Abstract
Background
Ictal neck pain is a frequent symptom reported by half of migraine patients. It is unknown if neck pain is caused by peripheral or central mechanisms. Neck muscle stiffness can be investigated with ultrasound shear wave elastography.
Objectives
To determine if migraine patients with ictal neck pain have stiffer neck muscles interictally compared with patients without ictal neck pain and controls.
Methods
This was a cross-sectional study investigating neck muscle stiffness, pressure pain thresholds and neck pain symptoms in 100 migraine patients recruited from a tertiary headache center and 46 controls.
Results
Patients with ictal neck pain had increased mean neck muscle stiffness interictally compared to both migraine patients without ictal neck pain (p = 0.018) and controls (p = 0.036). Muscle stiffness was negatively correlated with pressure pain thresholds in the neck in migraine patients with ictal neck pain (r = −0.292, p = 0.042). There were no differences in mean pressure pain thresholds between migraine subgroups.
Conclusions
Migraine patients with ictal neck pain have stiffer neck muscles interictally compared with migraine patients without ictal neck pain and controls measured with ultrasound shear wave elastography. The increased stiffness could be due to local alterations in the neck muscles.
Keywords
Introduction
The origin of migraine headaches is the subject of intense research and interest. During migraine attacks, neck discomfort, stiffness, and pain are very common features (1,2). Interestingly, pressure pain thresholds in the neck have been found to be reduced in migraine patients even when they are not experiencing an attack (3). Nociception from neck muscles may contribute to migraine headaches experienced in the first trigeminal branch (4) via central convergence in the trigeminal cervical complex (5). A recent experimental study showed how peripheral nociceptors traverse the occipital muscles before innervating the posterior dura (6). This suggests a functional anatomical link between intracranial and extracranial structures that may be important in the development of migraine pain. Peripheral nociceptors have been suggested as a possible contributor to migraine pain (7) and may have an important causative role in patients with neck pain associated with migraine attacks. Still, it is unknown if neck pain directly contributes to migraine pain or is a consequence of it. Only a few MRI studies have reported hyperintense signals in the trapezius muscle in migraine patients (8,9), but the implications of these signal changes are not fully known. Thus, neck pain in migraine is, despite being a very frequent symptom, an under-investigated field of research in migraine. Muscle stiffness can be non-invasively quantified using shear wave elastography (SWE) (10–12). SWE studies have demonstrated increased muscle stiffness in different muscle pain conditions (13–16), but muscle stiffness measurement in migraine patients has not been performed previously.
This study therefore aimed to measure muscle stiffness in migraine patients in the interictal phase and in healthy controls. We hypothesized that migraine patients with ictal neck pain would have stiffer neck and shoulder muscles compared with migraine patients without ictal neck pain and controls.
Methods
Study population
One hundred migraine patients with or without aura were investigated interictally in a cross-sectional study. Patients met the migraine criteria from the International Classification of Headache Disorders 3rd edition (17). Headache specialists recruited migraine patients from the Danish Headache Center between July 2017 and August 2018. Patients were stratified into categories defined by sex and five-year age intervals and frequency matched with 46 controls in a 2:1 ratio. Controls had never experienced migraine, had less than one headache day per month and had no first-degree relatives with migraine. Exclusion criteria for the study group were concurrent primary or secondary headaches, except for tension-type headache. Exclusion criteria for both groups were self-reported injuries in the head or neck region, prior neck trauma causing pain, any extracephalic or extracervical muscle or pain conditions, pregnancy or lactation. All individuals were instructed to reschedule if: A migraine attack occurred within 48 hours prior to examination, they had taken simple analgesics within 24 hours or taken triptans or opioids within 48 hours of the examination. Upper body exercises within 48 hours of the examination were also not allowed. Stable preventive migraine treatment was not an exclusion criterion in the study group. Data on headache characteristics, total and local tenderness scores, stimulus-response function, and cold and heat pain thresholds have previously been published as part of a larger project with the aim to investigate the role of pericranial muscles in migraine pathophysiology (18). This study was registered at clinical-trials.gov (Identifier: NCT03626805).
