Abstract
Objective
Studies have shown it is possible to elicit a tension-type headache episode in 15 to 30% of healthy individuals following a tooth-clenching or stress-inducing task. Despite this, no studies have attempted to understand why some healthy individuals develop a headache episode while others do not.
Methods
The present randomized, single-blind, controlled study recruited 60 healthy participants who participated in a 30-minute tooth-clenching task and 10 participants who participated in a control task. Before the tasks, participants had their pericranial tenderness and pain modulation profiles (wind-up ratio and conditioned pain modulation) assessed. Two hours later, pericranial tenderness and pressure pain thresholds were assessed as well as any developing temporomandibular disorders. Pain diaries were kept for 24 hours to register any developing pain or headache.
Results
Participants with a decrease in pericranial tenderness after the tooth-clenching task were less likely to develop headache when compared to participants without. Pain modulation profiles could not predict who developed headache and who did not. Finally, no difference was found between groups for developing temporomandibular disorders. No difference in frequency of participants who developed headache was found between the tooth-clenching and the control task.
Conclusions
In conclusion, it was shown that increased pericranial tenderness was not required to trigger an episode of tension-type headache in healthy participants. Furthermore, pain modulation profiles could not predict who developed headache and who did not. Finally, activation of descending inhibitory pathways, as assessed by decreases in pericranial tenderness, was protective against the development of headache. These findings provide new insights into the pathophysiology of experimentally-induced tension-type headache.
Introduction
Peripheral activation of muscle nociceptors, for example following involuntary muscle contraction, has been reported to be involved in the development of muscle pain but also in the development of episodes of TTH (1–3). However, very few studies have looked into the role of muscles in the development of episodes of TTH in healthy individuals. The few studies assessing this have used a static isometric contraction task of the masticatory or cervical muscles as a way to experimentally trigger headache in healthy individuals (2,4–6). This method has been used supposing that it may lead to increased pericranial tenderness (PCT) and in turn trigger a headache episode in susceptible individuals (2). In these studies, between 15 and 30% of healthy individuals developed a headache consistent with TTH (2,4). Neufeld et al. included a placebo task and found that both TTH patients and healthy individuals were as likely to develop headache following the placebo task as after a tooth-clenching task (4). This indicates that tooth-clenching did not play a specific role in the development of headaches and the reasons for their occurrence remain unknown (4). Importantly, in these studies, the headaches that developed following these tasks were not assessed according to the diagnostic criteria for temporomandibular disorders (DC/TMD) (7) and could possibly be classified as myalgia of the temporalis muscle or headache attributed to TMD.
Previous studies have attempted to phenotype patients based on their facilitation and inhibition qualities related to nociceptive processing such as building pain modulation profiles (PMP) (8,9). This phenotyping has been able to predict which patients will respond to different treatments (9–11). However, to our knowledge, PMP have not been used to predict which individuals will develop a headache episode following a headache-inducing task and who will not.
With this in mind, we aimed to use a tooth-clenching task to trigger headache in healthy participants and assess if their PMP as well as changes in PCT could predict who develops headache and who does not. Finally, we aimed to assess what kind of headache and pain develop according to both the International Classification of Headache Disorders 3rd edition (ICHD-3) and DC/TMD.
Material and methods
Participants
Seventy participants (33 women and 37 men) with a mean age of 28.6 years (SD ± 11.7) participated in this study. Participants were recruited by advertising in flyers around the Aarhus University campus, internet pages of the Section of Orofacial Pain and Jaw Function (http://odont.au.dk/om-odontologi/sektioner/kof/) and internet volunteer recruiting systems (www.sona-systems.com, forsoegsperson.dk, www.sundhed.dk). Participants were screened for TMD in accordance with the DC/TMD (7). The inclusion criteria for this study were that participants should be: a) adults above 18 years of age, and b) healthy as determined by the investigator (FE). The exclusion criteria for this study were: a) positive screening for TMD according to the DC/TMD, b) other orofacial pain or pain elsewhere in the body, c) report of any kind of headache, with the exception of a maximum of 12 TTH days in the past year, d) use of analgesic or anti-inflammatory medication in the 24 hours preceding the experiment day in order to avoid any risk of including a patient with myalgia or headache.
