Abstract
Objective
The interplay between neuronal innervation and other cell types underlies the physiological functions of the dura mater and contributes to pathophysiological conditions such as migraine. We characterized the extensive, but understudied, non-arterial diffuse dural innervation (DDI) of the rat and Rhesus monkey.
Methods
We used a comprehensive integrated multi-molecular immunofluorescence labeling strategy to extensively profile the rat DDI and to a lesser extent that of the Rhesus monkey.
Results
The DDI was distributed across a dense, pervasive capillary network and included free nerve endings of peptidergic CGRP-expressing C fibers that were closely intertwined with noradrenergic (NA) sympathetic fibers and thin-caliber nonpeptidergic “C/Aδ” fibers. These newly identified C/Aδ fibers were unmyelinated, like C fibers, but expressed NF200, usually indicative of Aδ fibers, and uniquely co-labeled for the CGRP co-receptor, RAMP1. Slightly-larger caliber NF200-positive fibers co-labeled for myelin basic protein (MBP) and terminated as unbranched corpuscular endings. The DDI peptidergic fibers co-labeled for the lectin IB4 and expressed presumably excitatory α1-adrenergic receptors, as well as inhibitory 5HT1D receptors and the delta opioid receptor (δOR), but rarely the mu opioid receptor (µOR). Labeling for P2X3, TRPV1, TRPA1, and parasympathetic markers was not observed in the DDI.
Interpretation
These results suggest potential functional interactions, wherein peptidergic DDI fibers may be activated by stress-related sympathetic activity, resulting in CGRP release that could be detected in the circulation. CGRP may also activate nonpeptidergic C/Aδ fibers that are likely mechanosensitive or polymodal, leading to activation of post-synaptic pain transmission circuits. The distribution of α1-adrenergic receptors, RAMP1, and the unique expression of the δOR on CGRP-expressing DDI fibers suggest strategies for functional modulation and application to therapy.
Introduction
The functions of the dura mater as a sense organ are poorly understood, especially in light of its implicated contribution to the pathophysiology of migraine, as well as other types of headache. Anatomical studies of the innervation and microvascular structure of the dura have demonstrated that it is richly innervated and highly vascularized (1–7). In this study, we profile the multi-molecular characteristics of the extensive, non-arterial, diffuse dural innervation (DDI), which is distributed across a dense network of dural capillaries and appears to be far more extensive and autonomous from the innervation associated with the major meningeal arterial arbors. These detailed molecular characteristics of the DDI provide insight into how this neuronal and capillary network may normally function at a pain-free subconscious level, as well as in pathophysiologic conditions.
Migraine is a neurologic disorder characterized by multiple phases, including premonitory symptoms, aura, nausea, sensory sensitivity (light, sound, odor), pain, and in some patients, cephalic and extracephalic cutaneous allodynia. Collectively, these symptoms suggest complex sensory integration involving both peripheral and central mechanisms (8–13). Interest in the dura mater as an essential peripheral contributor to migraine has largely focused on the peptidergic CGRP-containing C-fiber innervation to the meningeal arterial arbor (10,14–21). Much evidence supports the conclusion that CGRP plays a key role in migraine. Elevated levels of CGRP in blood plasma and CSF have been demonstrated both during migraine attacks, and in chronic migraine patients between attacks. Experimental infusion of CGRP has been shown to induce migraine in migraineurs (9,16,17,22–24). The efficacy of the triptan family of serotonergic (5-HT1B/1D/1F) agonists and of botulinum toxin peripheral injection in treating migraine is thought to be due, in part, to their ability to inhibit CGRP release (25–28). CGRP receptor antagonists, and peptide or receptor neutralizing antibodies, are also clinically effective in acute and preventative migraine therapy (29–33). However, CGRP is a potent vasodilator and globally mediates cardiovascular function, cutaneous sensation, and tissue homeostasis throughout the body (31,32,34–44). Therefore, strategies for migraine therapeutics that would limit CGRP regulation to the dural innervation are of particular interest.
Here, we demonstrate that the DDI overlies an extensive capillary bed within the rat dura mater and contains vast CGRP-containing peptidergic C fibers. These peptidergic fibers are closely intertwined with an even more extensive noradrenergic (NA) sympathetic innervation and a unique nonpeptidergic innervation that is unmyelinated, characteristic of C fibers, but express labeling for 200kD neurofilament protein (NF200) usually indicative of Aδ fibers. A comparable DDI and capillary maze was confirmed in Rhesus monkey dura. Importantly, the DDI peptidergic fibers express α1 adrenergic and delta opioid (δOR) receptors, but only rarely the mu opioid (µOR) receptor, while the non-peptidergic C/Aδ fibers uniquely express the CGRP co-receptor, RAMP1. These results indicate a complex inter-axonal signaling that, together with non-neuronal cells, underlies both the physiological and pathophysiological functions of the dura mater.
Materials and methods
The experimental design is a variant of the INTiDYN ChemoMorphometric™ Analysis (CMA) platform of comprehensive, integrated multi-molecular immunofluorescence previously applied to serial cryostat sectioning of three-dimensional tissues, in which successive sections are rotated across a slide series such that each slide has multiple sections spaced throughout the specimen (45–50). This approach enables the integrated use of multiple immunolabel and staining combinations across the set of slides, and leverages microanatomical features and landmarks to standardize analyses and distinguish between different types of innervation and other tissue components. The CMA platform has been validated in our previous studies of cutaneous innervation in species ranging from mouse to human, under normal, experimental, and disease conditions, providing insight into the normal stimuli that might be relevant to sensory function (42,46,47,49,51–54). Rodent dura mater is a very thin, nearly two-dimensional membrane that cannot be serially sectioned, but is amenable to whole mount labeling. In this case, we used the highly predictable arborization of meningeal vessels to divide cranial duras from each rat into four standardized sectors for integrated multimolecular CMA. However, Rhesus monkey dura is a substantially thicker structure than rodent dura, and was amenable to sectioning for CMA.
Study objectives
ChemoMorphometric Analysis (CMA) antibody specifications.

All primary antibodies are polyclonal unless indicated otherwise
No detectable labeling
Labeled some innervation affiliated with arterioles
Labeled subsets of fibroblasts
Summary of the different types and proportions of the non-arterial diffuse dural innervation (DDI) of the rat.

CGRP C fibers, NF nonCGRP C/Aδ fibers, and NA sympathetic fibers are intimately intertwined.
Relative proportions: The number of + signs is indicative of the relative proportions of the various types of innervation
Biomarkers: The presence of a + or − sign indicates whether immunolabeling for the antigen was present or absent in the various types of innervation.
A “++” designation indicates that immunolabeling was present in at least half of the innervation.
A “+!” designation indicates that immunolabeling was in the vast majority, but not all of the innervation.
A “?” indicates that the presence of immunolabeling could not be definitvely determined.
CGRP C fiber innervation: At least two, and potentially three, subtypes of CGRP C fibers were detected among the intrinsic dura innervation.
1. Most coexpressed immunolabeling for δOR.
2. A small proportion coexpressed immunolabeling for µOR, but it was not certain whether they also coexpressed immunolabeling for δOR.
3. A small proportion lacked immunolabeling for δOR or µOR.
¥Could not determine whether α2A or Nav1.7 immunolabeling was restricted to a particular subset of CGRP C fiber innervation.
Labeling rationale
Previous detailed CMA of cutaneous innervation was used to help define the profiles observed among the DDI. Briefly, cutaneous sensory innervation can be broadly divided to include myelinated versus unmyelinated, and peptidergic versus non-peptidegic fibers (45,47,66). Cutaneous myelinated sensory innervation typically co-expresses immunoreactivity (IR) for the 200kD neurofilament protein (NF200) as well as myelin basic protein (MBP), and normally consists of larger caliber axons that are classified physiologically as Aβ fibers and substantially smaller caliber axons classified as Aδ fibers (45,47,50,66). Unmyelinated cutaneous sensory innervation has an especially thin caliber axon that is MBP negative and typically lacks NF200, and these are classified as C fibers. Most Aβ fibers, most Aδ fibers, and most C fibers do not express CGRP-IR and are referred to as nonpeptidergic. Cutaneous peptidergic sensory innervation does express CGRP-IR and also usually SP, and constitutes a large minority proportion of C fibers and a small minority of Aδ fibers. Nearly all cutaneous vasculature has associated peptidergic sensory innervation that can release CGRP and SP in the periphery following antidromic stimulation. In rats, cutaneous nonpeptidergic C fibers typically have an affinity for the isolectin B4 (IB4) that is usually lacking among peptidergic C fibers, and among these thin nonpeptidergic fibers many are classified as nociceptors (53,67,68).
Cutaneous autonomic innervation is unmyelinated, and includes sympathetic and parasympathetic fibers. Sympathetic cutaneous innervation typically contains and releases noradrenaline (NA) and immunolabels for the enzyme dopamine-β-hydroxylase (DβH) and a co-expressed neuropeptide, NPY (50,69). The sympathetic NA innervation is a major component of cutaneous vascular innervation, and both NA and NPY are potent vasoconstrictors. Cholinergic innervation typically immunolabels for vasointestinal peptide (VIP), vesicular acetylcholine transporter (VChAT), peripheral choline acetyl-transferase (pChAT), and neuronal nitric oxide synthase (nNOS). Acetylcholine, VIP, and NO mediate vasodilatation, but cholinergic innervation appears to be a relatively small contingent of the innervation to cutaneous vasculature (46,70,71).
