Abstract
Background
Erenumab was effective and well tolerated in a pivotal clinical trial of chronic migraine. Here, we evaluated efficacy and safety of monthly erenumab (70 mg or 140 mg) versus placebo in the subgroup of patients who had previously failed preventive treatment(s) (≥ 1, ≥ 2 prior failed medication categories) and in patients who had never failed.
Methods
Subgroup analyses evaluated change from baseline in monthly migraine days; achievement of ≥ 50% and ≥ 75% reduction in monthly migraine days; and change in monthly acute migraine-specific medication days. Adverse events were evaluated for each subgroup.
Results
Treatment with both doses of erenumab resulted in greater reductions in monthly migraine days (primary endpoint) at Month 3 (treatment difference [95% CI], never failed subgroup: −2.2 [−4.1, −0.3] for 70 mg and −0.5 [−2.4, 1.5] for 140 mg; ≥ 1 prior failed medication categories subgroup: −2.5 [−3.8, −1.2], for 70 mg and −3.3 [−4.6, −2.1] for 140 mg; ≥ 2 prior failed medication categories subgroup: −2.7 [−4.2, −1.2], for 70 mg and −4.3 [−5.8, −2.8] for 140 mg). Similar results were observed in the monthly acute migraine-specific medication days endpoint, and in the achievement of ≥ 50% and ≥ 75% reduction in monthly migraine days. There were no new or unexpected safety issues.
Conclusion
Erenumab showed consistent efficacy in chronic migraine patients who had failed prior preventive treatments and was well tolerated across subgroups.
Keywords
Introduction
Few of the currently used preventive treatments for migraine have evidence from randomized, double-blind, placebo-controlled clinical trials; even fewer have evidence for the prevention of chronic migraine (CM), with the exception of topiramate and onabotulinumtoxinA (currently the only drugs approved for CM prevention). All current preventive medications have been repurposed from other indications rather than specifically targeting a pathophysiological pathway known to be relevant in migraine (1–3).
Across the migraine spectrum, low persistence (≤20% of patients at 12 months) and adherence rates (81% of patients had gaps of > 90 days in their migraine prophylaxis in the first year) for oral migraine preventive therapies result in frequent switching, re-initiation, or complete cessation of preventive therapies. As patients cycle through preventive therapies, these discontinuation rates increase (4,5). A high unmet need exists for new, effective, safe, and tolerable preventive therapies for patients who have failed existing preventive treatments due to either insufficient efficacy and/or poor tolerability.
Erenumab (AMG334) is a fully human monoclonal antibody (mAb) that selectively targets and blocks the canonical calcitonin gene-related peptide (CGRP) receptor (6). In a 12-week randomized, double-blind, placebo-controlled study (NCT02066415), erenumab (70 mg and 140 mg) reduced monthly migraine days (MMD), increased the likelihood of achieving ≥ 50% reduction from baseline in MMD (i.e. ≥ 50% responder rate), and reduced monthly acute migraine-specific medication days (MSMD) in patients with CM. The safety profile for erenumab was comparable to placebo (7).
No data are available to inform clinicians on the efficacy and tolerability of preventive medications in patients who have failed treatment with prior preventive therapies. To contribute to clinical decision making, we present here efficacy (change from baseline in MMD and MSMD; ≥ 50% and ≥ 75% responder rates) and safety results of erenumab (70 mg and 140 mg), administered monthly by subcutaneous (SC) injection from a prespecified subgroup analysis of CM patients who had previously failed preventive treatment(s) (≥ 1 prior failed medication category and ≥ 2 prior failed medication categories) and in patients who had never failed preventive treatment. Additional post hoc analyses were performed to evaluate efficacy of erenumab versus placebo in patients who had failed ≥ 3 medication categories.
Methods
The CM study
The main study design, methods and statistical analyses were reported previously (7). In brief, this was a multicenter, randomized, double-blind, placebo-controlled, 12-week parallel-group study of erenumab 70 mg and 140 mg SC monthly in adult patients with CM (≥ 15 headache days/month; ≥ 8 migraine days/month).
Prior preventive treatments and rates of treatment failure a .

Note: Beta blockers include atenolol, bisoprolol, metoprolol, nadolol, nebivolol, pindolol, propranolol, and timolol. SNRIs, serotonin-norepinephrine reuptake inhibitors, include venlafaxine, desvenlafaxine, duloxetine and milnacipran.