The sample size was calculated with a risk of type I error of 5% and an estimate of the inter-individual variation of 30% for the total tenderness scores (TTS), a minimal relevant difference to detect set at 20% and a sampling ratio of one participant that develops headache to five participants that do not.
The study was approved by the Central Denmark Region Research Ethics Committee (1-10-72-235-17) and conducted in accordance with the Declaration of Helsinki.
Study design
The present randomized, single-blind, controlled study consisted of a first session that lasted approximately two hours and a second session two hours later that lasted approximately one hour (Figure 1). In the first session, participants were assessed for TMD according to the DC/TMD (7). Following this, their PCT was assessed by a modified TTS. Next, their conditioned pain modulation (CPM) and wind-up ratio (WUR) were assessed in both the anterior temporalis and thenar regions. After these baseline assessments, a tooth-clenching task was performed. Two hours after the tooth-clenching task, the participants returned to the laboratory and were assessed for TMD development, headache development, TTS and pressure pain thresholds (PPT). Before leaving the laboratory, the participants were asked to rate in a diary, every 2 hours, any head, facial and/or bodily pain they experienced in the 24 hours following the tooth-clenching task. Ten out of the 70 participants were recruited for a control task, which was the same as the tooth-clenching task with the exception that participants were asked to look at a blank screen and not clench their teeth. They were not told that looking at the screen was a control task. The same examiner (FE) performed all assessments and experimental procedures at the Department of Dentistry and Oral Health, Aarhus University. Participants in both tasks were not informed that the study was focusing specifically on headache, to avoid bias.
Overview and timeline of the tooth-clenching task. The control task was performed with the same design, but participants stared at a screen without clenching their teeth.
Clenching and control task
The clenching task consisted of participants using a visual feedback signal to clench their teeth at 10% of their maximum voluntary contraction for three sets of 10 minutes each with a 1-minute interval between sets. The 10% maximum voluntary contraction was determined by taking the average of three 2-second epochs of maximum voluntary contraction from the temporalis muscle using EMG surface electrodes (4). The control task consisted of participants looking at a blank white screen during three sets of 10 minutes each with a 1-minute interval in between. Participants were told to have their teeth apart and jaw relaxed at all times. Participants were monitored to assure compliance with the task by an examiner (FE). At the end of both tasks, the participants were asked to rate any pain, fatigue and stress they experienced during the task on a 0 to 10 numeric rating scale (NRS), where 0 indicated “none whatsoever” and 10 indicated the “most imaginable”. Participants were blind to whether they were participating in the active or control task and simple randomization was done using randomization.com.
Total tenderness score
The participants were evaluated for PCT according to a modified version of the TTS (12,13). The muscles evaluated for PCT were the frontal, temporalis, masseter, splenius capitis and trapezius. Furthermore, the TTS was divided into a MasticatoryTS (masseter and temporalis muscles) and a CervicalTS (splenius capitis and trapezius muscles). All muscles were palpated for 2 seconds with a manual palpometer (Palpeter®, Sunstar Suisse SA) of 1.0 kg of force. Participants rated the sensation on a 0–100 scale with 0 being “no sensation whatsoever”, 1 to 49 being “sensation of pressure but the stimulus is not painful, 50 being “barely painful” and 100 being “the most painful imaginable” (14,15). This was done to ensure that participants were able to rate both painful and non-painful sensations and as such give a more comprehensive assessment of the mechanical sensitivity of the pericranial muscles. The TTS was repeated before and after the task and two times at each instance.
Conditioned pain modulation
CPM is a non-invasive test of the endogenous pain-inhibitory system using a heterotopic noxious conditioning stimulation paradigm. The protocol for this study was similar to one used in a previous study (16). Briefly, PPT were assessed in triplicate using a handheld pressure algometer (Somedic Production AB, Sollentuna, Sweden) before, during, and after application of a noxious conditioning cold stimulus (cold pressor test: immersion of the participant's foot in ice water maintained at 5–10℃) and a non-noxious conditioning stimulus (neutral water maintained at 26–28℃) (17). The primary outcome was the relative changes in PPT to baseline; the order of the water temperature was randomized using randomization.com.
Wind-up ratio
WUR was assessed as per a previously used protocol (18). In summary, the perceived intensity of a single pinprick stimulus was compared to a series of 10 repetitive pinprick stimuli of the same intensity at a rate of one pinprick per second. The participant was instructed to give a pain rating for the single pinprick and then a rating for the 10th pinprick. For this, the participants used a ‘0–100’ NRS where 0 indicated “no pain” and 100 indicated the “most intense pain imaginable”. The whole procedure was repeated three times.
