Abstract
Background
Stress is commonly reported to contribute to migraine although mechanisms by which this may occur are not fully known. The purpose of these studies was to examine whether norepinephrine (NE), the primary sympathetic efferent transmitter, acts on processes in the meninges that may contribute to the pain of migraine.
Methods
NE was applied to rat dura using a behavioral model of headache. Primary cultures of rat trigeminal ganglia retrogradely labeled from the dura mater and of rat dural fibroblasts were prepared. Patch-clamp electrophysiology, Western blot, and ELISA were performed to examine the effects of NE. Conditioned media from NE-treated fibroblast cultures was applied to the dura using the behavioral headache model.
Results
Dural injection both of NE and media from NE-stimulated fibroblasts caused cutaneous facial and hindpaw allodynia in awake rats. NE application to cultured dural afferents increased action potential firing in response to current injections. Application of NE to dural fibroblasts increased phosphorylation of ERK and caused the release of interleukin-6 (IL-6).
Conclusions
These data demonstrate that NE can contribute to pro-nociceptive signaling from the meninges via actions on dural afferents and dural fibroblasts. Together, these actions of NE may contribute to the headache phase of migraine.
Background
Migraine is the third most common disease worldwide and is one of the top 10 most disabling disorders (1). For a substantial number of migraine patients, currently available treatments either lack efficacy or lead to intolerable side effects, which contributes to the disabling nature of this condition. Development of new therapeutics has been slow, due in part to a poor understanding of the underlying pathology contributing to migraine.
One of the most consistently reported factors contributing to migraine is stress (2). For example, 59% of 200 patients in one study (3) and 80% of 1750 patients in another (4) reported stress as a trigger for common migraine. In both of these studies, stress was the most commonly reported trigger. In fact, analysis of data from more than 7000 patients concluded that stress is the number one reported trigger for migraine (5). The timing of attacks in relation to stress is variable as 59% of patients reported migraines during stress while 70% had migraines triggered after resolution of stress (6). When the types of stress among patients were differentiated, too much work (84%), marital problems (36%), and economic problems (34%) were found to be among the leading stressors triggering migraines (7). Stress has also been reported as the most common trigger in patients with familial-hemiplegic migraine (FHM) with 32% reporting migraines during and 37% after stress (8) (all other trigger factors in FHM patients were lower incidence). Cumulative stress appears to exert a greater influence as two consecutive days of moderate or greater stress in a row increases risk of migraine while low stress on the prior day with moderate or greater stress on the subsequent day is associated with less risk (9). An interesting observation from this study is that when stress is moderate or greater on the preceding day, but low on the subsequent day, migraine risk is still elevated. This is consistent with a recent study finding the greatest risk of migraine in the six to 18 hours following resolution from stress (10). These latter observations suggest a complex relationship between stress and migraine. Nonetheless, the studies described above demonstrate a clear link, at least as reported by patients, between stress and migraine.
How and where stress contributes to migraine is not well understood as it can affect diverse physiological processes throughout the body via increases in circulating stress hormones (e.g. cortisol, epinephrine) and activation of sympathetic efferents. While many hypotheses have been proposed as potential pathophysiological mechanisms of migraine pain, afferent input from the meninges is most likely necessary for the initiation of headache (11). Circulating stress hormones may act within the meninges to alter nociceptive signaling. Additionally, the meninges are innervated directly by sympathetic efferents from the superior cervical ganglion (12–14) and sympathetic outflow can directly act on cell types in the meninges following release of sympathetic transmitters such as norepinephrine (NE). Further, NE has been shown previously to have pro-nociceptive actions on peripheral sensory neurons (15). Thus, under conditions of stress, the sympathetic nervous system may contribute to processes within the meninges leading to headache.
The purpose of these studies was to examine the effects of NE on headache-related processes in the meninges using preclinical rat models. Experiments were designed to assess the effects of NE application on excitability of dural afferents in vitro as well as to determine whether direct application of NE to the dura mater produces headache-related behavior in rats. In addition, we recently published that dural fibroblasts may participate in headache as they can release pro-inflammatory cytokines that activate/sensitize dural afferents (16). Experiments described here also examined whether dural fibroblasts respond to NE in a manner that may promote headache. Findings from these studies indicate that NE has direct actions both on dural afferents and dural fibroblasts, either of which may contribute to headache following acute or chronic stress.
