Abstract
Background
Migraine is a primary headache disorder involving dysregulation of central and peripheral pain pathways. Medical treatment is often limited by drug side effects, comorbidities and poor compliance. This makes neuromodulation an ideal option for migraine treatment. Cefaly® is a transcutaneous electrical neurostimulator designed specifically for migraine treatment. It results in external trigeminal nerve stimulation of the supraorbital and supratrochlear nerves. External trigeminal nerve stimulation is effective for acute and preventive migraine treatment and may result in normalization of dysregulated pain pathways.
Objective
Our objective was to provide a narrative review of the neuroanatomical and pathophysiological basis of external trigeminal nerve stimulation for migraine treatment and to provide the rationale behind the choice of the electrical parameters used for external trigeminal nerve stimulation.
Methods
We reviewed external trigeminal nerve stimulation clinical trial publications, basic science neurostimulation literature, publications describing pathophysiological mechanisms in migraine, and documentation used in the application for the Food and Drug Administration approval of external trigeminal nerve stimulation.
Results
The electrical parameters used for external trigeminal nerve stimulation were chosen to maximize safety and efficacy. Critical parameters include generator characteristics, pulse shape, pulse duration, pulse frequency and session duration and frequency. We explain the rationale behind determination of each parameter. There is evidence of dysregulated central and peripheral pathways in migraine and evidence that external trigeminal nerve stimulation may normalize function of these pathways.
Conclusion
External trigeminal nerve stimulation is a safe and effective Food and Drug Administration-approved option for the acute and preventive treatment of migraine. The electrical parameters were optimized specifically for external stimulation of the trigeminal nerve to maximum safety, comfort and efficacy.
Introduction
Migraine is a common primary headache disorder affecting approximately 10% of the world population (1,2). It is the leading neurological cause of disability and overall the seventh most common cause of disability globally (3). The pathophysiology of migraine involves an abnormal processing of sensory stimuli, including hyperexcitability and a sensitization of trigeminal and central pain pathways (4). Limits to medical treatment of migraine include adverse reactions to medications, drug interactions, medication-overuse headache (MOH) and contraindications based on medical comorbidities. In patients affected by these limitations, a non-medication, device-based migraine treatment, such as external trigeminal nerve stimulation (eTNS, Cefaly®) may be a reasonable option for many patients. Some patients simply prefer non-pharmacological treatments.
Objective
Our objective was to provide a narrative review of the neuroanatomical and pathophysiological basis for eTNS in the treatment of migraine and a rationale for the determination of the electrical parameters used.
Methods
We reviewed eTNS clinical trial publications, basic science neurostimulation literature, publications describing pathophysiological mechanisms in migraine, and documentation (5) used in the application for the Food and Drug Administration (FDA) approval of eTNS.
Background
Transcutaneous electrical nerve stimulation (TENS) has been used to treat acute chronic pain disorders, including neuropathic pain (6–8). Since migraine patients have decreased sensory and pain thresholds for electrical stimulation (cutaneous allodynia), standard TENS applied to the head can be painful, as well as cumbersome (9). Percutaneous occipital nerve stimulation (ONS) has some efficacy for migraine treatment; however, it is invasive and adverse effects such as persistent implant site pain are common (10,11).
A standard TENS device consists of a battery-powered generator connected to a set of skin electrodes delivering electrical impulses of adjustable amplitude, frequency and duration. Patients typically use a trial and error approach in determination of each parameter setting. The International Association of Pain (IASP) describes two primary forms of TENS. Conventional TENS, which is most common, uses low intensity, high frequency (50–100 Hz) electrical stimulation of short duration (50–200 μs). Acupuncture-like (AL) TENS, typically reserved for non-responders to conventional TENS, uses high intensity, low frequency (2–4 Hz) electrical stimulation of long duration (100–400 μs) (12).
Cefaly® (CEFALY Technology, Belgium) is an external trigeminal nerve stimulator (eTNS) of the supraorbital and supratrochlear nerves (see Figure 1). It was developed specifically for migraine and was approved by the FDA in 2014 for preventive treatment and in 2017 for acute treatment. It generates biphasic rectangular micro-impulses with an electrical mean equal to zero, delivered with a 30 mm × 94 mm self-adhesive electrode placed over the area of the distribution of the supratrochlear and supraorbital branches of the ophthalmic division of the trigeminal nerve. Over a 20-minute treatment session, preprogrammed electrical pulses are delivered at a width of 250 µs, frequency of 60 Hz and maximum intensity of 16 mA.