Pressure pain threshold
A hand-held electronic pressure algometer (Somedic Production AB, Sollentuna, Sweden) was used to assess the PPT in the anterior temporalis muscle and the thenar region. This was done by applying increasing forces with the algometer perpendicularly to the skin at a continuous rate of approximately 30 kPa/s by visualizing in a digital readout of ramp rate until the participants indicated the perception of pain by pressing on a button connected to the algometer. For each region the PPT were assessed in triplicate, both before the clenching task as part of the CPM protocol and two hours after the task.
Pain diary
Participants were asked to fill out a pain diary, where they recorded their pain for the masseter and temporalis region, headache and pain in any other body region on a 0 to 10 NRS where 0 indicated “no pain whatsoever” and 10 indicated the “most intense pain imaginable”. They were asked to record their scores every 2 hours up to 24 hours after the task.
Classification of headache and TMD
Diagnostic questions present in pain diary.

Participants were instructed on how to use the diary before they left the laboratory by the same examiner (FE). For the purpose of this study, patients were instructed that if they developed pain in the temple area and this pain was modified by jaw function, they should rate this as temporalis pain and, if the pain was not modified by jaw function, they should rate it as a headache. This was to ensure that participants with myalgia of the temporalis and/or headache attributed to TMD were not diagnosed as TTH. Finally, assessment of any TMD was done at both sessions according to the DC/TMD by physical examination of all participants (7,21). For the purposes of this study, participant report of muscle pain (from the pain diary) will be referred to as masseter or temporalis pain, depending on which muscle is affected, and myalgia, as diagnosed according to the DC/TMD by the investigator (FE), will be referred to as myalgia.
Subgrouping of participants
Subgrouping of participants into four distinct PMP was done with CPM (normal/impaired) and WUR (normal/facilitated) as variables, as has been previously reported (8). The four distinct PMP were as follows: Double pronociceptive (impaired CPM and facilitated WUR); inhibitory pronociceptive (impaired CPM and normal WUR); facilitatory pronociceptive (normal CPM and facilitated WUR), and antinociceptive (normal CPM and normal WUR). For statistical analysis purposes, participants were divided into pro-nociceptive and anti-nociceptive. For the TTS, a decrease following the tooth-clenching task was considered normal.
WUR data for each participant was Z-transformed and a facilitated WUR was considered if the Z-score was higher than 1.96, as per Rolke et al. (18). For the CPM and the TTS data, the smallest detectable change (SDC) (22); that is, the smallest change in score that can be detected above the measurement error, was used as the cut-off point. For the calculation of the cut-offs, we used the three repeated PPT measurements that were taken at baseline and the two repeated TTS, also taken at baseline, to determine the standard error of the measurement
Statistical analyses
Since the purpose of this study was to assess differences between participants who developed headache and those who did not, the data from the six participants that developed myalgia and/or headache attributed to TMD were not analysed. Data were assessed for normality by using Q-Q plots. All data were normally distributed with the exception of the WUR data. For analysis of WUR, the data were log transformed and assessed again using Q-Q plots, at which point it was normally distributed. The primary outcome parameters for this study were: Headache developed after the tooth-clenching task (yes/no) and changes in TTS before and after the tooth-clenching task (TTS difference from baseline).
To ascertain differences between participants who developed headache and the ones who did not, repeated measures analysis of variance (ANOVA) was used. 2-way ANOVA was used to test for differences in sensations immediately after the tooth-clenching task with the factors of group (headache and no-headache) and type (pain, fatigue and stress). 4-way ANOVA was used to test differences in TTS with the factors of task (tooth-clenching and control), group (headache and no-headache), type (TTS, MasticatoryTS and CervicalTS) and time (before and after task). 4-way ANOVA was used to test for differences in CPM effect with factors of task (tooth-clenching and control), group (headache and no-headache), site (temple and thenar) and temperature (cold and neutral). 3-way ANOVA was used to test differences in WUR with the factors of task (tooth-clenching and control), group (headache and no-headache) and site (temple and thenar). 4-way ANOVA was used to test differences in PPT with the factors of task (tooth-clenching and control), group (headache and no-headache), time (before and after task) and site (temple and thenar). When ANOVA was used, following Bonferroni correction, p-values of less than 0.006 were considered significant for the main factors and interactions to correct for multiple comparisons. Moreover, Tukey's HSD tests were used to adjust for multiple comparisons for the post hoc comparisons.
