Abstract
Background
Efficacy of galcanezumab in chronic migraine has been demonstrated in a pivotal Phase 3 study. Here, we assess efficacy in patients who have failed ≥2 and ≥1 prior migraine preventives for efficacy and/or safety reasons, and in those who never failed.
Study design/methods
REGAIN (NCT02614261) was a Phase 3, randomized, double-blind, placebo-controlled study in patients with chronic migraine. Patients were randomized 2:1:1 to receive placebo, galcanezumab 120 mg/240 mg once monthly during a double-blind treatment period lasting three months. Subgroup analyses were conducted among patients who failed ≥2 and ≥1 prior preventives and who never failed previously. Outcomes assessed were change from baseline in number of monthly migraine headache days, proportion of patients with ≥50% and ≥75% response (reduction in monthly migraine headache days), change in number of monthly migraine headache days with acute medication use and change in patient functioning per Migraine-Specific Quality of Life Questionnaire Role Function Restrictive (MSQ RF-R) domain score.
Results
Treatment with galcanezumab versus placebo resulted in significant improvements (p < 0.01) in overall reduction (Months 1–3) from baseline in the number of monthly migraine headache days in patients with prior failures (LS mean change [SE]: ≥2 prior failures: galcanezumab 120 mg: −5.35 (0.71); galcanezumab 240 mg: −2.77 (0.66); placebo: −1.01 (0.54); ≥1 prior failures: galcanezumab 120 mg: −5.53 (0.60), galcanezumab 240 mg: −3.53 (0.59); placebo: −2.02 (0.49). Similarly, significant results were seen with galcanezumab versus placebo for ≥50% and ≥75% response rates, reductions in acute medication use and improvements in MSQ RF-R domain score. In the subgroup with no prior preventive failures, results were statistically significant for the 240 mg galcanezumab group versus placebo on all outcome measures, and for the 120 mg group on the reduction in migraine headache days with acute medication use. There was also a higher placebo response observed in the patients with no prior preventive failures.
Conclusion
Galcanezumab is consistently efficacious versus placebo in reducing monthly migraine headache days and several other key outcomes in patients with chronic migraine who have failed ≥2 or ≥1 preventives previously. In the subgroup with no prior failures, greater numerical differences were seen with galcanezumab, but statistical separation from placebo varied by dose and outcome.
Clinicaltrials.gov identifier number
NCT02614261.
Introduction
Patients with chronic migraine experience headache for ≥15 days per month, ≥8 of which are classified as migraine headache days (MHDs) (1). Chronic migraine is associated with substantial headache-related disability, high number of comorbid medical and psychiatric conditions, poor quality of life and worldwide societal burden (2,3). Economic costs of chronic migraine are high, comprising both direct healthcare costs and indirect costs from productivity loss (3). Across multiple countries, patients treated with current oral preventive medications for chronic migraine are at risk of not experiencing meaningful reductions in migraine headache due to poor adherence, high discontinuation rates and treatment switches (4–7). In the US, approximately 50% of patients with chronic migraine discontinue a newly prescribed preventive medication within 60 days, regardless of drug class (5), and up to 80% are no longer on therapy after 12 months (6). Among patients with chronic migraine taking oral preventive treatment, side effects and lack of efficacy are the most common reasons for discontinuation (4,5). Patients with chronic migraine on average switch preventives four times (4) and discontinuation rates are greater among patients who cycle through additional oral preventives (4,5). This can lead to patients relying solely on acute medications to manage the disease. Patients who are eligible for preventive treatment but rely on acute treatments alone are at risk of medication overuse and associated disease progression, leading to higher levels of disability (8–11). The risk of medication overuse headache and associated disability is particularly high among patients with chronic migraine (12,13). When stratified by frequency of headache days in a month, migraine-related disability levels are consistently greater among patients with a history of failure and switches to several migraine preventives when compared to patients on their first preventive treatment (14). Therefore, patients with chronic migraine who have failed prior preventive treatments for efficacy reasons, safety/tolerability reasons, or both, are an important subgroup with high unmet need and in whom it is important to understand therapeutic effects of novel preventive treatment options.
Galcanezumab, a humanized monoclonal injectable antibody that binds to calcitonin gene-related peptide (CGRP), belongs to a new class of molecules that is specifically targeted for migraine, unlike current oral preventive classes that were developed for other indications (15). The efficacy and safety of galcanezumab in patients with chronic migraine have been demonstrated in a Phase 3 RCT with a 3-month double-blind treatment period (REGAIN) (16). In this study, galcanezumab 120 mg or 240 mg versus placebo administered once monthly significantly (p < 0.001) reduced the mean number of monthly migraine headache days (MHDs). Both doses of galcanezumab were also superior to placebo for proportion of patients with ≥50% and ≥75% mean reduction in monthly MHDs, change in monthly MHDs with acute medication use and improvements in patient functioning. In REGAIN, 77.8% of patients had a history of prior migraine preventive treatment.
The objectives of this subgroup analysis of REGAIN were to evaluate efficacy outcomes for galcanezumab (120 mg and 240 mg) versus placebo in patients who had failed ≥2 or ≥1 prior preventive treatments for efficacy and/or safety reasons and in patients who had never failed preventive treatments previously.
Methods
Study design and patients
REGAIN was a Phase 3, placebo-controlled study of galcanezumab in patients with chronic migraine and was conducted at 116 study sites in 12 countries. The study comprised a 3-month double-blind treatment period, a 9-month open-label extension period and a 4-month post-treatment (follow-up) period. Eligible patients were randomized 2:1:1 to receive subcutaneous injections of placebo, galcanezumab 120 mg (with a loading dose of 240 mg) or galcanezumab 240 mg once every month during the double-blind treatment period (16).