External trigeminal nerve stimulator placed over the supraorbital and supratrochlear branches of the ophthalmic division (V1) of the trigeminal nerve. Use of this copyrighted image was approved by Cefaly Technology.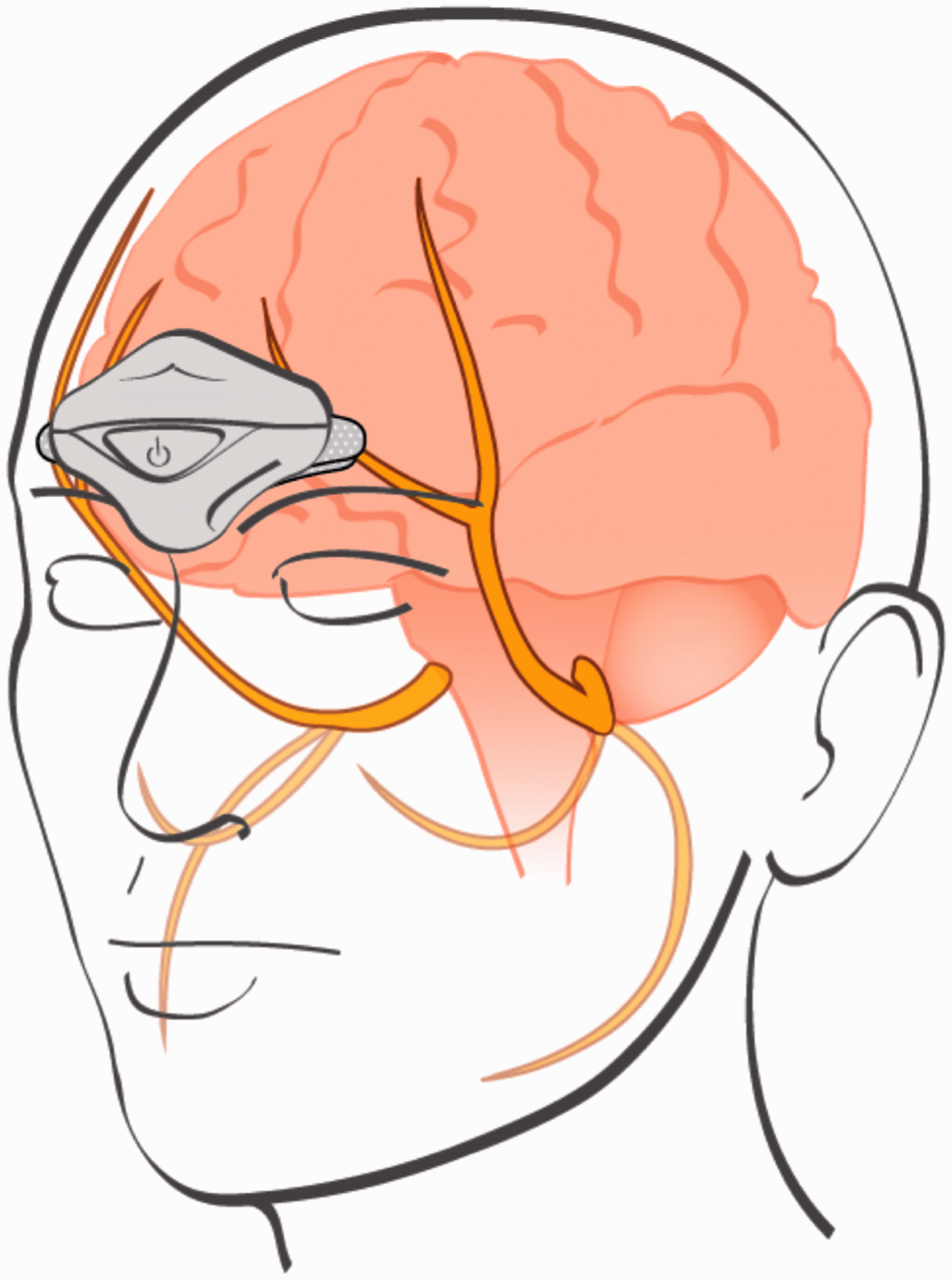
Clinical evidence for eTNS
In a randomized, double-blind sham-controlled eTNS trial (Prevention of Migraine with Cefaly, PREMICE), 67 migraine patients were preventively stimulated 20 minutes daily for 3 months. The therapeutic gain for the 50% responder rate and reduction of migraine days in the verum compared to sham group was modest at 26.1% and 12%, respectively. The 50% responder rate for change in migraine days for the verum and sham group was 38% and 12%, respectively. Moderate improvement occurred in 59% of the verum group and 27% of the sham group. Verum stimulation also significantly decreased monthly migraine attacks, headache days, and acute medication use compared to sham at three months. Efficacy results may have been skewed in favor of the verum group, given a lower mean age (−4.47 years) and shorter disease duration (−3.46 years) compared to the sham group. Authors were unable to prove that partial unblinding of patients did not occur. Of the 90 intended stimulation sessions, compliance was limited to 55.5 sessions (61.7%) in the verum group and 49 sessions (54.4%) in the sham group (13).
