Abstract
Objective
To investigate the temporal changes of circadian rhythmicity in relation to the disease course in patients with cluster headache.
Methods
In this multicenter study, patients with cluster headache were recruited between September 2016 and July 2018. We evaluated the patients for circadian rhythmicity and time of cluster headache attacks in the current bout and any experience of bout-to-bout change in circadian rhythmicity. We analyzed the patterns of circadian rhythmicity in relation to the disease progression (the number of total lifetime bouts, grouped into deciles).
Results
Of the 175 patients in their active, within-bout period, 86 (49.1%) had circadian rhythmicity in the current bout. The prevalence of circadian rhythmicity in the active period was overall similar regardless of disease progression. Sixty-three (46.3%) out of 136 patients with ≥2 bouts reported bout-to-bout changes in circadian rhythmicity. The most frequent time of cluster headache attacks was distributed evenly throughout the day earlier in the disease course and dichotomized into hypnic and midday as the number of lifetime bouts increased (p = 0.037 for the homogeneity of variance). When grouped into nighttime and daytime, nighttime attacks were predominant early in the disease course, while daytime attacks increased with disease progression (up to 7th deciles of total lifetime bouts, p = 0.001) and decreased in patients with the most advanced disease course (p = 0.013 for the non-linear association).
Conclusions
Circadian rhythmicity is not a fixed factor, and changes according to the disease course. Our findings will be valuable in providing a new insight into the stability of functional involvement of the suprachiasmatic nucleus in the pathophysiology of cluster headache.
Introduction
Cluster headache (CH) is a primary headache disorder characterized by excruciating, strictly unilateral headaches that are accompanied by cranial autonomic symptoms (1). Circadian and circannual rhythmicity is also a characteristic of CH (1,2). Patients with CH experience extremely severe headaches and psychiatric comorbidities (3–5). While the pathophysiology and treatment of CH have been increasingly investigated, the disease course is relatively unknown.
Circadian rhythmicity is considered a key feature of CH, strongly suggesting hypothalamic involvement in the pathophysiology of this disorder (6). Sex-related differences in the timing of CH attacks have been recently suggested (7). However, changes of circadian rhythmicity have not been well investigated in previous studies. This might be because a long-term longitudinal follow-up study with detailed data on CH is lacking, which may limit the ability to study changes in CH features in relation to the disease course. In our previous study regarding the natural course of untreated CHs in a retrospective cohort of CH patients who were lost to follow up for >5 years, patients with long-term remission (i.e. no recurrence for >5 years) had a lower baseline prevalence of circadian rhythmicity than patients with active recurrence (i.e. recurrence of cluster periods within 5 years) (8). Furthermore, patients with active recurrence had a longer disease duration, and more frequently reported a change in their circadian rhythmicity during their disease course. From these findings, we hypothesized that circadian rhythmicity might change in association with disease course and reflect the disease activity of CH. Thus, we questioned if circadian rhythmicity of CH attacks evolves or decreases according to the disease course.
In this study, we aimed to investigate the pattern of circadian rhythmicity in relation to disease progression. In CH patients participating in a prospective, multicenter registry study, the prevalence and characteristics of circadian rhythmicity were compared between patients with different numbers of total lifetime bouts. Within-individual changes of circadian rhythmicity were also investigated.
Methods
Study design
This study is a part of the Korean Cluster Headache Registry Study (KCHR), which is a prospective, longitudinal, multicenter, observational study that enrolled consecutive patients with CH from 15 hospitals (13 university hospitals, including eight tertiary and five secondary referral hospitals, and two general hospitals) in Korea. CH symptoms, manifestations, and comorbidities were comprehensively collected. Patient recruitment was started between September 2016 and February 2017 following the IRB approval in each hospital. Patients who visited the participating hospitals during the study period and were diagnosed with CH were recruited for the KCHR study. The diagnosis of CH was made by KCHR investigators (experienced neurologists with expertise in headache disorders) based on the most updated criteria from the International Classification of Headache Disorders (ICHD) at the time of enrollment (i.e. 3rd edition, beta version or 3rd edition) (9,10). The study protocol was reviewed and approved by the Institutional Review Board of each participating hospital, and written informed consent was obtained from every patient prior to recruitment.
