Abstract
Objective
We present long-term follow-up results and analysis of stimulation sites of a prospective cohort study of six patients with chronic cluster headaches undergoing deep brain stimulation of the ipsilateral posterior hypothalamic region.
Methods
The primary endpoint was the postoperative change in the composite headache severity score “headache load” after 12 months of chronic stimulation. Secondary endpoints were the changes in headache attack frequency, headache attack duration and headache intensity, quality of life measures at 12, 24, and 48 months following surgery. Stimulating contact positions were analysed and projected onto the steroetactic atlas of Schaltenbrand and Wahren.
Results
There was a significant reduction of headache load of over 93% on average at 12 months postoperatively that persisted over the follow-up period of 48 months (p = 0.0041) and that was accompanied by a significant increase of reported quality of life measures (p = 0.03). Anatomical analysis revealed that individual stimulating electrodes were located in the red nucleus, posterior hypothalamic region, mesencephalic pretectal area and centromedian nucleus of the thalamus.
Conclusions
Our findings confirming long-term effectiveness of deep brain stimulation for chronic cluster headaches suggest that the neuroanatomical substrate of deep brain stimulation-induced headache relief is probably not restricted to the posterior hypothalamic area but encompasses a more widespread area.
Introduction
Cluster headache (CH) is classified as a trigeminal autonomic cephalgia (Headache Subclassification Committee of the International Headache Society, 2004) (1). In 10–15% of patients with CH, attacks can occur on a daily basis with no significant period of remission for over a year, which is then referred to as chronic CH (CCH). When CCH is unresponsive to medication, different neurosurgical procedures such as occipital nerve stimulation (ONS), sphenopalatine ganglion stimulation and deep brain stimulation (DBS) are considered (2–5).
Pioneering work by Leone and co-workers led to a first attempt to treat CCH with deep brain stimulation (DBS) of the posterior hypothalamus in 2001 with long-term pain relief (6). Over the years, the selected target has been slightly modified in several open-label studies by the same as well as other groups and different names have been introduced such as ventral tegmental area stimulation and mamillo-tegmental fasciculus stimulation. Renaming the initially proposed target region contributes to some sort of confusion in the literature but also reflects the emerging controversy about the underlying anatomical structures that mediate the effects of high-frequency stimulation (7,8). So far, over 100 cases of DBS for CCH worldwide have been published, reporting significant headache reduction in about 50–100% of patients depending on the chosen outcome measures and selected target (8–10). Here, we present the results of a prospective series of six patients with therapy-refractory CCH that underwent DBS. Stimulation effects on headache frequency, duration and severity and quality of life measures as well as the stimulation sites are investigated in detail over a median long-term follow-up of more than 90 months. Stimulating electrode position analysis of clinically effective DBS revealed involvement of a widespread area beyond the posterior hypothalamus and ventral tegmental area.
Patients and methods
We selected patients who fulfilled the International Classification of Headaches Disorders-II classification for CCH with highly disabling, medically refractory symptoms for at least 2 years. The selection criteria proposed by Leone et al. in 2004 were applied to all patients (11). All included patients failed to respond, had contraindications against or were intolerant to at least five of the following medications in combination: Verapamil, lithium, methysergide, carbamazepine, oxcarbazepine, topiramate, gabapentin, amitriptyline or sumatriptan. Prior to surgery, all patients were discussed in a multidisciplinary team and underwent extensive preoperative neuropsychological evaluations and an MRI to exclude structural abnormalities. All patients provided written and informed consent to the procedure and data analysis and the study was approved by the local institutional review board.
Surgical procedure
We applied the atlas-target coordinates of the posterior hypothalamus (LAT 2 mm, AP −6 mm and VERT −8 mm with reference to the mid-commissural point) according to Leone et al. (6). The surgical procedure was described in detail before (12). In summary, patients underwent DBS implantation under local anaesthesia without intraoperative microelectrode recording. A preoperative high-resolution CT scan was co-registered to the patient’s preoperative MRI head. The DBS-lead model 3389 (Medtronic, Minneapolis, USA) was implanted in all patients. A second high-resolution CT scan was performed to confirm lead location and rule out intracerebral haemorrhage. Lead tip position and stimulating contact (SC) position were calculated with reference to the midcommissural point (MCP) and a 2D-vector of error was calculated as previously described (13).
Outcome measures and follow-up
Patients were followed up regularly and data recorded prospectively. Stimulation parameter settings, medications, headache attack frequency, headache attack duration, headache severity, quality of life (QoL) measures, health status and adverse effects as well as complications were recorded at each follow-up visit. These measures were collected preoperatively and at 12, 24, and 48 months postoperatively. No systematic follow-up was conducted thereafter but patient outcome data were included in the long-term follow-up where possible.