REGAIN included patients aged 18–65 years with a diagnosis of chronic migraine (with or without aura), as defined by the International Classification of Headache Disorders 3rd edition, beta version (ICHD-3 beta) criteria (17). Patients experienced ≥15 headache days per month, of which at least eight were MHDs. Exclusion criteria included failure to respond to three or more classes of migraine preventives at maximum tolerated dose for at least 2 months. This treatment-failure exclusion only applied to those preventives with level A or B evidence based on the American Academy of Neurology/American Headache Society treatment guidelines (18) and botulinum toxin A or B. Therefore, patients could potentially have failed more than three different classes of preventive medications as long as those treatment failures were not in the above list of medications. Patients could take allowed concomitant medication for acute treatment as needed during the study, with the following exceptions: Restricted use of opioid- and barbiturate-containing medications (no more than 3 days per month) and injectable steroids (a single dose was allowed only once during the study in an emergency setting). Allowed concomitant acute treatments included acetaminophen (paracetamol), nonsteroidal anti-inflammatory drugs (NSAIDs), triptans, ergotamine and its derivatives, isometheptene mucate, dichloralphenazone and acetaminophen combination (Midrin), or any combination thereof.
The study protocol was reviewed and approved by the appropriate institutional review board for each of the study sites. The study was conducted according to good clinical practice and the Declaration of Helsinki guidelines. Prior to undergoing any study procedure, patients provided written informed consent.
Study objectives, measures and assessments
The primary objective was to assess if at least one dose of galcanezumab was superior versus placebo in overall mean change from baseline in the number of MHDs across the double-blind period. An MHD was defined as a calendar day, on which a migraine headache or probable migraine headache lasting ≥30 minutes occurred with features meeting ICHD-3 beta criteria. Key secondary outcomes included the following measures for each galcanezumab dose group compared with placebo during the double-blind treatment period:
Mean proportions of patients with ≥50% and ≥75% reduction in monthly MHDs. Overall mean reduction from baseline in monthly MHDs with acute medication use for migraine or headache. Mean change from baseline at Month 3 in the Role Function-Restrictive domain score of the Migraine-Specific Quality of Life Questionnaire version 2.1 (MSQ-RFR). This score ranges from 0 to 100, with higher scores indicating improved patient functioning.
Subgroup analyses of the above outcome measures were performed for the following patient sub-groups:
Patients who failed at least two prior preventives for efficacy and/or safety reasons. Patients who failed at least one prior preventive for efficacy and/or safety reasons. Patients who never failed any prior preventives. This included patients who had not taken any preventive previously (treatment-naïve) and patients who had taken a preventive but did not fail for efficacy reasons, safety reasons, or both.
The subgroups described above were identified to more closely address the medical question, “does galcanezumab offer benefit to patients who have failed at least one prior preventive or at least two prior preventives?” The efficacy gains with galcanezumab versus placebo across smaller subgroups (e.g. Failed 1 or Failed 2 prior preventives) were comparable, which allowed them to be pooled for better statistical power (not shown). For example, the subgroup with ≥2 prior preventive failures was comparable in efficacy to the subgroup with one prior preventive failure, which led these to be pooled to create the subgroup with ≥1 prior failure.
The primary purpose of these analyses was to evaluate the three main subgroups described above. The number of preventive failures in these subgroups referred to the number of individual medications failed in the past 5 years and did not refer to classes of medications. Also, there was no restriction as to which types of medications could count as a treatment failure in these subgroups, and failures could be due to either efficacy or safety/tolerability reasons. Note that this definition of treatment failures for the subgroup analyses differs from the previously described exclusion criterion of efficacy failures to preventives with Level A or B evidence from ≥3 classes (see Study design and Patients), which was based on lifetime history, not just the past 5 years. Thus, it was still feasible to explore an additional subgroup with ≥3 prior preventive failures for efficacy and/or safety reasons.
Headache data were collected using an electronic patient-reported outcomes (ePRO) diary where the patient logged in daily to answer questions. The ePRO diary data were used to categorize days as MHDs (16) and to record monthly MHDs with acute medication use. MSQ-RFR assessments were made using a self-report slate device during site visits. Acute headache medication overuse during the 30-day prospective baseline period was determined based on the following thresholds adapted from the ICHD-3 beta guidelines (17): i) triptan use for ≥10 days, ii) NSAID or aspirin for ≥15 days, iii) acetaminophen/paracetamol for ≥15 days, iv) ergotamine or its derivative for ≥10 days, v) ≥2 of the above medication classes for ≥10 days and vi) total days with drug use from at least two of the above categories for ≥10 days.
Statistical analyses
Analyses were performed on the intent-to-treat population, which included all patients who received at least one dose of galcanezumab or placebo. Subgroup analyses for repeated continuous and binary measures were conducted using restricted maximum likelihood-based mixed models with repeated measures (MMRM) and generalized linear mixed model, respectively. Overall mean change from baseline, which was the average mean change from baseline across Months 1–3, was estimated from the model. Response rates were calculated as the mean percentage of responders using the categorical, pseudo–likelihood-based repeated-measures analysis assessing overall response rate across Months 1, 2, and 3. Treatment-by-subgroup interactions were included in the models. Baseline acute medication overuse status (presence or absence) was included within the statistical model.