Methods
Animals
Male Sprague-Dawley rats (35–100 g for dural fibroblast cell culture, 150–175 g for patch clamp studies, 250–300 g for behavioral studies) were maintained in a climate-controlled room on a 12-hour light/dark cycle with food and water ad libitum. All procedures were performed in accordance with the policies and recommendations of the International Association for the study of Pain, and the National Institutes of Health guidelines for handling and use of laboratory animals, and were approved by the Institutional Animal Care and Use Committee of the University of Arizona.
Surgery
Retrograde tracer injection
Dural afferents were identified as previously described (17). Briefly, animals were anesthetized, two 3 mm holes were made in the skull, and 5 μl of Fluorochrome (4% in synthetic-interstitial fluid (SIF), Fluorochrome LLC) was applied onto the dura. The holes were covered with bone wax. Undamaged dura at the injection sites was evaluated at the time the animals were sacrificed and only data from animals with intact dura and no signs of damage were used for further analysis.
Dura cannulation
Dura cannulae were implanted as previously described (17). Briefly, animals were anesthetized, the skull was exposed, and a 1 mm hole (2 mm left of sagittal suture and 2 mm anterior to lambdoid suture) was made to carefully expose the dura. A guide cannula (Plastics One) was inserted into the hole and sealed into place with glue. Two additional holes were made to receive stainless-steel screws (Small Parts), and dental acrylic was used to fix the cannula to the screws. A dummy cannula (Plastics One) was inserted to ensure patency of the guide cannula. Animals were housed separately after surgery and allowed six to eight days of recovery.
Cell culture
Trigeminal ganglion (TG) culture
Seven days following Fluoro-Gold application, TG were removed, enzymatically treated, and mechanically dissociated as previously described (18,19). Rats were anesthetized, sacrificed, and the TG were removed. Ganglia were incubated in papain (Worthington) followed by collagenase type II (Worthington). Ganglia were then triturated and plated. Cells were cultured in a room-temperature, humidified chamber in L-15 medium with 10% fetal bovine serum (FBS), 10 mM glucose, 10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) and 50 U/ml penicillin/streptomycin. Cells were used within 24 hours of post-plating.
Dural fibroblast culture
Rats were anesthetized, sacrificed, and the dura mater removed. Dura mater were incubated in collagenase A (Roche) and collagenase D (Roche) with papain (Roche) for 40 to 50 minutes. To eliminate debris, 70 µm cell strainers (BD Biosciences) were used. The dissociated cells were resuspended in DMEM/F12 (Invitrogen) containing pen-strep (Invitrogen), GlutaMax and 10% FBS (Hyclone). The cells were plated and incubated at 37℃ in a humidified CO2 incubator. Dural fibroblasts were plated at a density that would achieve confluency by day 3 post-plating.
Fibroblast-conditioned media collection
Confluent cultures of dural fibroblasts were treated with 10 µM NE (Sigma) or vehicle for one hour. After treatment, cells were washed three times with media (DMEM/F12 with 1X GlutaMax) to remove the NE. Next, 500 µl of media was added to the dishes to collect factors released from fibroblasts. The media was kept in the culture dishes for 12 hours and then collected. Samples were dialyzed to remove salts, lyophilized, and kept in −20℃ for behavior or enzyme-linked immunosorbent assay (ELISA).
Western blotting
Confluent dural fibroblast cultures were washed in DMEM/F12 for 30 minutes before treatment. NE (10 µM) was applied to the culture for 15 minutes and then protein was extracted from cells in lysis buffer (50 mM Tris HCl, 1% Triton X-100, 150 mM NaCl, and 1 mM ethylenediaminetetraacetic acid (EDTA) at pH 7.4) containing protease and phosphatase inhibitor mixtures (Sigma) with an ultrasonicator on ice, and cleared of cellular debris and nuclei by centrifugation at 14,000 relative centrifugal force (RCF) for 15 minutes at 4℃. Fifteen micrograms of protein per well were loaded and separated by standard 7.5% or 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Proteins were transferred to Immobilon-P membranes. Blots were incubated with primary antibody overnight at 4℃ and detected the following day with appropriate secondary antibodies. Signal was detected by enhanced chemiluminescent (ECL) substrate on chemiluminescent films. Densitometric analyses were performed with Image J software (NIH).
