Abstract
Background
There is limited research on the association of sleep problems with International Classification of Headache Disorders (ICHD-II)-defined headache subtypes in youth, particularly from community-based samples. This cross-sectional study examines the associations of sleep patterns, symptoms and disorders with specific headache subtypes among adolescents from the general population of the United States.
Methods
The sample includes 10,123 adolescents in the National Comorbidity Survey – Adolescent Supplement, a face-to-face survey of adolescents aged 13–18 years in the continental USA. Headache subtype diagnoses were based on modified ICHD-III criteria, and mood and anxiety disorders were based on Diagnostic and Statistical Manual of Mental Disorders criteria. The associations of self-reported sleep patterns and insomnia symptoms and headache status were estimated with multivariate regression models that adjust for demographic characteristics and comorbid anxiety and mood disorders.
Results
There was no significant difference in bedtime between youth with and without headache. However, adolescents with migraine headache reported significantly shorter sleep duration (p = .022) and earlier wakeup time (p = .002) than those without headache. Youth with any headache, particularly migraine, had significantly more sleep disturbances than those without headache. With respect to headache subtypes, youth with migraine with aura were more likely to report difficulty maintaining sleep, early morning awakening, daytime fatigue, and persistent insomnia symptoms than those with migraine without aura. However, these associations were largely accounted for by comorbid anxiety and mood disorders. There was a monotonic increase in the number of insomnia symptoms with increasingly restrictive definitions of migraine.
Conclusion
Pervasive sleep disturbances in adolescents with headache have important implications for the evaluation, treatment and etiology of pediatric headache.
Introduction
Headaches and sleep problems are common among children and adolescents (1–9) and are associated with significant functional impairment (5,10,11). These problems have been shown to co-occur, but the mechanism underlying their comorbidity has not been established. Whereas several studies have shown that acute sleep disturbances may trigger acute headaches in both adults (12) and in children (13–15), other research has shown that sleep problems may be a complication of headache. Moreover, sleep has been shown to alleviate acute symptoms of migraine attacks (16).
Studies of clinical samples of youth have found significantly more abnormal sleep behaviors and symptoms including co-sleeping, day naps, problems initiating and maintaining sleep, daytime sleepiness, and poor sleep hygiene than controls without headache (17–23). Headache has also been associated with specific sleep disorders, including insomnia (19), disorders of circadian rhythm (14) and disordered breathing (24,25). For example, one study revealed that more than half of children and adolescents with headache assessed with polysomnography had sleep-disordered breathing. However, other studies that employed objective measures of sleep such as actigraphy found no group differences in sleep patterns of 8–12-year olds with and without headaches (26). These discrepancies may be due to heterogeneous headache groups, differences in assessment methods, and/or sample characteristics (i.e. age, demographics).
To date, only two studies have examined sleep disorders in non-clinical samples of youth (14,27). In both studies, youth with headache had more sleep-related problems including increased daytime sleepiness, decreased sleep quality, and increased sleep disturbances, than their peers without headache. Moreover, even though the study by Bruni and colleagues found no difference in sleep schedule or sleep duration between children with and without headache, they reported that insufficient sleep was the most frequently reported trigger for headache. Therefore, while the association between headache and sleep has been well established, the exact nature of this relationship has not been fully elucidated.
Potential explanations for a link between sleep symptoms/disorders with headache could include common biological factors (i.e. neurophysiologic and neuroanatomic substrates) (3,28), shared behavioral or sociodemographic characteristics, or common risk factors for sleep and headache such as comorbid mood and anxiety disorders that have been consistently shown to be associated with both headache and sleep disturbances (29–31). For example, our previous research on community samples has shown strong associations between short sleep duration and later weekday bedtime with mood, anxiety, substance use and other behavior disorders (32), and between migraine with anxiety and mood disorders in children and adolescents (33).
The goal of this study was to examine the association between headaches in general and specific headache subtypes including migraine with and without aura in the National Comorbidity Survey – Adolescent Supplement (NCS-A), a nationally representative sample of US adolescents. The specific aims are: (a) to describe the prevalence of headache subtypes among sleep patterns in US adolescents by demographic characteristics and status of past year anxiety and mood disorders; (b) to examine associations of sleep patterns and insomnia symptoms among youth with ICHD-III-defined headache subtypes; and (c) to evaluate whether sleep and headache patterns are associated with the comorbid mood and anxiety disorders.