In a small (n = 24) prospective, uncontrolled trial, preventive eTNS in episodic migraineurs with low frequency of attacks (mean 4.5 days/month) naive to preventive medications, the 50% responder rate at 60 days for decrease in migraine attacks and migraine days was 81% and 75%, respectively (14). No adverse effects (AEs) were reported in either of the two aforementioned studies. It should be noted that the findings of these studies are not applicable to more disabled migraine patients with high frequency of attacks, as these patients were not included.
In a small open-label, uncontrolled, prospective study of daily preventive eTNS in 23 chronic migraineurs, 35% of patients experienced both a 50% reduction in monthly migraine days and 50% reduction in monthly medication use at 4 months. Four patients (17.4%) dropped out of the study, three of whom did so due to an inability to tolerate eTNS (one reported worsened headache and two reported the development of neck pain) (15).
In a 30-patient open-label trial of eTNS for acute treatment of migraine, mean pain intensity was reduced by 57% after 1 hour of treatment and 77% of patients reported > 50% improvement in pain. No AEs were reported, however two of the 34 patients (5.9%) were not enrolled due to failure of a tolerance test secondary to the presence of allodynia (16). Given that cutaneous allodynia is reported in approximately two-thirds of patients with migraine (17), further studies including a representative sample of allodynic migraineurs should be completed to assess the true tolerability of eTNS among the general migraine population.
In an unpublished (data presented in abstract form only) multi-center, double-blind, randomized sham-controlled trial of acute eTNS in 106 patients with episodic or chronic migraine, (Acute Treatment of Migraine with eTNS, ACME), mean reduction of pain scores after 1 hour of treatment was −59% in the verum group and −30% in the sham group. Complete pain freedom was achieved at 1 hour in 29% of the verum group and 6% of the sham group. Of note, blinding of sham versus verum stimulation has been validated in a separate study of 128 patients (18).
In a survey of 2313 patients using eTNS for a trial period of 40 days, 54.4% reported satisfaction and were willing to purchase the device. AEs were reported in 4.3%. Two percent of patients were unable to tolerate the treatment due to local pain or paresthesia, 0.82% reported sleepiness, fatigue or insomnia, 0.52% reported headache after the stimulation, and 0.09% reported a local skin allergy (19).
Trigeminal nociception
The trigeminal pathway plays an integral role in the pathophysiology of migraine. In order to explain the rationale for trigeminal electrostimulation we review basic neuroanatomical and physiological components of this system.
Sensory nerve fiber types.

Second order neurons from the TCC project in the trigeminothalamic tract to supratentorial central pain centers of the thalamus (ventroposteromedial and dorsomedial nuclei), periaqueductal gray (PAG), and the hypothalamus. Third-order neurons from the thalamus project to the insular, anterior cingulate and somatosensory cortex. The cortex is involved in the higher function processing, modulation, and perception of pain (22).
In addition, supratentorial central pain structures are responsible for inhibiting pain via a descending modulatory pathway. The process, called diffuse noxious inhibitory control, is activated by afferent A-delta and C fiber stimulation (23). The PAG can inhibit the nociceptive second order neurons of the spinothalamic tract (24). The prefrontal and posterior cingulate cortex, nucleus raphe magnus, and rostroventral medulla have been shown to play a role in the descending modulation of pain via connections with the thalamus (25).
