Abstract
Aim
To investigate the influence of clinical and demographic features on diagnostic delay in cluster headache patients, in order to discuss diagnostic pitfalls and raise disease awareness.
Methods
A large, well-characterized cohort of 400 validated cluster headache patients from the Danish Cluster Headache Survey, diagnosed according to ICHD-II, were investigated. ANOVA was applied to investigate differences in diagnostic delay between groups. Selected independent variables were assessed in relation to diagnostic delay using a gamma regression model.
Results
Diagnostic delay was significantly reduced for each decade of cluster headache onset from 1950–2010 (p < 0.001). Onset after 1990 was associated with shorter diagnostic delay (OR = 0.28, p < 0.001), whereas attack duration > 180 minutes (OR = 1.62, p < 0.034), migraine-like features (OR = 1.30, p < 0.043) and nocturnal attacks (OR = 1.39, p < 0.021) were associated with prolonged diagnostic delay. Further, diagnostic delay decreased with age of onset (age < 20: 13.8 years, age 20–40: 5.4 years and age > 40: 2.1 years, p < 0.001).
Conclusion
Diagnostic delay was reduced for every decade investigated, whereas some atypical cluster headache features were associated with prolonged diagnostic delay. Better medical education and more disease awareness are needed to prevent misdiagnosis and prolonged diagnostic delay.
Introduction
Cluster headache (CH) is a primary headache disorder characterized by very severe, unilateral, orbitally located pain attacks accompanied by autonomic symptoms and/or restlessness (ICHD-III)(1). Despite clear diagnostic criteria (1), diagnostic delay (DD) is often reported to be very long: In a prior study from 2017 from our group, the mean time from CH onset until a CH diagnosis was established to be approximately 6 years (2). This is in line with previous studies, summarized in a recent review, reporting a DD ranging from 3.6 to 9 years (3).
The consequence of a long DD is first and foremost that many CH patients receive misinformation and wrong treatment (4,5). For instance, a UK study showed that 40% underwent unnecessary invasive procedures (6). Furthermore, CH has substantial socioeconomic implications for both patients and society due to sick leave, use of medical services and hospitalization (7). According to an American study, about 20% of a CH cohort had lost their job due to the disease (8). To treat CH effectively, a correct diagnosis must be provided early in the disease phase, as the treatment of CH differs from the treatment of other primary headache disorders.
In order to reduce DD, more education of medical doctors and awareness of CH in the general population are consistently mentioned as key elements (5,9,10). Still, very little is known about which clinical factors influence DD. Only a few studies have attempted to identify such factors, and with diverging results. One study found that the presence of migraine-like features (MLF) such as nausea, vomiting, photo- and phonophobia prolonged DD (11); however, another study was not able to demonstrate this relationship (12). Additionally, it has been shown that onset of CH symptoms after the age of 40 years leads to shorter DD (10).
The aim of this study was to investigate the relation between DD and a wide variety of demographic and clinical characteristics in order to raise more disease awareness and prompt an earlier CH diagnosis.
Methods
The study was designed as a cohort study consisting of 400 CH patients. Data derives from a questionnaire consisting of 362 questions divided into seven parts: Headache diagnosis, burden of the headache, treatment, sleep, demography, lifestyle habits and physical activity. It took approximately one hour to complete the survey. The collection of data was conducted in the time period of 2012–2017, and the detailed methods have previously been outlined (13). In short, patients completed the questionnaire, after which they had a structured interview in person or by telephone, with a physician, a trained medical student or a trained study nurse, to rule out ambiguities and to validate the headache diagnoses.
The protocol was approved by the Capital Region of Denmark Ethical Committee (H-2-2012-016) along with other studies. However, after these studies were finalized, the questionnaire-based study did not need formal approval according to Danish law. The local ethics committee was informed of the continued data collection and had no objections to it (file number: 17008910).