Semi-structured interview and definitions
All individuals underwent a semi-structured interview including questions about neck symptoms before being examined (Figure 1). The semi-structured interview was performed to confirm migraine diagnosis according to the ICHD-3 classification, establish headache burden and to differentiate between patients with and without ictal neck pain. Ictal neck pain in the study groups was defined as pain, soreness or stiffness of the neck, which could occur 0–48 hours before, during, or 0–24 hours after a migraine attack. Additionally, neck pain had to occur in two-thirds or more of their migraine attacks. Interictal neck pain was defined as neck pain during the last year unrelated to migraine attacks. Patients with very frequent neck pain who reported significant worsening of neck pain associated with migraine attacks were classified as having ictal neck pain. Controls were asked if they had experienced neck pain during the last year.

Procedures: The study was conducted in the following order. i) Semi-structured interview; ii) areas were marked for shear wave elastography and pressure pain threshold. Blue marks the neck area and red marks the trapezius muscle; iii) pressure pain threshold examination with a pressure algometer; iv) muscle stiffness measurement with ultrasound shear wave elastography.
Examinations
All individuals were examined at the Danish Headache Center in a quiet and temperature-controlled room. Two areas were marked bilaterally on the trapezius muscle (one-third of the length between the spinous process of C7 and acromion) and in the neck (over the transverse process of C5). The areas were chosen based on a study from our group (10) and previous differences in pressure pain thresholds between migraine patients and controls (19). The markings were used during both ultrasound SWE and pressure pain threshold examinations. Ultrasound SWE examination was performed with individuals sitting relaxed in a comfortable chair, similar to previous studies (20,21).
Muscle stiffness assessment by shear wave elastography
The technique is based on the ultrasound probe emitting a push pulse, which deforms the tissue. This causes shear waves in the tissue that travel perpendicular to the push pulse, and the speed of these waves can be measured (11,20). The speed is dependent on tissue stiffness, with a high speed corresponding to stiff tissue. It has previously been shown that muscle stiffness of the trapezius, splenius capitis and semispinalis capitis muscles can be assessed with SWE (20). We (JH) recorded the muscle stiffness with a linear 9 MHz ultrasound probe (9L) and the Logic E9 scanner, both from GE Healthcare (Chalfont St Giles, UK). Measurements were performed with the probe parallel to the muscle fibers, which were visualized with B-mode (Figure 2). We measured the previously marked areas at the trapezius muscle and the neck. The neck muscles