Dysfunction of trigeminal nociceptive pathways in migraine
Functional MRI studies have shown an abnormal cycling pattern of trigeminal activation secondary to nociceptive stimuli. There is lower activation interictally with increasing activation preictally compared to controls, possibly reflecting an increased susceptibility to migraine attacks (26). There is greater fMRI BOLD activation in the anterior cingulate cortex in migraine patients following noxious heat stimulation to the trigeminal nerve compared to controls (27). There is a lack of normal habituation with repeated trigeminal nociceptive blink reflex (nBR) interictally compared to controls, consistent with abnormal trigeminal nerve mediated pain modulation in migraine (28,29). This reflects a derangement of normal trigeminal nerve modulation in migraine and a role for targeting the trigeminal system with peripheral electrostimulation for treatment. In a pilot study of 10 patients with migraine treated with 20 minutes of eTNS, an immediate decrease in the amplitude of the nBR was observed (30).
Introduction to electrostimulation
Neuromodulation for the treatment of pain has the goal of decreasing peripheral excitation and increasing central inhibition (31). Electrical nerve stimulation is one of the most widely used forms of neuromodulation.
In 1965, Melzack and Wall first described the “gate control theory of pain”, the mechanism by which activation of low threshold A-beta fibers inhibit nociceptive fibers via GABAergic spinal interneurons (32,33). This mechanism was the basis for TENS treatment, used for decades in various pain conditions but only recently studied in migraine.
Central pain modulation and gain control
While mechanisms of the gate control theory were not experimentally entirely confirmed, it transformed our idea of pain mechanisms and was the basis of new pain treatment modalities such as TENS. Since then, a new theory has been introduced, based on the nociceptive system’s ability to modulate pain sensitivity at peripheral, spinal, or supraspinal levels, called “gain control” (34). Historically, discussion of spinal cord mechanisms of central excitability had been prominent; now it is clear that central descending pathways have a predominant role in spinal hyperexcitability (35). Mild stimulation results in adaptation and decreased responses (habituation). In contrast, peripheral sensitization reduces the threshold for activation of the transient receptor potential cation channel subfamily V member 1 (TRPV1) receptors, creating an increased response to repeated mild noxious stimuli. While fatigue predominates for mild stimuli, sensitization predominates for noxious stimuli (34).
Nociceptive neurons in the dorsal spinal cord exhibit a temporal summation called “wind-up”. Increased responses occur when C-fiber input reaches a frequency of 3 Hz, and this phenomenon is characteristic of “wide-dynamic range” neurons. Wind-up is thought to compensate for peripheral fatigue but is short-lived, and when prolonged leads to long-term depression. It can also lead to central sensitization of wide-dynamic range neurons.
Central sensitization is a longer-lasting process involving second-order wide-dynamic range neurons, which have spinothalamic projections. Impulses from peripheral nociceptors activate second-order neurons, resulting in ipsilateral cephalic allodynia (when non-noxious stimuli become noxious). Second-order neurons then sensitize third-order neurons, resulting in contralateral head and ipsilateral forearm allodynia (36). Glial cell activity around second order neurons is believed to play a role in central sensitization. Following an injury to the first order neuron, wide-range high threshold nociceptors increase the response to noxious stimuli. This peripheral sensitization results in central sensitization. This is similar to the mechanism of long-term potentiation; both utilize glutamatergic transmission. Central sensitization is initially maintained by peripheral sensitization from prolonged C-fiber stimulation; later it can become activity independent of peripheral input in part by mechanisms of gene transcription.
BOLD-fMRI has demonstrated larger responses to brush and thermal stimulation in the posterior thalamus of rats during a migraine attack with extracephalic allodynia. The spread of allodynia outside the typical distribution of pain is mediated by sensitized thalamic neurons (37).
The brainstem delivers both tonic and stimulus-invoked descending inhibitory input to spinal nociceptive neurons, using serotonin (5-HT) and norepinephrine (NE) as neurotransmitters. NE acts at spinal alpha-2 receptors, while serotonin may be facilitatory through spinal 5-HT3 receptors and inhibitory through spinal 5-HT7 receptors (35). The brainstem receives ascending nociceptive input leading to activation of the network called diffuse noxious inhibitory control (DNIC). This leads to a widespread pain inhibition outside the area of stimulation.