Patients
Patients participating in the study were recruited with the following inclusion criteria: Diagnosis with ECH or CCH according to ICHD-II (14); age between 18 and 65 years; and with the ability to distinguish CH attacks from other primary headache attacks. The exclusion criteria were as follows: Patients diagnosed with another primary or secondary headache disorder ( ≥ 14 days per month) and patients not able to communicate in Danish. Patients with attacks lasting longer than 180 min were not excluded from the study.
The patients were mainly recruited from a registry from the Danish Headache Center, a highly specialized tertiary headache center.
Sampling and statistical analysis
To explore the relation between certain clinical characteristics, other variables such as history of familial CH and comorbid headache disorders and DD, the literature was searched for previous publications on DD. On the basis of these findings and clinical experience, several independent variables were identified to assess their relation to the dependent variable DD (calculated by subtracting the year of CH diagnosis from the year of first CH attack), using a gamma regression model. Gamma regression was applied because of the skewed distribution of DD. Odds ratios were established by applying the exponential function to the coefficients.
The investigators selected clinical characteristics believed to be relevant for DD. The following variables were identified: a) Age, calculated from the patients' personal social security number ( < 20 years, 20–40 years, > 40 years); b) onset (onset before or after 1990 and according to decade); c) MLF (reporting photo-/phonophobia or neither); d) restlessness or agitation (reporting restlessness/agitation or neither); e) attack duration (15–180 minutes or longer); f) very severe pain intensity reported on a four point Likert scale (reporting mild/moderate/severe or very severe pain); g) alternating side of attacks (yes/no); h) educational level (primary/lower secondary school, vocational education/academy/bachelor's degree, master's/PhD degree, enrolled in education, another level of education); i) family history of CH (reporting genetic relatives assumed to have CH or not); j) current co-existing migraine.
In addition, ANOVA (analysis of variance) was used to investigate the difference in mean DD between groups (decade of onset, age of onset, educational level). p-values were calculated as two-tailed, and the results were regarded statistically significant if the p-value was < 0.05. The statistical calculations were performed with STATA/SE 15.1.
Results
In total, 644 CH patients were invited to participate in the study; three died before returning the questionnaires, 231 were non-responders, and 10 patients withdrew their consent.
Overall, the study consisted of 400 patients (response rate 62.1%) of which 67.0% were men and 33.0% were women, resulting in a 2:1 male-female ratio. ECH was diagnosed in 63.2% of the cases, CCH in 36.8%. The mean age of participants was 46.2 years, ranging from 19–65 years. Mean DD was 6.3 years (range 0–47 years, median 3 years).
Clinical characteristics
Clinical features are presented in Figure 1. Attack duration was longer than 180 minutes in 7.5% of patients, 63.8% (the same for men and women) of patients reported one or more MLF, and 73.5% reported accompanying restlessness/agitation during attacks. Very severe pain intensity was reported by 61.7% and a total of 22.3% reported alternating attack sides either between or within bouts. Seventy-seven percent had nocturnal attacks and co-existing migraine was present in 12.3% of the patients. Only 1.75% reported not having autonomic symptoms during CH attacks.
Selected clinical features relevant for diagnostic delay and their prevalence in the cohort.
Onset and demographics
Decade of CH onset and diagnostic delay.