measured were the splenius capitis and semispinalis capitis. Eight B-mode images and shear wave measurements were recorded for each area (Figure 2). An observer (LTK) blinded for headache diagnosis and characteristics subsequently analyzed the shear wave measurements for quantification; a circular region of interest was placed covering as much of the depth of the muscle as possible, and the system calculated the mean shear wave speed within that region of interest. The mean speed of the eight measurements was calculated for each area. Finally, the mean of the right and left sides was calculated.
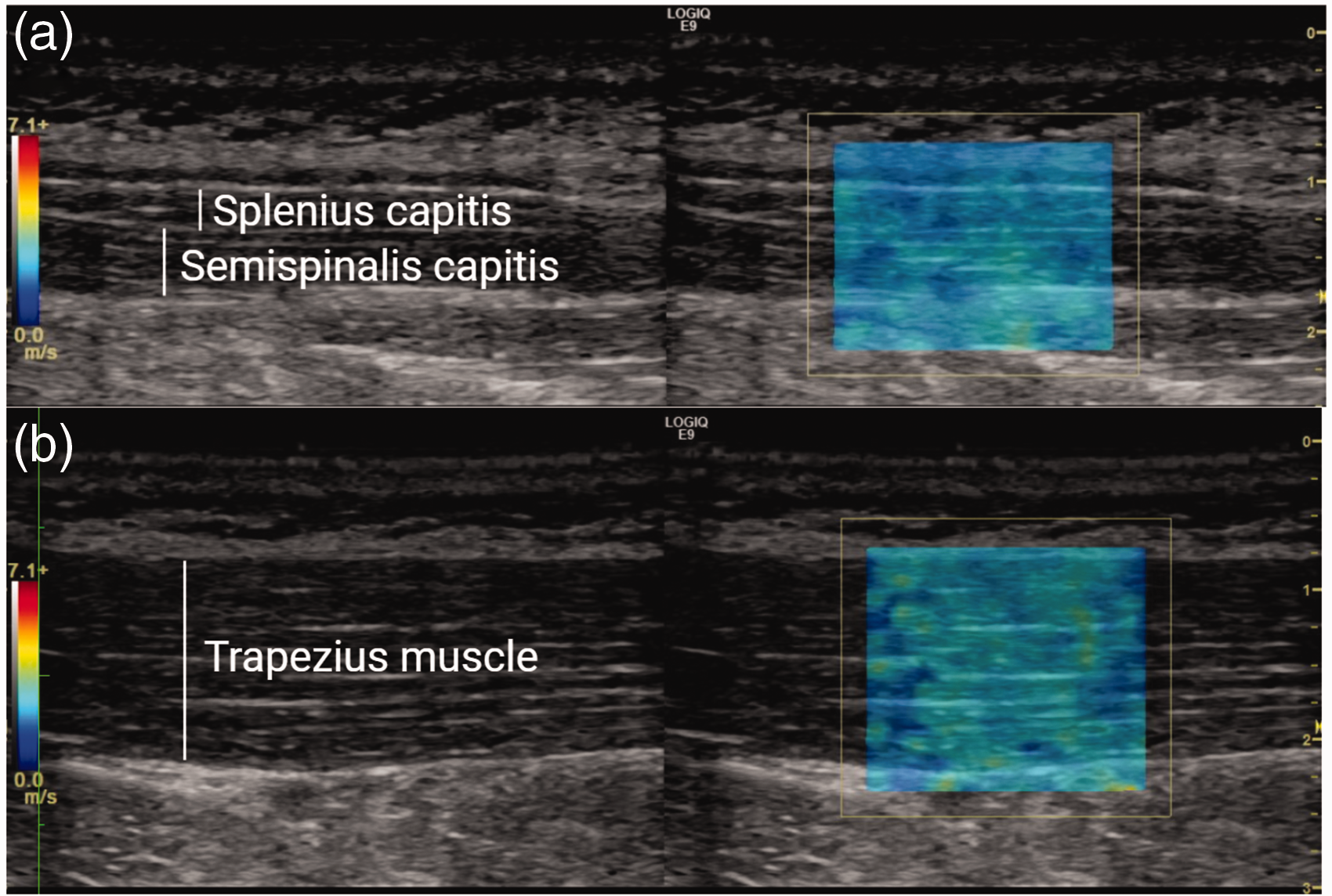
Ultrasound images of a migraine patient. (a) shows the neck muscles and (b) shows the trapezius muscle. The left side shows the B-mode pictures. The elastograms are shown on the right side, where shear wave speeds are shown color coded. The image is without the circular region of interest in the elastogram used for quantification. Blue represents slow speeds and red represents high.
Pressure pain threshold
Pressure pain threshold was measured at three bilateral areas: The marked areas (at the trapezius muscle and neck) and over the temporalis muscle. We used a mechanical pressure algometer (FPK20, Wagner Instruments, USA). Before actual testing, the test was demonstrated at the thenar region. The pressure algometer had a contact area of 1 cm2, and the pressure was increased by 0.5 kg/cm2/s (22). The individuals were instructed to say “stop” when they felt a sensation of pain. A mean value of three recordings at each area was calculated.
Statistics
We calculated our sample size based on data from a prior study that showed a mean shear wave speed of 3.36 m/s and a standard deviation of 0.75 m/s in the semispinalis muscle (20). According to this, two groups of 40 individuals would be enough to detect a 20% difference at 5% significance (two-tailed) with 80% power (23). Data were also available for the splenius and trapezius muscles but had smaller standard deviations (20). Demographics are presented as median and quartiles. Demographics of controls and migraine subgroups were compared pairwise using the Mann Whitney U-test or Fisher’s exact test. Pressure pain threshold, elastography data and headache frequency were logarithmically transformed when calculating correlations to achieve normalized data.
The primary hypothesis was that migraine patients with ictal neck pain would have stiffer muscles than patients without ictal neck pain. The secondary hypotheses were: a) Migraine patients with or without ictal neck pain have stiffer muscles than controls; b) pressure pain thresholds differ between migraine subgroups and controls; c) muscle stiffness correlates with headache frequency and pressure pain threshold. Additionally, we wanted to examine the association between age and muscle stiffness in migraine patients, when correcting for sex, BMI, and ictal neck pain.
