Abstract
Background
Often concussion/mTBI triggers a chronic headache syndrome called persistent post-traumatic headache (P-PTH) that can last from months to years post-injury, and produce significant disruption of childhood education, social interaction and development. Although prevalent and highly disabling, P-PTH is underrepresented in headache and pain research and lacks clear definition and pathophysiology. Clinical presentation of P-PTH frequently resembles that of other headache disorders, like migraine, yet the pathophysiological mechanisms are distinct and not fully understood, making the disorder difficult to treat in the clinical setting.
Methods
In a retrospective analysis of 1506 pediatric patients attending Boston Children’s Hospital clinics, demographic trends, symptom features, and the influence of sex on clinical presentation of PTH are presented. We compare clinical characteristics of P-PTH with a published cohort of migraine patients to evaluate the clinical features that are unique to P-PTH.
Results
Findings show that despite equivalent representation of sex in the clinic, P-PTH is expressed more in females than males and is weighted towards somatic symptoms. Relative to migraine, PTH is less associated with a family history of headache.
Conclusions
The ability to identify persons with PTH can help manage risk factors and identify persons likely to develop persistent post-concussion symptoms.
Introduction
Concussions, also referred to as mild traumatic brain injuries (1) are acute traumatic events that largely resolve within the hours to days after injury. However, the acute recovery from a concussion may not be complete, with 15–25% of individuals experiencing some level of persistent concussion symptoms (2,3) and females experiencing symptoms more severely than males (1). These symptoms can include difficulty with working memory, depression, or general fatigue that may interfere with school (4), work (5) or play (6), especially within the pediatric setting. Post-traumatic headache represents one of the major chronic post-concussion symptoms, with one study reporting that 7.8% of children continue to experience headaches at 3 months post-injury (7). Another study conducted with children 18 years and younger concluded that 13.7% of school-aged patients and 11% of overall patients were experiencing symptoms at 3 months post-injury and 2.3% remaining symptomatic at 1 year post injury (8). The high incidence of persistent post-traumatic headache (PTH) and associated morbidity demand further understanding of this clinical condition and its unique clinical features.
The proper clinical management of a concussion is contingent upon a detailed understanding of its clinical features and their longitudinal progression. To date, updated consensus documents on concussion in sport (1) have shown that proper recognition of symptoms including headache after a concussion are critical towards the immediate identification and removal of athletes or patients from stressors (e.g. sport or classrooms). The immediate identification of such symptoms in the acute setting is critical, as the presentation of specific symptoms in the immediate post-concussion period can be risk factors for the persistence of symptoms beyond the 1 month post-concussion period. Headaches are one of the largest risk factors for the persistence of symptoms in persons with a concussion (9). Notably, the distinction between headaches emerging from a concussion and those from a non-concussive event are unclear. Some research has shown a potential link between concussion and a family history of migraine headache (10); however, imaging literature supports a distinction between persistent post-traumatic headache and migraine (11). A thorough understanding of the clinical features of post-traumatic headache is required for proper clinical treatment.
Here, we chose to execute a cross-sectional retrospective chart review (RCR) as they can prove useful when investigating clinical presentations of a specific disease, disorder, or patient population to inform future questions for related prospective studies. RCRs also allow data extraction from large-scale sample sizes with significant statistical power to inform questions for prospective clinical research studies that cannot obtain such numbers. A previous RCR by Wilcox et al. (12) focused on two factors, sex and age, in the clinical presentation of pediatric migraine. With over 300 migraine patients included for data collection and analysis, they revealed that the sex distribution of migraine significantly changed at puberty from a 1:1 ratio female to male to a 2:1 ratio, indicating a genetic or hormonal change in female neurophysiology at puberty (12). The RCR by Wilcox et al. (12) provides a strong foundation for successfully analyzing and communicating retrospective data while answering basic yet crucial questions about PTH presentation in children and adolescents. We envisage this RCR will provide headache and sports medicine fields with novel fundamental findings on the clinical presentation of pediatric PTH.