Patients completed a pain diary over 7 days prior to follow-up-appointments to document pain frequency and durations as well as pain characteristics. Headache severity was measured on the numeric rating scale (NRS) for pain (0 being free of pain, 10 being the worst imaginable pain). Individual scores were averaged over the observation period. We calculated the headache load as a headache severity composite score adapted from Akram et al. as Σ(severity × duration (min)) of all headache attacks occurring over the 7-day period (9). Health-related QoL was assessed based on the Short Form-36 (SF-36) questionnaire including both physical and mental components. Patients were asked to assess their general health-status on a visual analogue scale from 0 to 100 (with 0 being the worst imaginable health status and 100 being healthy without any restrictions).
DBS programming
Open-label programming was initiated usually after 6 weeks following surgery to allow time for any initial stun-effect to settle. All devices were programmed with a frequency of 180 Hz, a pulse width of 90 ms and an amplitude of 2 V. Voltage was adapted according to elicitation of stimulation-induced side effects (usually double-vision, oscillopsia, vertigo). Stimulation parameters were adjusted if patients were not responding adequately to stimulation.
Primary and secondary outcome measures
The primary endpoint was the postoperative change in the composite headache severity score “headache load” after 12 months of chronic stimulation. Secondary endpoints were the changes in headache attack frequency, headache attack duration and headache intensity, QoL according to the SF-36 questionnaire and general health status at 12, 24, 48 and > 60 months following surgery as well as headache load after 24, 48 and > 60 months following surgery.
Statistical analysis
Data was analysed by applying descriptive nonparametric statistics using GraphPad Prism 6.01 (La Jolla, USA). Data are presented as mean ± range if not otherwise indicated. Statistical differences of preoperative and 12-month postoperative headache load scores were tested by applying the Wilcoxon signed-rank test. Differences of pre- and postoperative headache scores, SF-36 scores and health status scores at different postoperative follow-up points was tested by applying a matched value one-way ANOVA of repeated measurements (Friedman test). Adjusted p-values for post-hoc multiple comparisons were applied. A p-value < 0.05 was considered statistically significant.
Results
Patient characteristics before surgery.
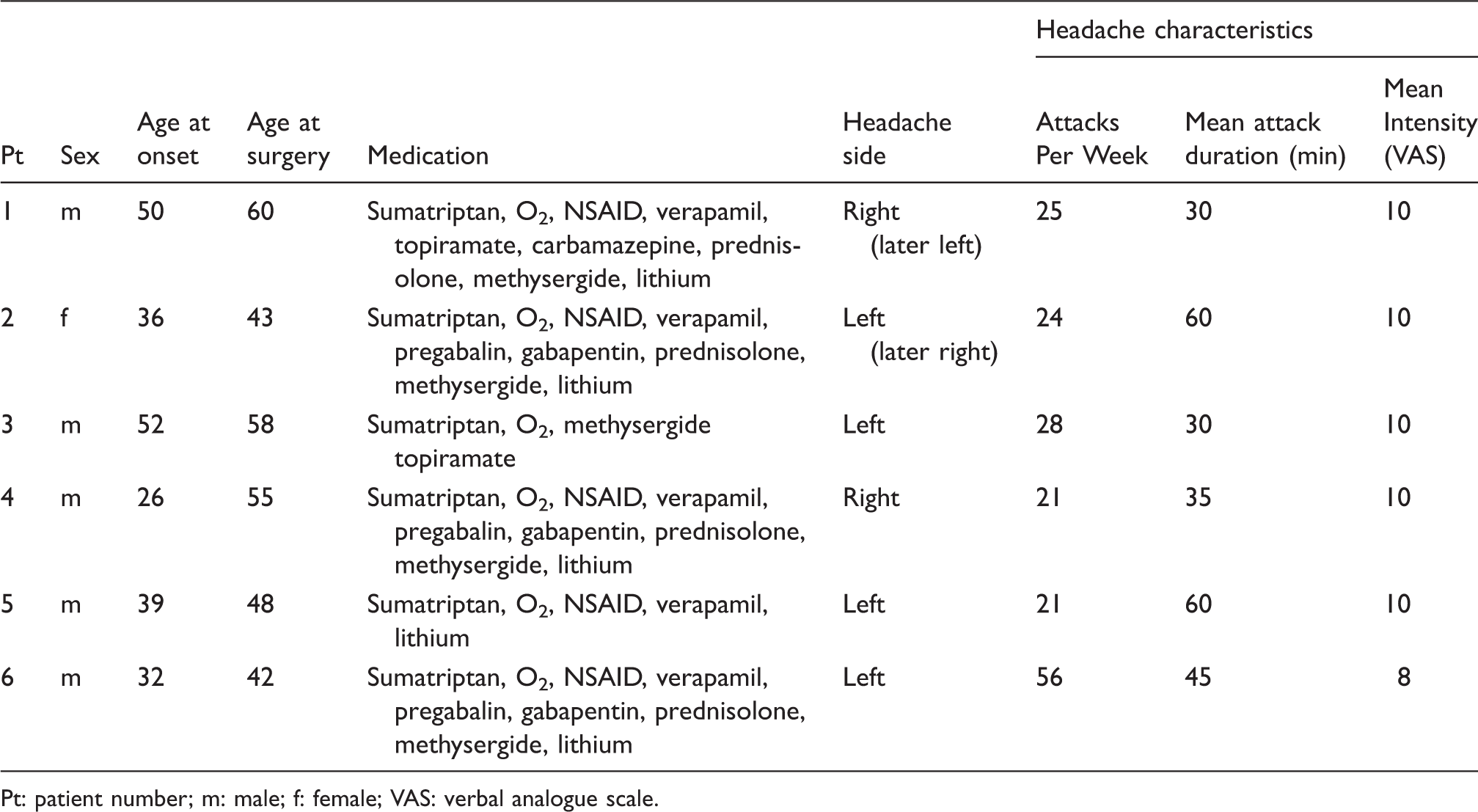
Pt: patient number; m: male; f: female; VAS: verbal analogue scale.