Animals
Adult, male SD rats (Harlan Laboratories Inc., Indianapolis, IN, USA) weighing 300–330 g at the time of tissue collection were maintained three per cage in a climate-controlled room at 22 ± 2℃ with free access to food and water on a 12-hour light/dark cycle (lights on at 7am), with all studies conducted during the light cycle. All experimental procedures were approved by the Institutional Animal Care and Use Committee of the University of Arizona.
Dura harvesting and fixation
Whole duras and dural sectors from both crania in 20 Sprague-Dawley rats were used for the CMA innervation panel. To obtain the dura, rats were overdosed with sodium pentobarbitol and perfused intracardially with ice-cold 4% paraformaldehyde (PFA) in 0.1M phosphate buffered saline (PBS) at pH 7.4. Following fixation, the rats were decapitated, the heads were halved along the midsagittal plane, and all soft tissues except the duras were removed from inside and outside the crania. The skulls with attached dura were then immersed in 4% PFA for an additional four-hour postfix at 4℃. The skulls were then rinsed and stored in cold PBS. Within the next few days, the duras were dissected from the skull using a scalpel to score the perimeter along the margin of the tentorium (caudal), the superior sagittal sinus (dorsal), the anterior margin of the cranial fossa (rostral), and the lateral margin of the trigeminal ganglia (ventral). The duras were removed under a dissecting microscope by scraping against the bone from superior to inferior with fine-tipped forceps. The trigeminal ganglia were also excised from the floor of the orbit. After collection, the removed duras and ganglia were stored in PBS at 4℃.
Immunofluorescence labeling procedure
All antibodies were diluted in PBS containing 1% bovine serum albumen with 0.3% Triton X-100, which was also used for two-hour pre-incubations and two-hour rinses between primary and secondary antibody incubations (see Table 1). Whole duras and dura sectors were incubated under primary antibodies for two days with gentle agitation and refrigeration. Incubations in secondary antibodies were undertaken for four hours at room temperature. Cocktails of antibodies raised in different species of mice, rabbits, and sheep were used for double label combinations. Appropriate cocktails of goat and/or donkey raised secondary antibodies were used that were conjugated with either Cy3 for red fluorescence or Alexa 488 for green fluorescence. The antibody solutions also contained DAPI to label cell nuclei in blue fluorescence. After immunolabeling, the duras were rinsed for four hours in PBS and mounted under coverslips in 90% glycerin in PBS, where they could be maintained for long-term storage at −20℃. Coverslips could also be floated off in PBS for future additional labeling with other antibodies as desired and feasible.
Imaging and digital capture
Fluorescent images were captured utilizing an Olympus BX51-WI microscope equipped with conventional fluorescence filters (Cy3: 528–553 nm excitation, 590–650 nm emission; Cy2/Alexa488: 460–500 nm excitation, 510–560 nm emission; DAPI: 330–350 nm excitation, 450–470 nm emission); a Hamamatsu ER, DVC high-speed camera; linear focus encoder, and a three-axis motorized stage system interfaced with Neurolucida software (MBF Bioscience, Essex, VT). High resolution, seamless, whole dura montages were created with the 10X and 20X objectives using the automontage and auto-focus feature of Neurolucida (Figures 1 and 2). Figures were composed with Photoshop (Adobe, San Jose, CA). In general, we favored low contrast images that captured the full range of labeled pixels as seen through the microscope with minimal contrast enhancement.
An overview of the rat dura mater and its innervation as seen in a whole mount montage of high-resolution immunofluorescence images of the entire innervation labeled for PGP (green) and the peptidergic sensory innervation double labeled for CGRP (red, see #1, Table 2). Cell nuclei are counterstained with DAPI. The meningeal nerve enters the ventral caudal aspect of the dura along with the middle meningeal artery. Some sensory and sympathetic innervation is intimately associated with the artery and its branches. Numerous small nerves radiate into the dura from all around its perimeter and provide a dense, diffuse innervation throughout the dura, which is not associated with the arterial arbor. The tributaries of the middle meningeal artery have a consistent pattern that provided a landmark to cut duras into four standardized sectors (broken lines) which were used to conduct four pairs of multi-molecular double label combinations per dura and eight combinations per rat. Scale bar = 500 µm. A 12 × enlargement of the sector shown in the small white rectangle reveals the dense diffuse innervation (DDI) labeled with PGP (green), of which a vast proportion are peptidergic sensory axons labeled for CGRP (red) (#1, Table 2). Scale bar = 500 µm.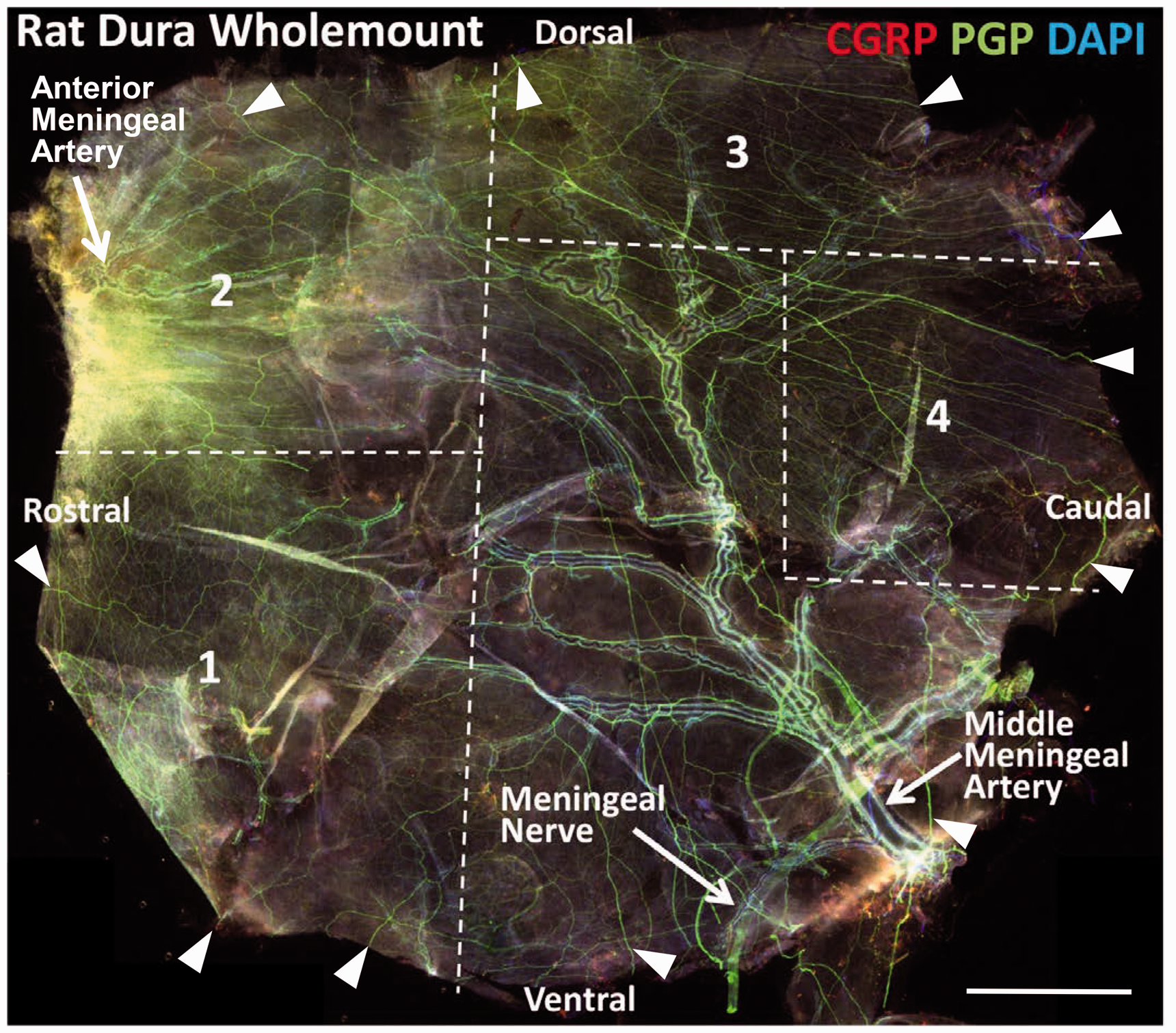

Multi-molecular profiling of dural innervation
Peptidergic versus non-peptidergic innervation
In order to map the arterial arbors and identify comparable sectors for comprehensive multi-molecular analyses of the DDI, whole mounts of 10 duras from 10 different rats were labeled in their entirety with sheep anti-CGRP and rabbit anti-PGP9.5 (PGP), a pan-neuronal enzyme that has been shown to be expressed in all types of peripheral innervation under normal conditions (Figure 2). Double labeling for both CGRP and PGP was interpreted as peptidergic sensory innervation. This was confirmed, and other molecular characteristics defining potential subtypes were assessed through double label combinations of sheep or rabbit anti-CGRP combined with the lectin IB4 and a variety of antibodies against other antigens (Table 1) applied to at least three dura sectors from three different rats for each combination (Figures 3–9).