In this retrospective chart review (RCR), we examined demographic features and symptom characteristics of pediatric PTH to further understand this complex disorder with two specific aims: To identify demographic/biological trends and clinical features of PTH, and to compare clinical manifestations of PTH with those of a migraine cohort previously reported by Wilcox et al. (12). We hypothesized that, like migraine, PTH will occur most frequently in female adolescents (age 12–17) and that a greater proportion of PTH patients will report a family history of headache. We also predicted that symptoms common in migraine, such as nausea, irritability, blurred vision, photophobia, and/or phonophobia would also be commonly reported symptoms in PTH, and that unique clinical manifestations would help distinguish PTH patients from acutely recovering concussion patients and migraine patients.
Methods
Overview
We performed a retrospective chart review for pediatric patients seen in Boston Children’s Hospital Sports Medicine outpatient clinics over a one-year period between 1 June 2015 and 1 June 2016 (see Figure 1). The cohort sample consisted of patients who underwent an evaluation for post-concussive symptoms (PCS) within the one-year period. These patients were screened for PTH and included or excluded accordingly (Table 1). Patients were identified according to the ICHD-3 criteria for PTH, followed by a thorough analysis of patient demographics and symptom features. Approval from the Boston Children’s Hospital Institutional Review Board was obtained prior to conducting the review.
Experimental workflow. Data for the RCR was extracted from Boston Children’s Hospital Sports Medicine physician notes documented in PowerChart electronic medical record. The time frame for this RCR was a one-year period between June 2015 and June 2016. A total of 1506 patients reporting concussion-like symptoms were screened for the presence of post-traumatic headache lasting greater than 3 months and meeting the ICHD-3 and inclusion criteria. In all, 157 patients met these criteria and were included for further data collection. A further 1349 patients were removed for meeting exclusion criteria. Participant demographics. The relative incidence of PTH (10.4%) is less than that of migraine (22.7%). Age distribution is more heavily dominated by adolescents (12–17 years) in PTH. The sex distribution is almost identical, with a significant female dominance in both PTH and migraine. Many more migraine patients than PTH patients reported a positive family history of headache.

Database searches
Informatics for Integrating Biology and the Bedside (i2b2) and PowerChart databases were used for the RCR. i2b2 is a biomedical computing program used for categorizing patient data, providing researchers and clinicians with an efficient means to sort through patient charts. An i2b2 query was used to determine the number of concussion clinic visits to BCH Sports Medicine from 1 June 2015 to 1 June 2016. PowerChart is an electronic medical record database used by several institutions, including BCH, to file clinic charts, physician notes, and other patient data. Sports Medicine clinic notes and Post-Concussion Symptom Score (PCSS) forms from PowerChart were the main resources for data collection.
Inclusion and exclusion criteria
The International Classification of Headache Disorders (ICHD) has stated that “to be consistent with ICHD-II diagnostic criteria for chronic post-traumatic headache and with the time interval used in the diagnoses of other secondary headache disorders, 3 months is the time interval after which headache attributed to trauma or injury to the head is considered persistent” (13). Patients were screened and included if they met the following criteria: Age ≥ 8 and ≤ 21 years old; headache lasting more than 3 months; 3–12 month PCSS in chart; formal concussion diagnosis; concussion within 3–12 months of clinic note; headache related to concussion in line with ICHD-3 criteria (determined at time of clinical examination); no prior headache disorder diagnosed; meets ICHD-3 PTH criteria. Demographic and symptom data were collected for included patients. Specifically, the following information was collected for each patient meeting the inclusion criteria: a) Sex, race, age (at time of injury), height, weight, BMI; b) significant medical history, previous and current medications, headache history, family headache history, number of lifetime concussions; c) date of injury, cause/mechanism of injury, loss of consciousness (LOC), prescribed medications for concussion symptoms, post-concussion symptoms score (PCSS total scores (initial visit score and 3-month to 12-month post-injury score)); d) 3 month to 12 month PCSS data, including: Headache, pressure in head, neck pain, nausea, photophobia, phonophobia, balance, dizziness, blurred vision, being more emotional, irritability, sadness, nervousness/anxiety, confusion, fogginess, difficulty concentrating, difficulty remembering, not feeling right, feeling slowed down, drowsiness, fatigue, difficulty falling asleep, sleeping more than usual.