Primary outcome
The preoperative headache load was 10045 (3360–20160). At 12 months, the mean headache load was significantly reduced to 780 (0–4080; p = 0.03). At 12 months, five patients had at least a more than 90% reduction of the headache load compared to before surgery and one patient experienced a 44% reduction of the headache load.
Secondary outcome
Headache load remained significantly reduced at 24 months (1175) and 48 months (118) postoperatively (p = 0.0041). Throughout the follow-up period until 48 months, every patient experienced at least 44% reduction of the headache load (Figure 1). Similar results were found for frequency, duration and intensity for headache attacks that remained significantly reduced postoperatively over the follow-up period of 48 months. Five patients experienced at least 90% and 1 patient 44–98% headache reduction at subsequent follow-up appointments until 48 months. Three out of four patients with a follow-up of over 68 months and two out of three patients with a follow-up over 114 months remained completely pain free, whereas the other one experienced significant long-term pain alleviation (headache load reduction of 97%). Table 2 summarises other headache-related secondary outcome measures.
Headache load (a), headache frequency (b), headache attack duration (c) and headache attack intensity (d) according to the verbal analogue scale (VAS) preoperatively and at 12, 24 and 48 months respectively. Individual lateral (LAT), anterior-posterior (AP) and vertical (VERT) coordinates (in mm) of the stimulating contact position relative to the midcommissural point (MCP), chosen stimulating contact (SC) and 2D-vector of error of the study patients.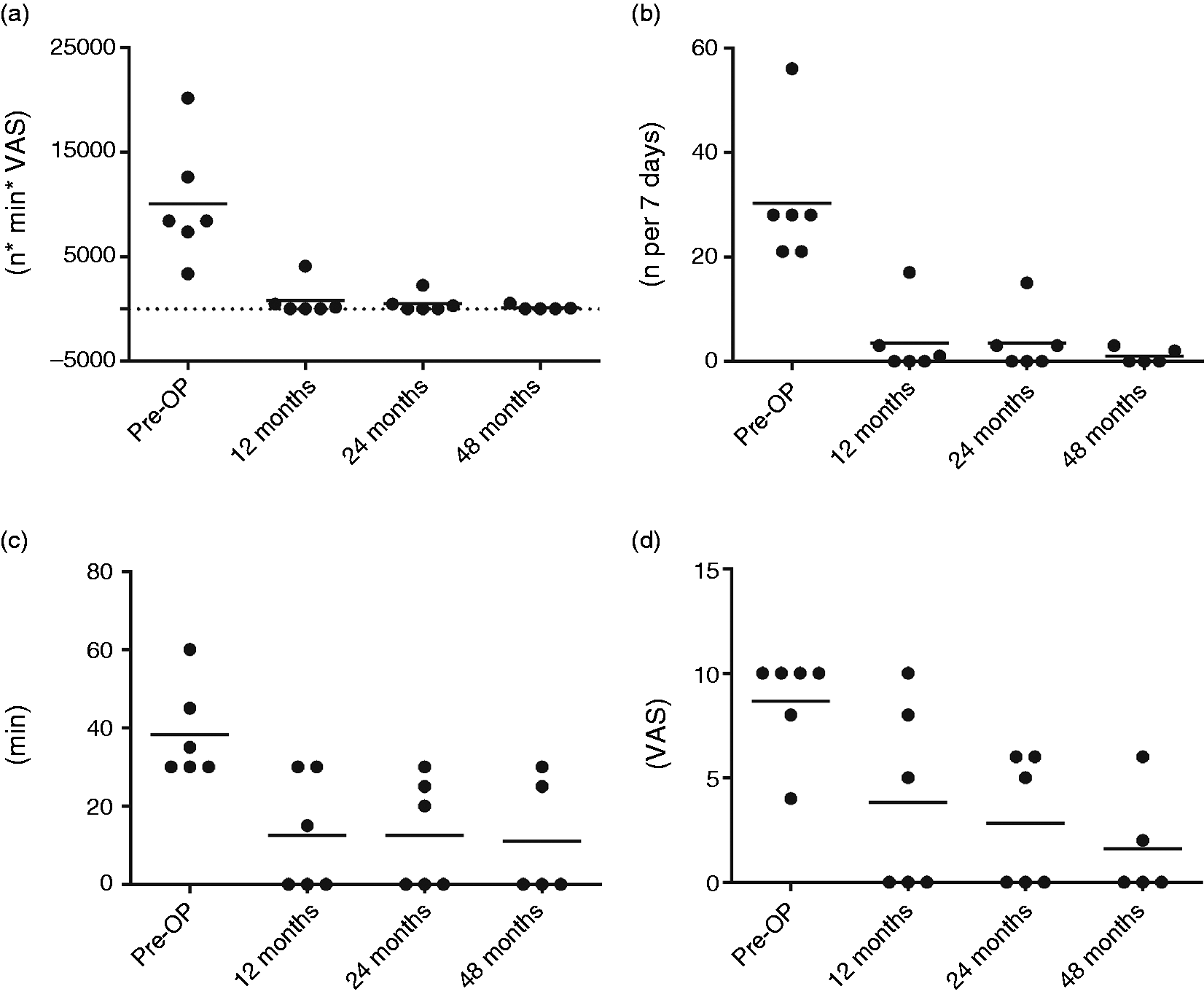