General linear models were used for testing the primary hypothesis and secondary hypotheses 1 and 2. These analyses were adjusted statistically for sex (categorical variable), age and BMI (continuous variables) by including them in the general linear model as covariates. The adjustments were determined beforehand since muscles alter with age (24), and muscle stiffness has been shown to be affected by sex (25,26) and BMI (13). Pairwise comparison was conducted with least significant difference. The assumptions for a general linear model were tested with the following methods: Collinearity using variance inflation factor (all below 1.1); homogeneity of variance with Levene’s test and graphically with residuals plotted against the predicted values; visualization of the linear relationship with residuals plotted against covariates; verification of normal distribution of residuals using Q-Q plots. Secondary hypothesis 3 was tested using Pearson’s correlation coefficients. To verify that data were normally distributed, we used the Shapiro-Wilk test. During the analysis we found an association between age and muscle stiffness in migraine patients. This was examined further post hoc using a general linear model. The statistical analysis was performed using IBM SPSS statistics version 25 for Windows (Chicago, IL, USA). The level of significance was set at 5%. Two-tailed testing was performed.
Results
A total of 100 migraine patients and 46 controls were examined. Fifty-two percent of all patients had ictal neck pain, out of which 24 (46%) also had interictal neck pain. Forty-seven percent of all patients had interictal neck pain, while 14 (30%) of controls had had neck pain during the last year (Table 1). Forty-three percent of patients had tension-type headache at the time of examination. Interictal neck pain and migraine with aura were equally prevalent in the two migraine subgroups (Table 1). The blinded observer (LTK) discarded six trapezius measurements and nine neck measurements due to ultrasound signal noise, measurements assessed not to be adequately parallel to the imaged muscle fibers, or fewer than eight sufficient measurements per muscle.
Demographics of 100 migraine patients from a tertiary headache center and 46 controls. Patients are divided according to whether they had ictal neck pain or not. No group differences were found.

The 100 migraine patients participating in the study were recruited out of 270 patients referred from headache specialists at the Danish Headache Center. One hundred and seventy migraine patients were excluded or did not participate due to: Current medication changes (n = 3), neck/shoulder injuries (n = 17), extracephalic and extracervical muscle or pain conditions (n = 11), not fully meeting the ICHD-3 migraine criteria (n = 16), medication overuse headache (n = 12), coexisting cluster headache (n = 1) or trigeminal neuralgia (n = 1), not wanting to participate (n = 58) and practical reasons that made participation impossible (n = 51).
Muscle stiffness
Migraine patients with ictal neck pain had stiffer neck muscles compared to migraine patients without ictal neck pain (mean difference 0.48 m/s, 95% CI: 0.08–0.87, p = 0.018). Migraine patients with ictal neck pain also had stiffer neck muscles than controls (mean difference 0.40 m/s, 95% CI: 0.03–0.78, p = 0.036). There was no difference in neck muscle stiffness between migraine patients without ictal neck pain and controls (mean difference 0.08 m/s, 95% CI: −0.31–0.47), p = 0.681). We found no differences between groups in stiffness of the trapezius muscle (see Table 2 and Figure 3).
Migraine patients without ictal neck pain compared to patients with ictal neck pain. Muscle stiffness (shear wave speeds) of the neck muscles and trapezius muscle adjusted for age, sex, and BMI.
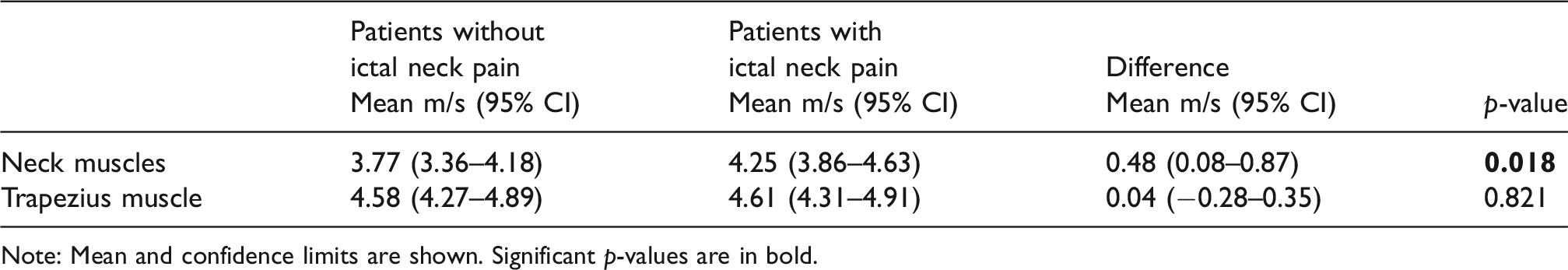
Note: Mean and confidence limits are shown. Significant p-values are in bold.