Statistical analysis
Research findings are outlined and compared using a descriptive statistical analysis. Biological trends and symptom distribution were analyzed using pie charts, bar graphs, means and standard deviations.
Results
Incidence of PTH
We screened concussion patients from a one-year period to examine the demographic and clinical features of PTH. Based on previous data, we expected that between 7% to 15% of concussion patients would develop PTH (7,8). We found that, of the 1506 concussion patients screened from the one-year period, 157 (10.4%) developed PTH and were included for data collection and analysis (Figure 2). The cohort sample included patients with a formal concussion diagnosis, headache descriptors meeting the ICHD-3 criteria for PTH and having a 3- to 12-month PCSS form available in PowerChart. Of the included subjects, only 12 had no prior history of concussion; all subjects presented with their most recent concussion being the cause of the PTH. Other subjects ranged from one to five prior concussions (1 = 84; 2 = 30; 3 = 18; 4 = 11; 5 = 2). The most recent concussion was noted as precipitating the PTH symptoms. Participants were on average evaluated 122.8 days (SD = 38) after date of injury and 142 were sport-related injuries. See Supplemental Table 1 for complete breakdown of medications and mechanisms of injury for each subject).
Exclusion criteria. Ten percent of concussion patients met the inclusion criteria for this RCR (a) and the majority of excluded patients acutely recovered under 3 months (3mo) following concussion (b). The 157 included patients represent PTH patients experiencing headache symptoms greater than 3 months and meeting the ICHD-3 and inclusion criteria. Fifty-nine percent of excluded patients experienced headache for under 3 months, meaning they were acutely recovered. Only 3% of patients were excluded due to the presence of a previous headache disorder. Other exclusionary reasons (n = 511) include insufficient PowerChart notes, being outside the specified age range, headache unrelated to concussion (i.e. influence of previous health disorder or unparalleled stress), or no formal concussion diagnosis.
Reported symptoms by gender. Data was subdivided by sex and the percentage of patients reporting individual symptoms was calculated. More females report pressure in head, nausea, blurred vision, and anxiety/nervousness. More males report confusion, fogginess, difficulty concentrating, and drowsiness.

Sex distribution in concussion
To confirm demographic claims about PTH, it is essential to first understand the demographic distribution of concussion. We noted the sex of all screened concussion patients and found an even distribution of male and female concussion patients (n = 1506): 786 (52%) were female and 720 (48%) were male (Figure 3). In contrast, the sex distribution of patients experiencing PTH was 113 (72%) female and 44 (28%) male. These data suggest that PTH is most prevalent in females and mirrors findings from migraine (17).