Regarding QoL measurements, the mean preoperative physical component summary (PCS) of the SF-36 was 30.0 (20.0–44.8) and the mean preoperative mental component summary (MCS) was 21.1 (8.2–38.1). At 12 months postoperatively, mean PCS was significantly increased to 47.8 (26.7–60.7; p = 0.03) and mean MCS was significantly increased to 56. 1 (48.4–64.8; p = 0.03). With respect to the different subcomponents of the SF-36, there was a significant increase in Body Pain and Vitality over the follow-up period of 48 months. The other subcomponents of the SF-36 were not significantly increased over this period of time (See supplements).
General health status improved from a 39% (20–70%) prior to surgery to 68% (5–95%), 67.5% (5–95%) and 71% (5–100%) at 12, 24 and 48 months respectively (p = 0.07).
All patients were on preventive medications (verapamil, methysergide, lithium) prior to surgery. Only one patient continued to take preventive medication after 12 months and throughout the follow-up period until the last follow-up appointments. However, he reduced the dose of verapamil and stopped taking gabapentin. Of the remaining five patients who stopped preventive medications after surgery, four did not require any intravenous sumatriptan throughout the follow-up period.
Long-term follow-up and general observations
DBS implantation led to a complete relief of headache attacks in three patients after chronic stimulation and to a significant reduction to an almost headache free state in one patient. In two patients, significant headache relief was achieved following DBS with a reduction of approximately 88–92 % of headache load. Two patients developed CH attacks on the contralateral side after an interval of 24 and 48 months of significant reduction or pain freedom post-surgery. An immediate postoperative headache suppressive effect without active stimulation was noted in six patients (stun effect). The most frequent side effect was double-vision and oscillopsia that was stimulation-amplitude dependent and reversible in all patients. Lowest thresholds to induce double vision were noticed in patients 2, 3 and 4. Patient 2 experienced an irreversible dysarthria after implantation of the second DBS lead on the right side. No effects on autonomic functions (altered blood pressure, heart rate, sweating, bowel or bladder dysfunction) were recorded. However, no systematic investigations (blood pressure monitoring on- and off-stim, post-void bladder scan) were done. A summary of the history of each patient’s postoperative course is provided in the supplementary material.
DBS lead location
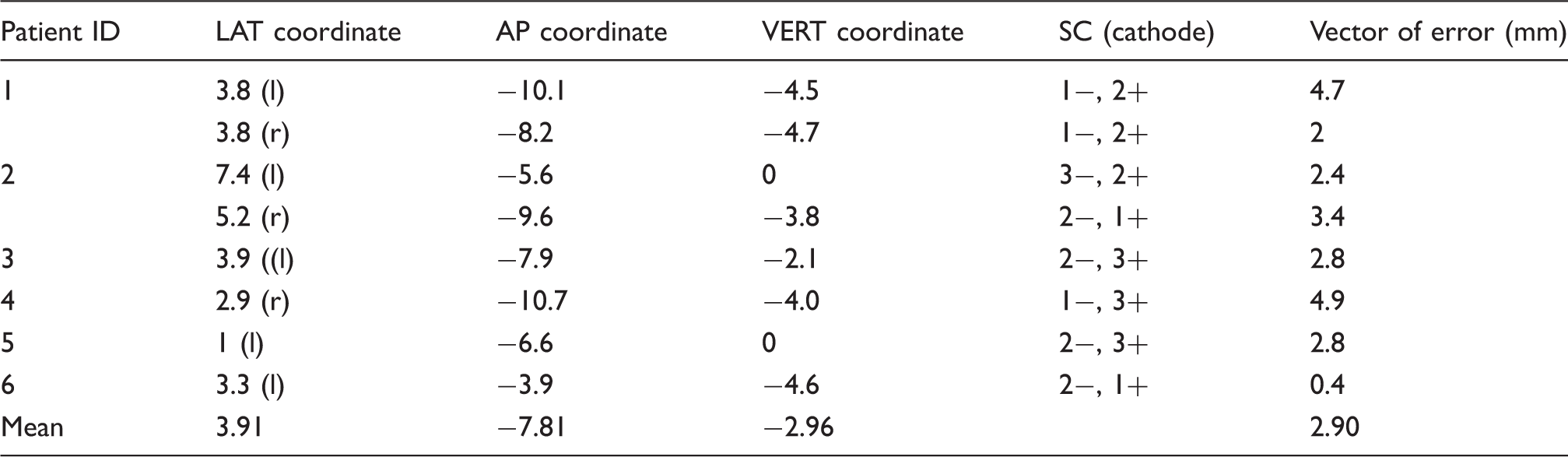
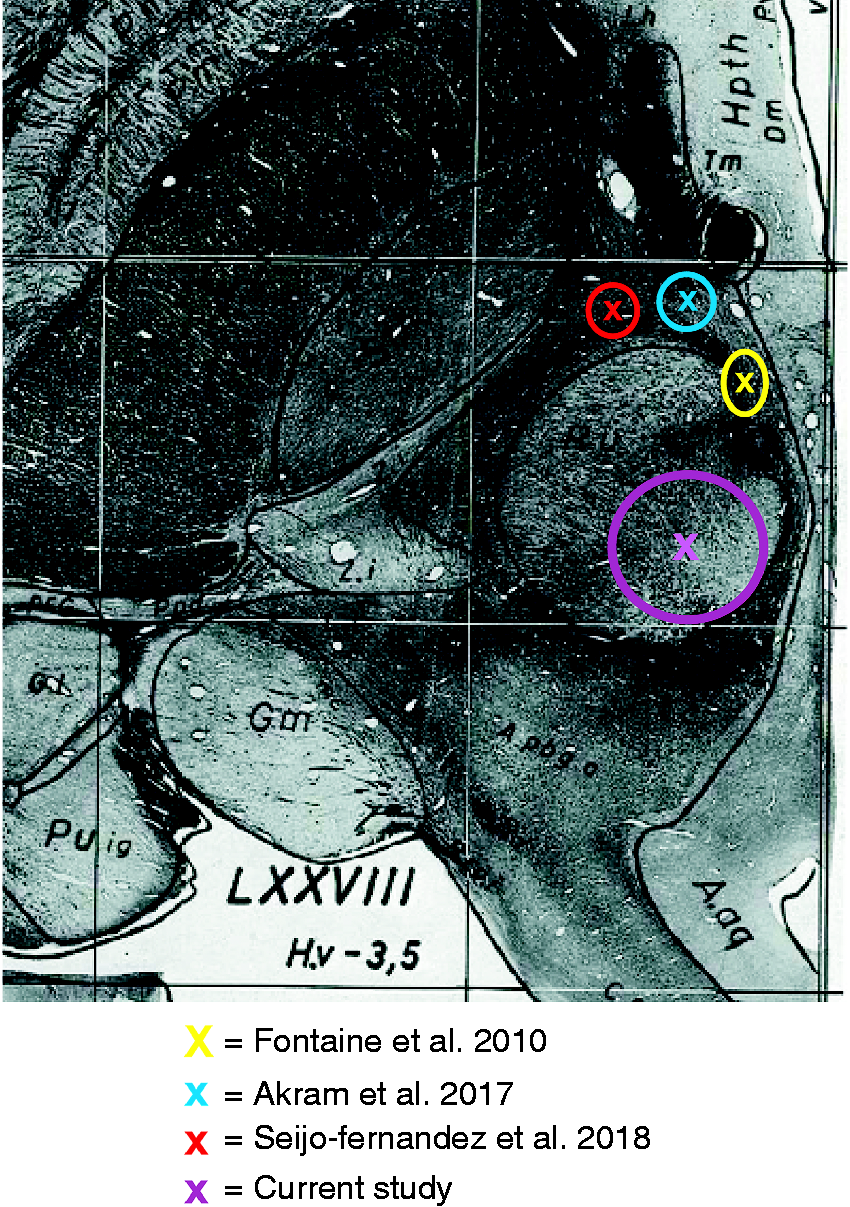
The SC positions of individual patients are shown in Figure 2. Table 2 summarises the individual stimulating contact (SC) positions. The mean coordinates of the SC were LAT 3.0 mm (range 1 mm to 7.4 mm); AP −7.8 mm (range −3.9 to −10.7 mm) and VERT −1.9 mm (range −4.7 to 0 mm). The mean stereotactic coordinates of the SC projected onto the red nucleus of the stereotactic atlas of Schaltenbrandt and Wahren (Figure 3) (14).