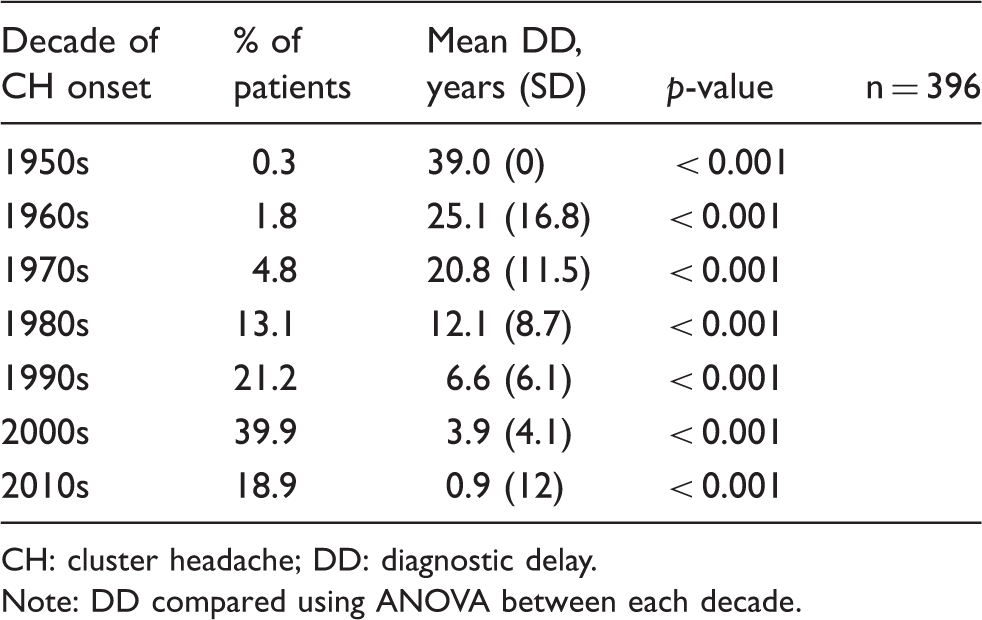
CH: cluster headache; DD: diagnostic delay.
Note: DD compared using ANOVA between each decade.
CH onset: Age of CH onset ranged from 3–65 years with a mean of 31.4 years of age. When divided into groups, 21.8% of the patients had onset < 20 years of age, 50.1% between 20–40 years of age and 28.1% > 40 years of age. DD was longest for the youngest age group and became shorter with increasing age (13.8 years for onset < 20 years of age, 5.4 years for onset at 20–40 years of age, and 2.1 years for onset > 40 years of age, p < 0.001) (Figure 2).
Age of CH onset and diagnostic delay.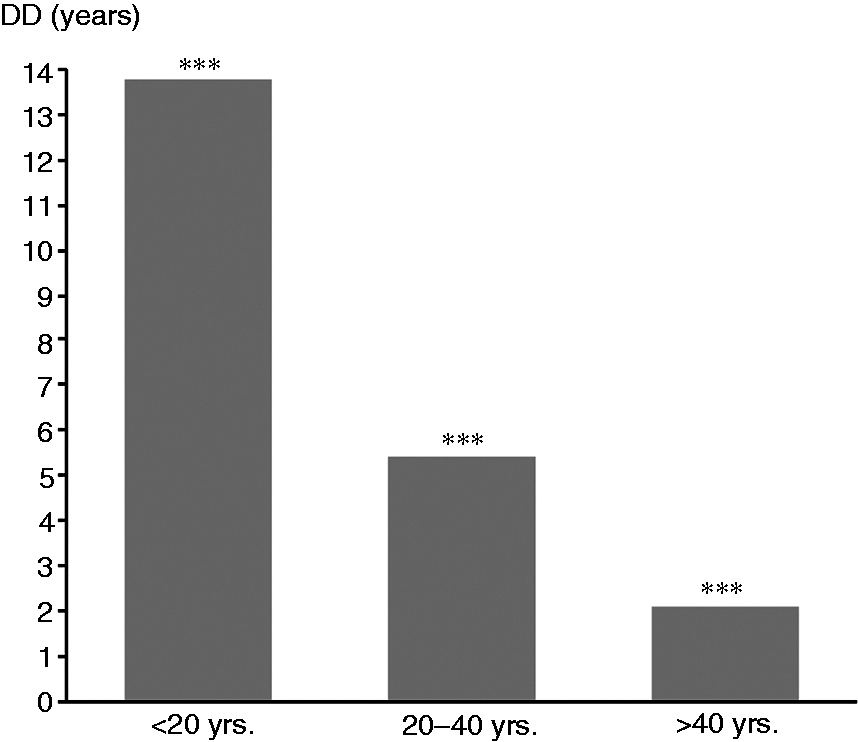
Educational level: For 11.0% of patients, the highest educational degree obtained was a primary or lower secondary school degree, for 41.5% it was a technical, trainee or associate degree, for 38.0% it was a bachelor's, masters or a higher degree, 4.5% were still studying and 5.0% reported another level of education. Educational level was not associated with DD (4.5 years for secondary school degree, 6.8 years for short higher education, 5.7 years for long higher education, 10.3 years for another level of education and 6.1 years for being enrolled in education, p = 0.069).