Categories are not mutually exclusive and patients may contribute to more than one category; bpercentages represent the number of patients with any prior preventive treatment compared to the overall study population (placebo n = 286; erenumab 70 mg n = 191; erenumab 140 mg n = 190; total n = 667); cpercentages represent total number of patients with category-specific prior preventive treatment failure compared to the number of patients who had received prior treatment; dother includes butterbur/feverfew/magnesium (≥ 600 mg/day)/riboflavin (≥ 100 mg/day), clonidine/guanfacine, cyproheptadine, methysergide, pizotifen, carbamazepine and gabapentin.
Patients were excluded if they had no therapeutic response to more than three preventive treatment categories (i.e. no reduction in headache frequency, duration or severity after administration of the medication for at least 6 weeks, based on the investigator’s assessment) (7). History of partial response (i.e. lack of efficacy) or tolerability issues did not constitute no therapeutic response.
The exclusion criterion was defined in the protocol as no therapeutic response in prophylaxis of migraine after an adequate therapeutic trial to > 3 of the preventive medication categories. No therapeutic response is defined as no reduction in headache frequency, duration or severity after administration of the medication for at least 6 weeks at the generally accepted therapeutic dose(s), and is based on the investigator’s assessment.
Subjects do not meet this exclusion criteria if:
the subject discontinued the medication prior to achieving a therapeutic response due to adverse events related to the medication based on investigator opinion, the subject did not receive an adequate dose of the medication for at least 6 weeks
For a given preventive medication taken previously, the case report form gave the following response options for ending the medication: Insufficient efficacy; poor tolerability; prophylactic medication no longer necessary; other. Thus, while a patient was to be excluded if they had no therapeutic response to > 3 preventive categories as defined above, they were not excluded for insufficient efficacy or poor tolerability (which were recorded based on the investigator’s judgment) and as such, an individual patient could have had ≥ 3 treatment failures and also no therapeutic response to ≤ 3 preventive treatments.
The primary endpoint was change in MMD from baseline to Month 3 of the double-blind treatment phase. The protocol and patient consent information were approved by all relevant ethical review boards, all patients gave written informed consent, and the study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice.
Subgroups
Subgroups were defined on the basis of prior migraine preventive treatment failure either for lack of efficacy and/or unacceptable tolerability, as recorded by the investigator. The number of prior preventive treatment failures for any given subject was based on medication categories. The never failed group included treatment-naïve patients and patients who had been exposed to a preventive treatment but did not fail it due to lack of efficacy and/or unacceptable tolerability.
The following were classified as migraine preventive treatment categories: Topiramate; beta blockers (e.g. propranolol or metoprolol); tricyclic antidepressants (e.g. amitriptyline or nortriptyline); divalproex sodium or sodium valproate; calcium channel blockers (e.g. flunarizine or verapamil); serotonin-norepinephrine reuptake inhibitors; botulinum toxin; antihypertensives (lisinopril or candesartan); or other medications.
Endpoints and statistical analyses
Efficacy analyses of the never failed, ≥ 1, and ≥ 2 prior preventive treatment failure(s) subgroups were considered as prespecified and were planned before the unblinding of treatment assignment. Safety analyses for all subgroups and efficacy analyses of the subgroup with ≥ 3 prior preventive treatment failures were post hoc.
Efficacy analyses comprised the primary endpoint of change from baseline in MMD and key secondary endpoints: Achievement of ≥ 50% and ≥ 75% reduction from baseline in MMD, and change from baseline in monthly acute MSMD (e.g. the use of triptans or ergots).
Each erenumab group (70 mg or 140 mg) was compared to placebo (reference group). For continuous endpoints, adjusted analyses utilized a generalized linear mixed model, which included treatment, visit, treatment by visit interaction, the two stratification factors (region and medication overuse status) and baseline value as covariates, and assumed a first-order autoregressive covariance structure. Observed data were used in analyses without imputation for missing data. For dichotomous endpoints, odds ratios were estimated from a stratified Cochran-Mantel-Haenszel test after imputation of missing data as nonresponse. The main study was not designed or powered to compare differences in efficacy between subgroups. Subgroup analyses included here were not adjusted for multiplicity under a pre-specified hypothesis testing procedure. Statistical significance was determined by comparing descriptive p-values with a nominal significance level at p ≤ 0.05.
