Abstract
Introduction
Familial hemiplegic migraine 2 is a pathology linked to mutation of the ATP1A2 gene producing loss of function of the α2 Na+/K+-ATPase (NKA). W887R/+ knock-in (KI) mice are used to model the familial hemiplegic migraine 2 condition and are characterized by 50% reduced NKA expression in the brain and reduced rate of K+ and glutamate clearance by astrocytes. These alterations might, in turn, produce synaptic changes in synaptic transmission and plasticity.
Memory and learning deficits observed in familial hemiplegic migraine patients could be ascribed to a possible alteration of hippocampal neuronal plasticity and measuring possible changes of long-term potentiation in familial hemiplegic migraine 2 KI mice might provide insights to strengthen this link.
Results
Here we have investigated synaptic plasticity in distinct hippocampal regions in familial hemiplegic migraine 2 KI mice. We show that the dentate gyrus long-term potentiation of familial hemiplegic migraine 2 mice is abnormally increased in comparison with control animals. Conversely, in the CA1 area, KI and WT mice express long-term potentiation of similar amplitude.
Conclusions
The familial hemiplegic migraine 2 KI mice show region-dependent hippocampal plasticity abnormality, which might underlie some of the memory deficits observed in familial migraine.
Keywords
Introduction
Familial hemiplegic migraines (FHMs) are monogenic subtypes of migraines with aura characterized by transient hemiplegia during the aura phase. FHMs are considered channelopathies (1) caused by genetically heterogeneous autosomal dominant mutations, which are classified in three types. The type 1 (FHM1) is caused by missense mutations in the gene CACNA1A, encoding the pore-forming α1 subunit of the human voltage-gated CaV2.1 (P/Q-type) calcium channel; the type 2 (FHM2) is characterized by mutations in the ATP1A2 gene, which encodes the catalytic α2 subunit of the Na+/K+ adenosine triphosphatase (ATPase) (NKA) whereas mutations in the SCNA1A gene, encoding the α1 subunit of the voltage-gated NaV1.1 sodium channel, are responsible for the type 3 (FHM3) (1–3). Memory and cognitive impairment have been described in many families affected by the FHM pathology (2,4).
LTP represents the major experimental model to study the synaptic changes underlying cognitive processes and memory. The alteration of hippocampal plasticity could be involved in memory deficits and altered cognitive processes found in FHM patients, as well as underlying the cognitive changes present in a mouse model of FHM1 (5). Additionally, this FHM1 experimental mouse model shows enhanced glutamatergic neurotransmission, which is causally linked to enhanced susceptibility to cortical spreading depression (6).
The heterozygous W887R/+ FHM2 knock-in (KI) mice (7) are characterized by a loss-of-function mutation in the gene encoding the α2 subunit of NKA (3), which, in the adult brain, is almost exclusively expressed in glial cells. Expression of α2 NKA is reduced to half in this FHM2 animal model (7,8). Recent findings demonstrate the relevant functional link between the glutamate transporter (GLT1) and the α2 NKA pump in the proximity of glutamatergic synapses. Interestingly, the mutation in the ATP1A2 gene causes a reduction of GLT1 in perisynaptic astrocytic processes that mirrors the reduction of α2 NKA, and it reduces the rate of glutamate and K+ clearance during neuronal activity in the FHM2 model (8). In the present study, we have investigated whether the mouse model of FHM2 can express abnormalities of hippocampal synaptic plasticity recorded from distinct subregions of this structure.