A histofluorescence and immunofluorescence overview of major basic features of the dura structure and innervation. In this and all subsequent figures, pairs of double labeling are shown in panels with the labeled antigens listed above the panels. Panels may include separate images for the red and green labeled antigens and the merged images for both antigens. Arrowhead indicators are red or green if only one of the antigens is detected, or yellow if both antigens co-localize. (a) Histofluorescence with the isolectin IB4 (red) labels the endothelial linings of a dense capillary network (open arrowhead) distributed throughout the dura. IB4 also co-labels virtually all of the overlying diffuse peptidergic sensory innervation, which is labeled for CGRP (yellow arrowheads; #1, Table 2). (b) Double labeling for anti-NF200 (red) and anti-PGP (green) reveals that a substantial proportion of the total innervation expresses NF200 (yellow arrowheads; #3 and 4, Table 2). Some innervation lacks NF200 (green arrowheads). See Figure 5 for details. (c) A large proportion of the total innervation co-expresses immunoreactivity for DβH and NPY (yellow arrowheads), indicating that this is noradrenergic sympathetic innervation (#5, Table 2). See Figure 6 for details. Scale bar = 100 µm for (a) and (b), 50 µm for (c). Detection of a nonpeptidergic C fiber innervation and Nav1.7 expression in peptidergic innervation. (a) Virtually all of the innervation that labels for CGRP also co-labels with IB4 (yellow arrowheads; #1, Table 2). As shown at 2 × higher magnification in the inset, sparse fibers are only labeled with IB4 (red arrowheads). This nonpeptidergic innervation lacks labeling for NF200 and MBF as well as DβH and NPY (not shown), indicating that it consists of C fibers (Table 2, #2). (b) Nav1.7 immunolabeling was detected on some innervation that coexpresses CGRP (yellow arrowheads), but not on other CGRP fibers (green arrowheads; #1, Table 2). Some fibers labeled for Nav1.7 lack CGRP and may be sympathetic (red arrowheads; #5, Table 2). Scale bar = 50 µm.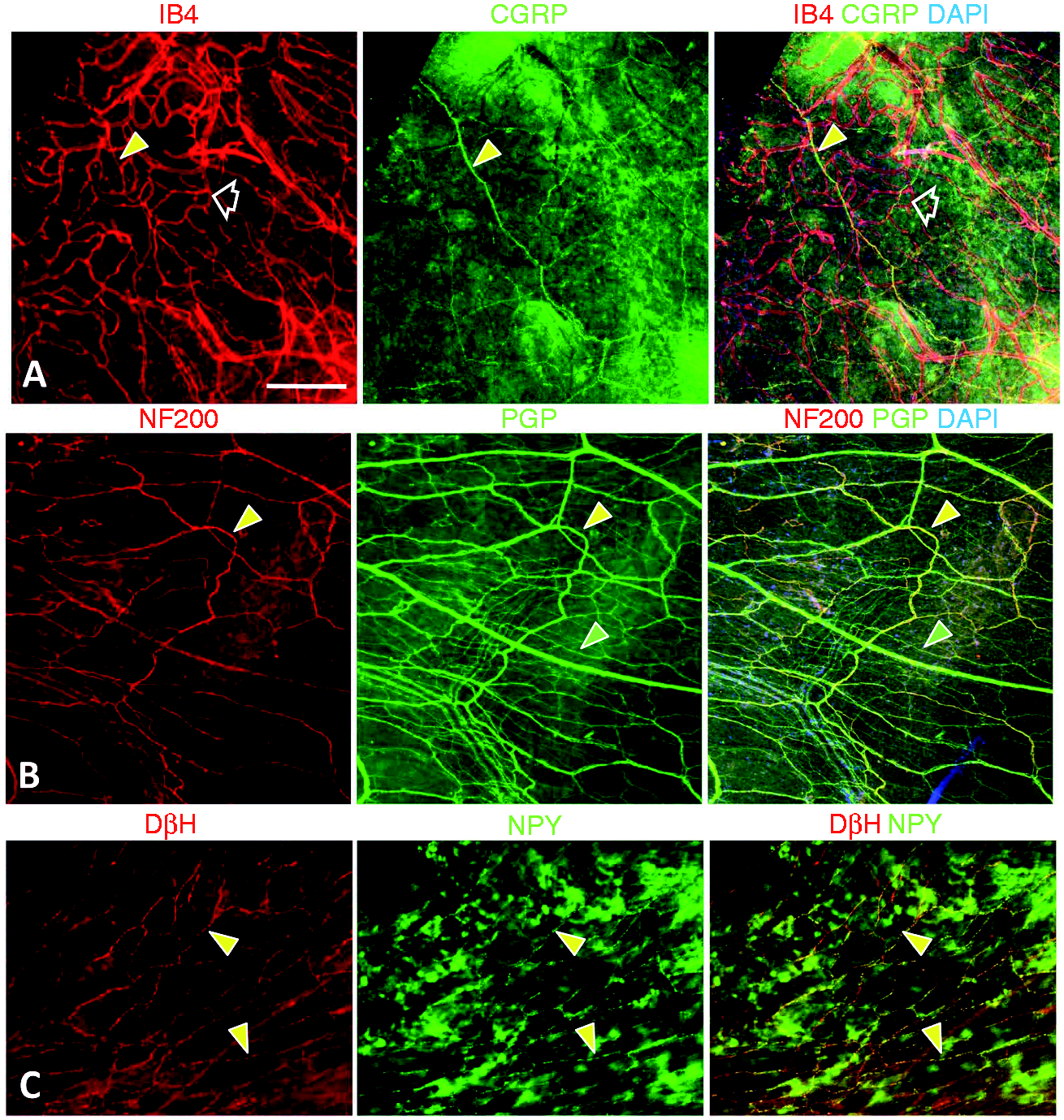

Myelinated versus unmyelinated innervation
Another 10 whole duras from 10 different rats were double labeled for anti-NF200 and anti-PGP (Figure 3b). In studies of normal cutaneous innervation, NF200-IR was consistently co-expressed with myelin basic protein (MBP)-IR. Double labeling with mouse anti-NF200 and rabbit anti-MBP antibodies was used on dura sectors to determine whether the NF200-labeled innervation was myelinated or if any was unmyelinated (Figure 5). Mouse and rabbit anti-NF200 antibodies as well as rabbit anti-MBF antibody were used in combinations with a variety of antibodies against other antigens to profile other molecular characteristics and potential subtypes (Figures 3b and 5). Any innervation that lacked NF200-IR was interpreted as unmyelinated sensory C-fibers or autonomic fibers, which was confirmed with other label combinations.