Adverse events (AEs) were tabulated for the subgroup without prior preventive treatment failure and the subgroups with ≥ 1, ≥ 2 and ≥ 3 prior preventive treatment failure(s).
Results
Characteristics of prior preventive treatment and treatment failure
In the overall study population, 73.8% (492/667) of patients had previously received preventive treatment (Table 1). The majority of these patients had previously failed at least one preventive treatment, and they account for nearly 70% of the overall study population. Lack of efficacy (73.2%; 360/492) was slightly more common than unacceptable tolerability (66.3%; 326/492) (categories were overlapping). Rates of treatment failure were remarkably similar across drug categories, ranging from 86.0–91.9%, with the exception of botulinum toxin (66.5%) and serotonin-norepinephrine reuptake inhibitors (78.7%).
Baseline characteristics of subgroups
Baseline demographics and clinical characteristics by treatment group.
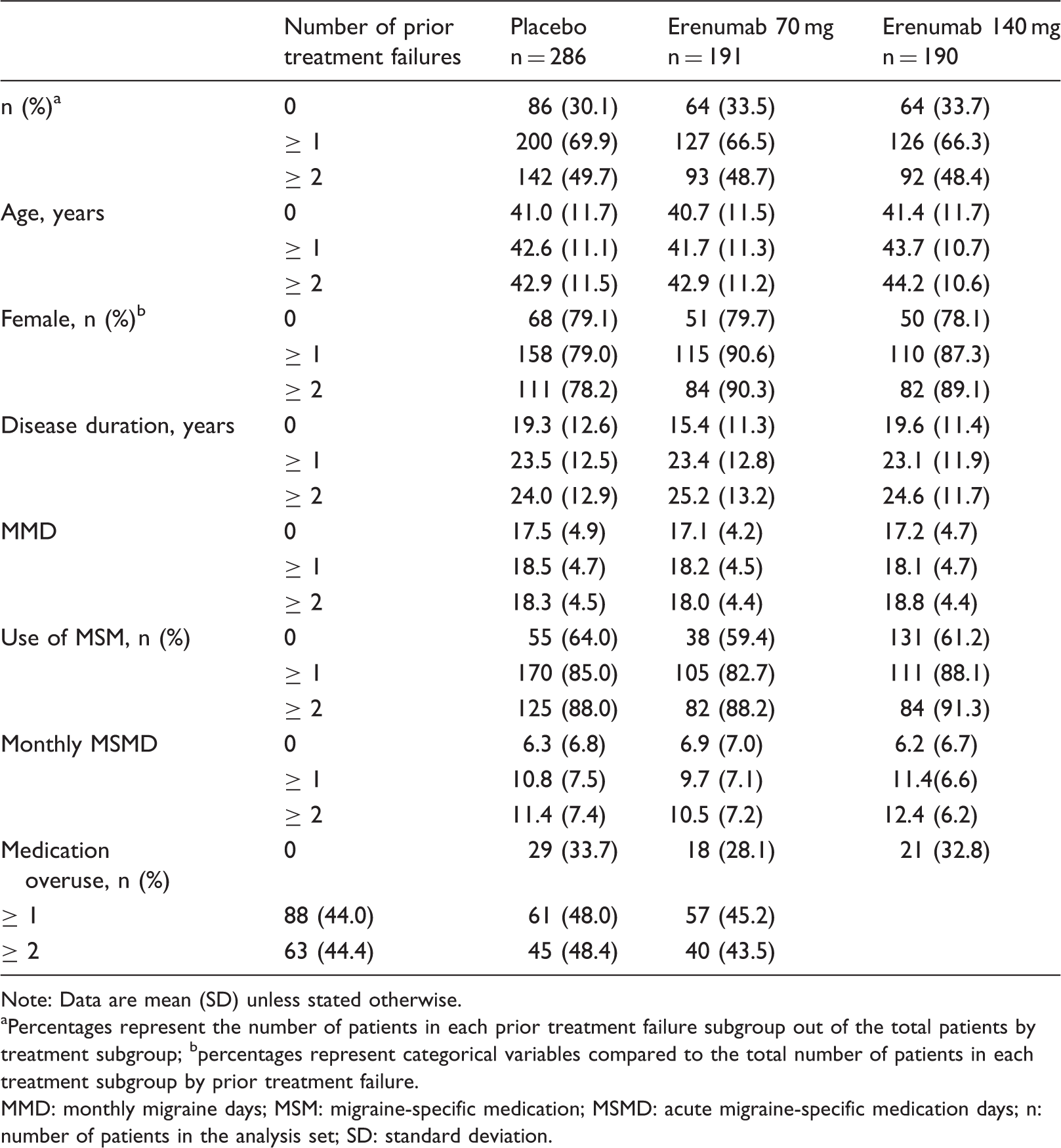
Note: Data are mean (SD) unless stated otherwise.