Methods
Electrophysiology
Electrophysiological studies were performed in accordance with the European Community Council Directive of September 2010 (2010/63/EU) and the Institutional Animal Care and Use Committee of the University of Perugia. The heterozygous W887R/+ FHM2 KI (n = 6) and wild-type (WT) (n = 4) male mice (2 months old) were decapitated and the brain rapidly removed and immersed for 2 min in ice-cold artificial cerebrospinal fluid (ACSF) containing (in mM): 126 NaCl, 2.5 KCl, 1.2 MgCl2, 1.2 NaH2PO4, 2.4 CaCl2, 10 glucose, and 25 NaHCO3, continuously bubbled with 95% O2 and 5% CO2, pH = 7.4. Hippocampal horizontal slices were cut (400 µm thick), placed to recover in oxygenated ACSF at 30℃ for 30 min and then at room temperature for another 1 hour before experimental recordings. Horizontal slices were preferred, to maximize the number of slices obtained from each animal. One slice was then transferred into the recording chamber and submerged in ACSF at a constant rate of 2.5 mL/min at 30℃. Before running the experiment, we recovered the hippocampal slices in the recording chamber for an additional 30 min, to let them adapt in the new environment in terms of temperature, O2 levels and mechanical stress. For extracellular experiments, the stimulating electrode was inserted into the Schaffer collaterals or in the perforant path fibers and the recording electrode, made of borosilicate glass capillaries filled with 2 M NaCl (resistance 10–15 MΩ), was placed into the stratum radiatum of the Cornu Ammonis 1 (CA1) or in the Dentate Gyrus (DG) close to the granular layer, respectively. Stimuli of 0.1 Hz, 10 μs duration, and 20–30 V amplitude evoked field excitatory post-synaptic potentials (fEPSPs) that in the CA1 were 50–70% of maximum slope and in the DG included a population spike (PS) that was 50% of maximum amplitude. The PS amplitude was defined as the average of the amplitude from the peak of the early positivity to the peak negativity and of the amplitude from the peak negativity to peak late positivity. While for analyzing the DG PS the amplitude is the typical parameter considered, for the CA1 fEPSP the initial slope of the EPSP waveform is more often measured to reduce contamination from other sources of current flow in the slice. Traces were filtered at 3 KHz, digitized at 10 KHz, and stored on a personal computer (PC). An Axoclamp 2B amplifier (Molecular Devices, San Jose, CA, USA) was used for extracellular recordings. The input-output (I-O) curves were constructed by plotting the stimulus intensity against the mean of slope of fEPSPs and the amplitude of PS respectively for the CA1 and DG regions. Paired-pulse of hippocampal synaptic transmission was obtained by pairing electric stimuli at increasing interstimulus intervals (50–300 ms) during extracellular recordings. Paired-pulse ratios were calculated by dividing values of the second post-synaptic response by the first one. After recording a stable baseline for 10 min, LTP was induced by a high frequency stimulation (HFS) at 100 Hz (1 s) consisting of one train of stimuli for the CA1 area and three trains (5 min intervals) for the DG area. The initial slope (for the CA1) and the PS amplitude (for DG) of the extracellular fEPSPs were used to assess alterations in the synaptic strength. Data analysis was performed off line using Clampfit 10 (Molecular Devices, San Jose, CA, USA). Values given in the text and figures are mean ± standard error (s.e). Two-way ANOVA or Student’s t-test was used for statistical analysis. The significance level was established at *p < 0.05
Results
Long term potentiation of synaptic transmission as well as input-output and paired-pulse responses in the hippocampal CA1 region are similar in FHM2 KI and wild-type mice
To test the hypothesis of possible alteration of hippocampal plasticity, we performed extracellular recordings by stimulating Shaffer Collaterals and evoking characteristic CA1 field potential (fEPSP) in KI FHM2 and wild type (WT) mice. CA1 population excitability was assessed by measuring extracellular fEPSP slope (mV/ms) responses at increasing stimulus intensities (increased stimulation 2 V for each step). The input-output curves obtained were not significantly different between groups (FHM2 mice, n = 7 slices (sl); WT, n = 8 sl; p = 0.9, F(15,215) = 0.0824) (Figure 1(a)). In addition, the paired-pulse ratio responses, at different intervals, were similar between FHM2 (n = 8 sl) and WT mice (n = 8 sl) (Figure 1(b), (c)). After 10 min stable control baseline, high frequency stimulation (HFS; one 100 Hz train, for 1 s) produced a stable LTP, which (at 50 min post-HFS) was similar in FHM2 (n = 8 sl) and WT mice (n = 7 sl, p = 0.76; F(30,390) = 0.79) (Figure 1(d), (e)).
LTP is induced in CA1 hippocampal area of FHM2 mice. Graphs show the input-output curves (a) and paired pulse ratios (b) of KI FHM2 (ATPa2R887/+) and wild type (WT) groups of mice plotted together. Representative traces of paired pulse facilitation recorded in CA1 slices (c) of KI FHM2 and WT mice. Time-course of the LTP (d) recorded from CA1 fEPSP appears unaltered between the groups after the high frequency stimulation (HFS) protocol. (e) Representative traces before HFS protocol and 50 min after tetanic stimulation recorded from hippocampal area.