Stimulating contact positions are projected onto the patient’s preoperative MRI. Stimulating contact positions were located in the red nucleus (n = 4), anterior to the red nucleus at the junction of the periventricular grey of the posterior hypothalamic and ventral tegmental area (n = 1), posterior to the red nucleus in the pretectal area at the junction of the central tegmental tract and the periaqueductal grey (n = 1), in the posterior hypothalamic region (n = 1) and at the junction of the centromedian and parafascicular nucleus of the thalamus (n = 1). Mean stimulating contact positions projected onto the stereotactic atlas of Schaltenbrandt and Wahren. The x represents the mean position, the diameter of the circle represents the 95% CI in the medial-lateral and anterior-posterior axis.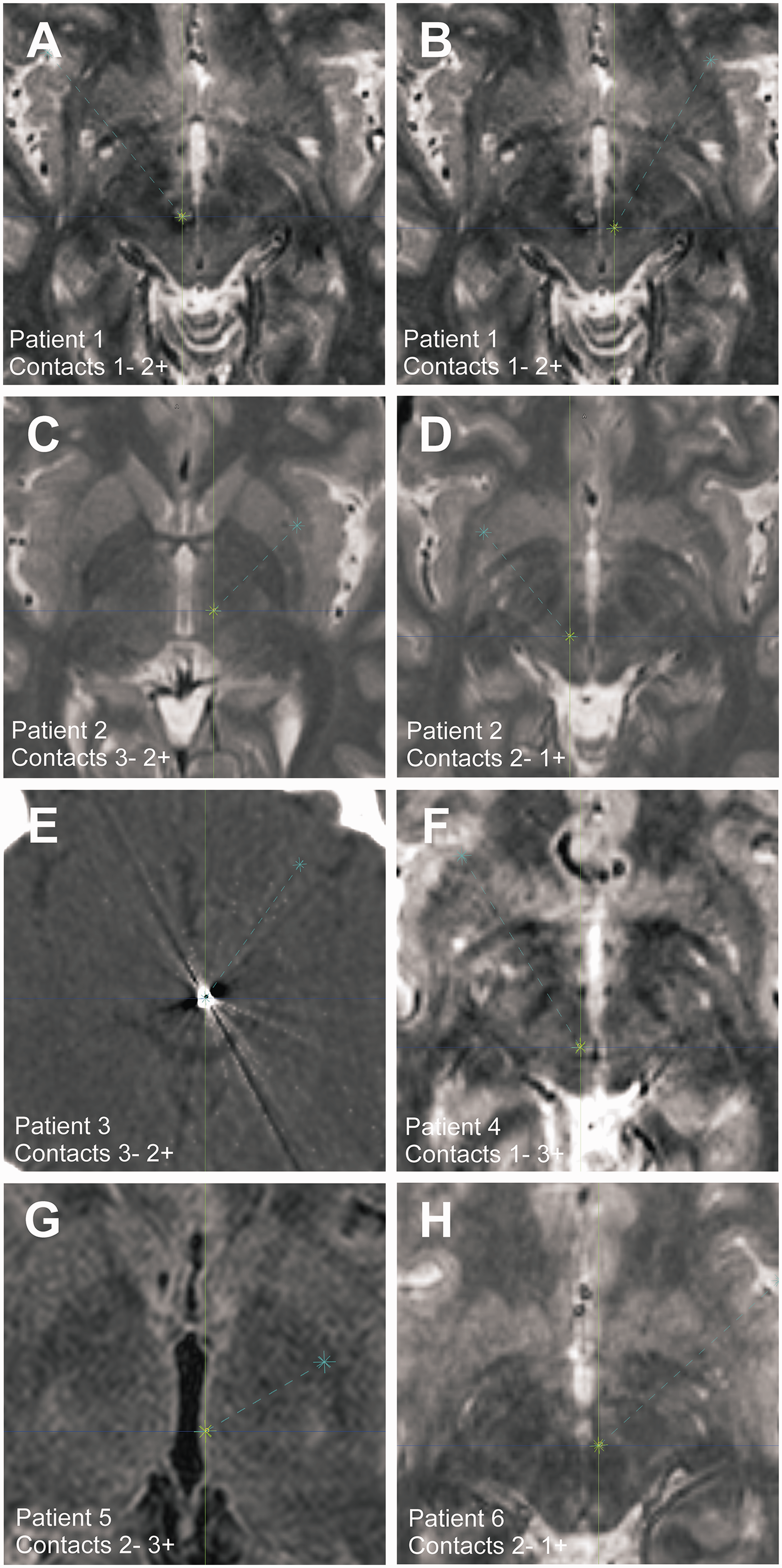
Discussion
We present postoperative results and long-term follow-up data of up to 11 years of a prospective series of six patients undergoing DBS implantation for chronic CH. The primary outcome measure was the postoperative change in the composite headache severity score “headache load” after 12 months following surgery. Headache load was significantly reduced at 12 months in all patients and remained significantly reduced over the follow-up period until 48 months. Five patients experienced at least 90% and one patient 44–98% headache reduction at subsequent follow-up appointments with no poor responders to surgery. Three out of four patients with a follow-up of over 68 months and two out of three patients with a follow-up over 114 months remained completely pain free, whereas the other one experienced significant long-term pain alleviation. Furthermore, patients experienced a significant improvement of QoL measurements following surgery. Although statistically not significant, patients described a marked improvement of their general health status following surgery. Anatomical analysis of the stimulating contact position showed some remarkable findings compared to the existing literature. The mean location of the stimulating contacts projected onto the red nucleus; however, there was a considerable variability of the stimulating contact position in the presented cohort, with individual electrodes located at the border of the red nucleus, posterior hypothalamic region, mesencephalic pretectal area and centromedian thalamus.