Percentages represent the number of patients in each prior treatment failure subgroup out of the total patients by treatment subgroup; bpercentages represent categorical variables compared to the total number of patients in each treatment subgroup by prior treatment failure.
MMD: monthly migraine days; MSM: migraine-specific medication; MSMD: acute migraine-specific medication days; n: number of patients in the analysis set; SD: standard deviation.
Efficacy
Greater reductions from baseline in MMD were seen for both doses of erenumab compared with placebo in the prior treatment failure subgroups (≥ 1, ≥ 2 failed prior medications; Figure 1(a) and (b)). Overall, treatment differences at Month 3 for erenumab versus placebo were numerically higher in patients with ≥ 1 or ≥ 2 failed prior preventive treatments than in patients with no prior treatment failure, with the greatest differences in patients that failed ≥ 2 prior preventive medications, particularly for 140 mg (difference in least squares mean [LSM] 95% confidence interval [CI] erenumab 70 mg vs. placebo, −2.7 [−4.2, −1.2], p < 0.001; erenumab 140 mg vs. placebo, −4.3 [−5.8, −2.8], p < 0.001) (Figure 1(b), bottom panel). The treatment difference was driven by a lower placebo response in patients with prior treatment failure. In patients with prior treatment failure, erenumab 140 mg showed consistently higher efficacy than 70 mg in both subgroups (≥ 1 and ≥ 2 treatment failures). Post hoc subgroup analyses of patients with ≥ 3 prior treatment failures were consistent with the results of the prespecified subgroup analyses. In this subgroup, erenumab 70 mg and 140 mg reduced MMD from baseline across all time points, with the greatest numerical improvements at the higher erenumab dose at Month 3 (difference in LSM [95% CI]: erenumab 70 mg vs. placebo, −2.5 [−4.3, −0.8], p = 0.005; erenumab 140 mg vs. placebo, −4.1 [−5.8, −2.3], p < 0.001).
Change from baseline in monthly migraine days over 3 months (a) and placebo-adjusted change from baseline in monthly migraine days at Month 3 (b). In panel (a), data are LSM (95% CI) change from baseline in MMD by treatment groups. In panel (b), data are differences in LSM (95% CI) change from baseline in MMD, erenumab versus placebo at Month 3. Adjusted analysis utilized a linear mixed model, which included treatment, visit, treatment by visit interaction, stratification factors (region and medication overuse status), and baseline value as covariates and assumed a first-order autoregressive covariance structure.
The proportion of patients reaching ≥ 50% reduction from baseline in MMD was greater in patients treated with erenumab 70 mg and 140 mg compared with placebo across all subgroups (0, ≥ 1, and ≥ 2 failed prior medications; Figure 2). Again, a lower placebo response was observed in patients with prior treatment failure. In patients with prior treatment failure, erenumab 140 mg showed consistently higher efficacy than 70 mg in both subgroups (≥ 1 and ≥ 2 treatment failures). Post hoc subgroup analyses of patients with ≥ 3 failed preventive medications showed consistent efficacy of erenumab 70 mg and 140 mg in responders, with a ≥ 50% reduction in MMD at Month 3 (n [%]: placebo, 15 [15.3]; erenumab 70 mg, 23 [34.8]; erenumab 140 mg, 25 [38.5]; odds ratio [95% CI]: erenumab 70 mg vs. placebo, 3.0 [1.4, 6.3], p = 0.004; erenumab 140 mg vs. placebo, 3.5 [1.6, 7.4], p = 0.001).