The details of the KCHR study are published in our previous studies (11–13). In brief, the KCHR study comprises several sub-studies, including this study. All participants were evaluated and interviewed by investigators. Comorbid depression and anxiety were assessed using the Korean version of the Patient Health Questionnaire-9 (PHQ-9) and the General Anxiety Disorder-7 item scale (GAD-7) (14,15). Patients enrolled in their active, within-bout period were followed up after 2 ± 2 weeks (visit 2: Verification of diagnosis and early treatment response), at 3 months (visit 3: Verification of remission and early treatment response), at 1 year (visit 4: Verification of recurrence), and annually up to 5 years thereafter (visits 5–8: Verification of recurrence). Follow-up evaluations were performed either by a clinic visit or by phone. When the active bouts remitted, patients underwent an evaluation for symptoms and functioning in their between-bout periods.
Assessment of circadian rhythmicity
For this study, data obtained between September 2016 and July 2018 were used. Circadian rhythmicity was assessed at the baseline visit and at visit 2. At the baseline visit, circadian rhythmicity of the current bout was determined by investigators by asking patients if their attacks in the current bout had a trend toward occurring at the same time of day. All patients were instructed to recall and check all the chronological times of their CH attacks experienced in the current bout and mark one of the most frequent times of CH attack. In case of uncertainty (e.g. a visit at the earliest phase of the bout), it was recommended that the patients were observed without preventive or transitional treatment for more than 1 week (at least 2 weeks of observation was recommended if available), and the presence of circadian rhythmicity and time of CH attacks were ascertained at visit 2. In such cases, the times of CH attacks were also retrospectively recorded at visit 2.
Patients with ≥2 lifetime bouts were asked if they experienced any change in the pattern of circadian rhythmicity during their disease course. Patients were instructed to choose one of four answers: a) stationary: No change between bouts; b) developing: Becomes more prominent as disease progresses; c) decreasing: Becomes less prominent as disease progresses; and d) variable: Different from bout to bout. In patients with primary chronic CH, overall changes throughout the disease course were investigated.
Statistical analysis
Categorical data are presented as numbers (percentages), and continuous variables are summarized as median (interquartile range [IQR]). Student's t-test, Chi-square test, and Fisher's exact test were used to compare variables between two groups. The number of total lifetime bouts was used to represent disease progression. It was categorized into 10 groups (deciles) for categorical analysis, otherwise, raw numbers were used for continuous analysis. To analyze the association between the presence of circadian rhythmicity and total lifetime bouts, we created bar graphs of circadian rhythmicity according to deciles of total lifetime bouts, and then compared the presence of circadian rhythmicity between the most advanced (10th decile) versus the other (1st to 9th decile) groups using the Chi-square test. Scatterplot and bar graphs were used to show the distribution of time of the most frequent attacks in relation to disease progression. The time of CH attacks was grouped into four periods (7 am to 12 pm, 1 pm to 6 pm, 7 pm to 12 am, and 1 am to 6 am) and daytime versus nighttime (7 am to 6 pm vs. 7 pm to 6 am) for categorical analyses, otherwise, it was used as a continuous variable. To statistically test if the heterogeneity of the time of most frequent CH attacks differs in relation to disease progression, we performed the Fligner-Killeen test to compare the homogeneity of variances for total lifetime bouts among four periods of the time of most frequent CH attack. Post-hoc analyses were performed, and the Sidak method was used in correction for multiple testing. In addition, daytime versus nighttime predilection was analyzed by using bar graphs of daytime/nighttime according to deciles of total lifetime bouts, and the generalized additive model was used to statistically test the non-linear relationship identified in the bar graph. Statistical analysis was performed using the Stata 16.0 software (StataCorp LLC, College Station, TX, USA) and SAS 9.4 software (SAS Institute Inc., Cary, NC, USA). A two-tailed p-value < 0.05 was considered statistically significant.