A Chi-square test was used to assess differences between groups (headache, no-headache) for the PMP (anti-nociceptive and pro-nociceptive). Fischer's exact test was used to test for differences in headache frequency between the tooth-clenching task and the control task and to test for differences between groups for TTS and DC/TMD diagnosis. Furthermore, odds ratios and confidence intervals were also calculated. For the binomial data, p-values were considered significant if less than 0.05.
Results
Upon inclusion into the study, 60 participants were randomly assigned to the tooth-clenching task and 10 to the control task. In the 24 hours following the tooth-clenching task, 35 of the 60 participants did not develop any pain or headache, 19 developed headache and six developed pain in the masseter and/or temporalis muscles that was modified by jaw function. Of these six, four could be classified as headache attributed to TMD. In the control task, three of the 10 participants developed headache and seven participants did not develop any pain or headache. All headaches that developed were compatible with a diagnosis of probable TTH (19). Characteristics of the headaches that developed following the tooth-clenching task can be seen in Table 2 and the frequency of the different pain types that developed over time can be seen in Figure 2.
Number of participants that developed each type of pain at each recording period according to pain diaries. Number of participants that developed each criterion for the diagnosis of TTH according to pain diaries. Approximate percentage in brackets (n = 19).

Pain, fatigue and stress following the tooth-clenching task
A significant difference was found for the factor type (p < 0.006, ANOVA). Post-hoc analysis showed that significantly higher scores were found for fatigue and stress during the tooth-clenching task when compared to pain (p < 0.05, Tukey post-hoc). No difference was found between groups or the interaction group × type (p > 0.006, ANOVA).
Wind-up ratio comparison between the headache and no-headache groups
No significant differences were seen in WUR for any of the assessed parameters (p > 0.006, ANOVA) (Figure 3).
WUR and CPM values for the headache and no-headache groups and PPT for before and after tooth-clenching task for the headache and no-headache groups. Vertical bars show mean values. Error bars show SD. No significant differences were seen for any of the parameters (p > 0.0063, ANOVA).
Conditioned pain modulation comparison between the headache and no-headache groups
Significant differences were noted for the factors of temperature and the interaction site × temperature (p < 0.006, ANOVA). No significant differences were seen between groups (p > 0.006, ANOVA) (Figure 3).
Pressure pain thresholds comparison between the headache and no-headache groups
Significant differences were seen for the main factors time and site (p < 0.006, ANOVA). No significant differences were seen between groups (p > 0.006, ANOVA) (Figure 3).
Pericranial tenderness comparison between the headache and no-headache groups
Significant differences in tenderness scores were seen for type and type × time × group (p < 0.006, ANOVA). In the post-hoc analysis, it could be seen that MasticatoryTS were significantly higher than CervicalTS (p < 0.05, Tukey post-hoc). Furthermore, a significant decrease in all three tenderness scores between the two sessions was seen for the no-headache group (p < 0.05, Tukey post-hoc) but not for the headache group (p > 0.05, Tukey post-hoc). No significant differences were seen for tenderness scores at baseline between groups (p > 0.006, ANOVA) (Figure 4).
Tenderness scores for before and after the tooth-clenching task for both the headache and no-headache groups. Vertical bars show mean values. Error bars show SD. A significant decrease in total, masticatory and cervical tenderness scores was seen for the no-headache group after the tooth-clenching task when compared to before the tooth-clenching task (*p < 0.001, Tukey post-hoc).
When looking at individual differences between groups for changes in TTS, it could be seen that there was a significantly higher percentage of participants that had a decrease in TTS below the cut-off point in the no-headache group when compared to the headache group (p < 0.05; OR: 9.45; 95% CI = 2.6–34.3, Fischer's exact test) (Figure 5).