Long-term potentiation at the perforant path synapses in the dentate gyrus is increased in FHM2 KI mice, while input-output and paired-pulse responses are similar in FHM2 and wild-type animals
An enriched expression of GLT1 in the DG (9,10) has been previously reported. Thus, we sought to perform a deeper electrophysiological analysis in this hippocampal region, stimulating perforant pathway fibers and recording PS in DG. The analysis of input-output curves did not show any significant changes between FHM2 (n = 7 sl) and WT mice (n = 8 sl) (p = 0.99; F(15,182) = 0.208) (Figure 2(a)). Moreover, no significant change has been found in the paired-pulse experiments for all the intervals used (FHM2, n = 8 and WT, n = 10) (Figure 2(b)). The HFS protocol (three 100 Hz trains, 5 min interval) induced in the DG of FHM2 mice (n = 6 sl) a significantly increased LTP in comparison to WT (n = 8 sl). In fact, 50 min after HFS application the PS measured in FHM2 mice were increased (435.6 ± 39.83) compared to WT (290 ± 29.65) (interaction between groups p < 0.0001; F(21,168) = 3.34) (Figure 2(d), (e)).
LTP is induced in the DG hippocampal area of FHM2 mice. The input-output curves (a) and paired pulse ratios (b) of KI FHM2 (ATPa2R887/+) and (WT) groups of mice appear unaltered between the groups and are plotted together. Representative traces of paired pulse facilitation, recorded in DG, (c) for of KI FHM2 and WT mice. (d) Time course of PS amplitude recorded from slices in the KI FHM2 and WT groups of mice showing an increase of long-term potentiation (LTP) in DG after delivery of the high frequency stimulation (HFS) protocol (***p < 0.001). (e) PS representative traces before HFS protocol and 50 min after tetanic stimulation recorded from the DG hippocampal area.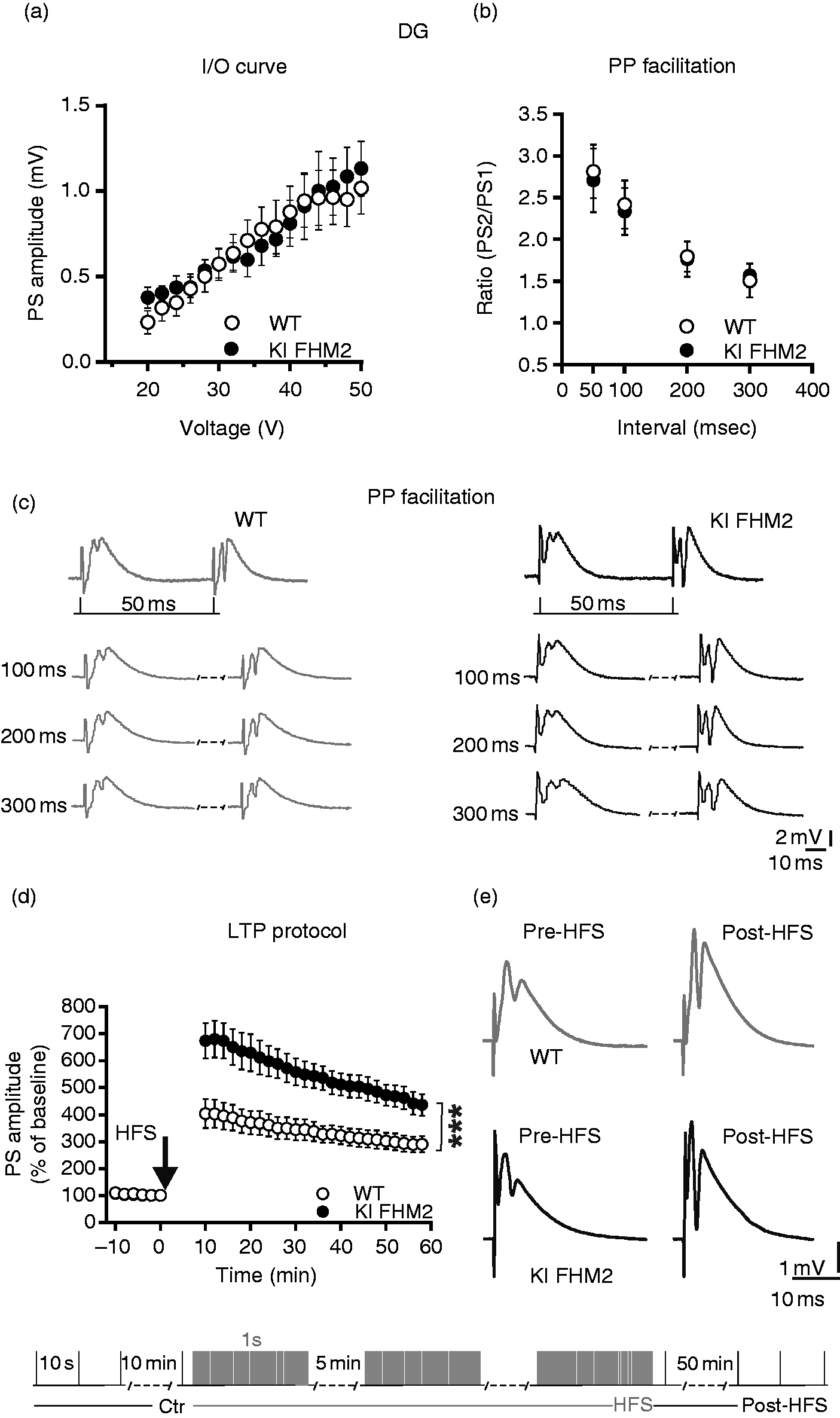
Discussion
The main finding of the present study is that the LTP induced in the DG of FHM2 mice is abnormally increased in comparison with control animals. Conversely, in the CA1 area KI and WT mice express this form of synaptic plasticity of similar amplitude. Abnormal increase of LTP might be associated with reduced cognitive functions; in fact, it could even lead to a detrimental effect on mental performance (5). FHM2 is characterized by a mutation in the ATP1A2 gene, which causes a significant reduction of NKA expression in the brain (8). In FHM2-KI mice, glutamate clearance by astrocytes during neuronal activity is slower and the density of GLT-1a in the membrane of astrocytic processes surrounding cortical excitatory synapses is greatly reduced in the FHM2 mutants, mirroring the reduced expression of the α2 NKA protein (8). Interestingly, the