Electrophysiology
Whole cell patch-clamp experiments were performed on isolated rat TG using a MultiClamp 700B (Axon Instruments) patch-clamp amplifier and pClamp 10 acquisition software (Axon Instruments). Recordings were sampled at 5 kHz and filtered at 1 kHz (Digidata 1322A, Axon Instruments). Pipettes were pulled using a P-97 puller (Sutter Instruments) and heat polished using a microforge (MF-83, Narishige). Series resistance was typically <7 MΩ and was compensated 60%–80%. All recordings were performed at room temperature. A Nikon TE2000-S microscope equipped with a mercury arc lamp (X-Cite® 120) was used to identify Fluoro-Gold-labeled dural afferents. Data were analyzed using Clampfit 10 (Molecular Devices) and Origin 8 (OriginLab). Cell sizes were not significantly different among groups (NE: 39.52 ± 2.03 pF vs vehicle: 40.19 ± 1.64 pF p > 0.05). The pipette solution contained (in mM) 140 KCl, 11 ethylene glycol tetraacetic acid (EGTA), 2 MgCl2, 10 NaCl, 10 HEPES, 2 MgATP, and 0.3 Na2GTP, 1 CaCl2 pH 7.3 (adjusted with N-methyl glucamine), and was ∼315 mosM. External solution contained (in mM) 135 NaCl, 2 CaCl2, 1 MgCl2, 5 KCl, 10 glucose, 10 HEPES, pH 7.4 (adjusted with N-methyl glucamine), and was ∼300 mosM. Freshly thawed supernatant (600 µl) was added to the TG culture for 15 minutes and then replaced with bath solution before recording as performed previously (16). Fluoro-Gold-positive neurons were identified and patched immediately after replacement of supernatant with bath.
Behavioral testing
Behavioral testing methods were identical to those used previously (16). Rats were acclimated to suspended Plexiglas chambers with a wire mesh bottom. Ten microliters of vehicle or testing solution was injected through an injection cannula (Plastics One) cut to fit the guide cannula. Doses of NE were chosen based on prior studies injecting NE in vivo in rats and humans (20,21). Withdrawal thresholds to probing of the face and hindpaws were determined at one-hour intervals after administration. A behavioral response to calibrated von Frey filaments applied to the midline of the forehead, at the level of the eyes, was indicated by a sharp withdrawal of the head. Paw withdrawal (PW) thresholds were determined by applying von Frey filaments to the plantar aspect of the hindpaws, and a response was indicated by a withdrawal of the paw. The withdrawal thresholds were determined by the Dixon up-down method. Maximum filament strengths were 8 and 15 gm for the face and hindpaws, respectively. Behavioral testing was performed by an investigator blinded to the treatment groups.
ELISA
Confluent dural fibroblasts were stimulated with NE for one hour and conditioned media collected as described above with the following exceptions. First, media containing NE was discarded, cells were then washed three times with media, 600 µl media was again added to the dishes for 12 hours and collected. Conditioned media collected from dural fibroblasts cultures was centrifuged at 4℃ for 15 minutes at 16,000 × g to remove any debris or dead cells and transferred to a fresh tube and used as samples. A rat IL-6 DuoSet ELISA Development SYSTEM (R & D Systems) was used for detecting the interleukin-6 (IL-6) levels in the conditioned media. The supernatant was de-salted to concentrate factors released from fibroblasts. Supernatant was collected, immediately put on ice, and centrifuged at 4℃ for 15 minutes. Supernatants (400 µl of the initial 600 µl volume) were transferred to a desalting cassette (Slide-A-Lyzer Dialysis Cassette, Thermo Scientific) following the instructions provided by the manufacturer. The supernatant in the cassette was then removed, lyophilized, and resuspended in 100 µl DMEM/F12 media. ELISA assay procedures were performed according to manufacturer instructions.
Data analysis
All data are presented as means ± SEM unless otherwise noted. Electrophysiology data were analyzed using linear regression for a significant difference in slopes of the lines connecting the numbers of action potentials. Behavioral studies among groups and across time were analyzed by two-factor analysis of variance (ANOVA) for the treatment and time. Data were converted to area over the time-effect curve and analyzed with a one-factor ANOVA. ELISA data were analyzed by a two-factor ANOVA for treatment and time. Statistics were run using GraphPad Prism (GraphPad Software Inc, La Jolla, CA, USA).