Methods
The NCS-A is a nationally representative face-to-face survey of 10,123 adolescents aged 13–18 years in the continental USA. The survey was conducted between February 2001 and January 2004 in a dual-frame sample that included subsamples from households (n = 924) and schools (n = 9224) with a combined response rate of 75.6%. Twenty-five non-student residents were excluded from the analysis. Adolescents were interviewed by trained lay interviewers affiliated with the Survey Research Center at the University of Michigan. Information concerning the sampling strategy, participation rates and questionnaires in the NCS-A can be found in detail elsewhere (34,35). The data were weighted to adjust for the differential probability of selection of adolescents within the school and household samples, for differential nonresponse, and for residual differences between sample and US population on the cross-classification of sociodemographic characteristics. All recruitment and consent procedures were approved by the human investigation committees of Harvard Medical School and the University of Michigan.
Measures
Headache (HA)
ICHD-III criteria were used to define increasingly restrictive headache status: No headache (No HA), non-migraine headache (Other HA), migraine headache with or without aura (MIG), and the two subtypes of migraine without aura (MO), and migraine with aura (MA). Any headache (All HA) included migraine (MIG) and non-migraine headache (Other HA). The entry criterion for headache was endorsement of the question in the NCS-A Chronic Condition section “Did you have frequent or very bad headaches or receive any treatment for it at any time during the past 12 months?”. The interview included all of the criteria for migraine with the exception of (a) a minimum of five lifetime attacks or (b) aggravation by, or causing avoidance of, routine physical activity (36). Migraine with aura was defined by the presence of spots, lines, or heat waves and a partial loss of vision before the headache. The interview did not include all possible criteria for aura. However, it has been shown that visual manifestations represent 99% of aura symptoms, whereas sensory and aphasic features are less common.
Sleep duration and bedtime
Total sleep duration in hours and bedtime on weekdays were self-reported. Wakeup time was calculated using reported bedtime and adding sleep duration.
Insomnia symptoms
Four insomnia symptoms during the past 12 months and duration in weeks were assessed in the NCS-A interview. Adolescents were asked whether they had a period lasting for two weeks or longer in the past 12 months when they had any of following sleep disturbances: a) Difficulty initiating sleep (‘‘…problems getting to sleep, when nearly every night it took you two hours or longer before you could fall asleep?’’); b) difficulty maintaining sleep (‘‘…problems staying asleep, when you woke up nearly every night and took an hour or more to get back to sleep?’’); c) early morning awakening (‘‘…problems waking too early, when you woke up nearly every morning at least two hours earlier than you wanted to?’’); and d) daytime sleepiness (‘‘…problems feeling sleepy during the day?’’). We employed four definitions of sleep problems that included: a) insomnia symptoms: Presence of any one of the four insomnia symptoms cited above; b) daytime fatigue: Presence of nonrestorative sleep plus one other insomnia symptom; c) persistent insomnia: Insomnia symptoms that lasted 12 weeks or longer; d) number of insomnia symptoms: Sum of 1–4 insomnia symptoms. However, this interview, which focused on insomnia symptoms, did not include sufficient information to make a diagnosis according to the full criteria of insomnia in the Diagnostic Statistical Manual, Fifth Edition (DSM-5).
Anxiety and mood disorders
Adolescents were administered a modified World Health Organization Composite International Diagnostic Interview (CIDI Version 3.0), a fully-structured, clinically validated instrument for use by trained lay interviewers. Diagnoses of any 12-month DSM-IV anxiety (agoraphobia, generalized anxiety disorder, panic disorder, specific phobia, social phobia, post-traumatic stress disorder, separation anxiety disorder) and any mood disorders (major depressive disorder, dysthymia, bipolar disorder I/II) were based on adolescent reports using the DSM-IV criteria.
Sociodemographic characteristics
Information on sex, age (13–14, 15–16, 17–18 years), race/ethnicity (Hispanic, non-Hispanic black, non-Hispanic white, other), and parental education (less than high school, high school graduate, some college, and college graduate or higher) was collected during adolescent interviews. Family income was normed against federal poverty level using poverty index ratio (PIR ≤ 1.5 poor, ≤ 3.0, ≤ 6.0, >6.0).