Failures to prior preventive treatments were determined on the basis of reasons for stopping prior preventives listed on the prior therapy case report form, which collected any migraine preventive medications that had been taken in the past 5 years. Failure to prior preventive treatment was defined as cessation of drug for efficacy-related reasons (“nonresponse” or “inadequate response”) or safety/tolerability reasons. Preventive treatments reported by the clinical investigative sites were further restricted to medications identified in the treatment guidelines as having been investigated for preventive use and having at least level C evidence of efficacy based on American Academy of Neurology/American Headache Society treatment guidelines; all reported data are specific to this criteria (18,19). All statistical analyses were conducted using SAS software (SAS Institute Inc, Cary, NC, USA). Two-sided p-values ≤ 0.05 were assumed to be statistically significant.
Results
Baseline demographics and disease characteristics
Distribution of patients by number of prior treatment failures in REGAIN.

GMB: galcanezumab; PBO: placebo.
Patient demographics and baseline disease characteristics in overall population and in subgroups with prior failures in REGAIN study.

p < 0.05 for differences vs. placebo.
For MSQ RF-R- domain scores: PBO: n = 546; GMB 120 mg: n = 272; GMB 240 mg: n = 272; whole population: 1090.
Baseline medication overuse was based on use of acute medication reported in the ePRO diary.
ePRO: electronic patient-reported outcome; GMB: galcanezumab; MHD: migraine headache day; MSQ RF-R: Role Function-Restrictive domain score of the Migraine-Specific Quality of Life Questionnaire version 2.1; PBO: placebo; SD: standard deviation.
Reasons for discontinuation of previous migraine prevention therapy in patients who failed ≥1 prior preventives in REGAIN.
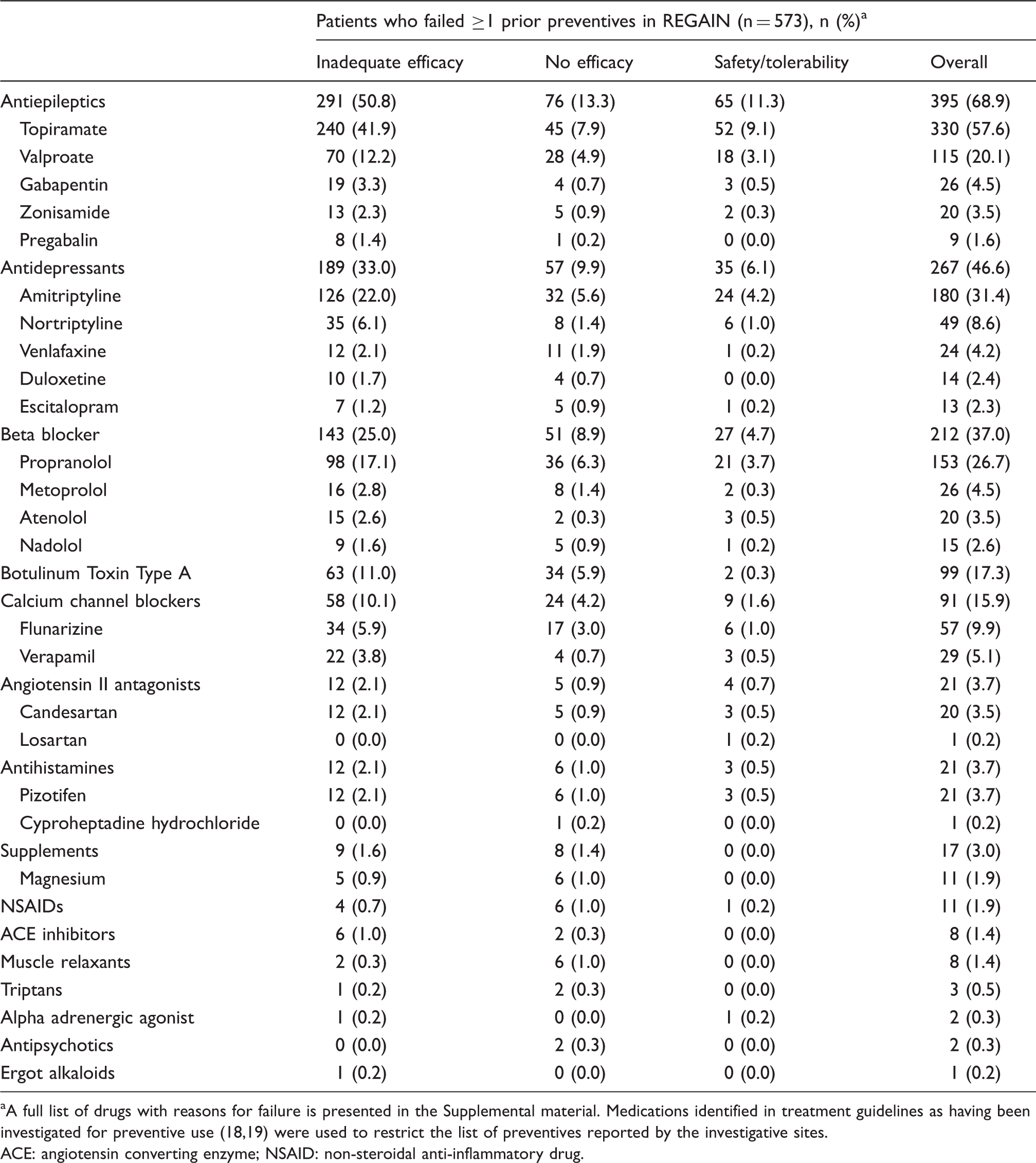
A full list of drugs with reasons for failure is presented in the Supplemental material. Medications identified in treatment guidelines as having been investigated for preventive use (18,19) were used to restrict the list of preventives reported by the investigative sites.