Postoperative outcome
Despite a growing number of positive case series and prospective trials, the effectiveness of this treatment is still a matter of debate and controversy. According to the American Headache Society Guideline, DBS for CCH was classified as level B negative evidence (possibly ineffective) (15). This is mainly due to a failed randomized placebo-controlled double-blind trial of 11 patients with CCH (16). In the double-blind randomized phase, symptom reduction of stimulation was compared to sham-stimulation over a period of one month. During the randomized phase, no significant change in primary and secondary outcome measures was observed between active and sham stimulation. At the end of the open phase, however, six out of 11 responded to the chronic stimulation (weekly frequency of attacks decrease of 50%), including three pain-free patients. A limitation of the study is that the randomised phase was set too early and too short. Previous studies found that the stimulation effects on headache attack reduction fully develop only after a few weeks to months while acute stimulation is not effective in resolving acute headache attacks (6,10,17,18). On the other hand, growing evidence from large prospective observational studies suggests effective long-term headache reduction after DBS. For instance, in their series of 17 patients with a median follow-up time of 8.7 years, Leone et al. noted a stable and long-lasting almost pain-free condition in 35% of the patients and a significant postoperative reduction in the frequency and duration of headache attacks in another 35%, while 30% continued to have daily headache attacks and despite reduction of pain intensity were considered non-responders (65% responder rate) (3). In a multi-centre study of six patients, Bartsch et al. reported on long-term headache attack reduction to an almost pain-free extent in three of the patients, while one patient, who initially responded showed a relapse of pain attacks and two did not respond (50% responder rate) (17). In a prospective study of six patients, Schoenen et al., two out of five patients were completely pain-free, one showed a marked reduction in the frequency of pain attacks, while two exhibited a relapse (60% responder rate) (19). Unfortunately, one patient died in this study due to a perioperative complication. More recently, Akram et al. presented data of their prospective open-label uncontrolled trial of 21 patients with DBS for CCH (9). There was a reported headache load reduction of 68%, and 81% of the patients experienced at least a 30% reduction of the headache load during a median follow-up period of 18 months (81% responder rate). Furthermore, Seijo-Fernandez et al. published a series of 15 patients undergoing DBS for CCH with a median follow-up time of 61.3 months (8). They reported long-term complete pain suppression in 33%, and a relevant pain reduction in the remainder of the patients (100% responder rate). Our results are in line with these previous positive findings and underline the clinical effectiveness of DBS for CCH. Despite the difficulty of directly comparing the cited studies due to different outcome measures, follow-up and target selection, there seems to be a clear trend in the literature of a marked long-term reduction after DBS of CCH.
Electrode locations and anatomical structures mediating stimulation effects
In their original work, Leone et al. aimed to target the posterior hypothalamic grey (6). This target was selected based on imaging studies that suggested an involvement of the posterior inferior hypothalamic grey matter during CH attacks (20,21). These findings formed the basis of the hypothesis that overactivity in the posterior inferior hypothalamus constitutes a causal factor of CH attacks that can consequently be suppressed by high-frequency stimulation and led to the pioneering first successful DBS implantation in the posterior hypothalamus in 2001 by Leone et al.6. In a consecutive case series of 17 patients published by the same group, the authors changed their initial target after five implanted patients to a more anterior and superior position (2 mm lateral, 3 mm posterior and 5 mm below the midcommissural point) based on their findings that stimulation via electrodes located closer to the midcommissural point led to better outcome. These target coordinates have been widely used by other groups in consecutive studies (3,16,17,19). However, postoperative stimulating electrode coordinates are rarely reported in the literature. That this would be crucial to draw further conclusions about the actual stimulation site is indicated by our own data. Although we used the original target coordinates proposed by Leone et al. (2 mm lateral, 6 mm posterior and 8 mm below the midcommissural point) our actual position of the stimulating contacts was on average more posterior (−7.8 mm) and superior (−1.9 mm) than intended and showed in general a considerable spread around the average contact position. Analysis of our targeting error showed that three DBS leads had a targeting error of more than 3 mm with a predominantly posterior shift of the actual lead position compared to the intended target. We cannot explain this tendency of the targeting error. On the background of the reported occurrence of third ventricular haemorrhage and the delicate area of DBS implantation in this patient cohort, it was decided not to revise the leads immediately but to wait to see how the patients would respond clinically to the stimulation.