Results
Dural application of NE produces cutaneous allodynia
A preclinical in vivo migraine model was used to evaluate whether dural NE causes mechanical allodynia of the face and hindpaws. Application of NE (30 µM; 50 ng in 10 µl) to the dura produced a significant time-dependent and reversible decrease in withdrawal thresholds to tactile stimuli applied to the face and the hindpaws (Figure 1(b)–(d)) compared with vehicle (SIF). Lower doses of NE (10 µM; 17 ng in 10 µl) also produced significant reduction in withdrawal thresholds to tactile stimulus applied to the hindpaw but not to the face (Figure 1(c) and (d)). NE (1 µM; 1.7 ng in 10 µl) did not produce reductions in withdraw thresholds in either the hindpaw or the face in response to tactile stimulation (Figure 1(c) and (d)). These data indicate that direct application of NE to the dura mater produces behavioral responses consistent with headache.
Application of norepinephrine (NE) to the dura dose dependently elicits cutaneous allodynia. Withdrawal thresholds to tactile stimuli applied to the face were measured in rats before and for five hours after dural application of NE in (a) (1 µM, n = 7) and (b) (30 µM, n = 16) or pH 7.4 synthetic-interstitial fluid (SIF) (n = 16). For facial responses, two-factor analysis of variance (ANOVA) indicated a significant effect of both treatment and time. Treatment F(1,150) = 8.06, Time F(5,150) = 5.42. Data were converted to area over the time-effect curve (AOC). NE dose-dependently decreased the withdrawal threshold both in the face (c) and the hindpaws (d). The raw data time course for hindpaw allodynia is not shown. Significant differences among means (**p < 0.01, ***p < 0.005) for each group were determined by analysis of variance followed by Dunnett’s post hoc test.
NE sensitizes dural afferents in vitro
To explore whether cutaneous allodynia induced by NE is due, at least in part, to actions on dural afferents, we examined the effects of NE using patch-clamp electrophysiology. Slow ramp currents from 0.1 to 0.7 nA with a Δ = 0.2 nA were injected to retrogradely labeled trigeminal neurons (i.e. identified dural afferents) over one second to mimic slow depolarization. Dural afferents treated with NE for 15 minutes before recording showed a significant increase (p < 0.005) in the number of spikes compared to dural afferents treated for the same amount of time with vehicle (Figure 2). A direct sensitizing action of NE on dural afferents indicates that NE release in the meninges can be pro-nociceptive and may contribute to the behavioral responses described above via actions on sensory afferents.
Norepinephrine (NE) treatment induces hyperexcitability of dural afferents. Dural afferents treated with NE (10 µM) for 15 minutes show increased numbers of action potentials (a) compared with vehicle-treated dural afferents (b). Action potentials in (a) and (b) were elicited by one-second ramp current injections ranging from 0.1 to 0.7 nA in 0.2 nA increments as shown in (c). Differences in the mean numbers of action potentials among groups were analyzed by comparing the slopes of the lines for each group using linear regression (F(1,64) = 9.32, p < 0.005). Dural afferents treated with NE (red squares, n = 9) showed a significant increase in the number of action potentials compared to vehicle-treated groups (black circles, n = 8) (d).
NE activates intracellular signaling pathways in cultured dural fibroblasts
We recently showed that the predominant cell type in the dura mater, fibroblasts, can participate in the headache process by releasing pro-inflammatory cytokines in response to stimulation (16). Thus, we investigated whether dural fibroblasts respond to noradrenergic stimulation. NE (10 µM) was applied to cultured dural fibroblasts for 15 minutes, and phosphorylation of extracellular signal-related kinase (ERK) levels were examined using Western blotting. As shown in Figure 3, cultures treated with NE showed a significant (p < 0.001) increase in the ratio of ERK phosphorylation (pERK) to total ERK (pERK/tERK) compared to vehicle. This is consistent with prior reports in cardiac fibroblasts where noradrenergic stimulation leads to increased mitogen-activated protein kinases (MAPK) signaling pathways (22,23). These data indicate that noradrenergic receptor activation on dural fibroblasts initiates intracellular signaling cascades.