Data analysis
Descriptive statistics were used to summarize 12-month headache status by demographic characteristics and 12-month anxiety and mood disorder. Least square mean sleep duration, bedtime, and wakeup time during weekdays were estimated by headache status after adjusting for demographic characteristics and 12-month anxiety and mood disorder. Prevalence of insomnia symptoms by headache status was estimated. Logistic regression models were used to estimate the associations between headache subtypes and insomnia symptoms. Age and other demographic variables (sex, race/ethnicity, and family income) that were significantly associated with headache status in bivariate analyses (p < .10) were used as potential confounding factors in partially adjusted models. Past-year mood disorder and anxiety disorder were subsequently controlled in the fully adjusted models because of the overlap of these conditions. Ad hoc headache group comparisons were made between migraine with aura versus migraine without aura, migraine with or without aura versus no headache, and all headache groups versus no headache. Logistic regression coefficients were transformed to odds ratios for ease of interpretation. Two adolescents with missing headache information were included in the analysis and treated as no headache. Adolescents with missing data on sleep duration (n = 172, 1.3%) and/or bedtime (n = 252, 2.3%) were excluded from the analyses. Compared to the adolescents with complete sleep information, adolescents with missing data were older (mean = 15.5 (SE = .11) vs. mean = 15.2 (SE = 0.06), p < .01) but no significant differences in distributions of sex, race/ethnicity, family income, mood and anxiety disorder (all p-values < .05).
All of these analyses corrected for the complex sampling design and were weighted to adjust for differential probabilities of selection, non-response, and post stratification. Variance and 95% confidence interval estimates and Wald Chi-square tests were performed using the Taylor series method implemented in the SUDAAN version 11 (Research Triangle Institute, Research Triangle Park, NC). Statistical significance was based on two-sided design-based tests evaluated at the .05 level of significance.
Results
Twelve-month headache status by sample characteristics, NCS-A (n = 10,123).

MA: migraine with aura; MO: migraine without aura; Other HA: non-migraine headache; No HA: no headache; % (SE): weighted prevalence and standard errors; PIR: poverty index ratio.
Sleep patterns during weekdays associated with 12-month headache status, NCS-A n = 10,123.

MA: migraine with aura; MO: migraine without aura; MIG: migraine with/without aura; Other HA: non-migraine headache; HA: headache; M (95% CI): adjusted means and 95% confidence interval based on generalized linear regression models using exposure as 12-month headache status; outcome as sleep pattern variables (one at a time) controlling for demographic characteristics (age, sex, race/ethnicity, family income) and 12-month anxiety and mood disorders. For derived wakeup time variable, i.e. bedtime + sleep duration, models additionally adjusted for bedtime.
Table 3 provides estimates of 12-month prevalence of insomnia symptoms by headache subtypes. Compared to youth with migraine without aura (MO), those having migraine with aura (MA) were more likely to report difficulty maintaining sleep (OR = 2.54, 95% CI 1.32–4.88), early morning awakening (OR = 1.72, 95% CI 1.02–2.91), daytime fatigue (OR = 2.01, 95% CI 1.06–3.81), and persistent insomnia symptoms (OR = 2.03, 95% CI 1.02–4.05), after adjusting for demographic characteristics. After further adjustment for past-year anxiety and mood disorders, these associations were not statistically significant. Youth with any headache (All HA) had higher odds of various sleep disturbances than those without headache (No HA) (ORs ranged from 2.82 to 3.77), particularly among those with migraine (MIG) (ORs ranged from 2.87 to 4.32), after adjustment for demographics and for anxiety and depression. The difference between migraine with aura (MA) and migraine without aura (MO) in the number of insomnia symptoms was significant in the demographic adjusted models (OR = 1.89, 95% CI 1.03–3.44). The number of insomnia symptoms was significantly higher in youth with migraine headache (MIG) or with any headache (All HA) than in those without headache (No HA) in both partially and fully adjusted models. While the prevalence of insomnia symptoms did not differ between non-migraine headache (Other HA) and migraine without aura (MO), youth with migraine with aura (MA) were more likely to report insomnia and insomnia with daytime fatigue (Figure 1). After examining two-way interactions between headache status and potential confounders including age, sex, race/ethnicity, family income, anxiety and mood disorder in relation to insomnia symptoms, there were three statistically significant interactions between headache status and insomnia in the fully adjusted models: a) The association between migraine (MIG) and difficulty initiating sleep only occurred among youth with lower family income PIR≤ 6.0 (aOR = 2.03, 95% CI: 1.36–3.04); b) the association between headache (All HA) versus no headache (No HA) and non-restorative insomnia was significantly stronger in youth with 12-month anxiety (aOR = 1.78, 95% CI: 1.25–2.55) compared to those without a 12-month anxiety disorder (aOR = 2.83, 95% CI: 2.11–3.80); c) the association between persistent insomnia and migraine with aura (MA) compared to those without aura (MO) only emerged among youth with a 12-month mood disorder (aOR = 2.38, 95% CI: 1.11–5.13). However, there was an association between headache (All HA) and persistent insomnia regardless of mood disorder status.