Sex distribution. The sex distribution of concussion patients (a) PTH patients (b) and migraine patients (c) is outlined. A significant shift from even distribution in concussion to majority female in PTH is present. A female dominance is present in both PTH and migraine. Thus, concussions requiring medical attention occur equally in females and males, yet PTH and migraine develop most frequently in females.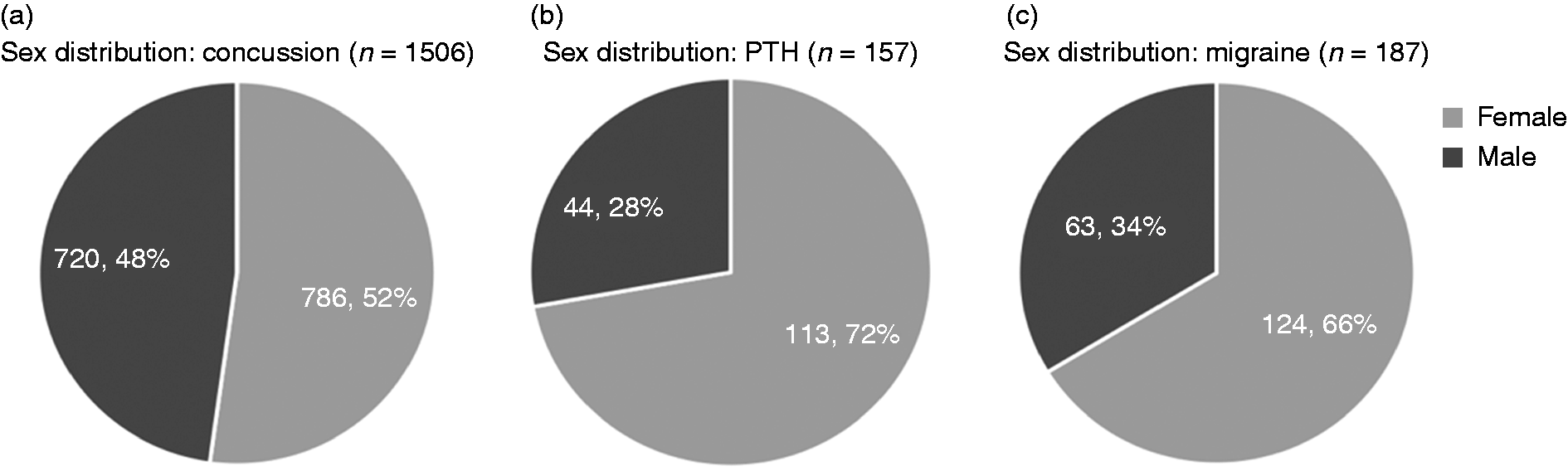
Age and family headache history
To compare PTH with migraine and understand differences between the two disorders, we paralleled PTH age distribution and family headache history reports to those of a published migraine cohort. We expected a greater adolescent dominance in PTH and an equal reporting of family headache history. Data extraction revealed an adolescent dominance in PTH (73%). However, only 18% of PTH patients reported a positive family headache history, which varies significantly from migraine (87%) (Table 1). Age distribution and adolescent dominance was expected; however, PTH patients reported a lower incidence of family headache history than expected. The relative incidence of PTH (10.4%) is less than that of migraine (22.7%). Age distribution is more heavily dominated by adolescents (12–17 years) in PTH. Many more migraine patients than PTH patients reported a positive family history of headache.
PTH symptoms and phenotype
In addition to demographic features, it is important to analyze clinical features and symptom distribution in PTH. The presence of each symptom type and its associated category was collected to analyze symptom trends in PTH. We expected to see a higher presence of migraine-like symptoms, most commonly found in the somatic domain, to be most prevalent in PTH. Symptom distribution of PTH was nearly even across domains with a slightly greater incidence of somatic symptoms (Figure 4). However, when broken down into specific symptoms, distribution varied with higher relative frequencies of headache, dizziness, balance, irritability, difficulty concentrating, and fatigue (Figure 4). These findings were expected, as headache and many associated symptoms reported in PTH have also been reported in migraine (17). Great variability exists in PTH symptom presentation, reflecting the complicated nature of the disorder. Somatic symptoms were examined further to understand headache characteristics and severities in PTH. PCSS forms allow patients to report symptom severities from 0–6. The reported severities of each somatic symptom were plotted to analyze relative frequencies and trends (Figure 6). Overall, headache proved to be the most severe somatic symptom, followed by pressure in the head. This was expected. PTH patients also reported low presence and low severity for nausea/vomiting, which was unexpected, as nausea is a common migraine symptom (Figure 4) (12). These data align with headache being the most debilitating symptom following concussion, specifically in patients experiencing prolonged symptoms.