Data availability
Anonymized data will be shared by request from any qualified investigator.
Results
Patients
Demographics and characteristics of patients according to circadian rhythmicity.

Data are presented as n (%) or mean (SD).
Abbreviations: BMI: body mass index; ICHD-3b: International Classification of Headache Disorders, 3rd edition, beta version; NRS: numeric rating scale; HIT-6: Headache Impact Test-6; PHQ-9: Patient Health Questionnaire-9; GAD-7: General Anxiety Disorder-7.
Calculated only in patients with ≥ 2 lifetime bouts.
In the case of first-onset CH with active periods lasting without 1 month of attack-free periods, but an observation period of less than 1 year.
Thirty-six patients with first-onset bout and one who could not recall the season of the previous bout (which was 13 years before the current bout) were excluded.
Prevalence of circadian rhythmicity according to the number of total lifetime bouts
When classified into deciles of lifetime bouts, the prevalence of circadian rhythmicity was similar among groups. The circadian rhythmicity was lowest (28.6%) in patients in the 10th decile group (>20 lifetime bouts) of total lifetime bouts (Figure 1); however, it was statistically insignificant when compared to the average prevalence in the other groups (10th decile vs. 1st to 9th decile groups: 28.6% vs. 50.9%, respectively; χ2 (1) = 2.577, p = 0.163). The number of total lifetime bouts did not correlate with advanced age (Spearman's rho = 0.029, p = 0.701).
Prevalence of circadian rhythmicity in the current bout in relation to the number of total lifetime bouts. The number of total lifetime bouts per patient is grouped into deciles. Thirty-six patients with first-onset CH were grouped into first and second deciles. The number of patients included, range and mean numbers of total lifetime bouts are summarized in the table.
Patient-reported changes in circadian rhythmicity between bouts
Bout-to-bout changes in circadian rhythmicity in patients with ≥ 2 bouts.
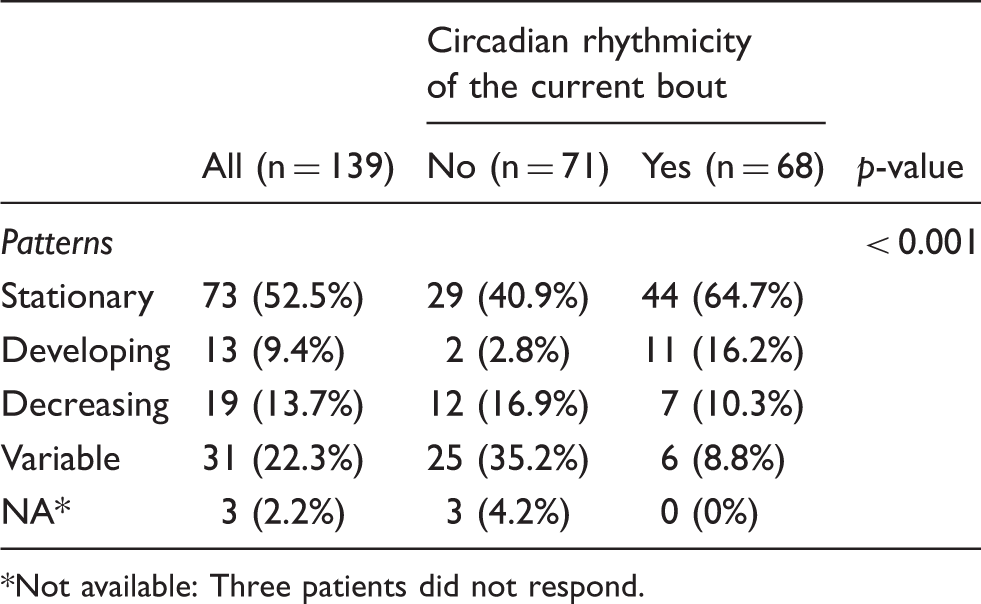
Not available: Three patients did not respond.