Family disposition: A total of 17.3% of patients had family members with CH and 44.3% had family members with migraine. Patients who had a positive family history of CH were associated with a 34.0% higher risk of a prolonged DD compared with patients without family disposition (p < 0.062).
Diagnostic delay and therapeutic mismanagement
Half of the patients (49.0%) were misdiagnosed before receiving CH as the correct diagnosis, and the most common misdiagnosis was migraine (n = 92, 23.5%), followed by tension-type headache (n = 76, 19.0%), and sinusitis (n = 56, 14.0%). On average, misdiagnosed patients had received 1.7 wrong diagnoses. The majority of patients (76.1%) were diagnosed in the secondary or tertiary health care sectors by a private neurologist (36.7%), a headache specialist at a headache clinic (27.9%) or a medical doctor working at a hospital (11.2%), and only 20.8% were diagnosed by their general practitioner.
Treatment prior to CH diagnosis: 81.3% were treated with over-the-counter analgesics, followed by 33.0% treated with stronger analgesics such as synthetic opioids or codeine. As many as 20.3% were treated with strong analgesics such as morphine and slow-release morphine and 19.8% had dental treatment such as extraction, drilling or root canal therapy of healthy teeth.
Regression model analyses
Factors associated with a prolonged DD were attack duration > 180 minutes (OR = 1.62, p < 0.034), MLF (OR = 1.30, p < 0.043) and nocturnal attacks (OR=1.39, p < 0.021). Onset after 1990 was associated with a shorter DD (OR = 0.28, p < 0.001). Less than very severe pain intensity, alternating attack side, absence of restlessness/agitation, co-existing migraine, and phenotype were not significantly associated with a prolonged DD (Table 2 and Figure 3).
Features associated with diagnostic delay according to gamma regression model. Risk of longer diagnostic delay related to clinical phenotype and demographics. Factors assessed in the gamma regression model. CH: cluster headache; ECH: episodic cluster headache.

Discussion
Our extensive observational study, composed of 400 CH patients, investigated causes of DD in CH. The main finding of the study was a reduced DD for every decade of CH onset. Further, important variables associated with shorter DD were onset after 1990 and onset > 40 years of age, whereas attack duration > 180 minutes, presence of MLF, and nocturnal attacks were associated with prolonged DD.
We reported an average DD of 6.3 years, which is comparable to other European investigations (4,5,9,12,16).
Clinical characteristics
DD has sometimes been divided into patient's versus physician's delay, referring to time from onset until first consultation, and time from first consultation until correct diagnosis, respectively (17). Until a diagnostic (bio-)marker of CH is discovered, the diagnosis of CH depends entirely on the medical doctor being able to obtain a thorough clinical history based on the diagnostic criteria (18). In other words, total delay may also be due to physician's delay. One study reported a physician's delay of 4.9 years (total DD not reported), and another reported a physician's delay of 2.9 years (total DD of 3.6 years) (4,17).
At times, even for headache specialists, it may be difficult to differentiate CH from migraine in patients presenting with atypical symptoms. Migraine was the most common misdiagnosis among our patients, which is in line with previous studies (4,8,16,17). A high rate of patients misdiagnosed with migraine naturally leads us to suspect that the presence of MLF may prolong DD. However, it remains uncertain whether an association between MLF and misdiagnosis of migraine exists or not (17). In the present study, 7.5% of patients experienced CH attacks exceeding 180 minutes. Long attack duration ( > 180 min) increased the risk of a prolonged DD with 62.0%. A study focusing on accompanying symptoms in CH identified that 10.0% without MLF, and 22.0% with MLF, had attacks lasting longer than 180 min (12). An increased physician's delay, because of confusion as to whether the patients actually suffer from migraine attacks (lasting between 4 and 72 hours), has been suggested to be the explanation for misdiagnosis among CH patients (9,12). Another explanation for migraine to be the most common misdiagnosis could be the high proportion of CH patients with MLF. In the present study, 63.8% (the same percentage for both men and women) reported MLF during CH attacks; other studies report MLF to be present in 61.6% and 46.0% of patients (16,19). MLF prolonged DD in the current study, which is also confirmed by both a previous Dutch study and a Greek study (11,16). However, other studies defining MLF as also being vomiting, nausea and osmophobia did not find the same relation (12,17,19). The fact that MLF is a common feature among CH patients (20) underlines the importance of further studies to enlighten the relation between MLF and DD.