Insomnia by headache status in 13-18-year-old adolescents in NCS-A. Prevalence of insomnia symptoms was significantly higher in youth with migraine headache or with any headache than those without headache. While insomnia did not differ between other headache and migraine without aura, youth with migraine with aura were more likely to report insomnia and insomnia with daytime fatigue. Insomnia symptoms associated with 12-month headache status, NCS-A n = 10,123. MA: migraine with aura; MO: migraine without aura; MIG: migraine headache with or without aura (i.e. MA or MO); Other HA: non-migraine headache; All HAs: all headache (i.e. MA, MO, NMH); No HA: no headache; SE: standard error; aOR (95% CI): adjusted odds ratio and 95% confidence interval based on logistic regression models using exposure as 12-month headache status, outcome as insomnia symptoms (one at a time) controlling for ademographic characteristics (age, sex, race/ethnicity, family income) and badditionally controlling for 12-month anxiety and mood disorders; cThree or four insomnia symptoms vs. all others (0,1,2 symptoms).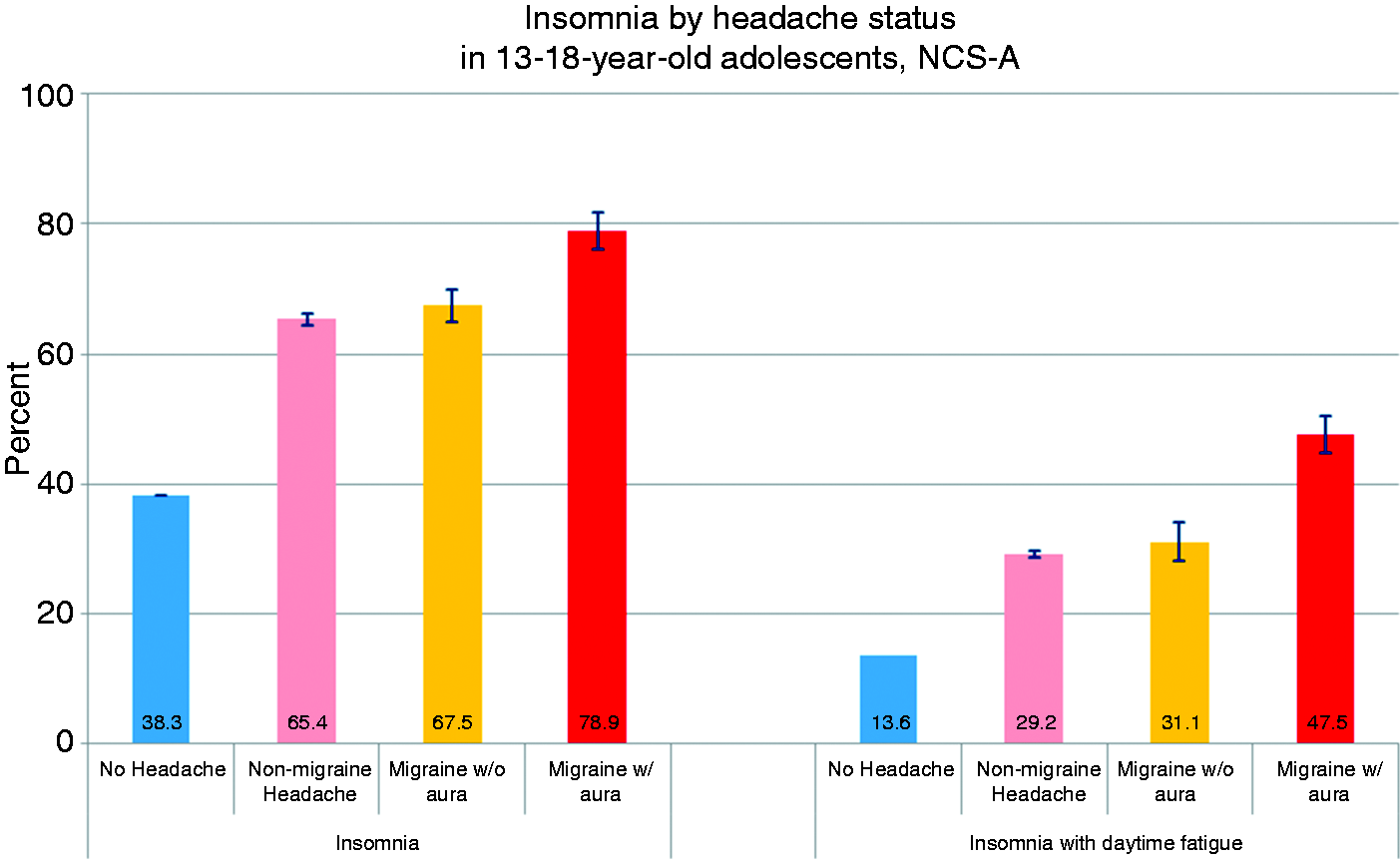

Discussion
These findings confirm earlier studies that support a connection between sleep problems and migraine (18–23), particularly migraine with aura. In the largest population-based study to date, we found significant associations between insomnia symptoms among youth with headaches, with a monotonic increase with increasingly restrictive definitions of migraine. Youth with headache, particularly migraine, had shorter sleep duration and earlier wake up times than those without headache. There was also a strong and consistent association between headache and insomnia-related complaints among youth, as described in previous research (37,38). The reduction in the strength of the associations between sleep problems with headache subtypes after controlling for mood and anxiety disorders suggests that sleep problems may be partially attributed to comorbidity with these conditions. These findings have important implications for understanding the etiology and the evaluation and treatment of migraine.