The periaqueductal gray (PAG) and the rostroventral medulla (RVM) are important centers in the modulation of spinal gain control. The RVM receives descending input from the PAG and is modulated by the hypothalamus, amygdala and the cerebral cortex (32). Thus, cognitive processes can modify ascending input, as occurs with the placebo effect. The descending pathways effects are system-wide. Growing evidence suggests that chronic pain is a result of dysregulated descending inhibitory pain pathways. Disruption of this pathway may favor facilitation and sensitization, maintaining pain (34).
Peripheral stimulation of nociceptive C-fibers results in the release of excitatory amino acids such as glutamate and inflammatory neuropeptides such as calcitonin gene-related peptide (CGRP) and substance P at the spinal level (38). Extrasynaptic spread of these molecules facilitates spinal nociception (in part by glial cells) and results in sensitization of dorsal horn neurons. Activation of the NMDA receptor is critical for creating and maintaining sensitization of the glutamate receptor. Dorsal horn neurons develop an expanded receptive field, which results in hyperalgesia and allodynia. Inflammation upregulates ion transporters, resulting in excessive depolarization and cross excitation of high threshold nociceptive fibers. Sustained release of nociceptors such as glutamate, CGRP, and substance P causes dissociation of Mg from the NMDA receptor, increases synaptic efficacy, and induces Ca++ influx, resulting in sustained central sensitization (39). For this reason, electrostimulation of C-fibers should generally be avoided (40). It has also been suggested that electrostimulation of low threshold non-nociceptive afferents may induce a reversal of central sensitization and decrease allodynia and hyperalgesia (31).
Basic electrical and neurophysiological principles
Electric charge is measured in coulombs, named after the French academic Charles de Coulomb. One coulomb is equal to the amount of charge of 6.24 × 1018 electrons.
Elements, mainly metallic, which have a large number of free electrons are called conductors. The movement of negatively charged electrons through a conductor is an electric current (41). Electric current is described in terms of intensity and is measured in amperes. The quantity of negative charge transported per second by a current of one ampere is equal to one coulomb.
In the resting state, neurons maintain a net negative charge, or electrical potential inside compared to outside the cell membrane. This difference in electrical charge is powered by adenosine triphosphate (ATP), providing energy to pump charged ions such as potassium (K+) and sodium (Na+) against their respective concentration gradients. The net negativity within the cell created by this process is approximately −70 mV and is called the equilibrium or resting potential (42).
In order for a nerve to generate an action potential, it must be excited to the threshold level or about −50 mV. An external electrical impulse can be delivered through the skin (transcutaneous electrostimulation) to stimulate the nerve. The threshold potential for a sensory nerve is less than that of a motor nerve. This is the basis of electrostimulation for pain.
Principles of electrostimulation
In 1901, a Parisian electrophysiologist, George Weiss, described the “fundamental law of electrostimulation” (43). Weiss determined that the quantity of electric charge provided by the stimulation current was the most important factor in reaching the excitation threshold and generation of the action potential. He applied currents of variable intensity with variable pulse durations and found a linear relationship between the intensity of the current and pulse duration used to reach the excitation threshold (see Figure 2). Also referred to as the Weiss Law, he defined this relationship as the “fundamental formula of electrostimulation” (44).
Weiss Law; Q: charge necessary to reach stimulation threshold; i: experimentally defined coefficient; t: time.
Weiss Law

Weiss also determined that the shape of the impulse had no bearing on the stimulation effect. He also established the concept of a minimum amount of current (q) required to reach excitation independent of pulse duration. Louis Lapicque, a French neuroscientist, published a paper in 1907 that defined the minimum current intensity as “rheobase”.
Lapicque established a formula using the variables of current intensity and pulse duration to compare the excitability of different tissues (45). Even with an infinite duration of stimulation, a minimum current intensity equal to rheobase is necessary to reach threshold. “Chronaxie” is the term Lapicque introduced to define the minimum pulse duration to reach threshold when current intensity is twice that of rheobase (see Figure 3). The practical utility of chronaxie is as follows: A longer duration would consume current without substantially decreasing threshold; it allows for the use of smaller electrodes and minimal generator output, which increases safety (46). Lapicque called this the “strength-duration relationship”, the title he gave the formula used to compare the excitability of different objects (47), and this is defined as follows:
Strength-duration curve.