Potential functional inter-relationships between different subsets of sensory innervation that express CGRP or NF200. Areas shown in small white rectangles are shown at twice the magnification in accompanying insets in large white rectangles. (a) Double labeling for myelin basic protein (red) and NF200 (green) revealed that all of the myelinated axons express NF200 (yellow arrowheads) and have a relatively thicker caliber than all other axons that innervate the dura (#4, Table 2). Numerous thinner caliber axons express NF200 and lack MBP (green arrowheads; #3, Table 4). As shown in the lower left insets, NF200 axons that lack MBP can be detected in nerves running parallel to the myelinated NF200 axons, indicating that they are likely a unique type of fiber and not merely unmyelinated branches of the myelinated axons. The lower left inset also shows that the unmyelinated NF200 axons terminate as simple endings with few if any branches. (b) Double labeling for CGRP and NF200 indicates that these antigens are expressed almost entirely in separate types of axons (red and green arrowheads; #1, #3, and #4, Table 2) that are often closely intertwined (upper right insert). Faint CGRP labeling was rarely detected in some very thin NF200 with unmyelinated axons (#3, Table 2). The upper right inset also shows a simple branched ending of an NF200 fiber. (c) The myelinated NF200 fibers (yellow arrowheads) terminated as unbranched bulbous endings (green arrowheads) shown at a 2X higher magnification in the insets. (d–f) RAMP1 immunolabeling was only detected on axons that express NF200 (yellow arrowheads in (e); #3 and #4, Table 2), and was not expressed on sensory axons labeled for CGRP or noradrenergic sympathetic axons labeled for NPY (red and green arrows in (d) and (f); #1 and #5, Table 2). RAMP1-IR was also expressed among the diffuse capillary network and arterial smooth muscle (red arrows in d–f). Scale bar = 50 µm in (a) and (b), 25 µm in (c)–(f). Some potential functional interactions between noradrenergic sympathetic and CGRP expressing sensory innervation. (a) Double labeling for DβH and CGRP revealed that virtually every CGRP labeled sensory fiber (green arrowheads; #1, Table 2) was closely intertwined with a DβH labeled noradrenergic sympathetic fiber (red arrowheads; #5, Table 2). Many more DβH fibers were unaccompanied by a sensory axon, making the sympathetic innervation the largest contingent of the dura innervation. (b–e). Double labeling for CGRP and various alpha adrenergic receptors revealed that nearly all of the CGRP fibers expressed the α1B receptor (yellow arrowheads in (c)), and many but not all of the CGRP fibers also co-expressed labeling for α1A and α2A receptors (yellow and green arrowheads in (b) and (d)). Some fibers expressed α1A labeling without CGRP (red arrowheads in (b)). Double labeling revealed that α1A is co-expressed on at least some noradrenergic sympathetic fibers that label for NPY (yellow arrowheads in (e)). Likewise, α1A and NPY co-labeling resulted in some fibers that only labeled for α1A (red arrowheads in (e)) that are likely those that co-express CGRP (yellow arrowheads in (b)). α1B and α2A labeling was not detected on the sympathetic innervation (not shown). α2C was not detected on any of the dura innervation. α1A, α1B, and α2A antibodies all produced labeling among non-neuronal cells in the dura which, by their sheer numbers, likely include fibroblasts. α2A appears to be expressed on the capillary endothelial cells. Scale bar = 50 µm.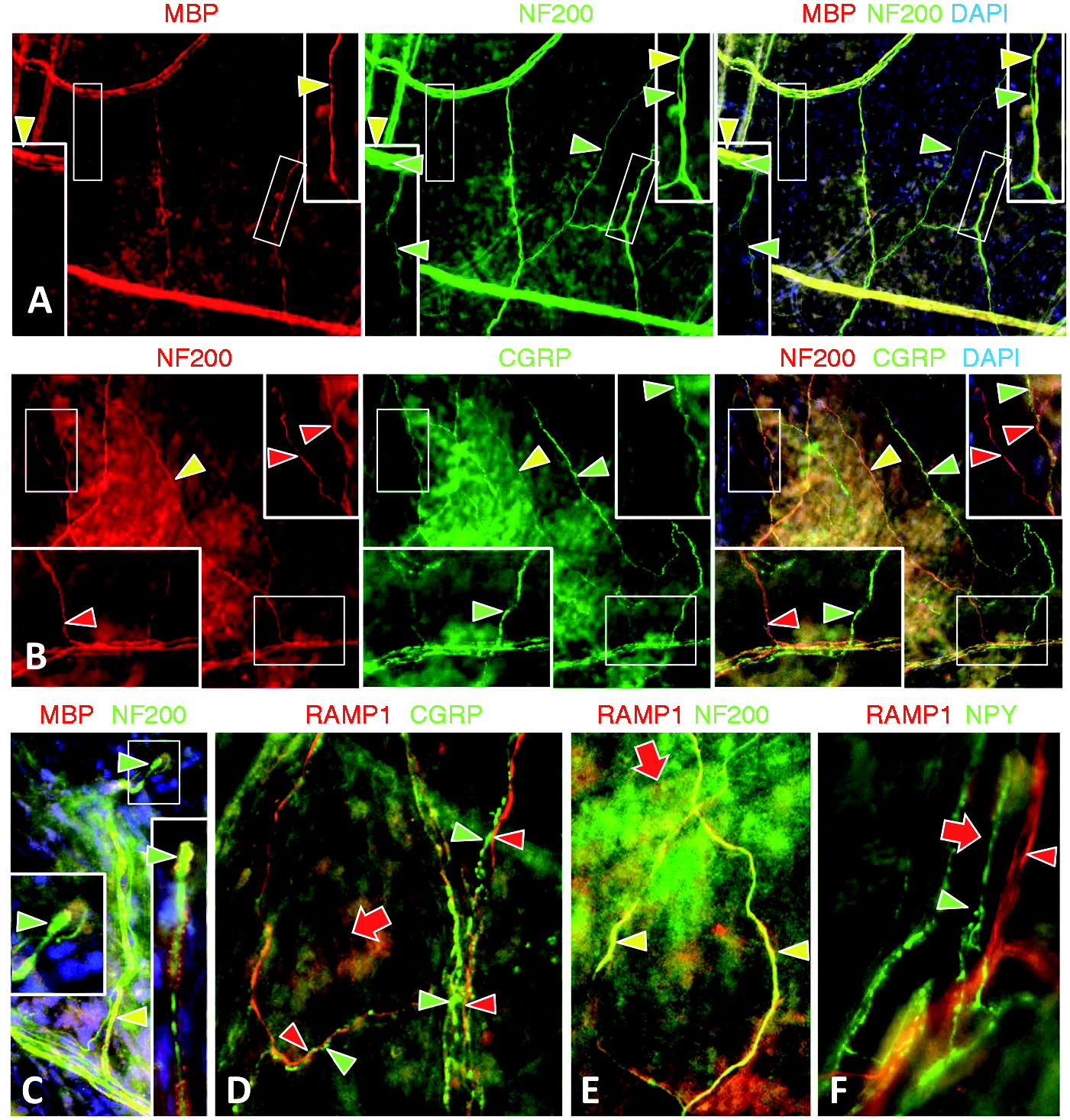
Autonomic innervation
Various antibody combinations were used to profile autonomic innervation. Rabbit anti-DβH and sheep anti-NPY antibodies were used to identify noradrenergic sympathetic innervation as distinct from all types of sensory and potential cholinergic presumptive parasympathetic innervation (Figures 3c and 7). Various rabbit antibodies against VIP, pChAT, VChAT, and nNOS were used to detect cholinergic innervation.
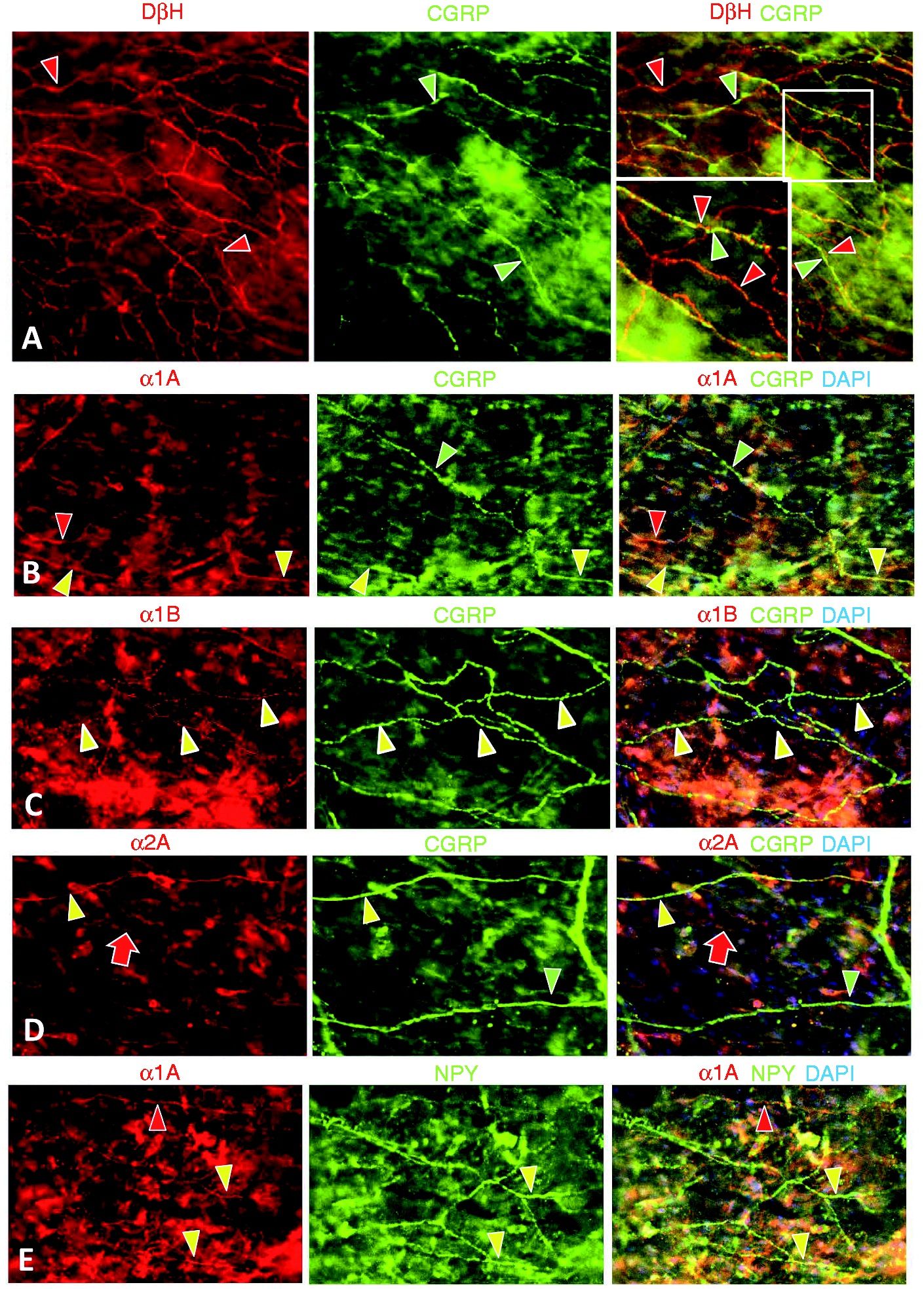
Serotonin (5-HT1D) receptor expression was not detected on NPY sympathetic fibers, but was detected on peptidergic sensory innervation in the dura (#1, Table 2). (a) Double labeling detected 5HT1D expression on fibers (red arrowheads) running intertwined with NPY fibers (green arrowheads). The areas shown in small white squares are shown at twice the magnification in accompanying insets, demonstrating an apparent ending with intertwining to the full extent. (b) 5HT1D (red arrowheads) and NPY (green arrowheads) remain segregated but intertwined among the fibers of DDI. (c) In contrast, double labeling detected 5HT1D expression on nearly all fibers colabeled with CGRP (yellow arrowheads). Note also the strong immunoreactiviy of CGRP and 5HR1D in additional cells of the dura. (d) Fine CGRP peptidergic fibers of the DDI coexpress 5HT1D (yellow arrowheads). Scale bar = 25 µm in (a) and (b), 50 µm in (c) and (d).
