEP897621 Supplemental Material3 - Supplemental material for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome
Supplemental material, CEP897621 Supplemental Material3 for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome by Wei-Hsiang Hsu, Shuu-Jiun Wang, Yen-Ming Chao, Chao-Jung Chen, Yen-Feng Wang, Jong-Ling Fuh, Shih-Pin Chen and Yun-Lian Lin in Cephalalgia
Supplemental Material
CEP897621 Supplemental Material4 - Supplemental material for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome
Supplemental material, CEP897621 Supplemental Material4 for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome by Wei-Hsiang Hsu, Shuu-Jiun Wang, Yen-Ming Chao, Chao-Jung Chen, Yen-Feng Wang, Jong-Ling Fuh, Shih-Pin Chen and Yun-Lian Lin in Cephalalgia
Supplemental Material
CEP897621 Supplemental Material5 - Supplemental material for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome
Supplemental material, CEP897621 Supplemental Material5 for Urine metabolomics signatures in reversible cerebral vasoconstriction syndrome by Wei-Hsiang Hsu, Shuu-Jiun Wang, Yen-Ming Chao, Chao-Jung Chen, Yen-Feng Wang, Jong-Ling Fuh, Shih-Pin Chen and Yun-Lian Lin in Cephalalgia
Footnotes
Author contributions
Study concept and design: YLL, SPC, SJW conducted and acquired data; analysis: WHH, YMC, CJC, YFW, JFL and SPC; drafting of the manuscript: WHH, YFW, JLF and SPC; review and editing the manuscript: YLL, SJW, SPC.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ministry of Science and Technology of Taiwan MOST 105-2320-B-039-055-MY3, 105-2811-B-039-026, 106-2811-B-039-014 and 106-2320-B-039-053 (to YLL and WHH); Brain Research Center, National Yang-Ming University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan (to SJW and SPC); Taipei Veterans General Hospital [V100E6-001, V106C-117] (to SWJ and SPC); MOST 104-2314-B-010-015-MY2, MOST 104-2314-B-075-006 -MY3, and MOST 107-2314-B-010-021 -] (to SWJ and SPC); and Ministry of Health and Welfare, Taiwan [MOHW 103-TDU-B-211-113-003, MOHW 104-TDU-B-211-113-003, MOHW 105-TDU-B-211-113-003] (to SJW). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