ACE: angiotensin converting enzyme; NSAID: non-steroidal anti-inflammatory drug.
Reductions in monthly migraine headache days
In patients who failed ≥2 or ≥1 prior preventive treatments, overall reductions from baseline in monthly MHDs were significantly (p < 0.01) larger with galcanezumab 120 mg or 240 mg versus placebo (Least Squares [LS] mean change [standard error [SE]]: ≥2 prior failures: galcanezumab 120 mg:−5.35 [0.71]; galcanezumab 240 mg: −2.77 [0.66]; placebo: −1.01 [0.54]; ≥1 prior failures: galcanezumab 120 mg: −5.53 [0.60], galcanezumab 240 mg: −3.53 [0.59]; placebo: −2.02 [0.49]) (Figure 1(a) and (b)). In the subgroup of patients who never failed prior preventives, overall reduction from baseline was numerically larger with galcanezumab 120 mg or 240 mg versus placebo; however, there was significance (p < 0.001) only for galcanezumab 240 mg versus placebo: galcanezumab 120 mg: −4.88 [0.75]; galcanezumab 240 mg: −6.58 [0.77]; placebo: −4.28 [0.70] (Figure 1(c)).
Monthly and overall LS mean changes from baseline in the number of migraine headache days per month during the treatment period in patients: (a) Who failed ≥2 prior preventives, (b) who failed ≥1 prior preventives and (c) who never failed any prior preventives, in REGAIN study.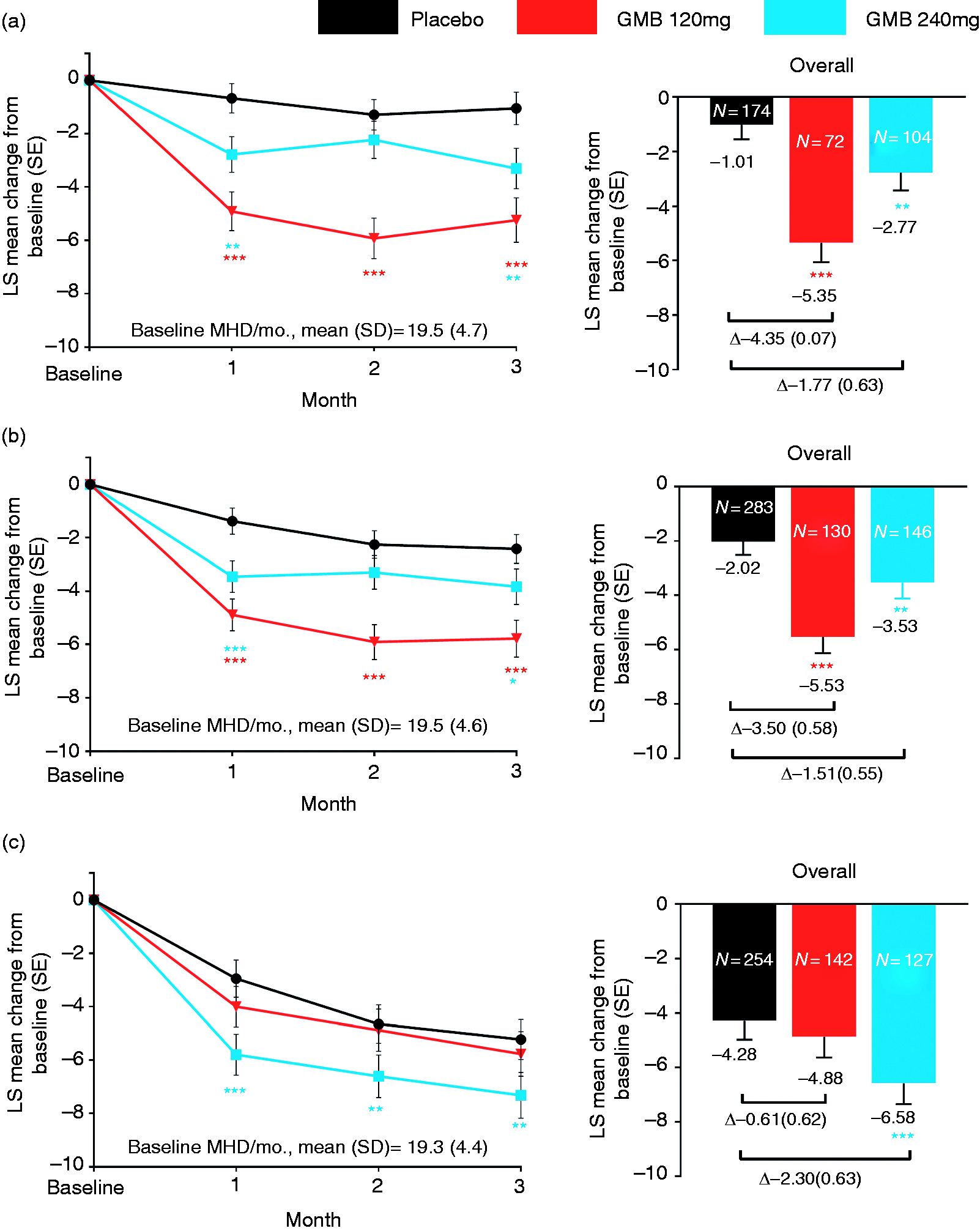
When considering MHD reductions by month, in patients with prior failures (≥2 or ≥1), treatment with galcanezumab 120 mg versus placebo led to statistically significantly (p < 0.001) larger reductions at each month of the treatment period (Months 1 through 3). In patients treated with galcanezumab 240 mg, monthly reductions were significantly (p < 0.05) larger versus placebo at Months 1 and 3 (Figure 1(a) and (b)). In the subgroup with no prior preventive failures, galcanezumab 240 mg versus placebo led to significant reductions (p < 0.01) at Months 1 through 3 (Figure 1(c)).