Individual TTS scores by group. A significantly higher number of participants in the no-headache group had a decrease in TTS below the SDC when compared to the headache group (p < 0.05; OR: 9.45; 95% CI: 2.6–34.3).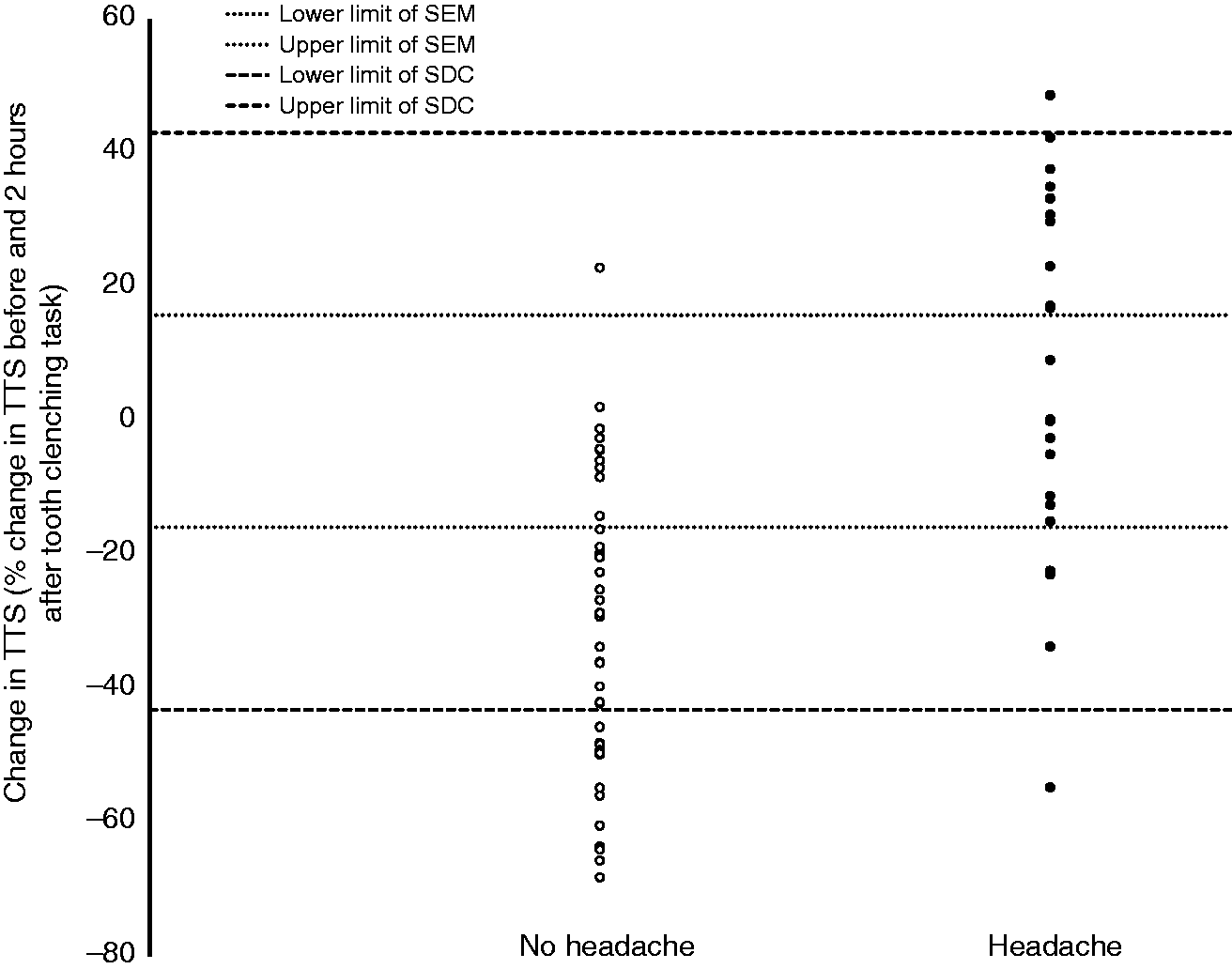
Pain modulation profiles comparison between the headache and no-headache groups
It was shown that there was no difference between groups for whether they had a pro-nociceptive or anti-nociceptive PMP (p > 0.05, Chi-square test) (Figure 6).
Scatterplot for the headache and no-headache group of WUR and CPM values. The WUR cutoff is illustrated by a dotted line. The CPM cutoff is illustrated by a dashed line. No difference was seen between the number of participants with a pro-nociceptive profile in the headache group when compared to the no-headache group (p = 0.51; OR: 0.826; 95% CI: 0.23–3.02).