Quantification of different types of DDI
Based on the various combinations of double labeling, the different types of DDI are summarized in Table 2. The relative quantities of different types of DDI were determined by the order in which the various paired combinations produced the most labeled fibers.
Trigeminal ganglion (TG) assessments
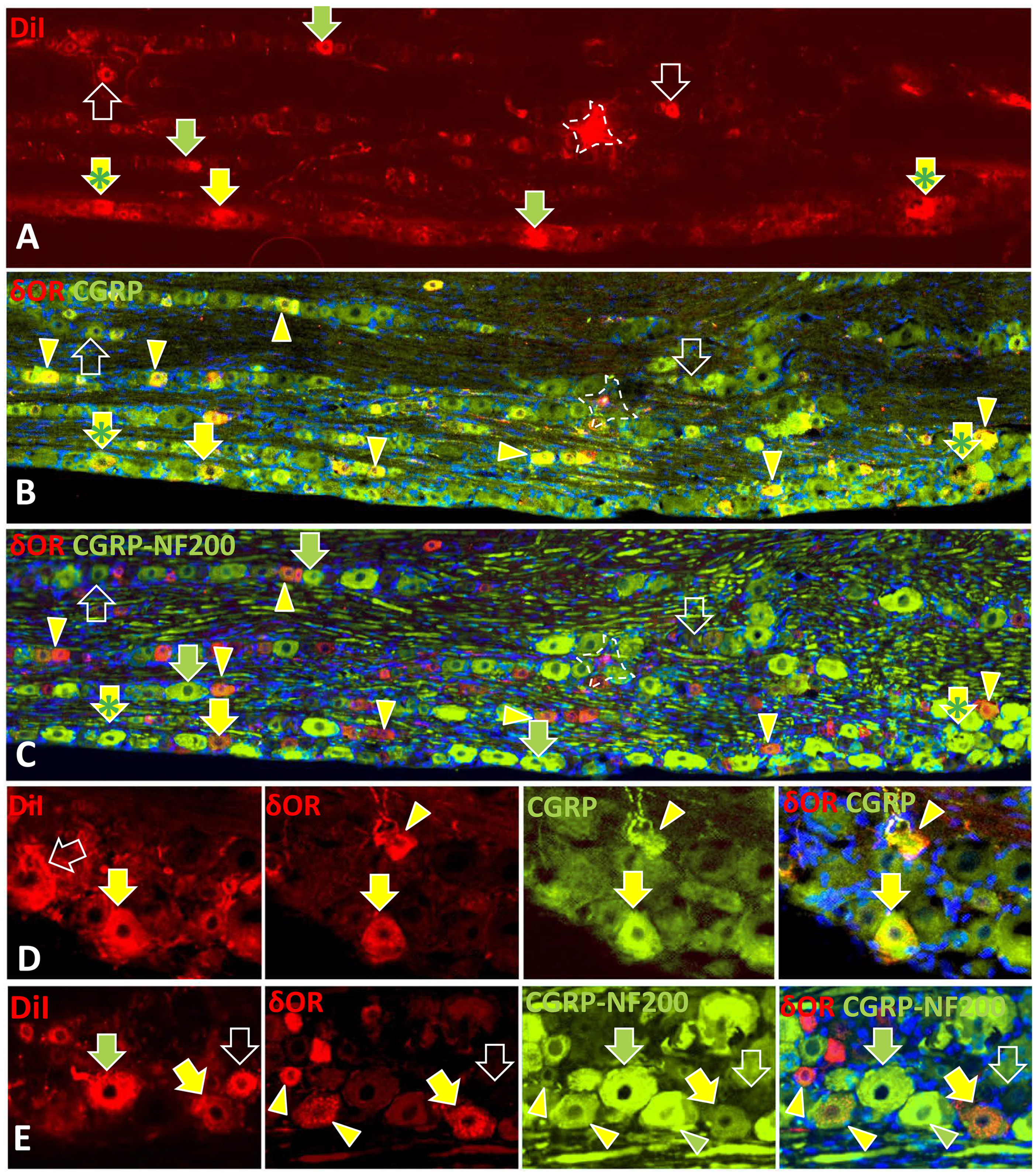
In order to confirm whether trigeminal neurons that definitively innervate the dura have immunolabeling characteristics consistent with the profiles of the innervation in the dura, retrograde labeling of TG neurons innervating the dura was done via the fluorescent tracer 1,1'-dioctadecyl-3,3,3',3'-tetramethylindocarbocyanine perchlorate (DiI) solution (20 mg/ml in PBS with 10% DMSO) applied onto the exposed dura as previously reported (72) . Several rats were anesthetized with ketamine/xylazine (80/12 mg/kg, i.p.), the scalp locally incised, and DiI crystals applied to the intact dura through four small holes in the cranium as previously described (72). Rats were subsequently housed individually in the animal facility for five to six days to allow the transport of the tracer to the somata in the TG. This method resulted in retrogradely-labeled neurons in both TG. The rats were euthanized and perfused transcardially with 4% PFA-PBS as described above. Soft tissues were removed from the halved skulls, the skulls were postfixed for four hours, and stored in buffer for subsequent removal of the duras and trigeminal ganglia. The trigeminal ganglia were immersed in cold 30% sucrose in PBS overnight and sectioned longitudinally by cryostat at a 14 µm thickness in a horizontal plane from inferior to superior. Alternating sections were rotated over five slides for detection of neurons labeled by DiI transported retrogradely from their axons terminating in the dura. Taking advantage of the fact that the DiI dissolved out of the sections during immunolabeling, the sections were first coverslipped with PBS and seamlessly montage imaged in their entirety for red fluorescence with a 10X objective using Neurolucida software automontage and autofocus. Neurolucida mapping routines were used to outline each section and the location and perimeter of each DiI labeled neuron, along with tissue morphology landmarks such as blood vessels and connective tissues of the TG. The coverslips were then removed and the TG sections were processed for double-label antibody combinations selected to confirm co-expression profiles observed on the dural innervation. The sections were re-montaged for immunofluorescence, and these montages were aligned with the outlines mapped for DiI in order to assess the immunolabeling characteristics of the DiI labeled neurons (Figure 10). The duras were also mounted on slides under PBS mounted coverslips to observe DiI labeled axons.
Multi-molecular profiling of Rhesus monkey dura innervation
In order to determine whether some key findings in the rat dura would translate to primates and possibly human, dura maters were obtained post mortem from two euthanized Rhesus monkeys from the primate center at the University of Washington and fixed by immersion for four hours in 4%PFA-BSA. The duras were stored in cold PBS and sectors ∼5 × 5 mm were excised for analysis. The Rhesus dura was too thick for antibody penetration and visualization as whole mounts, therefore, the sectors were serially sectioned by cryostat at a 20 µm thickness parallel to the dura surface. The sections were thaw mounted onto slides for double immunofluorescence labeling and imaging as described previously for skin biopsies (Figure 11) (48).
Results
Non-arterial diffuse dural innervation (DDI)
As shown in a dural whole mount preparation (Figure 1), we defined four sectors that could be separated for each dura, i.e., eight sectors for each rat. Our initial whole mount analyses of PGP labeling revealed numerous small nerves that radiated into the dura from the entire perimeter, in addition to the innervation associated with the major meningeal arterial vessels (Figure 1). These small nerves have been observed previously and create a plexus that provides a dense, diffuse innervation throughout the dura that is not associated with the major meningeal arterial or venule arbors (2,73,74). The diffuse dural innervation (DDI) appears mostly independent of the peri-arterial innervation, however there are anastomoses where branches from the small nerves distribute to arterial branches and arterial nerves contribute to the DDI. The distinct larger meningeal nerve enters the ventral-caudal aspect of the dura along with the middle meningeal artery, and the fibers closely parallel and intimately associate with, the large middle and smaller anterior meningeal arteries and their branches. While a few small nerves originated from the middle meningeal nerve to distribute to the DDI, most of the middle meningeal nerve supplies the peri-arterial innervation to the middle meningeal artery and its branches. Although in the literature the DDI appears often and is described as having free nerve ending terminations among the fibrous connective tissue of the dura, an underappreciated anatomy remains regarding the large and copious capillary bed among the dura. Here, we hypothesize that the DDI creates a large interface with the dural capillary bed and provides a major surface area of interaction for peptidergic regulation of vascular dynamics, rather than simply the cerebral or main meningeal vessels.