The pattern of change was different between patients with versus without current circadian rhythmicity (χ2 (4) = 25.221, p < 0.001; Table 2). When compared to patients with current circadian rhythmicity, patients without circadian rhythmicity at the current bout more often experienced bout-to-bout variability in circadian rhythmicity (35.2% vs. 8.8%, Table 2). Patients with circadian rhythmicity reported a stationary pattern (“no change”) more frequently than those without circadian rhythmicity (64.7% vs. 40.9%; Table 2).
Bout-to-bout changes in circadian rhythmicity according to the number of bouts experienced (2 vs. > 2 bouts).
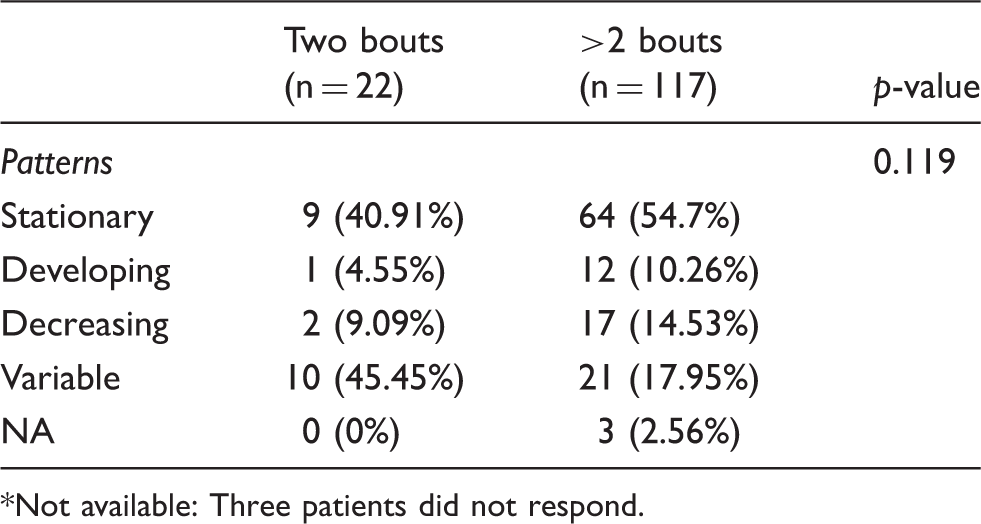
*Not available: Three patients did not respond.
Time of day for cluster headache attacks
Figure 2(a) shows all the times of CH attacks experienced in the current bout in all patients. The peak frequency was recorded at 10 am and 3 pm, followed by 10 pm and 2 am. When restricted to patients with circadian rhythmicity, similar peaks were observed (Figure 2(b)). Similarly, the time of most frequent occurrence was 10 am in the total study population (Figure 2(c)), as well as in patients with circadian rhythmicity (Figure 2(d)), followed by 2 am in both groups.
Time of CH attacks. (a),(b) All the times of CH attacks experienced in the current bout (a) in all patients and (b) in patients with circadian rhythmicity. (c),(d) Time of the most frequent occurrence in the current bout, marked by (c) all patients and (d) patients with circadian rhythmicity.