Proportion of patients reaching ≥ 50% reduction from baseline in monthly migraine days over 3 months. Data are proportion of patients reaching ≥ 50% reduction from baseline in MMD by treatment groups. Data inside bars at Month 3 are odds ratio (95% CI) versus placebo. The adjusted odds ratios and p-values are obtained from a Cochran-Mantel-Haenszel (CMH) test after the missing data are imputed as nonresponse, stratified by stratification factors (region and medication overuse status). The same analysis is repeated at each visit. p-values for pairwise comparisons are nominal p-values obtained from the CMH test using data including placebo and the corresponding erenumab dose group.
The proportion of patients reaching ≥ 75% reduction from baseline in MMD was greater in patients treated with erenumab 70 mg and 140 mg compared with placebo over 3 months across all subgroups (0, ≥ 1 and ≥ 2 failed prior medications; Figure 3). A higher placebo response was noted for this endpoint in patients without prior treatment failure relative to the treatment failure subgroups. In patients with prior treatment failure, erenumab 140 mg showed consistently higher efficacy than 70 mg in both subgroups (≥ 1 and ≥ 2 treatment failures). Post hoc subgroup analyses of patients with ≥ 3 failed preventive medications showed consistent efficacy of erenumab 70 mg and 140 mg in responders with a ≥ 75% reduction from baseline in MMD at Month 3 (n [%]: placebo, 4 [4.1]; erenumab 70 mg, 8 [12.1]; erenumab 140 mg, 15 [23.1]; odds ratio [95% CI]: erenumab 70 mg vs. placebo, 3.4 [1.0, 11.9], p = 0.048; erenumab 140 mg vs. placebo, 7.5 [2.2, 24.8], p < 0.001).
Proportion of patients reaching ≥ 75% reduction from baseline in monthly migraine days over 3 months. Data are proportion of patients reaching ≥ 75% reduction from baseline in MMD by treatment groups. Data inside bars at Month 3 are odds ratio (95% CI) versus placebo. The adjusted odds ratios and p-values are obtained from a Cochran-Mantel-Haenszel (CMH) test after the missing data are imputed as nonresponse, stratified by stratification factors (region and medication overuse status). The same analysis is repeated at each visit. p-values for pairwise comparisons are nominal p-values obtained from the CMH test using data including placebo and the corresponding erenumab dose group.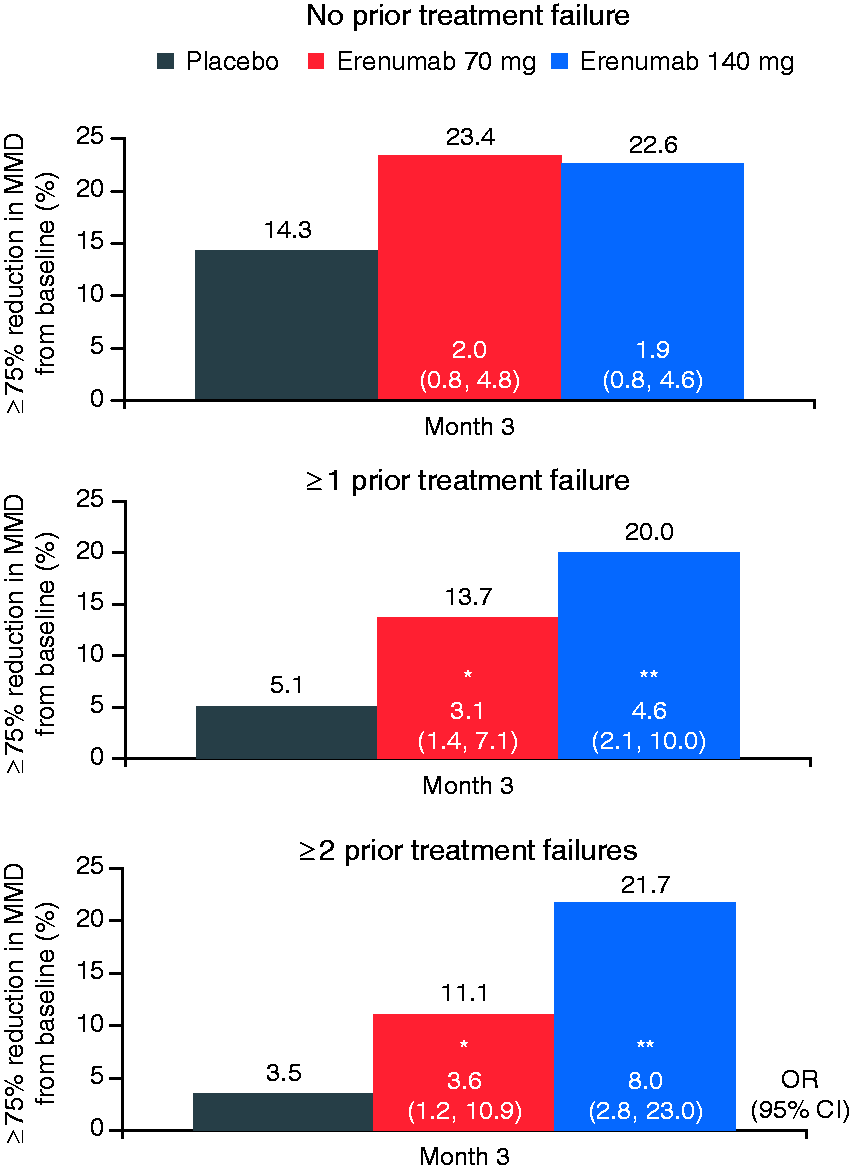
Treatment with erenumab 70 mg and 140 mg reduced monthly acute MSMD compared with placebo across all subgroups (0, ≥ 1 and ≥ 2 failed prior medications; Figure 4). Greater reductions in monthly MSMD were observed in patients treated with both doses of erenumab in the failed ≥ 1 and ≥ 2 subgroups versus patients with no prior treatment failure, which may in part be attributable to the higher baseline use of MSMs in the failed subgroups. Placebo response was higher for the non-failed subgroup on this endpoint than in patients who previously failed preventive treatment, though this is not as striking as for the other two endpoints.