PTH symptom distribution. Post-concussive symptoms are categorized into somatic, vestibular, emotional, cognitive, and sleep domains in the Post-Concussion Symptom Score sheet (PCSS). A nearly even distribution of symptom types was reported for PTH patients, indicating that a significant presence of associated symptoms accompanies post-traumatic headache (a). Headache, dizziness, irritability, difficulty concentrating, and fatigue were among the most prevalent symptoms ((b)–(f)). Sex-based PTH symptom distribution. Symptoms were noted on the PCSS form. A greater percentage of females report somatic, vestibular, and emotional symptoms. More males report cognitive and sleep symptoms than females.

Sex differences in PTH symptoms
Our data confirm that (i) PTH is more prevalent in females, implying a sex-related difference in PTH development and (ii) some PTH symptoms are more prevalent than others. Therefore, we compared symptom distribution and severity between males and females to investigate potential biological differences in PTH symptomology. Based on previous research demonstrating a female dominance in anxiety, especially during migraine, we expected to see a female majority reporting emotional symptoms in PTH (12,14). We also expected to observe a male dominance in cognitive symptom reporting, since cognitive disorders such as attention-deficit/hyperactivity disorder (ADHD) are more prevalent in males (15), and that sex distribution for individual somatic symptoms would resemble those associated with migraine, as reported by Wilcox et al. (12). This research reported a female dominance in phonophobia and a male dominance in nausea/vomiting (12). Overall, percentages of somatic, vestibular, and emotional symptoms reported between females and males were similar. More males reported cognitive and sleep symptoms (Figure 5). When broken down into individual symptoms, more females reported pressure in the head, nausea/vomiting, blurred vision, and anxiety/nervousness. More males reported confusion, fogginess, difficulty concentrating, and drowsiness (Table 2). These findings agree with our predictions that more females would report anxiety and more males would report cognitive deficits. However, these findings differ from migraine since an equal distribution of females and males reported phonophobia, and more females reported nausea/vomiting.
Male versus female symptom severity
Since the percentage of patients reporting specific symptoms in PTH varies between males and females, we wanted to compare symptom severities between males and females. We expected an overall equal reporting of symptom severities between males and females; however, we also expected average severities for emotional and cognitive deficits to vary between the two groups. Data reveal relatively equal symptom severities reported between females and males with slightly greater severity reported by females for pressure in the head, nausea/vomiting, vestibular symptoms, and anxiety/nervousness (Figure 6, Table 2). Symptom severities were noted on the PCSS form. Data was subdivided by sex and the means and standard deviations of individual symptom severities were calculated. Headache is the most severe symptom in males and females. Overall symptom severity did not differ between males and females. All findings were expected, except for the equal reporting of cognitive severities by the two groups. These data suggest that although the types of symptoms reported by males and females varies in PTH, relative severities of these reported symptoms are nearly equal.
Somatic symptom reporting. Patients reported symptom severities on the PCSS. The PCSS scale ranges from 0 (no symptoms) to 6 (severe symptoms). Headache is not only the most prominent somatic symptom, it is reported as the most severe as well, with a distribution closer to normal compared to the skewed distribution of the other five somatic symptoms.
Discussion
Overview of results
The purpose of this chart review was to retrospectively investigate the clinical features of pediatric post-traumatic headache including biological and symptom trends. Our first goal was to identify the demographic and clinical features of PTH in a pediatric setting and explore the data to determine if a difference in prevalence existed between males and females. Our second goal was to explore the clinical manifestations of PTH within the context of a recently published migraine cohort for the similitude of the two clinical conditions. Findings provide insight into the unique nature of PTH relative to migraine in terms of headache characteristics and clinical presentation. We discuss our findings within the context of sex differences and clinical presentation.