Time of day for cluster headache attacks in relation to the number of total lifetime bouts
Figure 3(a) shows a scatterplot between the time of most frequent occurrence and the number of total lifetime bouts. In patients with circadian rhythmicity, those in earlier disease course showed a relatively even distribution of the time of most frequent occurrence. With disease progression, the distribution showed a trend toward dichotomy into hypnic and midday occurrence in the current bout (Figure 3(a)). This trend was statistically significant according to the Fligner-Killeen test for the homogeneity of variance of total lifetime bouts between the attack time groups (χ2 = 8.491, DF = 3, overall p = 0.037; Figure 3(b)): The afternoon (1pm to 6pm) and hypnic (1am to 6am) groups showed greater variance (SD 6.57 and 8.14), whereas the evening (7pm to 12am) group had the least variance (SD 3.79), implicating that afternoon and hypnic attacks occur throughout the disease course, whereas evening attacks are prominent only during the earlier disease course. Post-hoc analyses showed higher variances of total lifetime bouts in the afternoon and hypnic group compared to the evening group (SD 6.57 vs. 3.79, p = 0.036; SD 8.14 vs. 3.79, p = 0.025, respectively). However, these associations were not significant after correction for multiple testing (adjusted p = 0.089 and 0.104, respectively).
The time of most frequent occurrence in relation to disease progression. (a) A scatterplot of time of most frequent occurrence and the number of total lifetime bouts. (b) Box-and-whisker plot of total lifetime bouts in different time groups. The greatest variance was noted in the afternoon (1 pm to 7 pm) and hypnic (1 am to 7 am) groups, while the least variance was found in the evening (7 pm to 1 am) group.
Figure 4(a) shows the time of most frequent occurrence, grouped into daytime (7am to 6pm) and nighttime (7pm to 6am), in relation to the disease progression. The nighttime predilection was predominant in the earliest (1st to 2nd deciles) disease duration group. The daytime predilection increased as the disease progressed (up to the 7th decile) (Spearman's rho = 0.448, p = 0.001). In the 8th and 10th decile groups, daytime predilection dropped, and nighttime attacks became predominant again. This non-linear association between the daytime attack predilection and the number of lifetime bouts was statistically significant (χ2 = 8.753, DF = 2, p = 0.013; Figure 4(b)).
Daytime vs. nighttime predilection of CH attack in relation to disease progression. (a) Proportions of daytime and nighttime predilection in patient groups of number of lifetime bouts. (b) The relationship between daytime predilection and the number of total lifetime bouts.
Discussion
The main findings of our study are as follows: a) the prevalence of circadian rhythmicity did not differ according to the disease progression, although circadian rhythmicity was less frequently reported in patients with the longest disease duration; b) in half of all patients with CH, circadian rhythmicity was not fixed, but varied from bout to bout; and c) the pattern of circadian rhythmicity differed according to the disease course. A trend toward dichotomy into hypnic and midday occurrence was observed as the number of lifetime bouts increased. Daytime attacks increased in correlation with the number of lifetime bouts during the early and middle disease course, whereas nighttime attacks were predominant in patients during the earliest or most advanced disease course.
Previous studies reported circadian rhythmicity in 58–82% of patients with CH (4,16–19). Our study showed a relatively lower prevalence of circadian rhythmicity, because we assessed the circadian rhythmicity only in the current bout in order to minimize the possibility of recall bias. Considering that 38 patients who did not have circadian rhythmicity in the current bout reported a change in circadian rhythmicity during their disease course, the estimated lifetime experience of circadian rhythmicity increased to 70.9% in our study. This discrepancy suggests that a substantial degree of within-subject variation exists in circadian rhythmicity. This hypothesis is further supported by our finding that half of our patients reported a change in their circadian rhythmicity between bouts.
We hypothesized that circadian rhythmicity may evolve or regress as the disease progresses. However, the overall prevalence of circadian rhythmicity in the index bout remained unchanged regardless of disease progression. Rather, the pattern of time predilection changed in relation to the number of lifetime bouts. Earlier in the disease course, the time of most frequent occurrence was diverse, particularly in patients with first-onset CH. With disease progression, there was a trend toward a dichotomy into nocturnal and midday attacks. This may suggest that the suprachiasmatic nucleus is involved to a certain degree in active bouts regardless of disease duration; however, its impact on the attack timing may change as the disease progresses. Combined together with our finding that the presence of circadian rhythmicity substantially changes within individuals and the pattern of circadian rhythmicity differs between individuals, we inferred that the involvement of the suprachiasmatic nucleus might not be stationary between bouts. It may imply that the role of the suprachiasmatic nucleus is additional, not essential, in the pathophysiology of CH, or its functional impact might be influenced by other unmeasured factors such as internal or external homeostasis or treatment.