Change from baseline in monthly acute migraine-specific medication days over 3 months (a) and placebo-adjusted change from baseline in acute migraine-specific medication days at Month 3 (b). In panel (a), data are LSM (95% CI) change from baseline in monthly MSMD by treatment groups. In panel (b), data are differences in LSM (95% CI) change from baseline in monthly MSMD, erenumab versus placebo at Month 3. Adjusted analysis utilized a linear mixed model, which included treatment, visit, treatment by visit interaction, stratification factors (region and medication overuse status), and baseline value as covariates and assumed a first-order autoregressive covariance structure.
The proportion of patients with missing data in the primary and secondary endpoints at the end of the double-blind phase (week 12) is 4.8%, 4.3%, and 2.3% in the never failed, ≥ 1 and ≥ 2 failed prior medication categories subgroups, respectively.
Tolerability
Adverse events by prior treatment failure status.

AE: adverse event; SAE: serious adverse event.
Discussion
In this prespecified subgroup analysis, erenumab 70 mg and 140 mg were shown to be efficacious and well tolerated in CM patients with prior treatment failure compared with placebo. Consistent with the results of the main primary analysis (7), erenumab versus placebo significantly reduced MMD and MSMD. More patients achieved ≥ 50% and ≥ 75% reductions in MMD from baseline across all treatment failure subgroups.
Treatment differences for erenumab versus placebo were numerically greater in patients with ≥ 1 or ≥ 2 failed preventive medications than in patients with no prior treatment failure, particularly for the 140 mg dose. In addition, the treatment differences in the patients who had failed existing preventive medications were driven by a lower placebo response than in patients without treatment failure, particularly for the migraine frequency-related endpoints (i.e. change in MMD, ≥ 50% and ≥ 75% response rates). In addition, most migraine attacks last several days (∼4 days in CM) (8), and many patients, following their physician’s advice and due to pricing issues, reserve triptans for a “really bad headache” or “only when severe,” and nonsteroidal anti-inflammatory drugs for the first day and beyond. Although MMD are reduced, triptan users continue to use a triptan on the first day of the attack, and therefore reductions and differences to placebo are less striking.