Double labeling combinations with anti-PGP revealed that the small non-arterial nerves contained a variety of immunochemically distinct axons. The DDI morphology is an anastomosing plexus consisting of tortuous bundles of a few tightly intertwined, thin caliber axons of different types, of which nearly all terminate as sparsely branched free nerve endings (Figures 3–8, Table 1). The vast majority of the multi-axonal bundles contained one or more CGRP labeled axons (Figures 2–7). Fine caliber axons that labeled for NF200 were also widely distributed among most of the anastomosing bundles (Figures 3b and 5a).
Double label combinations revealed that CGRP and NF200 labeled fibers were often tightly intertwined even to the level of the definitive branched endings (Figure 5b). However, very few fibers co-expressed NF200 and CGRP, indicating that nearly all of these are separate types of innervation. Both the CGRP and NF200 fiber types lacked co-expression of adrenergic and cholinergic immunochemical properties, indicating that they were sensory innervation. The CGRP labeled fibers did not express immunolabeling for MBP as well as NF200, defining that they were unmyelinated peptidergic C fibers (Figures 5a, b; #1, Table 2).
Surprisingly, NF200 labeled fibers consisted of some that co-labeled with MBP, indicating that they were myelinated, however the overwhelming majority lacked MBP and were presumably unmyelinated. The presence of MBP positive and negative NF200 fibers running parallel to each other within the nerves and occasionally splitting off from nerves, sometimes as intertwined pairs of axon fibers (Figure 5a), indicates that unmyelinated fibers are not terminal branches of the myelinated fibers, but are separate types of NF200 sensory innervation (#3 and #4, Table 2). Moreover, the thinner NF200 positive, MBP negative fibers could be traced to sparsely branched free nerve endings (Figure 5a, lower left insert) whereas the slightly larger caliber MBP positive fibers terminated as single unbranched bulbous endings (Figure 5c). The lack of myelin on the thinner NF200 positive axons is a characteristic of C fibers, however, in the skin, virtually all such thin caliber NF200 axons are myelinated, indicative of Aδ fibers. Therefore, this newly identified, unmyelinated thin-caliber NF200 positive component of the DDI may be a unique type of C/Aδ fiber (#4, Table 2). On the other hand, the myelinated NF200 positive DDI axons are the caliber of a cutaneous Aδ fiber, but their unbranched, bulbous terminals are more characteristic of the distinctive morphological endings of much larger caliber cutaneous Aβ fibers.
To test whether there were additional types of unmyelinated innervation that lacked CGRP or NF200, double labeling was conducted with the isolectin-B4, which has been shown to detect nonpeptidergic C fibers in rat skin. Interestingly, among the rat DDI, virtually all CGRP positive fibers co-labeled for IB4 (Figure 4a), whereas, in the skin, IB4 and CGRP labeling are usually on separate types of nonpeptidergic and peptidergic C fibers. No NF200 positive fibers labeled with IB4. Sparse IB4 labeled fibers were present that lacked CGRP, indicating that there was a small contingent of nonpeptidergic NF200 negative C fibers (#2, Table 2). Importantly, IB4 also labels endothelial cells and revealed that the rat dura contains a vast and diffuse network of anastomosing capillaries (Figure 4a). The DDI was spread across this dural capillary bed and could be visualized in the same focal plane, but was not intimately associated with the capillary contours.
Labeling with anti-DβH revealed that all of the DDI plexus contained a contingent of NA sympathetic fibers, of which nearly all co-labeled for NPY (Figure 3c). Virtually all CGRP fibers were closely intertwined with an NA fiber right to the level of their termination (Figure 6a). However, a large proportion of NA fibers were present that were not accompanied by any sensory fibers, making the NA sympathetic fibers the largest contingent of the DDI. Immunolabeling for VIP, pChAT, VChAT, and nNOS failed to reveal any cholinergic parasympathetic contingent among the DDI, however several such fibers were detected among the innervation to the meningeal arterial arbor (not shown).
In summary, the DDI consists of at least five types of innervation, as detailed in Table 2. These include: (1) noradrenergic sympathetic fibers, which are the most extensive (#5); (2) peptidergic unmyelinated IB4-positive C fibers, which are the next most extensive (#1); (3) nonpeptidergic unmyelinated NF200 positive C/Aδ fibers, which are also a major contingent (#3); (4) nonpeptidergic myelinated NF200 positive Aδ/β fibers (#4), and (5) a small contingent of IB4 positive nonpeptidergic C fibers (#2).
Potential mechanisms for modulation and interaxonal signaling
The intertwining of multiple axonal types within the DDI plexus led us to assess potential molecular substrates for excitatory and inhibitory modulation especially involving inter-axonal signaling.
Expression of receptor activity modifying protein (RAMP1)
As is noted above, separate individual CGRP and NF200 fibers are frequently closely intertwined in the diffuse dural plexus. As had been shown previously, immunolabeling for RAMP1, which is essential for CGRP-specific signaling of the Calcitonin Receptor Like Receptor, was expressed uniquely on NF200 positive innervation. Our results indicated that RAMP1 is expressed on virtually all of the NF200 innervation of the DDI, especially the nonpeptidergic C/Aδ fibers (Figure 5d–f). This suggests that the CGRP fibers can exert an excitatory modulation of the nonpeptidergic C/Aδ innervation. RAMP1 was not expressed on closely intertwined NA innervation, nor on the CGRP fibers themselves. RAMP1 was also present on the smooth muscle of arterial walls, as shown previously, and was particularly well distributed among the diffuse network of dural capillaries (Figure 5d–f).
Expression of adrenergic receptors
As was shown above, virtually all of the CGRP fibers were closely intertwined with NA sympathetic fibers (Figure 6a). Having previously observed α2C expression on CGRP fibers that are intermingled with NA innervation to human cutaneous arterioles, we assessed immunolabeling for a variety of adrenergic receptors among the DDI (Table 1). We failed to detect α2C among any of the innervation in the dura including the peri-arterial innervation. However, labeling for α1B occurred uniquely to, and on nearly all of the CGRP fibers (Figure 6c). Labeling for α2A occurred uniquely on only a minor subset of CGRP fibers (Figure 6d). Neither α1B nor α2A was detected on NA sympathetic fibers. By contrast, α1A was detected both on at least some CGRP fibers (Figure 6b) and on at least some sympathetic fibers (Figure 6e). None of the adrenergic receptors were detected on any NF200 innervation. These results indicate that sympathetic NA innervation may exert primarily an excitatory modulation of the peptidergic C fibers as well as some excitatory autoregulation. The antibodies for α1A, α1B, and α2A also produced labeling among many non-neuronal cells that have yet to be specifically identified, but likely include at least some dermal fibroblasts (Figure 6d). In addition, α2A appears to be expressed on the endothelial cells of the capillary network.
Expression of serotonin receptors
The clinical effectiveness of the triptans in treating migraine led us to investigate the expression of 5-HT1D receptors among the DDI plexus. Labeling for 5-HT1D was not detected on the NPY sympathetic innervation (Figures 7a, b), whereas 5-HT1D was co-expressed among most of the CGRP fibers (Figures 7c, d). Therefore, the therapeutic actions of the triptans may be mediated, in part, via inhibition of CGRP release among this vast DDI/capillary network.
Expression of opioid receptors
We next investigated the expression of µOR and δOR among the DDI plexus. Labeling for µOR was detected rarely and on only a small proportion of CGRP labeled fibers (Figure 8b), whereas the majority of CRGP fibers co-expressed labeling for δOR (Figure 8a). The δOR result was confirmed using three different antibodies, including a rabbit monoclonal antibody (Table 1). Because all of the µOR and δOR antibodies were raised in rabbit, we were not able to investigate whether both opiate receptors were co-expressed on some of the peptidergic fibers. However, a sizeable minority of CGRP fibers lacked labeling for either opioid receptor. Whether these differences in OR expression among the CGRP fibers of the DDI is indicative of different functional subtypes (#1a–c, Table 2) remains to be determined.
Opiate receptor expression on peptidergic sensory innervation in the dura. (a) Double labeling detected δOR expression on most (yellow arrowheads) but not all (green arrowheads) of the CGRP sensory innervation in the dura (#1a, Table 2). (b) Only rare CGRP fibers labeled for the μOR (yellow versus green arrowheads; #1b, Table 2). Scale bar = 50 µm in (a), 25 µm in (b).
Voltage-gated sodium channel Nav1.7 expression
Nav1.7 has been strongly linked to human pain disorders (75). Immunolabeling for Nav1.7 was detected on much, but not all of the CGRP innervation (Figure 4b). Nav1.7 labeling was expressed on some CGRP negative fibers, but was not co-expressed on NF200 innervation (not shown). Double labeling for NPY indicated that Nav1.7 was also expressed on some NA sympathetic fibers.
Growth associate protein (GAP)43 expression
GAP43 is expressed in all growing peripheral axons during development, especially at their growth cones, and is assumed to down regulate once the axons acquire their targets. In our previous assessments of cutaneous innervation, GAP43 was down regulated in myelinated axons consisting of both the Aβ and Aδ fibers that all co-express labeling for NF200 and MBP. However, GAP43 continued to be expressed among most of the mature cutaneous C fibers and sympathetic fibers. Among the DDI, GAP43 labeling was uniquely absent among nearly all of the NF200 positive innervation, including that which was myelinated and unmyelinated (Figure 9). Nearly all of the other DDI plexus fibers expressed GAP43 labeling, including peptidergic C-fiber and sympathetic innervation.