In patients with prior failures (≥2 or ≥1), differences in overall reductions in MHDs between galcanezumab and placebo were (LS mean change difference [SE]): ≥2 prior failures: galcanezumab 120 mg: −4.35 (0.70); galcanezumab 240 mg: −1.77 (0.63); ≥1 prior failures: galcanezumab 120 mg: −3.50 (0.58); galcanezumab 240 mg: −1.51 (0.55). These differences were smaller in patients with no prior failure; the smaller differences appeared to be driven by the larger placebo response in the no prior failure subgroup (Figure 1(a)–(c)).
Percentage of ≥50% and ≥75% responders for migraine headache days
Among patients who had failed ≥2 or ≥1 prior preventives, the estimated probability (SE) of achieving an overall ≥50% reduction in monthly MHDs from baseline was significantly greater (p < 0.01) with galcanezumab 120 mg or 240 mg versus placebo: ≥2 prior failures: galcanezumab 120 mg: 29.6% (4.7%); galcanezumab 240 mg: 18.7% (3.3%); placebo: 9.4% (1.9%); ≥1 prior failures: galcanezumab 120 mg: 31.2% (3.7%); galcanezumab 240 mg: 20.5% (3.0%); placebo: 11.3% (1.7%) (Figure 2(a) and (b)).
Mean percent of patients with ≥50% and ≥75% reductions in monthly migraine headache days during the treatment period in patients: (a) Who failed ≥2 prior preventives, (b) who failed ≥1 prior preventives and (c) who never failed any prior preventives, in REGAIN study.
In patients with ≥2 or ≥1 prior failures, the estimated probability (SE) of achieving a ≥75% response was larger with both galcanezumab doses versus placebo: ≥2 prior failures: galcanezumab 120 mg: 6.3% (2.2%); galcanezumab 240 mg: 5.0% (1.6%); placebo: 2.3% (0.8%); ≥1 prior failures: galcanezumab 120 mg: 7.0% (1.8%); galcanezumab 240 mg: 6.0% (1.6%); placebo: 2.7% (0.8%). All comparisons were statistically significant (p < 0.01) except for galcanezumab 240 mg versus placebo in the ≥2 prior failures subgroup (Figure 2(a) and (b)).
In the subgroup with no prior failures, the proportion of patients achieving ≥50% response or ≥75% response was significantly larger only for galcanezumab 240 mg versus placebo. In this subgroup, a larger placebo response was observed (Figure 2(c)).
Reduction in migraine headache days with acute migraine medication use
Among patients who failed ≥2 or ≥1 prior preventives, galcanezumab 120 mg or 240 mg versus placebo led to significantly (p < 0.05) larger overall reductions from baseline (LS mean change [SE]) in monthly MHDs with acute medication use for migraine or headache: ≥2 prior failures: galcanezumab 120 mg: −5.81 (0.69); galcanezumab 240 mg: −3.40 (0.65); placebo: −1.35 (0.53); ≥1 prior failures: galcanezumab 120 mg: −5.60 (0.57); galcanezumab 240 mg: −3.66 (0.56); placebo: −1.94 (0.46) (Figure 3(a) and (b)). In the subgroup with no prior failures, a larger placebo response was observed and significantly larger overall reductions from baseline were seen with galcanezumab 120 mg or 240 mg versus placebo (Figure 3(c)).
LS mean change in overall monthly migraine headache days with the use of acute migraine medications during the treatment period in patients: (a) Who failed ≥ 2 prior preventives, (b) who failed ≥ 1 prior preventives and (c) who never failed any prior preventives, in REGAIN study. Differences between individual galcanezumab dose groups and placebo are LS mean change difference (SE).
Change in MSQ RF-R domain score
Among patients with ≥2 or ≥1 prior failures, improvements from baseline at Month 3 in MSQ RF-R domain scores were significantly greater (p < 0.05) with galcanezumab 120 mg or 240 mg versus placebo. This indicated significantly greater improvements in patient functioning with galcanezumab versus placebo. LS mean change from baseline (SE) in these subgroups was: ≥2 prior failures: galcanezumab 120 mg: 19.13 (2.87); galcanezumab 240 mg: 19.24 (2.61); placebo: 10.67 (2.12); ≥1 prior failures: galcanezumab 120 mg: 21.63 (2.06); galcanezumab 240 mg: 19.22 (2.14); placebo: 13.64 (1.64) (Figure 4(a) and (b)). In the subgroup with no prior failures, significant improvements were seen only for galcanezumab 240 mg versus placebo, and similar to other outcomes, a larger placebo response was observed (Figure 4(c)).