Muscle stiffness: Comparison of controls and migraine patients with and without ictal neck pain. Muscle stiffness of the neck muscles and trapezius muscle adjusted for age, sex, and BMI. Mean and confidence limits are shown.
In migraine patients with ictal neck pain, headache frequency correlated with stiffness of the trapezius muscle (r = −0.309, p = 0.031). These correlations were not found in the other groups or muscle regions (p > 0.05). Stiffness of neck muscles and the trapezius was positively associated with age in migraine patients (p = 0.029 and p = 0.035 respectively), when corrected for sex, BMI, and ictal neck pain.
Post-hoc, we compared muscle stiffness of the 47 (47%) patients with interictal neck pain with those without and found no difference in muscle stiffness with or without adjustment for ictal neck pain (p > 0.5). We also looked at whether headache at the time of examination affected the results, and we found no association (p > 0.2). Additionally, we compared the muscle stiffness of the pain-side with non-pain side in patients with unilateral migraine and found no differences (p > 0.3).
Pressure pain threshold
There were no differences in pressure pain thresholds in migraine patients with or without ictal neck pain (Figure 4). The pressure pain thresholds for both migraine subgroups were lower at all three areas than for controls (p < 0.02). A negative correlation was found between pressure pain thresholds at the neck and stiffness of the neck muscles in patients with ictal neck pain (Table 3).

Pressure pain thresholds: Comparison of controls and migraine patients with and without ictal neck pain. Pressure pain thresholds of pericranial muscles corrected for age, sex and BMI. Mean and confidence limits are shown.
Pearson’s correlations between muscle stiffness (shear wave speeds) and pressure pain thresholds.

Note: Significant p-values are in bold.
Discussion
The major novel finding was that ultrasound SWE showed stiffer neck muscles interictally in migraine patients with ictal neck pain compared to migraine patients without ictal neck pain. The study also showed that neck pressure pain thresholds and muscle stiffness correlated only in migraine patients with ictal neck pain.
Sensory neurons from both the neck and head converge and synapse with second-order neurons in the trigeminal cervical complex (5), which probably accounts for the very widespread locations of migraine pain. The trigeminal cervical complex neurons also project to and receive input from different brain areas such as the hypothalamus, thalamus and cortex (27,28). If the increased neck muscle stiffness in the present study was primarily centrally driven, we would expect to be able to demonstrate differences in pressure pain threshold in several locations between migraine patients with or without ictal neck pain (Figure 4). However, we found no differences in pressure pain thresholds suggesting similar central sensitization and modulation between migraine patients with or without ictal neck pain. The neck muscle stiffness could alternatively be caused directly by alterations in muscles such as inflammation (29) or trigger points (15), which may activate sensory neurons and thereby contribute to the development of migraine pain. In the following, we will discuss possible mechanisms underlying increased neck muscle stiffness in migraine patients with ictal neck pain.
Recent migraine studies have shown evidence of peripheral alterations in migraine patients. For example, one study found elevated inflammatory markers, such as interleukin-6 (IL-6), in the calvarial periosteum of chronic migraine patients with neck pain (30). Interestingly, IL-6 has also been demonstrated to activate and sensitize both meningeal and muscle nociceptors (31–33). Focal T2 MRI hyperintensities have been demonstrated at neck muscle trigger points in migraine patients (9), and palpation of neck muscle trigger points causes migraine-like headaches (34). Furthermore, an injection of hypertonic saline into neck muscles causes pain in both C2 and trigeminal innervated head areas (35). Previous studies of muscle with SWE reported increased stiffness in muscular pain conditions, such as chronic neck pain (13) and chronic lower back pain (14). This has also been demonstrated in very localized conditions such as myofascial trigger points (15), where the stiffness correlated with and was increased in areas of muscle involvement. Collectively, these findings suggest local peripheral changes, such as muscle inflammation or dysfunctional inflammatory regulation, as possible causes of migraine pain and muscle stiffness. In contrast to the neck muscles, we found no differences in the trapezius muscle, which may be due to local changes in the neck or muscle specific differences.