Norepinephrine (NE) induces extracellular signal-regulated kinases (ERK) phosphorylation (pERK) in cultured dural fibroblasts. Dural fibroblast cultures were treated with 10 µM NE for 15 minutes. A representative blot is shown in (a). NE induced a significant increase in the pERK/total ERK (tERK) ratio (p < 0.001, n = 6) compared to vehicle treated cells (b).
Dural fibroblasts release IL-6 in response to NE stimulation
We have shown previously that IL-6 sensitizes dural afferents, produces cutaneous allodynia when applied to the dura (22), and that dural fibroblasts release IL-6 in response to lipopolysaccharide (LPS) (16). Others have shown that NE increases IL-6 mRNA via MAPK pathway activation in cardiac fibroblasts (23). Thus, we next asked whether exposure of dural fibroblasts to NE causes the release of IL-6. Application of NE to dural fibroblasts produced a time-dependent increase in the presence of IL-6 in the conditioned media as measured by ELISA (Figure 4). After one hour of treatment, there was little to no IL-6 release both in vehicle-treated and NE-treated dural fibroblasts cultures. However, at 12 hours (one hour stimulation with NE, 12 hours of collection in fresh media) there was significant IL-6 released from fibroblasts in the NE-treated group but not in the vehicle-treated group. Thus, IL-6 is released from dural fibroblasts in response to NE stimulation and may subsequently activate/sensitize meningeal afferents.
Dural fibroblasts release interleukin-6 (IL-6) in response to norepinephrine (NE) stimulation. Dural fibroblast cultures were treated with NE (10 µM) or vehicle for one hour, washed, fresh media was applied and conditioned media was collected after 12 hours. Supernatants were desalted and lyophilized into powder before being resuspended and analyzed. IL-6 content from vehicle-treated and NE-treated media was measured by enzyme-linked immunosorbent assay (ELISA). Mean IL-6 levels were compared by a one-way analysis of variance (ANOVA) (F(3,19) = 7.771, *p < 0.0001, n = 6).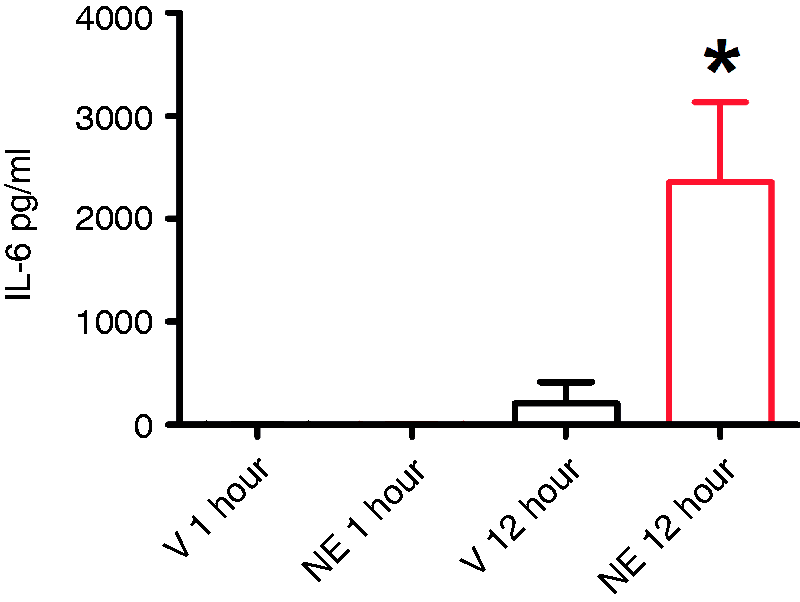
NE-conditioned fibroblast culture media induces cutaneous allodynia
In order to determine whether dural fibroblasts release mediators that can produce headache-like responses in vivo, culture media taken from fibroblasts stimulated with NE or vehicle was applied to the dura. Dural fibroblast cultures were stimulated with either NE (10 µM) or vehicle, the stimulus was washed off, and fresh media was applied and subsequently collected. Application of media collected from NE-treated dural fibroblasts to the dura produced significant time-dependent reductions in withdrawal thresholds to tactile stimuli applied to the face or the hindpaws (Figure 5). Media from vehicle-treated dural fibroblasts did not produce allodynia. Thus, stimulation of dural fibroblasts with NE can promote the release of mediators that produce allodynia when applied to the dura of awake rats.