Strength-duration relationship

Rheobase and chronaxie were conceptually fundamental in the determination of the electrical parameters used for eTNS, particularly the pulse shape, duration and amplitude.
eTNS electrical parameter determination
We now describe the rationale behind the determination of the electrical parameters for eTNS: Generator characteristics; the shape, width, amplitude and frequency of the electrical impulse; and the session duration. The goal of stimulation is to deliver the most efficacious charge to the nerve in the most safe and comfortable fashion.
Generator characteristics
To achieve nerve stimulation, a specific quantity of electrical charge needs to be delivered to the nerve. The amount of charge that reaches the nerve depends on the voltage output of the generator and the resistance, or impedance, of the circuit (skin, electrode, subcutaneous tissues). Voltage is defined as the difference in potential electrical energy or charge between two points and the driving force of current against resistance (41). The current delivered by a constant voltage generator will vary with variable resistance; however, a constant current generator will maintain a continuous level of current despite changing resistance. Ohm’s law describes the relationship of voltage, current and resistance as follows (48):
Ohm’s Law
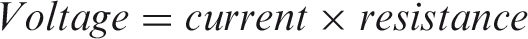
Based on impedance measurements made by Cefaly Technology (5), it was determined that a constant current generator designed for a maximum skin impedance of 2200 ohms would be needed for the eTNS circuit. Maintaining a constant current ensures precise delivery of the intended amount of current and impulse waveform to the nerve.
The most precise delivery of electrical impulse to a nerve is achieved by percutaneous electrical nerve stimulation (PENS). PENS requires the surgical placement of a stimulating electrode under the skin in direct proximity to the nerve. This technique bypasses the impedance of tissue between the skin and nerve. By designing the eTNS for the specific impedance of tissues overlying the supraorbital nerve and using constant current, a similarly precise impulse is delivered without the invasiveness of PENS.
Pulse shape
Since pulse intensity equivalent to rheobase is needed to achieve excitation, the most efficient pulse has a square waveform. There is no value in a gradually sloping intensity less than 90 degrees, as this unnecessarily prolongs pulse duration. Any intensity level less than rheobase is insufficient for excitation, therefore a 90-degree vertical impulse maximizes efficiency, comfort, and safety (see Figure 4).
Charge accumulated during t1-4 less than that of i = rheobase is insufficient to generate excitation, and that greater than i = rheobase unnecessarily delivers excessive charge, decreasing safety and efficiency.
The amplitude of the impulse should remain constant at the minimum intensity sufficient to produce stimulation. Spikes above this level decrease comfort by stimulating nociceptive fibers with higher thresholds (see Figure 5).
Pulse shape.
Direct current (DC) is the term used to describe electrical current with a single direction. Alternating (biphasic) current (AC) describes a current that reverses direction. Current is delivered to a tissue by electrodes. The anode is the positively charged electrode at which electrons leave the device and the cathode is the negatively charged electrode at which electrons enter the device. By convention, current flow is in the direction opposite to electron flow. In devices using DC or asymmetric AC, electrons build up around the cathode, resulting in ion accumulation and potentially tissue injury. eTNS uses a biphasic symmetrical impulse with an electrical mean of zero and therefore no net electron displacement (see Figure 6) (41).
Biphasic square wave impulse with electrical mean of 0, a positive S1 wave is equivalent to a negative S2 wave.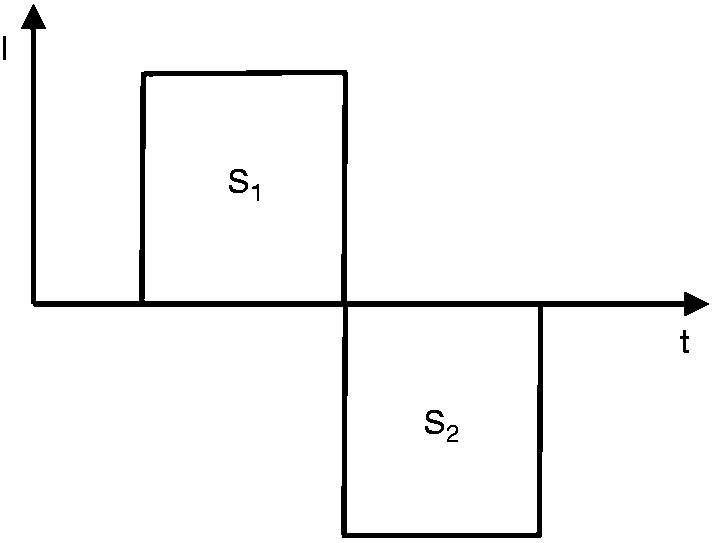
Pulse duration
To maximize patient comfort and safety, total energy delivered to tissues must be minimized. Electrical energy may be calculated as follows:

By substituting the value of current intensity (as defined by the Weiss Law), an equation with the following curve is generated (see Figure 7).