Surprisingly, almost one third of the cohort did not experience restlessness and agitation. However, they were not associated with either a prolonged or a decreased DD in the current study, as observed in a previous Greek study (16). In this cohort, autonomic symptoms were present in almost all patients and were therefore not included in the statistical model.
Phenotype (ECH/CCH) was not associated with DD in this study, as could have been suspected. Yet, some studies have reported that ECH patients have a prolonged DD compared to CCH patients and they propose that this is due to reluctance to contact the health care system when a bout has ended, as they may believe that their symptoms will never return (4,11,17). On the other hand, the periodicity is one of the characteristic features of CH and it could be hypothesized that the attack pattern of CCH could resemble more closely that of migraine.
Nocturnal attacks are a well-known feature associated with CH (18), even though not directly mentioned in the ICHD-III criteria (1). As many as 77.0% of patients experienced attacks during sleep, which is in line with previous studies (11,21). Nocturnal attacks were associated with prolonged DD, a finding also described in a Dutch study from 2003 (11). The relation may be explained by an increased patient's delay, because of the less intrusive nature of nocturnal attacks, meaning that patients wait longer to contact health professionals as long as their daily work life remains unaffected. On the other hand, nocturnal attacks are a very classical feature of CH, and would therefore be expected to result in a lower physician's delay. To the authors' knowledge, there are no studies that have investigated this.
DD was equal for both men and women, yet a previous study from our group established that the diagnostic process was more complicated for women than for men (2). Misdiagnosis was found to be more prevalent in women, and more women were not correctly diagnosed until seen in a highly specialized headache center. The incoherence with DD could be speculated to be due to sex differences in health seeking behavior.
Onset and demographics
Previous studies investigating DD over time have indicated that DD has improved (3,6); specifically, in a UK study, from 22.3 years in the 1950s down to 2.6 years in the 1990s (6). Our study confirms that DD decreases significantly for each decade investigated. Furthermore, onset after 1990 was associated with a shorter DD, which may be related to the ICHD criteria being published for the first time in 1988. In line with this, it has been suggested that patients today have better access to specialized neurologists that are more capable of diagnosing a primary headache disease correctly than before (6). In addition, more attention being paid to primary headache disorders and better education of medical students may have improved physician's delay. Furthermore, easier access to information about CH on the internet could very likely have led to more self-referred and self-diagnosed patients, ultimately improving patient's delay (5,6,16).
Comparable to our findings, early age of CH onset has been suspected to be associated with longer DD, because of the low prevalence and poor recognition of CH among children (10,17). Another explanation may be children's reluctance to seek treatment for a short-lasting attack-based headache disorder or that children my face difficulties expressing symptoms. Interestingly, patients who had onset above 40 years of age experienced the shortest DD. A possible explanation could be that patients over 40 years of age have a higher risk of other serious neurological disorders and therefore may be referred more quickly to neurological departments (10).
According to a Danish cohort study, one third of patients reported that CH had limited their working career (7). The risk of falling short in career or job opportunities could be hypothesized to lower patient's delay.
It was expected that DD would be shorter for patients with a positive family history of CH, because of the increased awareness of the disorder within the family and the knowledge of specific treatment possibilities. Nonetheless, there was no association between family disposition and DD. This could perhaps be explained by self-treatment among family members, leading to an increased patient's delay, but the questionnaire did not assess this further.