LS mean change from baseline at Month 3 in Migraine-Specific Quality of Life Questionnaire Role Function-Restrictive (MSQ RF-R) domain in patients: (a) Who failed ≥ 2 prior preventives, (b) who failed ≥ 1 prior preventives and (c) who never failed any prior preventives, in REGAIN study. Differences between individual galcanezumab dose groups and placebo are LS mean change difference (SE).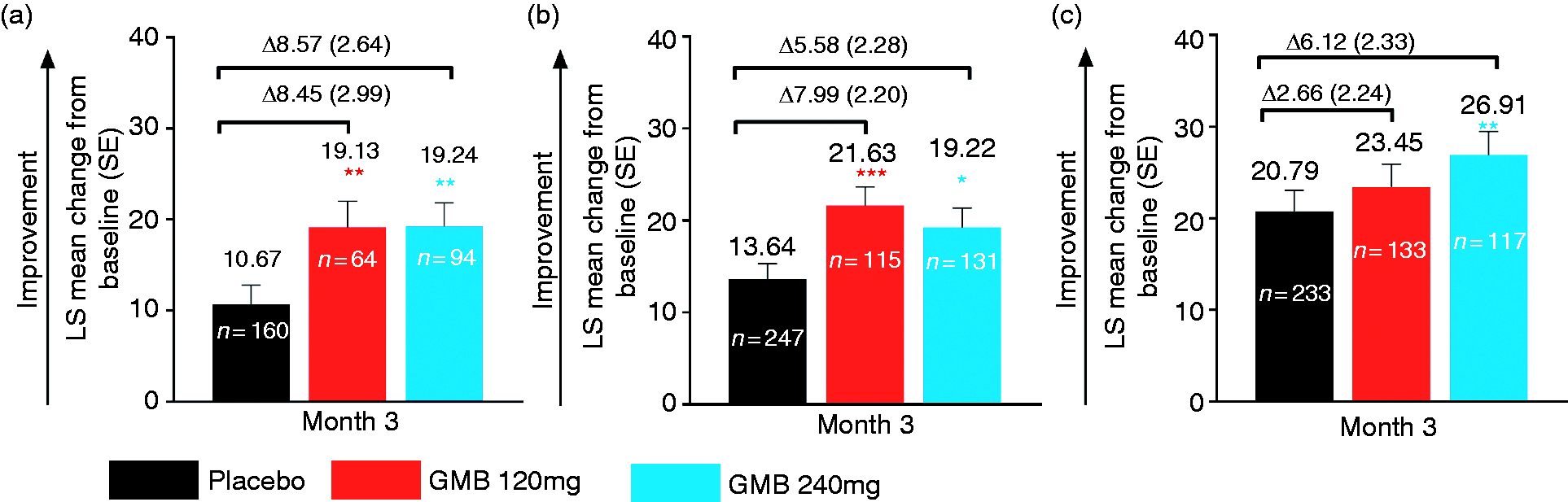
Exploratory outcomes in subgroup with ≥3 prior preventive treatment failures
In patients with ≥3 prior failures, overall reductions from baseline in monthly MHDs were significantly (p < 0.001) larger with galcanezumab 120 mg versus placebo: LS mean change (SE): galcanezumab 120 mg: −5.64 (0.97); galcanezumab 240 mg: −1.74 (0.91); placebo: −0.39 (0.76). For the overall ≥50% reduction in monthly MHDs, the estimated probability (SE) was significantly greater with galcanezumab 120 mg or 240 mg versus placebo: galcanezumab 120 mg: 29.1% (6.5%) (p < 0.001); galcanezumab 240 mg: 14.0% (3.7%) (p < 0.05); placebo: 6.1% (1.9%). For the outcome of ≥75% response rate, the analysis model did not converge. Overall reductions from baseline in monthly MHDs with acute medication use were: galcanezumab 120 mg: −6.01 (0.96) (p < 0.001 versus placebo); galcanezumab 240 mg: −2.34 (0.90) (not significant); placebo: −0.78 (0.75). Improvements from baseline at Month 3 in MSQ RF-R domain scores were: galcanezumab 120 mg: 20.34 (3.82) (p < 0.05 versus placebo); galcanezumab 240 mg: 16.24 (3.38) (not significant); placebo: 10.34 (2.74) (not shown).
Discussion
In this Phase 3 RCT in patients with chronic migraine, mean MSQ RF-R domain, MIDAS total and mean PGI-S scores at baseline reflected substantial functional impairment, very severe disability and markedly severe illness. The proportions of patients who had failed ≥1 prior preventives (51.5%) and ≥2 prior preventives (31.2%) at baseline are consistent with the high discontinuation rates reported with the current standard of care for migraine prevention (4,5). During the 1-month baseline period, a large proportion of patients met the criteria for acute medication overuse (63.8%), which is common among patients with chronic migraine, especially if they are not receiving an efficacious preventive treatment (22,23). Among patients who failed prior preventives, medications that were taken and failed included current standard of care and/or treatments with level A and level B evidence (18,19).
Efficacy results from these subgroup analyses demonstrate that galcanezumab is efficacious across multiple endpoints in patients with chronic migraine who failed ≥2 or ≥1 preventives previously. Patients who had a history of failing prior preventive treatments due to efficacy reasons and/or safety/tolerability reasons, and were treated with galcanezumab experienced significantly greater overall reductions in the number of MHDs per month, ≥50% and ≥75% response rates, overall reductions of MHDs with acute medication use, and improvements at Month 3 in patient functioning based on MSQ RF-R domain scores. Consistent results were seen in the small subgroup of patients with ≥3 prior failures, with the exception of ≥75% response rate, which could not be evaluated as the analytical model did not converge. Greater improvements in outcomes with galcanezumab versus placebo were also found in the subgroup of patients who had never failed a prior preventive treatment. However, the improvements were generally statistically significant only for galcanezumab 240 mg versus placebo. In the prior failure subgroups (≥2 or ≥1) as well as in the no prior failure subgroups, significant reductions in MHDs were seen with at least one of two galcanezumab doses versus placebo at Month 1, which continued through Month 3. Overall, this subgroup analysis demonstrates the consistent efficacious nature of galcanezumab among patients with chronic migraine, independent of prior preventive treatment failure status.