With respect to sleep patterns, we found a small but significant difference in bedtime and sleep duration on weekdays among adolescents with both subtypes of migraine and headache in general. This suggests that prior associations between increased sleepiness and headaches in previous clinical (18,19,21,22,25,39,40) and non-clinical (14,27) investigations may be partially attributable to less sleep on weekdays. Prior research has also shown that the frequency and duration of migraine attacks were associated with specific sleep disturbances such as sleep anxiety, parasomnias and bedtime resistance (40). This might explain the inability to fall asleep and shorter sleep duration despite there being no difference in bedtime for those with migraine. Furthermore, early onset sleep disorders have been found to predict headache persistence from infancy to childhood; for example, 78% of children with enduring headache had early childhood sleep disturbances compared to 25% of children with headache remission (41).
Our findings confirm those of two earlier community-based studies of youth that showed that children with all subtypes of headache reported more sleep problems than those without headache (14,27). Moreover, the former study showed that children and adolescents with headache also identified “bad sleep” and emotional distress as the most frequent causative factors for a migraine attack (14). In the latter study, children with headache, especially migraine, were also more likely to present with snoring, bedtime struggle, teeth grinding, sleep vocalization, as well as nightmares and sweating during sleep (27). While the questionnaires utilized in our study did not elicit the specific sleep disturbances included in prior studies, our findings regarding difficulty maintaining sleep, and non-restorative sleep among those with migraine, support these findings from prior community-based studies (14,27).