Patients across all subgroups had a similar mean age, were mostly female, and had similar MMD at baseline. Patients with prior treatment failures had a longer disease duration, higher use of MSM, and more medication overuse at baseline. The treatment effects (i.e. change in MMD) of erenumab 70 mg and 140 mg were accompanied by significant reductions in the number of days per month on which MSM were used in the erenumab groups versus the placebo group; additionally, greater reductions in monthly MSMD were observed with erenumab 140 mg.
For the primary endpoint (MMD), erenumab 70 mg was statistically different to placebo, though 140 mg erenumab was not. The result is unexpected and might be attributable to chance. The result might be due to the between-subject variability, since the randomization was not done within each subgroup. There might be some imbalance between treatment groups within each subgroup, though the observed baseline characteristics appear well balanced. Including a few “strong responders” in the 70 mg group may cause the high mean reduction in that group given the smaller sample size of the never failed subgroup compared to the ≥ 1 and ≥ 2 failed subgroups.
Most preventive medications for migraine have side effects deemed undesirable by patients, such as weight gain, drowsiness, fatigue, and dizziness, which contribute to failed adherence (9). Anxiety regarding the possibility of such side effects, as well as intolerance related to interactions with drugs prescribed to treat other medical conditions, may hinder compliance (10). Importantly, the benefits associated with erenumab versus placebo observed in the subgroups with prior preventive treatment failure were not accompanied by an increased incidence of AEs, and treatment was well tolerated. Although the overall rate of AEs was slightly higher in the subgroups of patients with prior preventive treatment failure than in the subgroup without prior treatment failure, the incidences of AEs were similar between placebo and erenumab groups within the subgroups, and very few patients discontinued the study due to AEs.
The results of the current analysis of erenumab in CM are consistent with the results of a study of erenumab in episodic migraine, in which 39% of patients had failed at least one previous preventive drug class (11), with greater placebo-adjusted treatment differences observed in the treatment failure subgroups compared with the overall population, again largely attributable to a lower placebo response. Together, results from these studies in EM and CM are consistent with lower expectation in patients who have tried and failed treatments, who might also represent a more severely affected population (12).
In a recent retrospective cohort study based on a large claims database, migraine patients who had previously used preventive medications reported poor persistence, with gaps in therapy early in the treatment, and were unlikely to switch prophylactic treatments; most discontinued prophylaxis by the end of the first year (5). Taken with the demonstration of efficacy, clinicians now have an evidence-based treatment showing efficacy for this population.
Limitations of this study are that this is a subgroup analysis from a clinical trial (with a strictly selected patient group), and it remains to be seen how these results translate to the real world. Further, the main study was not designed or powered to test the treatment difference within each subgroup. Therefore, the statistcal testing included in this analysis was not prespecified or adjusted for multiplicity.
The results of this analysis should help to inform clinical decision-making for physicians treating patients who have failed one or more preventive treatments for migraine. Further studies will be required to determine whether erenumab can provide relief in patients who have exhibited no therapeutic response to more than three classes of preventive treatment. To this end, the LIBERTY trial (NCT03096834) in episodic migraine patients with 2–4 prior preventive treatment failures will provide more high-quality data in this population with an important unmet need.
Conclusions
This is the first report showing efficacy, safety, and tolerability of a CGRP pathway antagonist in patients with CM with prior preventive treatment failure(s). A monthly erenumab injection at both 70 mg and 140 mg doses demonstrated consistently greater efficacy than placebo in patients with prior treatment failures (≥ 1, ≥ 2 and ≥ 3), with particularly greater clinical benefit observed for the erenumab 140 mg dose. Our prespecified and post hoc analyses support the utility of erenumab as a potential migraine preventive therapy for patients with CM who have failed other preventive therapies.
Clinical implications
Erenumab had consistent efficacy compared to placebo in patients with chronic migraine across prior treatment failure subgroups (≥ 1, ≥ 2 and ≥ 3). Patients in the non-failed subgroup had a higher placebo response, which resulted in a lower odds ratio compared to other subgroups. Erenumab was effective and well tolerated in patients with prior preventive treatment failure. Erenumab 140 mg gave a greater clinical benefit than 70 mg in this particular subgroup of patients.