relative impairment of glutamate clearance in FHM2-KI mice is activity dependent. In fact, the impairment of glutamate clearance was larger after stimulation of glutamate release with a train of pulses at high frequency than after stimulation with single pulses or a train of pulses at low frequency (8). This is consistent with our findings that in FHM2 KI mice LTP induced by HFS (100 Hz) is increased while field potentials evoked by single pulses (0.1 Hz) are unaltered.
GLT1, responsible for glutamate clearance, is not expressed ubiquitously. In fact, in the hippocampus, GLT-l is abundantly expressed in DG and CA3 regions whereas the expression in the CAl area is lower (10). It will be interesting to explore in future experiments the detailed region specificity of LTP changes in the hippocampus, including that at the mossy fiber-CA3 synapse. It has been previously reported that LTP was augmented in mice expressing the FHM1 mutation compared to WT in the CA1 hippocampal area. Interestingly, this synaptic alteration was associated with significant spatial memory deficits in contextual fear-conditioning and Morris water maze tests (5), showing that abnormal increase of LTP may be associated with reduced cognitive functions.
Our experiments did not reveal abnormalities of LTP recorded in the CA1 area of FHM2 mice. It will be interesting to establish with future behavioural experiments whether the plastic changes in a distinct hippocampal region induced by the FHM2 mutation also lead to cognitive alterations. This would support the hypothesis that region-dependent hippocampal plastic changes may underlie some of the memory deficits observed in familial migraine.
In conclusion, changes in α2 NKA activity, as arising from FHM2 mutations, might play a role in memory deficits observed in familial migraine, via glutamatergic dysregulation, pathologically increased long-term synaptic transmission, and deficits in energy metabolism.
Key findings
LTP in the dentate gyrus of FHM2 knock-in mice is abnormally increased relative to that of WT mice. LTP in the CA1 region is similar in WT and FHM2 mice. Input-output curves and paired-pulse ratio responses in both hippocampal CA1 and DG regions are similar in FHM2 and WT mice.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ministry of Education, Universities and Research (MIUR) – Progetto di Ricerca di Interesse Nazionale (PRIN) 2015 (prot. 2015FNWP34) (to PC), Italian Ministry of Health – Ricerca Finalizzata RF-2013-02356215 (to PC), and from Telethon Italy Grant GGP14234 (to DP). This work was partially supported by funding of the Italian Ministry of Health [ricerca corrente].