Increased muscle stiffness could also be caused by muscle tension (36,37). It has previously been suggested that increased muscle tension could be secondary to inflammation that acts via muscle spindles in muscle pain syndromes (38,39) or due to increased muscle nociception in tension type headaches (40). Neck muscle tension has previously been examined in migraine patients with EMG (41–47). Findings suggest that EMG activity is increased interictally in the cranial and upper cervical muscles during rest (41) and stressful mental tasks (42). Studies have also reported increased activity in the extensors of migraine patients during neck flexion (43,44), and increased activity compared to the generated force (45). In contrast, other EMG studies found no differences in the neck extensors of migraine patients at rest (42,46), during tasks or pain stimulation (46,47). Based on the findings in the present study, the diverse findings in the EMG studies may be caused by possible heterogeneity within and between migraine patient groups. Future studies may combine EMG and elastography to control for muscle tension as a contributor of muscle stiffness and pain. In the present study, we did find an association between age and stiffness, which shows that it is relevant to control for age in elastography studies. An association of age and muscle stiffness has also previously been demonstrated in an experimental study in rats measured with a force transducer (48). The underlying cause is likely to be structural changes such as fat infiltration and fibrosis (24) and collagen changes in the extracellular matrix of the connective tissue (49).
When comparing our results with the method study by Ewertsen et al. (20), we found higher SWE values. This could be caused by slight differences in the areas examined, and that examinations of pressure pain thresholds and muscle tenderness were conducted before SWE measurements. This is supported by a recent study of tension-type headache patients using a similar examination protocol, where SWE values were similar to ours (10). Also in support, it has previously been suggested that pressure applied to muscles could cause increased muscle hardness measurements in tension-type headache patients due to an idiomuscular reflex (50).
The present study has some limitations. The patients were recruited from a tertiary headache center and are therefore not comparable to the general population. The operator during examinations was not blinded for participants’ headache or neck pain group allocation. Whether individuals had neck pain at the time of examination was not reported and could have affected the measurements. SWE has shown a significant day-to-day variation in measurement of neck muscles (20). SWE measurements can also be influenced by the muscle fiber orientation (20), fascia, and muscle cross-sectional area (51). Thus, these factors could contribute to some variability in the present study. Also, it is unknown if SWE measurements are affected by measuring after examinations of pressure pain thresholds. Since neck muscles are small in volume, especially the splenius and semispinalis capitis muscles, we chose to measure across both muscles in the same recording. This could have made the recorded tissue area less homogenous and could have induced variability. Whether muscle inflammation would lead to increased muscle stiffness is not fully resolved. Thus, in idiopathic inflammatory myopathies MRI-verified edema was associated with decreased stiffness (52), which was also shown in a study investigating edematous thigh muscles after a 330 km endurance run (53). In contrast, eccentric exercise leads to increased stiffness, albeit temporarily (29). These elastography data were obtained under pathophysiological and physiological conditions that do not share mechanisms with migraine. To avoid such influences, patients were told to abstain from muscular activity prior to examination. Hence, there are multiple factors that could possibly contribute to changes in muscle stiffness, which suggests a need for further imaging and exploratory studies of neck pain in migraine patients.
Conclusion
This study demonstrates using SWE that migraine patients with ictal neck pain have stiffer neck muscles compared with patients without ictal neck pain measured interictally. These changes could be caused by local alterations in neck muscles, which could be an important component in the development and maintenance of migraine pain. Further studies are needed to investigate neck pain in migraine patients, which may result in the development of specific migraine treatments targeting the neck muscles.
Key findings
Migraine patients with ictal neck pain have stiffer neck muscles than patients without. Migraine patients with and without ictal neck pain have similar pressure pain thresholds. This suggests a peripheral cause for the muscle stiffness.
Footnotes
Acknowledgements
Figure 1 was created with biorender.com.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MA has received personal fees from Alder BioPharmaceuticals, Allergan, Amgen, Eli Lilly, Novartis, and Teva, and also participated in clinical trials as the principal investigator for Alder, Amgen, electroCore, Novartis, and Teva. MA also serves as an associate editor of Cephalalgia, associate editor of Headache and co-editor of the Journal of Headache and Pain. MA reports research grants from Lundbeck Foundation, Research Foundation of the Capital Region of Copenhagen, and Novo Nordisk Foundation. HWS has received speaking fees from Novartis, Lilly and Teva. HWS has also received a grant from Novartis. The other authors declare no conflicts of interest.
Ethics review board approval
The study was approved by the Capital Region’s Committee on Health Research Ethics in Denmark (protocol: H17010860) and conducted according to the Declaration of Helsinki. Written consent was obtained.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Candys Foundation and the A.P. Møller Foundation for the Advancement of Medical Science, and the Torben and Alice Frimodts Fond.
Study protocol
The study is registered at clinical-trials.gov with identifier NCT03626805