Difference in responses between galcanezumab and placebo for all outcome measures was numerically larger in the prior failure subgroups (≥2 or ≥1) compared with the no prior failure subgroup. This was likely driven by the lower placebo response in patients with prior failures (≥2 and ≥1) versus no prior failures, as the galcanezumab responses were comparable across subgroups. Similar to results reported here, in Phase 2 b and Phase 3 b studies of erenumab, a monoclonal antibody against CGRP receptor, lower placebo response was seen in patients with prior failure (≥2 or ≥1) versus no prior failure (24,25). The interplay between placebo response and patient expectations can be complex. The lower placebo response in patients with prior failures could reflect the lower expectations of this subgroup for a response when entering a clinical trial for an investigational treatment. On the other hand, the subgroup with a history of never receiving or never failing a preventive treatment may have more optimistic expectations (26,27).
For several outcomes, in the subgroup with prior failures, responses with galcanezumab 120 mg were numerically larger versus 240 mg. In the subgroup with no prior failures, responses to galcanezumab 240 mg were numerically larger versus 120 mg. This subgroup analysis was not powered to detect statistically significant differences between galcanezumab doses. The overall clinical trial population in REGAIN demonstrated no statistically significant differences between galcanezumab doses for any of the outcomes that are reported here (16), and similar results were observed in the overall populations of pivotal RCTs in episodic migraine (28,29). If the efficacy of galcanezumab doses is the same, as demonstrated in the overall populations, there is a 50% chance of one galcanezumab dose providing numerically larger responses than the other. Therefore, the differences observed between galcanezumab doses in the subgroups described here are more likely due to the small sample sizes and overall variability. Supporting this observation, at Month 1 in the subgroups with prior failure (≥2 or ≥1), reductions in monthly MHDs in the galcanezumab 120 mg dose group were larger than in the galcanezumab 240 mg group; however, at the beginning of Month 1, patients in both dose groups had received galcanezumab 240 mg as loading dose.
In previously published results from REGAIN analyzing the overall population, galcanezumab 120 mg and 240 mg met primary and secondary efficacy endpoints in patients with chronic migraine (16). Mean (SE) overall reductions in monthly MHDs in galcanezumab dose groups were similar across the overall population (16), and the subgroups described here, ranging from: overall population: −4.83 (0.44) to −4.62 (0.43); ≥2 prior failures: −2.77 (0.66) to −5.35 (0.71), ≥1 prior failures: −3.53 (0.59) to −5.53 (0.60); no prior failures: −4.88 (0.75) to −6.58 (0.77). Mean differences (95% CIs) for galcanezumab 120 mg vs. placebo and galcanezumab 240 mg vs. placebo were as follows: overall population, −2.09 (−2.92, −1.26) and −1.88 (−2.71, −1.05); ≥2 prior failures: −4.35 (−5.73, −2.96) and −1.77 (−3.00, −0.54); ≥1 prior failures: −3.50 (−4.64, −2.37) and −1.51 (−2.60, −0.42); no prior failure: −0.61 (−1.82, 0.60) and −2.30 (−3.54, −1.06). The fairly consistent nature of these results across subgroups with various histories of prior preventive treatment failures and the overall chronic migraine population is also observed for the secondary endpoints, including ≥50% and ≥75% response rates, reductions in MHDs with acute medication use, and improvements in patient functioning as measured using the MSQ RF-R domain score (16).
With regard to other research in patients with chronic migraine who failed prior preventives, erenumab has demonstrated significant improvements versus placebo across multiple outcomes in a Phase 2 b study (24). Mean differences (95% CIs) for erenumab 70 mg versus placebo and erenumab 140 mg versus placebo, respectively, were as follows for overall reductions in monthly migraine days: ≥1 prior failures: −2.5 (−3.8, −1.2) and −3.3 (−4.6, −2.1); ≥2 prior failures: −2.7 (−4.2, −1.2) and −4.3 (−5.8, −2.8); no prior failure: −2.2 (−4.1, −0.3) and −0.5 (−2.4, 1.5). Results with botulinum toxin A are mixed, with one study in patients with prior failures not demonstrating statistical separation from placebo in headache frequency reduction, and another study in patients with ≥3 failures demonstrating separation from placebo on multiple outcomes (30,31). However, the results reported here are comparable to the mean reductions in monthly migraine days (95% CI) reported in the overall population for botulinum toxin A over placebo at Week 24 in the Phase 3 PREEMPT study: −2.0 (−2.7, −1.3) (32).
The current results are also broadly comparable with results from Phase 3 RCTs in patients with episodic migraine with galcanezumab and erenumab. In Phase 3 studies of galcanezumab in patients with episodic migraine, in the overall population, mean difference (95% CIs) for galcanezumab over placebo for overall reductions in monthly MHDs (Months 1 through 6) ranged from −2.0 (−2.6, −1.5) to −1.8 (−2.3, −1.2) (28,29). In the LIBERTY study of erenumab, in patients with episodic migraine and with 2–4 prior preventive failures, mean difference for erenumab 140 mg over placebo for reductions in monthly migraine days (weeks 9–12) was −1.6 (−2.7, −0.5) (25).