Dural application of conditioned media from norepinephrine (NE)-treated fibroblasts elicits cutaneous allodynia. Withdrawal thresholds to tactile stimuli applied to the face (a) and the hindpaws (b) were measured in rats before and immediately after dural application of NE-treated conditioned media (red circles, n = 9) or vehicle-treated condition media (black squares, n = 8). For both facial and hindpaw responses, two-factor analysis of variance (ANOVA) indicated a significant effect both of treatment and time both of the face and hindpaw. Facial: treatment F(1,60) = 5.48, p < 0.05), time F(5,60) = 4.28, p < 0.005: Hindpaw: treatment(1,60) = 4.89, p < 0.05, time F(5,60)–5.32, p < 0.001. Withdrawal thresholds to tactile stimuli were measured for five hours and data were converted to area over the time-effect curve (AOC) in (c) and (d). Significant (*p < 0.05) differences among means for each group were determined by one-way ANOVA.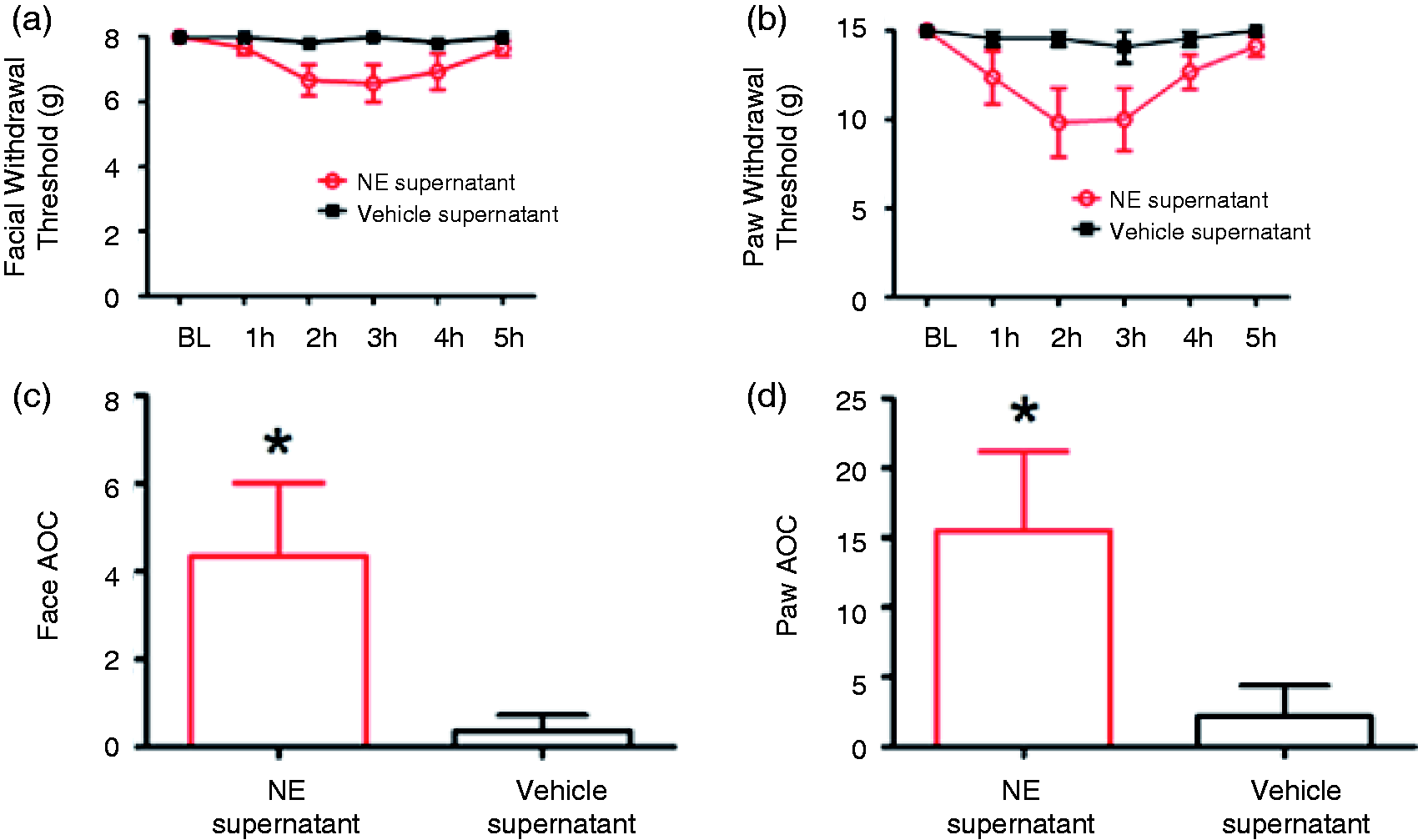
Discussion
Although stress is commonly reported to contribute to migraine (2,25–27), the mechanisms by which this may occur are not clear. We report here that the primary neurotransmitter released by sympathetic efferents, NE, induces headache-like behavior when applied to the meninges in a preclinical rat model of migraine. These studies show that the pro-nociceptive effects of NE can be mediated by actions on dural afferents as the excitability of identified dural afferents was increased in the presence of NE. Additionally, NE may contribute to headache by activating adrenergic receptors on dural fibroblasts and promoting the release of pro-nociceptive substances such as IL-6. Thus, one mechanism by which stress may contribute to migraine pathophysiology is via sympathetic outflow into the meninges and subsequent actions of NE on dural afferents and dural fibroblasts.