Supplemental Material
Supplementary table 1 -Supplemental material for Efficacy and safety of erenumab (AMG334) in chronic migraine patients with prior preventive treatment failure: A subgroup analysis of a randomized, double-blind, placebo-controlled study
Supplemental material, Supplementary table 1 for Efficacy and safety of erenumab (AMG334) in chronic migraine patients with prior preventive treatment failure: A subgroup analysis of a randomized, double-blind, placebo-controlled study by Messoud Ashina, Stewart Tepper, Jan Lewis Brandes, Uwe Reuter, Guy Boudreau, David Dolezil, Sunfa Cheng, Feng Zhang, Robert Lenz, Jan Klatt and Daniel D Mikol in Cephalalgia
Supplemental Material
Supplementary table 2 -Supplemental material for Efficacy and safety of erenumab (AMG334) in chronic migraine patients with prior preventive treatment failure: A subgroup analysis of a randomized, double-blind, placebo-controlled study
Supplemental material, Supplementary table 2 for Efficacy and safety of erenumab (AMG334) in chronic migraine patients with prior preventive treatment failure: A subgroup analysis of a randomized, double-blind, placebo-controlled study by Messoud Ashina, Stewart Tepper, Jan Lewis Brandes, Uwe Reuter, Guy Boudreau, David Dolezil, Sunfa Cheng, Feng Zhang, Robert Lenz, Jan Klatt and Daniel D Mikol in Cephalalgia
Footnotes
Acknowledgements
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MA received personal fees from Alder BioPharmaceuticals, Allergan, Amgen, Alder, Eli Lilly, Novartis and Teva. MA participated in clinical trials as the principal investigator for Alder ALD403-CLIN-011 (Phase 3 b), Amgen 20120178 (Phase 2), 20120295 (Phase 2), 20130255 (OLE), 20120297 (Phase 3), GM-11 gamma-Core-R trials, Novartis CAMG334a2301 (Phase 3 b), Amgen PAC1 20150308 (Phase 2 a) and Teva TV48125-CNS-30068 (Phase 3). MA has no ownership interest and does not own stocks of any pharmaceutical company. MA serves as an associate editor of Cephalalgia and co-editor of the Journal of Headache and Pain. MA is president-elect of the International Headache Society and General Secretary of the European Headache Federation. ST was an employee of Cleveland Clinic during this study and has research grants (no personal compensation) from: Alder, Allergan, Amgen, ATI, Dr. Reddy’s, ElectroCore, eNeura, ScionNeurostim, Teva, and Zosano; he has consulted for: Acorda, Alder, Alexsa, Allergan, Amgen, ATI, BioVision, Cefaly, Charleston Labs, DeepBench, Dr. Reddy’s, ElectroCore, Eli Lilly, eNeura, Gerson Lehman Group, Guidepoint Global, Neurolief, ScionNeurostim, Teva, Pfizer, and Zosano; he has stock options for ATI; a salary from the American Headache Society; and royalties from Springer. JLB received consulting fees, speaking fees, and/or research grants from Allergan, Amgen, Avanir, Depomed, Clinvest, Daiichi Sankyo, Pernix, Merck, Supernus, Teva, Arteaus, and Eli Lilly. UR has received consulting fees, speaking/teaching fees, and/or research grants from Allergan, Amgen, Autonomic Technologies, CoLucid, ElectroCore, and Novartis. GB has research grants from Teva, Novation, Amgen, and Allergan; consulting fees from Allergan, and Novartis; and he is self-employed. DD has consulting fees, and speaking and/or teaching fees from Allergan, Amgen, Biogen Idec, Novartis, Bayer, and Teva. JK is an employee of Novartis. SC, FZ, RL, and DM are employees of Amgen.
Contributors
All authors participated in the study design, implementation, and/or conduct of the study. All authors contributed to the review of the protocol and approved the final manuscript.
Ethical approval and conduct
The study protocol was reviewed by the Independent Ethics Committee or Institutional Review Board for each center. The study was conducted according to the ethical principles of the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Amgen, Thousand Oaks, CA, USA, and Novartis Pharma AG, Basel, Switzerland. Employees of the sponsors were involved in study design, data collection, analysis and interpretation. All authors, including those who are employees of the study sponsors, drafted and/or revised the manuscript and approved the final version for submission.
Patient information and consent
Informed written consent was obtained from each subject before conducting any procedures. Additional information on the study was provided verbally by the study investigator or in a written format.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