Diagnostic delay and mismanagement
According to this study, half of the CH patients were misdiagnosed, which is less than in other studies reporting prevalences as high as 70–80% (4,8,11,17). For comparison, a Turkish study has found that misdiagnosis occurs in 58.0% of migraine patients and 66.3% of tension-type headache patients (22). Additionally, the misdiagnosed patients received 1.7 wrong diagnoses, which is also less than proposed by other investigations, where it ranges from 2.1–3.9 (4,5,9). The number of wrong diagnoses was lowest in the most recent studies, indicating a positive development. In Denmark, recent national campaigns, an active and visible CH patient organization, and improved education of medical doctors may have played a vital role in the reduction of misdiagnoses.
More than half of our patients are diagnosed by neurologists or headache specialists at the tertiary Danish Headache Center. Only 20.8% are diagnosed by a general practitioner. These results correspond to a UK study (6), which indicates that most patients have to be referred to more specialized units to obtain correct diagnosis. Furthermore, many of our patients experience a wide variety of ineffective treatments before they receive CH diagnosis. Most disturbing is the high rate of patients being treated with morphine and morphine-like analgesics (20.3%), as these are mostly ineffective and entail considerable side effects (23). Further, patients who undergo unnecessary invasive dental procedures (19.8%) are in line with previous studies, but less than our finding of 42.4% in 2007 (6,7,11,17).
The strengths of the study were the large number of participants, the certainty of correct CH diagnosis, and the incorporation of a structured interview after the completion of the survey, which may help reduce recall bias. The broad spectrum of topics assessed in the questionnaire must also be considered a strength.
The limitations were the recruitment of participants from a tertiary headache center, possibly resulting in participation bias, risk of recall bias, not dividing DD into patient's and physician's delay and, finally, not assessing more details about MLF, including aura, nausea and vomiting.
Conclusion
The study has investigated the impact of demographics and clinical phenotype on DD in a large Danish cohort of CH patients. Recent onset of CH symptoms was associated with shorter DD and mean time to diagnosis decreased for every decade. National campaigns, internet access, improved basic medical education, and a higher proportion of patients seen by neurologist may have had a positive impact on DD. However, low age of onset, attack duration > 180 minutes, MLF and nocturnal attacks were associated with a prolonged DD. This implies that, even though DD has improved over time, more education of medical doctors and public awareness is needed to reduce DD for the large number of patients with somewhat atypical CH features.
Footnotes
Clinical implications
The diagnostic delay of CH has been reduced significantly with each decade of onset from 1950–2010.
CH patients with lower age of onset experienced significantly longer diagnostic delay than patients 20–40 years of age and patients > 40 years of age.
Clinical characteristics such as long attack duration, migraine-like features and nocturnal attacks were all associated with long diagnostic delay.
Acknowledgements
The authors would like to thank all the participants. Special gratitude is owed to study nurse Mette Frank Fisker for competent recruitment and data collection.
Author contributions
HF undertook data analyses and interpretation of data and drafted the manuscript; NL undertook data acquisition, data analyses and interpretation and critical revision of the manuscript for intellectual content; MB formed the study concept and design, undertook data acquisition and critical revision of the manuscript for intellectual content; AP undertook data acquisition and critical revision of the manuscript for intellectual content; RJ formed the study concept and design, interpretation of results, and undertook critical revision of the manuscript for intellectual content.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HF declared no conflict of interests. NL received a grant from the Tryg Foundation, travel grants from Candy Foundation, a grant from Cool Sorption, and is sub-investigator for studies funded by Autonomic Technologies (ATI) and Xenon Pharmaceuticals. MB has received honoraria from ATI and an unrestricted research grant from AGA, Linde-Gas. AP has been sub-investigator for a study funded by ATI. RJ has given lectures for Pfizer, Berlin-Chemie, Norspan, Merck and ATI; has been a member of the advisory boards of ATI, Medotech and ElectroCore; a principal investigator on clinical trials sponsored by Eli Lilly; and a principal investigator for ATI and Xenon Pharmaceuticals.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Danish Tryg Foundation.