The actions of NE on dural afferents and fibroblasts may serve distinct functions with different time courses in relation to sympathetic outflow. The actions on afferents may lead directly to modulation of nociceptive signaling. Sympathetic outflow clearly occurs during stressful events (28), which would stimulate the release of NE into the meninges. In response to stress, NE outflow from sympathetic efferents could thus contribute to headache by promoting afferent signaling (29). Although most studies find little effect of NE administration in behavioral pain models or on sensory neurons in vitro (15) (at least in normal animals, although see below), NE increases the excitability of colonic afferents and produces visceral hypersensitivity in a behavioral model (30). Additionally, very few studies have examined the effects of NE in the dura mater or in headache models. One of the only studies found a dramatic increase in prostaglandin E2 (PGE2) release from isolated rat dura (31), an effect that does not occur in rat skin (32). These studies indicate that NE may produce differential responses depending on the tissue and possibly the distribution of receptors.
In humans, sympathetic outflow alone does not cause pain in normal individuals (33), although NE injection into skin decreases heat pain thresholds (34). Further, acute stress is well known to be analgesic (35). Whether or not pain occurs in response to stress may depend on the setting as most preclinical studies indicate that sympathetic outflow or norepinephrine stimulation becomes pro-nociceptive after injury or inflammation (18,19,36–39). Human studies also show a pain-enhancing effect of stress when comorbid conditions such as depression or other pain states are present (40). Migraine patients do not have any obvious injury (although inflammation may be present (41)), but perhaps more relevant is the proposal that chronic stress is a contributing factor to migraine (42). Extensive experimental preclinical studies of chronic stress have shown decreased pain thresholds or increased responses to painful stimuli in many models (40). Further, chronic stress in rats can change β-adrenergic receptor signaling rendering stimulation with epinephrine pro-nociceptive (43), and chronic stress induces visceral hypersensitivity that is attenuated in the presence of beta-blockers (30). In migraine patients, chronic stress may cause maladaptive changes in the nervous system, enhancing the likelihood that stressful events will be pro-nociceptive.
A further complication to the link between sympathetic outflow in migraine patients and direct modulation of peripheral nociceptors is the observation that headaches do not typically present during stressful events in migraineurs, but following the resolution of stress (44,45). A recent study of 22 migraine patients covering 110 migraine attacks found that an increase in the likelihood of migraine attacks is not higher during stressful events but in the six to 18 hours following the removal of stress (10). One mechanistic explanation is that sympathetic outflow during the attack may not be pro-nociceptive in migraine patients, possibly due to inhibitory actions of NE at α-2 autoreceptors on sympathetic efferents, effectively decreasing its own release. A recent study in rodents found that stressors produced hyperalgesia only when α-2 receptors were blocked or were absent in knockout mice (46). For stress-induced migraines, there may be delayed changes in the activity of the afferent fibers, particularly as NE levels drop and feedback inhibition is lost, or there may be changes in expression of autoreceptors over the course of attacks. The exact temporal relationship between stress and the development of migraine attacks, as well as their underlying mechanisms, await future studies.