DC/TMD diagnosis comparison between the headache and no-headache groups
One individual in each group was diagnosed with myalgia according to the DC/TMD and, as such, no significant differences were seen between groups for DC/TMD diagnosis (p > 0.05, Fischer's exact test).
Differences between tooth-clenching task and control task
For TTS, CPM, PPT and WUR, no differences were found between tasks either as a main factor or as part of an interaction (p > 0.006, ANOVA). Furthermore, no differences were found for headache frequency between the tasks (p > 0.05, Fischer's exact test).
Discussion
The main findings of this study were that: a) PMP were not able to predict who developed headache and who did not following the tooth-clenching task; b) participants that had a decrease in TTS after the tooth-clenching task were less likely to develop headache when compared to participants whose TTS did not decrease; c) only two participants (one in each group) had a diagnosis of myalgia according to the DC/TMD following the tooth-clenching task.
Headache development following tooth-clenching and control tasks
As expected, headaches that developed after both tasks fulfilled the criteria for probable TTH. Probable, because we could not ascertain if participants fulfilled criterion A of the ICHD-3 diagnostic criteria for TTH, which was at least 10 episodes of headache (19). All other criteria were met. In this study, around 31% of participants developed headache following the tooth-clenching task and 30% developed headache following the control task. These numbers are within range of previous studies (2,4,6). Four of six participants that developed masticatory muscle pain had pain in the temporalis muscle, and they reported it to be modified by jaw function. As such, if examined at that time point, they could have been classified as either myalgia of the temporalis, per the DC/TMD, or headache attribute to TMD, per both the ICHD-3 and the DC/TMD (19,23). Previous studies that did not consider these two diagnoses may have included these participants in the TTH group, which could explain differences between this study and previous studies.
Pain modulation profile differences between the headache and no-headache groups
No difference in PMP was found between participants who developed headache and those that did not. This indicates that PMP, as assessed by CPM and WUR, could not predict which participants developed a TTH episode. This supports the notion that if individuals are healthy, as were the participants in this study, they will likely have normal PMP. Accordingly, changes in WUR and CPM in headache patients may occur as a consequence of frequent headaches rather than being the cause of frequent headaches (24,25).
Total tenderness scores assessment
In the present study, the TTS was used in a novel manner because it was felt that the traditional way of assessing TTS would not allow for an appropriate assessment of mechanical sensitivity of the pericranial muscles. Traditionally, TTS mixes different terms used interchangeably such as discomfort, pain and painful tenderness (12) that, together with a lack of standardization of the palpation force (2), can lead to erroneous results. These two reasons could explain why our results are in contrast to the findings by Jensen et al. (2), which is the only other study that has assessed TTS differences between healthy controls that developed headache and those that did not.
Tenderness scores, pressure pain threshold and myalgia diagnosis differences between the headache and no-headache group
The most striking result from this study was that participants not developing headache had a significant decrease in TTS following the tooth-clenching task but participants developing headache showed little change in their TTS. This is contrary to a previous study (2) that reported a slight increase in TTS following a tooth-clenching task in healthy individuals who did not develop headache. As previously explained, this could be due to different ways of assessing the TTS, the time point at which participants were assessed (5) as well as a smaller sample size in the aforementioned study. Moreover, since the above-mentioned study did not assess whether participants developed masticatory muscle pain, they may have included those who did in the no-headache group. Importantly, decreases in TTS found in the present study were due to a decrease in MasticatoryTS and CervicalTS, which indicates it is not a local phenomenon but rather due to a central mechanism such as the activation of descending inhibitory pathways. Finally, when looking at changes in TTS at the individual level (Figure 5), participants in the no-headache group mostly had a decrease in TTS, with 12 of them having a substantial decrease that was below the cut-off point. In the headache group, approximately 50% had a decrease in TTS while the other 50% increased, with only one participant showing an increase above the cut-off point. This further reinforces the idea that increased PCT as assessed by TTS did not seem to play a role in triggering TTH episodes. The change in PCT found in this study could indicate that participants who were able to engage their descending inhibitory pathways in response to noxious elements of the tasks, such as stress or visual disturbance, not only did not develop a headache but also had a decrease in PCT. This lack of ability to engage the descending inhibitory pathways was not shown in the CPM assessment, likely because it evaluates responses to exogenous painful stimuli and not other stimuli such as stress.