One of the most notable findings in our study was the tendency for migraine with aura to be more strongly associated with insommia symptoms than migraine without aura or non-specific headache subtypes. However, this association dissipated after controlling for comorbid anxiety and depression, suggesting that the common etiology between migraine and disordered sleep may be mediated by comorbid mood and anxiety disorders. Mood and anxiety disorders have been consistently associated with headache in general, and migraine in particular, in clinical (42) and community samples (31,43–46). The mechanisms underlying disturbances of sleep, mood and anxiety, and headache could result from shared sociodemographic characteristics, behavioral patterns, autonomic dysregulation, or biological correlates such as the autonomic nervous system, which has been implicated in both anxiety (47) and migraine with aura (48). Interestingly, anatomic substrates for both sleep and headache in the brainstem and diencephalon overlap, as do physiologic pathways involving dopamine, orexin, and melatonin (49).
Because of the cross-sectional nature of these data, we could not identify causal mechanisms for the association between sleep disturbances and headache. Sleep disturbances have been widely implicated as triggers of migraine in as many as 70% of children (50). Poor sleep could also be a consequence of headaches, as shown in previous studies in which migraine without aura was a sensitive risk factor for disorders of initiating and maintaining sleep (17). Besides these explanations for headache-sleep comorbidity, sleep complaints could also herald the onset of headache and represent a prodromal phenomenon.
Strengths of the current study include the use of a large representative sample of USA youth and the inclusion of diagnostic interviews for sleep, migraine, and mood and anxiety disorders as opposed to symptom checklists that have been used in many prior community surveys. The large sample size also enabled us to examine subgroups of youth with migraine with aura as a distinct category, which has not been previously examined. Limitations of this study include its cross-sectional nature resulting in inherently correlative data. Additionally, the relatively large number of tests conducted in this study may increase the probability that some of our associations are due to Type I error (false positive). Third, the diagnostic criteria for migraine and migraine with aura are only approximations of the International Classification of Headache Disorders, second edition (ICHD-III) criteria because a few of the required symptoms were not collected in these interviews. Headache in this study was limited to those youth who endorsed “severe and frequent” headache. Fourth, the full criteria for DSM-5 insomnia, including frequency and clinical impairment attributable to insomnia symptoms, were not collected during the interview; therefore, we refer to “insomnia symptoms” rather than to “insomnia disorder” throughout this report. Finally, the cross-sectional design precluded our ability to discern the order of onset of migraine, sleep problems, and anxiety/depression. Longitudinal research would help to inform the design of better strategies for prevention and management.
Our findings have important clinical implications. First, the recognition and treatment of sleep problems in people with migraine may diminish the negative impact of migraine on daily functioning. Pharmacologic, relaxation, and cognitive behavioral treatments aimed to improve sleep as well as strategies to improve sleep patterns may benefit migraine patients. Furthermore, if changes in sleep herald the onset of migraine, early intervention in this process could actually reduce the incidence of acute attacks of migraine. Our results argue for screening and management of sleep disturbance among people with headache regardless of whether their specific symptoms satisfy IHS criteria for a particular disorder, and further support paying additional attention to youth having migraine with aura. Our results also suggest that future studies should examine the potential overlap between sleep problems, migraine and comorbid mood and anxiety disorders. Systematic research using in time mobile tracking could provide insight into potential mechanisms for these associations.
Conclusions
Adolescents with severe headache had significantly more sleep problems than those without headache. Increased sleep problems among those with migraine with comorbid anxiety and mood disorders highlights the importance of comprehensive evaluation of the full spectrum of these regulatory systems in youth with headache. Further research should delineate the temporal relationships of disordered sleep with migraine and the effects of optimizing sleep on headache status and utilizing objective measures.
Public health relevance
Youth (13–18) with headache had poorer sleep (shorter duration, earlier morning awakening, more insomnia symptoms) than those without headache. The associations between headache and sleep problems were partially attributable to comorbid mood and anxiety disorders. This research adds to current evidence that pervasive sleep disturbances should be evaluated in adolescents with severe headache.
Footnotes
Acknowledgements
Funding for this study was provided by the National Institute of Mental Health (ZIAMH002808), which had no further role in study design, collection, analysis, interpretation of data, writing of this manuscript, and in the decision to submit this paper for publication. The authors alone are responsible for the content and writing of the paper. All authors declare no conflicts of interests. All authors have read and approved the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