A non-linear temporal pattern of daytime/nighttime propensity of CH attacks was also observed in correlation with disease progression. Night attacks were predominant early in the disease course and in the advanced course of CH. This pattern may indicate a possible evolvement and regression of diurnal expression of CH, the biological implication of which has not yet been determined. In addition to the internally driven rhythm of suprachiasmatic nuclei, factors such as hormones, light exposure, sleep interruption, or activity level, may be involved in the diurnal expression of cluster headache.
The exact mechanism of circadian rhythmicity has not been documented yet. Genetic polymorphism in the CLOCK (Circadian Locomotor Output Cycles Kaput) gene has been reported to be associated with diurnal rhythmicity of CH (20). The roles of sleep phase, melatonin, pituitary adenylate cyclase-activating peptide (PACAP), and orexin have been suggested as possible mechanisms of circadian rhythmicity in CH; however, their direct association with the timing of CH attacks has never been proven (21–26). Our findings suggest that a temporal evolution should be considered in the research into circadian rhythmicity of CH. Bout-to-bout changes in circadian rhythmicity should also be taken into consideration. This cannot be explained solely by genetic polymorphism; rather, it suggests changes in environmental and internal homeostasis might play a role in circadian rhythmicity of CH attacks.
In the current study, we obtained data from patients with active CHs. Our previous study on the natural history of untreated CH showed that patients with remitted CH had less circadian rhythmicity at baseline, and less bout-to-bout change in circadian rhythmicity during their disease course. Whether the evolution of circadian rhythmicity is a marker of active disease progression should be investigated in future, long-term observation studies. In this regard, the KCHR long-term follow-up study is ongoing in selected centers. The strengths of our study are as follows. A strength of the KCHR study is that the diagnosis and characterization of CH were well verified by experienced neurologists. Patients were longitudinally followed up, which enabled use to ensure the presence of circadian rhythmicity in patients who were uncertain about their pattern of CH attacks. However, our study also has limitations. First, most of the data were obtained from patients' recall of the current bout. This problem is attributed to two issues: The lack of definition of circadian rhythmicity in CH and the ethical issue. Currently, circadian rhythmicity has not been defined from a consensus, and it is unknown whether the circadian rhythmicity occurs exactly on a 24-hour basis. Therefore, it is doubtful if the use of prospective recording might ensure the circadian rhythmicity of CH. In addition, it might be unethical to delay transitional or preventive treatments for the prospective observation of attack occurrence. To reduce a possible recall bias, we instructed patients to answer only about the current bout, and only a small number of patients without a clear recall were prospectively observed without transitional and/or preventive treatment. Nonetheless, between-bout comparison was based on patients' recall, which might be prone to a recall bias. Second, our observational study can only be hypothesis generating, rather than hypothesis testing. The pathophysiological aspects of our findings should be further investigated in future studies. Third, we did not collect information regarding the association between previous treatment and changes of circadian rhythmicity. Finally, our cohort has a low prevalence of chronic CH. However, our data may provide more insight into the spectrum of episodic CH.
Clinical implications
In this multicenter study, half of patients with active cluster headache experienced circadian rhythmicity in the current bout. Circadian rhythmicity is not a fixed factor, and the presence of circadian rhythmicity varies from bout to bout. The pattern of circadian rhythmicity can change in association with disease progression.
Footnotes
Acknowledgements
We thank Ms. Miran Jung and Ms. Jinju Lee for aiding in data management. Dr. Soohyun Cho helped with patient recruitment.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