Negative results
Immunolabeling for a variety of other antigens of interest (Table 1) did not produce convincingly detectable label among the DDI. This included antibodies for TRPV1, TRPA1, and P2X3 that have been shown to label some types of cutaneous and dural innervation in rodents (58), and are presumed to play important roles in chemical and mechanical nociceptive transmission.
DiI retrograde labeling of trigeminal neurons
Double labeling of neurons in the trigeminal ganglion that were retrogradely labeled with DiI applied to the dura confirmed immunolabeling characteristics detected among the DDI (Figure 10). Of particular interest, many of the dural trigeminal neurons co-expressed immunolabeling for CGRP and δOR (Figures 10a, b, and d), and these neurons in turn had little or no additional labeling for NF200 (Figures 10c and e). Throughout the ganglia, virtually all neurons that labeled for δOR also co-expressed CGRP, even those not labeled with DiI. Dura neurons that labeled intensely for NF200 had little or no labeling for CGRP or δOR (Figures 10c and e). However, there were many neurons throughout the ganglion that co-expressed CGRP and NF200 but none of these were labeled with DiI from the dura. Some dural neurons were labeled for CGRP but lacked δOR (Figure 10b). Finally, there were rare dural neurons that lacked CGRP, δOR, or NF200 (Figure 10e).
Rhesus monkey dura innervation
Our double label combinations revealed that the Rhesus monkey dura also has areas with a diffuse maze of capillaries that distinctly label with the platelet/endothelial cell adhesion molecule (PECAM) (Figure 11a), which is accompanied by a non-arterial DDI that has similar combinations of intertwined peptidergic and nonpeptidergic sensory fibers and NA sympathetic fibers like those in the rat dura (Figures 11b–d). Importantly, the capillary network and DDI are not present equally throughout the monkey dura, but are located in wide zones that appear to flank the arterial arbors. CGRP labeled C fibers comprised a major proportion of the entire DDI innervation that labeled with anti-PGP, and individual axons terminated as free nerve endings having few branches (Figure 11b). Intertwined with the peptidergic C fibers were NA sympathetic fibers (Figure 11c) and small caliber nonpeptidergic fibers that labeled for NF200. Of particular interest, most of the CGRP fibers also co-labeled for δOR (Figure 11d).
GAP43 expression among the dura innervation. (a) GAP43 immunolabeling is widely expressed among the dural innervation. (b) and (c), (b) is an enlargement of the area in the white rectangle in (a) . GAP43 is expressed in all types of dura sensory and sympathetic innervation (yellow arrowheads in (b) and green arrowheads in (c)) except the sensory innervation that expresses NF200 (green arrowheads in (b) and red arrowheads in (c)). Scale bar = 100µm in A, 50µm in (b) and (c). Immunofluorescence characteristics of neurons in the trigeminal ganglion that were retrogradely labeled with DiI applied to the dura through a small hole in the cranium. (a–e) Arrows indicate cells that were initially labeled by DiI in sections of the trigeminal ganglia that were imaged before subsequent processing for immunofluorescence, which washed out the DiI. The broken outlines in (a–c) show a location contaminated by an aberrant DiI crystal that subsequently washed out. Arrowheads indicate cells that were never labeled by DiI but were subsequently labeled by immunofluorescence. (a–c) Identical sectors of complete image montages showing the three-step processing of a trigeminal ganglion section. (a) Prior to immunolabeling, the section was imaged in its entirety at 10× magnification, and montaged to map the location of all DiI labeled cells in the section. (b) Montaged images made subsequent to immunolabeling with rabbit anti-δOR and donkey anti-rabbit Cy3 (red) and with sheep anti-CGRP and donkey anti-sheep Alexa 488 (green), revealed several neurons that were originally labeled with DiI that were now double labeled for δOR and CGRP (yellow arrows in (a–c); e.g. #1a, Table 2). Yellow arrowheads show additional neurons double labeled for δOR and CGRP that were not initially labeled with DiI. (c) Montaged images were again made after incubating the sections for mouse anti-NF200 and donkey anti-mouse Alexa 488, which produces a much more intense and uniform green labeling than the prior labeling for CGRP. In these images, many of the neurons initially labeled with DiI were only labeled for NF200 (green arrows in (a) and (c)) and were not previously labeled for δOR or CGRP as shown in (b) (e.g. #3 and #4, Table 2,). In some cases, δOR and CGRP labeled neurons also appear to co-label for NF200 (yellow arrows with green asterisks). Some DiI labeled neurons had at best weak labeling for δOR, CGRP, or NF200 (open arrows; e.g. #2, Table 2). (d) and (e) Enlarged images of clusters of neurons in the trigeminal ganglia of rats in which the dura was labeled with DiI. As shown in the left-hand images, some neurons were initially labeled with DiI (arrows), which washed out during subsequent immunolabeling. (d) After imaging for DiI, these neurons were processed to detect δOR (red) and CGRP labeling (green). One original DiI neuron lacked δOR and CGRP labeling (open arrows) and was subsequently labeled for NF200 (not shown; e.g. #3 and #4, Table 2). Another DiI neuron was double labeled for δOR and CGRP (yellow arrows; e.g. #1a, Table 2). Two other neurons were not labeled with DiI, but were subsequently double labeled for δOR and CGRP (yellow arrowheads). (e) After imaging for DiI, these neurons were subsequently processed to detect δOR (red) and CGRP labeling (green) and were again processed for NF200 (more intense green). DiI was washed out, as seen in one neuron that subsequently lacked δOR, CGRP, or NF200 (open arrows; e.g. #2, Table 2). One neuron was initially labeled with DiI and subsequently only expressed NF200 (green arrows; e.g. #3 and #4, Table 2). Yellow arrowheads indicate additional δOR and CGRP neurons that were not labeled with DiI or NF200. Green arrowheads indicate an additional neuron that only labeled for NF200. Scale bar = 100 µm in (a–c), 50 µm in (d) and (e). Immunolabeling of 20 µm thick sections cut parallel to the surface of the Rhesus monkey dura also revealed a non-arterial DDI associated with a maze of capillaries. (a) PECAM labeling revealed a maze of capillaries (red arrowheads) branching from arterioles. (b) Double labeling for CGRP (red) and PGP (green) detected fibers of the DDI with and without CGRP (yellow and green arrowheads). (c) CGRP fibers in the DDI (green arrowheads) were individually closely intertwined with NPY-labeled sympathetic fibers (red arrowheads). (d) The CGRP innervation (green arrowheads) was also closely intertwined with thin caliber NF200 labeled fibers (green arrowheads). (e) CGRP fibers among the Rhesus monkey DDI colabeled for δOR. Scale bar = 50 µm in (a), (b) and (e), 25 µm in (c) and (d).

Discussion
The present study explored the diffuse dural innervation (DDI) of the rat dura mater, and to a lesser extent, that of the Rhesus monkey, in an effort to uncover potential points of physiological and pathological neurovascular regulation. We found that the DDI is far more vast than the arterial innervation, contains a majority of peptidergic fibers, and is distributed amongst a pervasive capillary network throughout the dura. This capillary network had been observed, but relatively underappreciated as a major component of dura vascularization (3,5,76). Thus, the vast DDI/capillary relationships would be a more likely source of the elevated serum levels of CGRP observed during and after migraine attacks than the lesser innervation of meningeal arterioles, which also have thicker smooth muscle walls. At least in the rat, where most of the dura could be observed in a single whole mount, the vast majority of the DDI was not merely derived from branches of the arterial innervation originating from the meningeal nerve, but was instead supplied by numerous small regional nerves converging into the dura from around the entire perimeter. Although these small nerves anastomosed extensively to form a seamless plexus, the separate nerve entries from around the perimeter suggest a regionotopic organization to the DDI that may have some functional autonomy from the arterial innervation. This may explain why there is a clear increase in dural CGRP release during migraine without an expected arterial dilatation (77).
Our results confirmed previous observations that the DDI is mostly composed of three major contingents of thin-caliber innervation (55). The largest contingent is NA sympathetic, originating from the superior cervical ganglia (2,4,74). The second major contingent consists of CGRP-expressing peptidergic innervation confirmed as C fibers by an absence of labeling for MBP and NF200 (2,4,74). The presence of CGRP-positive and NF200-negative sensory neurons in the trigeminal nerve was also confirmed by retrograde labeling from the dura. The third major, but lesser contingent consists of thin-caliber nonpeptidergic sensory fibers that express NF200 and lack CGRP, characteristics that were also confirmed by retrograde labeling among neurons in the trigeminal ganglion.