The clinical meaningfulness of these Phase 3 results with galcanezumab in patients with prior failures (≥2 or ≥1) and in the overall population can be inferred based on a recent adaptation of the IMMPACT framework for studies in chronic migraine (33). Clinically meaningful reductions in MHDs (≥1 day per month in patients with chronic migraine) are seen with galcanezumab, and these changes are significant versus placebo. The proportion of galcanezumab-treated patients achieving ≥50% response for overall reduction in MHDs is significantly greater versus placebo. In patients with chronic migraine, a ≥30% response for overall reduction in MHDs is considered clinically meaningful (33,34). With regard to MSQ RF-R domain scores, galcanezumab treatment led to increases that exceed established between-group minimally important differences (MIDs) (3.2, vs. placebo) and within-group MIDs (10.9, from baseline) (35,36). The discontinuation rates due to adverse events among galcanezumab-treated patients in the overall population are low in REGAIN (1%) and also low across all pivotal Phase 3 RCTs (EVOLVE-1 and -2, and REGAIN) of galcanezumab (<2.5%) (28,29,37). These discontinuation rates with galcanezumab are much lower than discontinuation rates reported for propranolol, amitriptyline and topiramate in RCTs, within 4 to 6 months of initiation (38). Overall, these results suggest that treatment with galcanezumab may lead to improved adherence, compliance, and hence better long-term patient outcomes.
There are multiple strengths in this subgroup analysis in patients with prior failures. The findings are from a global, multi-center, Phase 3 RCT. The documentation of failures of prior preventives in this study could be due to multiple reasons. The list of preventives that patients had taken and failed included those with level A or B evidence as per treatment guidelines for migraine prevention. Efficacy with galcanezumab in the prior failure subgroups was demonstrated not just in migraine frequency-related endpoints, but also in a key measure of patient functioning, namely MSQ RF-R domain score. Further, these results in a population with high unmet need and considered more difficult to treat are consistent with results in the overall population. However, these results are limited by the number of patients within each group of prior failures (≥2 or ≥1) and exclusion of patients who had failed three or more classes of preventives (level A or B evidence) at baseline. A study specifically designed to evaluate the effects of galcanezumab in patients with treatment-resistant episodic or chronic migraine who have failed previous migraine preventives (NCT03559257) may address this.
The results of this study are important, given that clinicians will need to evaluate the best treatment options for this subgroup of patients with high unmet need who have been treated previously with preventives but failed to respond or discontinued for safety- and/or tolerability-related reasons. Practitioners who are challenged with managing patients with chronic migraine with a history of prior preventive treatment failures may perceive that this subpopulation is less likely to respond to any preventive migraine treatment. This research evaluated multiple outcomes in addition to the reduction in MHDs per month, with consistent results across patients with different histories of prior preventive failures.
In conclusion, results reported here and results from the overall population reported previously (16) suggest that galcanezumab is efficacious for patients with chronic migraine who have previously failed other migraine therapies and may also be suitable among patients without prior preventive treatment failure. Thus, galcanezumab may offer a treatment option such that patients in this high unmet need subgroup are not at risk of managing their chronic migraine with only acute medications, which could lead to further exacerbation.
Clinical implications
In a Phase 3 randomized clinical trial (REGAIN), galcanezumab was efficacious versus placebo in patients with chronic migraine who failed ≥2 or ≥1 prior preventives for efficacy and/or safety reasons for several key primary and secondary outcomes including: reductions in monthly migraine headache days (MHDs), proportion of patients with ≥50% and ≥75% reduction in monthly MHDs, change in monthly MHDs with acute medication use and improvements in patients’ functioning per MSQ RF-R domain score. In patients who had never failed a prior preventive, results were statistically significant for galcanezumab 240 mg versus placebo on all outcomes, and for the 120 mg group on reduction in MHDs with acute medication use. A higher placebo response was seen in this subgroup. Results suggest that galcanezumab is efficacious in patients with chronic migraine who have previously failed other migraine preventive treatments and may also be suitable in patients without any prior preventive treatment failure. Galcanezumab may offer a treatment option in patients with chronic migraine with high unmet need who are at risk of managing their disease with only acute medications.
Supplemental Material
Supplemental material for Efficacy of galcanezumab in patients with chronic migraine and a history of preventive treatment failure
Supplemental Material for Efficacy of galcanezumab in patients with chronic migraine and a history of preventive treatment failure by Dustin D Ruff, Janet H Ford, Antje Tockhorn-Heidenreich, Matthew Sexson, Sriram Govindan, Eric M Pearlman, Shuu-Jiun Wang, Arif Khan and Sheena K Aurora in Cephalalgia
Footnotes
Acknowledgements
Qi Zhang, an employee of Eli Lilly and Company at the time of analysis, provided statistical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Eli Lilly and Company, Indianapolis, IN, USA. Dustin D Ruff, Janet H Ford, Matthew Sexson, Antje Tockhorn-Heidenreich, Sriram Govindan, Eric M Pearlman and Sheena K Aurora are employees of Eli Lilly and Company, and/or its subsidiaries, and hold company stock. Shuu-Jiun Wang has served on the advisory boards of Eli Lilly, Daiichi-Sankyo, and Taiwan Pfizer. He has received honoraria as a moderator from local companies (Taiwan branches) of Allergan, Pfizer, Eli Lilly, Bayer, and Eisai. He has received research grants from the Taiwan Minister of Technology and Science, Brain Research Center of National Yang-Ming University, Taipei Veterans General Hospital, and Taiwan Headache Society. Arif Khan was a principal investigator on the REGAIN study and has been a principal investigator for over 550 clinical trials with more than 60 pharmaceutical companies. He has no governmental or academic positions, except for adjunct, non-paid professorship at Duke University.
Trial registration
This study is registered with ClinicalTrials.gov, number NCT02614261.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