Summary of coordinates (in mm) of the stimulating contact position relative to the midcommissural point (MCP).
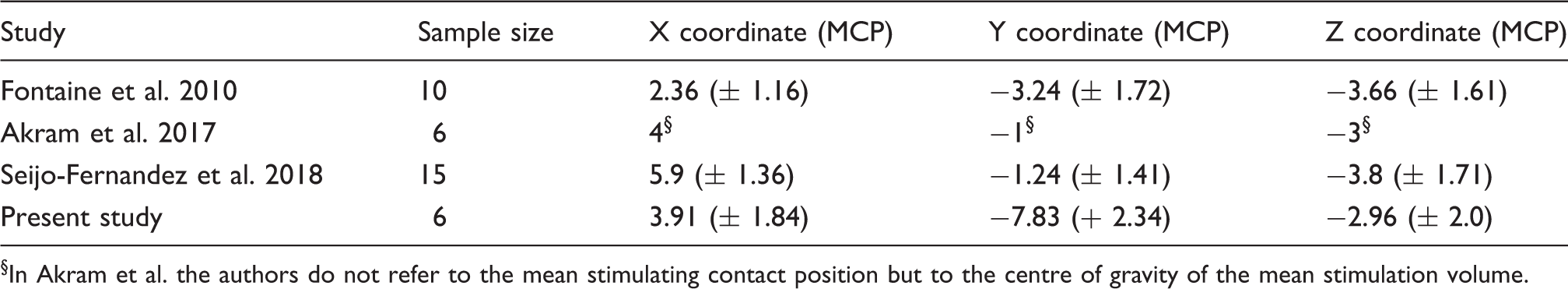
In Akram et al. the authors do not refer to the mean stimulating contact position but to the centre of gravity of the mean stimulation volume.
In summary, our findings are in line with previous studies suggesting that the neuroanatomical substrate of DBS-induced headache relief is probably not restricted to the posterior hypothalamic area but encompasses a more widespread area in the midbrain tegmentum and might even reach the centromedian thalamus. This is also supported by findings of structural connectivity analysis. Our own group could demonstrate widespread connections of the posterior hypothalamus to fronto-orbital cortex, reticular nucleus of the thalamus, superior cerebellar peduncle and cerebellum in a group of healthy control subjects (23). Similar findings were recently demonstrated in CCH patients in Akram et al., who found that the effective stimulated area was connected to the hypothalamus, prefrontal and mesial temporal regions as well as to widespread brainstem areas involving the parabrachial nucleus, nucleus of the solitary tract, periaqueductal grey and the region of the trigeminal nucleus (7). DTI analysis by Seijo-Fernandez et al. with effective DBS for CCH came to very similar conclusions that the stimulated area shows streamlines to the posterior mesencephalic tegmentum (8). In this respect, our findings of stimulating contact positions match the hypothesis that clinically effective DBS for CCH modulates a more widespread network of brainstem nuclei including the trigeminal nuclei, the periaqueductal grey matter, the hypothalamus, thalamus and cortical areas. Whether stimulation of the red nucleus itself plays a role in symptom reduction of DBS for CCH remains an interesting hypothesis that warrants more detailed research in the future.
Limitations
Our study has several limitations. First, the series is small, and our findings may not apply to a broader population. Assessment of the primary outcome was based on the headache load determined on the basis of a one-week pain diary and therefore does not reflect the headache reduction during the course of the whole year. We projected our mean electrode positions as well as those of other studies onto a stereotactic atlas and no systematic modelling of stimulation volumes in the normalised space was performed. However, this is far beyond the scope of our study and, so far, there are no detailed 3D atlases of the brain stem available for MNI space, at least to our knowledge. Furthermore, no connectivity analysis of our stimulation sites was performed that would have supported or disproved our hypothesis of a shared widespread network responsible for the stimulation effects. Future work needs to focus on more detailed anatomical analysis of electrode positions and their corresponding clinical effects and side effects to better define the neuroanatomical substrate of DBS-induced CCH alleviation, which can serve as a basis for bigger, multicentre, randomised controlled trials analysing the effects of DBS on CCH.
Supplemental Material
Supplemental material for Deep brain stimulation of chronic cluster headaches: Posterior hypothalamus, ventral tegmentum and beyond
Supplemental Material for Deep brain stimulation of chronic cluster headaches: Posterior hypothalamus, ventral tegmentum and beyond by Andreas Nowacki, Liz Moir, Sarah LF Owen, James J Fitzgerald, Alexander L Green and Tipu Z Aziz in Cephalalgia
Footnotes
Clinical implications
DBS for chronic cluster headaches leads to a significant reduction of headache load of up to 140 months of follow-up.
The neuroanatomical substrate of DBS-induced headache relief may not be restricted to the posterior hypothalamus and ventral tegmentum.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics or Institutional Review Board approval
This study was approved by the Oxford Research Ethics Committee (B: 11/SC/0229).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