In contrast to a prior report that the thin-caliber NF200 dura fibers were myelinated like those of presumptive Aδ cutaneous innervation, our double label combinations revealed that the vast majority in the DDI were unmyelinated, referred to here as C/Aδ. Both the peptidergic C fibers and nonpeptidergic C/Aδ fibers terminated as branched free nerve endings. Since the distance from the trigeminal ganglion to the dura is relatively short compared to that of most sensory nerves, the DDI NF200 fibers without myelin may be sufficient to fulfill the functional requirements of equivalent lightly myelinated cutaneous Aδ fibers. We did find a small DDI contingent of slightly larger-caliber NF200 fibers that were myelinated and terminated as unbranched bulbous endings, which would be more characteristic of much longer and larger-caliber cutaneous Aβ fibers (7). Very rare NF200 C/Aδ fibers co-expressed CGRP, like some cutaneous peptidergic Aδ fibers. We found no evidence of parasympathetic innervation among the DDI.
The peptidergic C fibers in the DDI of the rat were unusual in that virtually all had an affinity for the lectin IB4, whereas, in contrast, CGRP and IB4 labeling are mostly expressed on separate populations of cutaneous C fibers (50,57,67,68,78). Likewise, IB4-labeling and CGRP are mostly expressed on separate neurons in the dorsal root ganglia and trigeminal ganglia. However, the trigeminal ganglia had a much higher proportion that co-express both characteristics, which likely include the DDI peptidergic neurons (65,68,79). Additional subsets of peptidergic fibers included a very minor, sparse contingent of IB4 labeled but CGRP- and NF200-negative C fibers, consistent with the rare detection of similar retrogradely labeled trigeminal neurons. As shown previously in mice, Mrgprd receptors are expressed exclusively on nonpeptidergic IB4 neurons in DRGs, particularly those that supply nonpeptidergic C fibers that innervate the skin epidermis (61,80). Mrgprd fibers were absent in the mouse dura, consistent with our observed paucity of nonpeptidergic IB4 fibers in the rat DDI.
The stimuli that activate and determine the function of the various types of DDI sensory innervation under normal conditions are unknown. The unbranched bulbous endings of the few myelinated fibers would likely be low threshold mechanoreceptors. Free nerve endings of C and C/Aδ fibers have typically been implicated as various types of high threshold nociceptors, and/or low threshold thermoreceptors or chemoreceptors, with some being implicated as polymodal nociceptors (66). Previous electrophysiological assessments of dural afferents detected mechanosensitive properties and CGRP release (6). Given their location within the cranium, it seems unlikely that they would function as thermoreceptors. Using antibodies previously validated in rodent skin (59–61), we did not detect labeling in the DDI for P2X3, which was consistent with the reported expression of P2X3 on only a small proportion of IB4 labeled neurons in the trigeminal ganglion (81,82). Undoubtedly, some if not most of the DDI would likely contribute to normal metabotropic monitoring and blood flow regulation, although we did not detect the expected labeling for ASIC3, nor TRPV1, or TRPA1 (15,60,83). TRPA1 has been implicated in migraine as a transducer of environmental irritants, including components of cigarette smoke, acrolein, and formaldehyde. Additionally, umbellulone is known to provoke migraine, likely through a TRPA1 mechanism. The precise distribution of these channels remains to be determined (83–85).
We detected several preferentially expressed axonal signaling mechanisms that may exert a peripheral excitatory or inhibitory modulation of the relative sensitivity of the peptidergic C fibers and nonpeptidergic C/Aδ fibers. First, virtually every DDI peptidergic fiber was individually closely intertwined with a sympathetic fiber completely to their termination, albeit sympathetic fibers were more numerous and could distribute and terminate without a sensory fiber. Moreover, nearly all peptidergic C fibers co-expressed adrenergic α1 receptors, which are known to mediate excitatory effects via Gq coupling (86). The peptidergic C fibers predominately expressed the α1B receptor, and some the α1A receptor, suggesting that sympathetic activity could contribute to excitatory modulation of these fibers and possibly to enhanced CGRP release from the DDI among the capillaries (87,88). Moreover, the α1B receptor was also expressed on the sympathetic fibers, potentially mediating autocrine excitatory modulation. Only a few peptidergic fibers, and no sympathetic fibers, expressed the α2A, and none the α2C, receptors that are implicated in NA mediated inhibition (86,89). Interestingly, both the sympathetic and peptidergic fibers express GAP43, indicating that the density of their terminals may be dynamically maintained and subject to physical modification under stressful or immune challenge conditions (90–92).
Furthermore, the CMA results confirmed prior work demonstrating expression of the co-receptor for CGRP, RAMP1, on NF200-labeled dura innervation (55). Here, we further determined that RAMP1 was limited to the C/Aδ fibers that are closely intertwined with peptidergic C fibers. Therefore, potential sympathetic excitation of the peptidergic fibers may, in turn, result in an excitatory modulation of the C/Aδ fibers that could underlie the observed mechanical allodynia during migraine (6). Most of the peptidergic DDI fibers expressed the 5HT1D receptor, a known target for the triptan drugs supporting the possibility of CGRP inhibition as a part of the mechanisms mediating efficacy for these compounds (27,63) as well as a potential site of action of promising anti-CGRP peptide or receptor antibodies that do not penetrate the blood brain barrier (29–31). Additionally, consistent with indications that the delta opioid receptor (δOR) agonists may effectively treat migraine (93), most of the peptidergic DDI fibers expressed immunolabeling for δOR with three different polyclonal and monoclonal antibodies targeting different domains of δOR. Although there is some indication that existing δOR antibodies may be labeling an unknown molecule on cutaneous peptidergic C fiber neurons in mice (64), δOR expression has been confirmed on some cutaneous nonpeptidergic IB4 C-fibers (62), like the δOR and IB4 positive C fibers that are peptidergic in the dura. Unlike the common expression of µOR among cutaneous C fibers (62,64,94), µOR expression was rare among rat DDI, indicating that δOR peptidergic IB4 fibers may be unique to the dura. The paucity of µOR fibers in the DDI may underlie the relative lack of efficacy of µOR agonists in either the acute or prophylactic treatment of migraine headache, and suggests that any effect of µOR agonists in the treatment of migraine may reflect predominant actions within the brain, consistent with the preferential actions of these drugs on pain unpleasantness, rather than modulation of nociceptive transmission (95–97). It should be noted, however, that previous studies have demonstrated that systemic morphine or the mu opioid receptor selective peptide agonist DAMGO inhibited electrically-evoked CGRP release in the dura mater, while the δOR agonist DPDPE had no effect, suggesting other possible sites of µOR expression (98–100). Nevertheless, in view of indications that the δOR may be particularly relevant to treatment of chronic pain conditions, the prominent expression of the δOR in the DDI peptidergic innervation might allow for selective regulation of CGRP release by δOR agonists, including those that are peripherally restricted (101–103). The possible effectiveness of δOR agonists for acute migraine therapy has been previously suggested (93,104).
Summary
Our comprehensive multi-molecular CMA platform revealed several previously unknown characteristics of the diffuse dural innervation, apart from the innervation to the arterial network, which could underlie the mechanism of action of established and emerging novel therapeutics and provide potential targets for novel therapeutics. We note that while the present study emphasized characterization of the DDI, the arterial innervation is undoubtedly of physiological and pathological significance, and may represent sites of expression of molecular targets known to be relevant to cephalic pain including, for example, TRPV1, TRPA1 and ASIC channels. The relative contributions of the DDI and arterial innervation, as well as the cell bodies from which these fibers arise, will require additional study. Within the DDI, the expression of several neurosignaling molecules implies potential modulatory interactions between three major types of sensory and sympathetic innervation that are distributed throughout the dura and overlying a dense capillary network that would be a logical site of CGRP release into circulation, as is known to occur during migraine attacks. Activation of NA sympathetic fibers may activate peptidergic C fibers that express α1 receptors. By releasing CGRP, the peptidergic C fibers may in turn activate unique, previously undefined NF-positive C fibers that express receptors for CGRP. Activation of either, or all, of these C fiber types could be potential peripheral and post-synaptic contributors to migraine pain. Therapeutics inhibiting either the α1 or CGRP receptors or both could be a means of reducing this activation. Furthermore, the preferential expression of δOR on the DDI could be a potential target for a peripherally restricted δOR agonist that would inhibit the peptidergic innervation and CGRP release, providing an approach for acute migraine therapy.
Article highlights
The non-arterial diffuse dural innervation (DDI) is distributed across a dense capillary network. The DDI includes peptidergic CGRP-expressing C fibers that are intertwined with noradrenergic (NA) sympathetic fibers and thin-caliber nonpeptidergic “C/Aδ” fibers that express the CGRP co-receptor, RAMP1. DDI peptidergic fibers co-label for α1-adrenergic receptors, 5HT1D receptors and the delta opioid receptor (δOR). Expression of the mu opioid receptor (µOR) was rarely observed on DDI peptidergic fibers, and labeling for P2X3, TRPV1, TRPA1, was not observed. The anatomical relationships suggest potential functional interactions that could lead to stress-induced CGRP release and activation of pain transmission circuits. The distribution of α1-adrenergic receptors, RAMP1, and the unique expression of the δOR on CGRP-expressing DDI fibers, suggest strategies for anti-migraine therapeutic intervention.
Footnotes
Acknowledgements
We wish to thank Marilyn Dockum and Samantha Mall for their invaluable technical services. We also wish to thank Deborah Fuller of University of Washington Primate Center for providing Rhesus monkey dura mater.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the National Institutes of Health grant R01 NS 069575 and by Intidyn.