Risk of developing PTH
A 10.4% prevalence of PTH following concussion/mTBI agrees with prior reporting that 8–15% of patients report PTH following concussion/mTBI. This value should likely be slightly higher as many excluded patients continued to report headache at 3 months post-injury; however, these patients had insufficient/missing data or reported a previous diagnosis of headache and were therefore excluded from the study. A 10.4% prevalence of PTH agrees with the findings of Barlow et al. (8), reporting an 11–14% presence of post-concussive symptoms, including PTH, at 3 months in a pediatric concussion/mTBI cohort (2). The low presence of PTH at 3 months supports previous data claiming that concussion symptoms generally resolve within a few weeks post-injury (16). However, the small percentage of patients reporting PTH at 3 months or longer post-injury report varying demographic characteristics, mechanisms of injury, and medical histories. This implies that an underlying neurophysiologic mechanism may predispose some patients to PTH more than others, despite the mechanism or severity of the initial injury.
Sex differences
We note an equal sex distribution in persons who reported to the BCH sports medicine clinic. This finding is notable, given that it conflicts with prior research showing a greater incidence of concussion in females (17–20); however, it should be viewed in light of our primary search findings being sensitive to patients who presented to this clinic and does not distinguish individuals who were either symptomatic or received continuing care. After application of study exclusion criteria and consistent with prior findings, females were found to dominate the PTH cohort at 72% in this RCR, implying that females have a greater predisposition to PTH than males. This finding agrees with our hypothesis that females are more likely to report PTH. This finding also agrees with other studies, revealing a female dominance in both PTH and migraine (12,21). However, our data reveal an even larger percentage of females reporting PTH compared to migraine (72% vs. 66%), and a greater difference between males and females reporting PTH (12,21). The significant difference in females versus males presenting with PTH implies that an environmental, genetic, or hormonal effect differs between the two groups, putting females at a higher risk for developing PTH. Potential mechanisms explaining the uneven sex distribution could be the fluctuation in female hormones, especially in adolescents during puberty and menstruation. Studies have shown that migraine, a headache disorder with almost identical symptomology to PTH, is more common in females after puberty and that migraine onset may be due to fluctuation in sex hormones, specifically estrogen (12,22). These findings indicate that the post-pubertal fluctuation of hormones in females may contribute to the increased incidence of pediatric PTH in females.
PTH symptomology differs between males and females
Overall symptom presentation between males and females differed slightly. Results including increased cognitive deficits in males and increased anxiety in females align with our hypotheses. Males reported greater cognitive and sleep dysfunction, which may be due to sex-related differences in the development or blood flow regulation of the frontal lobe, cerebellum, and brain stem (23,24). More females reported pressure in the head than males. This may be due to increased muscle tension resulting from the injury itself or secondary to comorbid anxiety, which is also reported more frequently by females. A greater reporting of anxiety/nervousness in females is expected, as research has exposed a greater overall incidence of anxiety in females (18). A greater presence of nausea/vomiting in females was unexpected. This finding may be due to the fluctuation of hormones in female adolescents (25) or to changes in neuroendocrine function after TBI (26); however, research has yet to explicitly examine sex differences in the incidence of neuroendocrine dysfunction after mTBI. Although males and females report numerous combinations of somatic, vestibular, emotional, cognitive, and sleep functions, overall differences were observed between both groups. These findings indicate that PTH pathophysiology may differ between males and females.