No difference in PPT between groups or sessions was found. Lack of changes in PPT have been previously reported (4,26) but one study reported an increase in PPT in healthy participants who did not develop headache following a tooth-clenching task (2). Differences could be due to the different time points at which the PPT were assessed (5).
Finally, no differences were found between groups when assessing participants according to the DC/TMD, as only one participant in each group received a diagnosis of myalgia following the tooth-clenching task. This finding further points towards a lack of increased PCT in the development of TTH episodes in healthy participants.
Taken together, the results of this study indicate that TTH episodes in healthy participants develop irrespective of any kind of increase in PCT. This is contrary to general opinion (1,2,27) but it is not the first time that a lack of muscular involvement in TTH has been proposed (28,29). Furthermore, case reports in the literature have mentioned headache as the only pain complaint of patients with congenital insensitivity to pain, meaning that increased PCT could not be responsible for their headaches (29).
Comparisons of a tooth-clenching and control task
In this study, no difference was found in the percentage of participants that developed headache in the tooth-clenching task when compared to the control task. Furthermore, no difference was found between tasks for any of the other assessed parameters. This is in agreement with a previous study (4). Finally, six participants developed masticatory muscle pain following the tooth-clenching task, but this did not occur in the control task. Masticatory muscle pain was not taken into account in previous studies and, as such, we cannot compare these findings with previous studies. However, these results should be analysed with caution as we only included 10 participants in the control task.
Nevertheless, the data seem to indicate that tooth-clenching does not play a specific role in the development of TTH episodes (4) but can lead to muscle pain (3,30,31). Finally, since isometric contraction was not responsible for the development of headaches, it could be proposed that either stress from laboratory conditions, the visual input from looking at the screen, or a combination of the two were risk factors for the development of the TTH episodes. This is reinforced by the fact that both stress and visual stimuli have been shown to trigger headache in healthy controls and TTH patients (26,28).
Limitations
The major limitation in our study is the small number of participants (n = 10) that were recruited for the control task. Importantly, the control task comparison with the tooth-clenching task was not the main aim of the study. It was merely to show that tooth-clenching was not required for the development of headaches and that changes in TTS occur independently of tooth-clenching. The fact that three out of 10 participants developed headache and significant changes in TTS occurred following the control task confirmed this hypothesis, as well as confirming the results from a previous study (4).
It could be argued that one limitation is that participants did not fill out a headache diary before inclusion, which could have led to including participants with more than one headache day in the last 30 days due to recall bias. We feel that this most likely did not affect the results of the study and in most studies in which healthy participants are recruited to serve as controls for headache patients, they are not required to fill out a headache diary.
Finally, in this study, due to the multiple statistical tests that were done, we adjusted our p-value to avoid type I error, which is the possibility of finding false positives. When doing so, the risk of type II error increases, which is the possibility of finding false negatives. Despite this possibility, the lack of significance of the primary outcome parameters would not have changed as their p-values were bigger than 0.05. Furthermore, because we did not correct for the total number of tests, we feel that we reduced type I error without hugely increasing the risk of type II error (32).
Clinical implications
As stated above, the most common explanation for TTH is that increased PCT leads to TTH episodes and that sensitization of the central nervous system due to continued nociceptive stimuli from pericranial muscles causes conversion of episodic to chronic TTH (33). The results of this study contradict part of this explanation and could explain why many studies that have assessed the efficacy of physical therapy in TTH have not found a positive effect (34,35). As such, clinically, it would be important to define what constitutes increased PCT and ascertain which patients actually have increased PCT. It could then be inferred that TTH patients with increased PCT, most likely due to frequent headache episodes, would be the patients that would benefit from physical therapy.
Conclusion
In summary, following a tooth-clenching task, it was shown that increased PCT is not required to trigger an episode of TTH in healthy participants. Furthermore, PMP was not able to predict who developed headache and who did not. Finally, activation of descending inhibitory pathways, as assessed by decreases in TTS, may be protective against the development of a TTH episode.
Footnotes
Article highlights
Following a tooth-clenching task, increased PCT was not required to trigger an episode of TTH in healthy participants.
Pain modulation profiles could not predict who developed headache and who did not, following the provocation tasks.
Activation of descending inhibitory pathways, as assessed by decreases in PCT, may have been protective against the development of headache.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from “Den Midtjyske Bladfond”.