In addition to actions of NE on dural afferents, we also report that NE increases intracellular signaling in dural fibroblasts and promotes the release of pro-nociceptive substances including IL-6 from these cells. Stress and the resulting sympathetic outflow into the dura may also participate in the headache process via actions on dural fibroblasts and release of IL-6, a cytokine we have previously shown causes headache behaviors in rats (24). Although our prior study used a higher concentration of IL-6 (0.1 ng) than estimated to be present in the conditioned media (0.02 ng, given 2 ng/ml from the ELISA and a 10 μl dural injection volume), there are likely factors additional to IL-6 that may contribute to the behavioral results at lower IL-6 levels. Nonetheless, our findings with IL-6 are consistent with those observed in the cardiovascular system where sympathetic stimulation of cardiac fibroblasts can lead to increased IL-6 production via activation of noradrenergic receptors on these cells (47,48). Studies of cardiac fibroblasts also found that in addition to IL-6, these cells can release IL-1, tumor necrosis factor-α, nitric oxide, and transforming growth factor-β (49) consistent with IL-6 being among several factors that can activate/sensitize dural afferents. Of note, we observed significant IL-6 release from cultured fibroblasts at 12 hours (no significant release was observed at one hour), substantially longer than time points where effects of NE were observed on dural afferents (15 minutes) and in the behavioral headache model (beginning at one hour). A similar delayed action of NE has been observed in cardiac fibroblasts where increased IL-6 synthesis was observed only after eight or 12 hours of stimulation (47). Although the delayed release of IL-6 may be a common temporal pattern by which fibroblasts respond to NE, these cells may also release IL-6 more quickly in vivo. Based on our current data, it is not yet possible to differentiate the role NE acting on dural afferents vs NE acting on fibroblasts (via IL-6 release) plays in the behavioral responses observed, nor can we determine the time course of each of these effects. Identification of stimuli that act specifically on fibroblasts without any direct action on afferents will help resolve this issue.
Additionally, sympathetic outflow in the meninges and activation of noradrenergic receptors on fibroblasts may contribute to remodeling of the extracellular protein matrix, a function mediated primarily by fibroblasts (49–51). The dura mater consists of a dense network of proteins including collagen and fibronectin (52), proteins that are continually secreted and remodeled by fibroblasts. In the cardiovascular system, cardiac fibroblasts can change their production of extracellular matrix proteins in response to noradrenergic stimulation, a process contributing to cardiac fibrosis (53–55). Repetitive stress may alter the structure of the dura and this may have important consequences for headache pathophysiology.
Finally, these studies do not identify which receptor(s) mediates the observed effects. Prior studies of NE action on sensory afferents have found effects that occur primarily through alpha receptors (56,57) and studies on dorsal root ganglion reported the presence of α1A, α1B and α2C messenger RNA (mRNA) (58). Studies of NE effects on fibroblasts from other systems have implicated both alpha and beta receptors (59,60). Ultimately, the receptors that mediate the actions of NE on neurons and fibroblasts may be different and could be differentially targeted to determine how each cell type contributes to the observed behavioral responses. Future studies are required to more fully investigate the pharmacology of noradrenergic effects in the meninges.
Together, these studies show that meningeal NE contributes to pro-nociceptive processes via actions on both dural afferents and native dural fibroblasts. As stress is commonly linked to migraine, and NE is likely to be released in the meninges by sympathetic efferents during stress, these findings propose multiple ways in which stress via NE outflow may contribute to the headache phase of migraine. Uncovering how stress and NE interact with and change the anatomy and physiology of tissues relevant to headache will not only lead to better understanding of migraine, it may also lead to new therapeutic options.
Article highlights
Norepinephrine (NE) application to the dura mater produces headache behaviors in rats. NE sensitizes trigeminal afferents innervating the dura mater. NE application to dural fibroblasts promotes the release of pro-nociceptive substances including interleukin-6 (IL-6). Factors released from dural fibroblasts treated with NE produce headache behaviors in rats. Stress may contribute to headache via NE release in the meninges and subsequent actions both on dural afferents and dural fibroblasts.
Footnotes
Funding
This work was supported by National Institutes of Health (NIH) grant NS072204 and the Migraine Research Foundation.
Conflict of interest
None declared.