PTH vs. migraine
PTH and migraine share similar clinical presentation, but our data suggests they have pathophysiologic differences. The percentage of patients reporting positive family headache history was significantly lower in PTH than in migraine, suggesting that a genetic component is non-contributory in PTH, unlike migraine (12). This finding did not agree with our hypothesis that the majority of PTH patients would report a positive family headache history. Our study identified a female dominance in PTH, as in migraine, confirming our hypothesis that the disorder is more prevalent in females. Data also showed a consistent presence of migraine-like symptoms in PTH, which agrees with our hypotheses. These findings also imply that certain brain regions affected in migraine, specifically those regulating somatic symptoms, may also be affected in PTH (12). Contrarily, when comparing sex distributions in comorbid somatic symptoms between PTH and migraine, data revealed differences in the distribution of males and females reporting phonophobia and nausea/vomiting. Differences in family headache history reports and sex-related symptomology between PTH and migraine patients imply that, despite similar symptom presentation, the two disorders have distinct mechanisms.
PTH symptomology involves dysfunction of several brain systems
Our findings imply that several different neural networks and brain systems contribute to PTH symptomology. All patients reported headache, implying that concussion/mTBI likely triggers activation of the trigeminal system and changes in cerebral blood flow (23). Vestibular dysfunctions, such as difficulty balancing and dizziness, were also common in PTH implying a change in cerebellar functioning following concussion/mTBI (4). Emotional fluctuations causing irritability, sadness, and anxiety/nervousness in PTH patients are likely due partly to lifestyle changes required for treatment and partly to physiologic changes in the amygdala and hippocampus (27). Several cognitive dysfunctions were reported to be present in PTH, implying alterations in the frontal lobe and prefrontal cortex, controlling concentration and higher cognitive functioning and controlling memory in the temporal lobe (23). Lastly, sleep pattern disruptions and increased drowsiness/fatigue imply dysfunction of the hypothalamus and lower brain structures such as the cerebellum and brain stem (23,24). The multitude of symptoms reported and the potential dysfunction of several associated brain systems in PTH explains the debilitating nature of the disorder. These findings support the need for further investigation of PTH pathophysiology, targeting these brain regions and systems.
Limitations
Although this study offers novel findings, limitations must be addressed. The nature of patient-reported clinical data indicates subjectivity of symptom descriptors and pain severity. However, the PCSS form attempts to objectify symptom presence and severity by providing quantifiable symptom scores for each patient. Also, data was extracted from patient charts written by several different clinicians, meaning there is potential for unreported or misreported data. However, the conductance of clinical evaluations by a specialist staff from one clinic likely minimizes variability in patient reports. As data from the migraine cohort was retrieved from a published study, we were limited in the number of comparisons (e.g. patient phenotype) that could be performed. When determining family history of headache symptoms/disorders, we were not able to interview patients’ relatives or obtain clinical charts from family members. Lastly, data was obtained from a specialty clinic at a pediatric institution, indicating that findings may not be generalizable to the greater population of PTH patients. However, biological and symptom-related patterns are of importance for future studies, and findings agree with previous reports of PTH presentation (8,21).
Conclusions
In this retrospective study of pediatric PTH, we found a significant female dominance and presence of comorbid symptoms, some of which vary as a function of sex. Findings indicate that PTH pathophysiology may be influenced by sex-related biological factors and is likely clinically distinct from migraine. These findings have important implications for the development of effective treatments for PTH as well as return to play and return to school protocols that allow the safe resolution of activities in youth.
Clinical implications
PTH is expressed more in females than in males. PTH is less associated with family history of headache than migraine. Symptom distribution in persons with PTH is weighted towards somatic symptoms. Sex-dependent expression of PTH symptoms was present.
Supplemental Material
CEP896754 Supplemental Material - Supplemental material for Clinical features and sex differences in pediatric post-traumatic headache: A retrospective chart review at a Boston area concussion clinic
Supplemental material, CEP896754 Supplemental Material for Clinical features and sex differences in pediatric post-traumatic headache: A retrospective chart review at a Boston area concussion clinic by Haley McEvoy, David Borsook and Scott A Holmes in Cephalalgia
Footnotes
Acknowledgments
The authors would like to thank Dr. Hodkinson for his suggestions and guidance to the study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported in part by a grant to DB from National Institutes of Health (grant/award number: ‘#R501NS095655-04').
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