Any pulse duration longer or shorter than the time at the apex of the curve increases the energy requirement.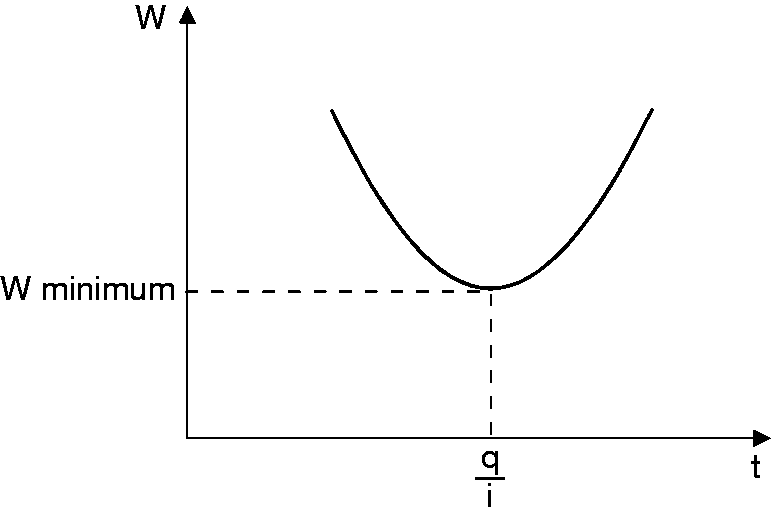
By derivative calculation using this curve, the duration (q/i) equivalent to the minimum energy may be calculated and is equivalent to chronaxie. Chronaxie was measured for the supratrochlear and supraorbital nerves of 15 adults and averaged 250 µS. This determined the pulse width value used for the device. Standard TENS devices use an adjustable pulse width from 50–500 µS (48). Chronaxie describes the excitability of tissues and is constant despite variable resistance (see Figure 8).
Chronaxie is constant despite variable resistance (R1-3).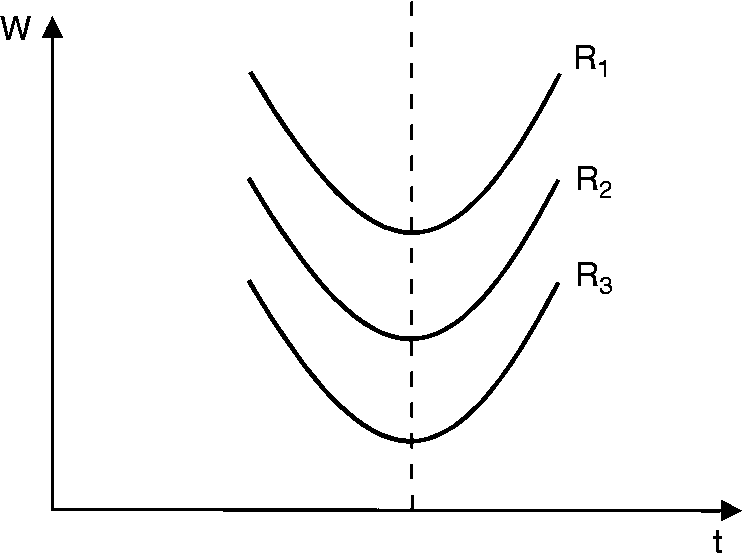
Pulse frequency
Data to support the use of high versus low frequencies is lacking. The natural action potential frequency for a sensory fiber ranges from 1–120 Hz. When a nerve is electrostimulated at a frequency higher than the natural rate of action potential discharge, fatigue occurs, which clinically manifests as paresthesia. High (> 60 Hz) pulse frequency TENS modulates delta and kappa opioid receptors at the spinal cord level and decreases activity of excitatory neurotransmitters such as glutamate and aspartate. This was supported by decreased TENS efficacy following blockade of spinal opioid receptors. Low (<10 Hz) frequency TENS modulates mu-opioid receptors and 5-HT2 and 5-HT3 receptors (12). Thus, in patients with chronic opioid use, TENS treatment may be inefficacious.
A conventional TENS device is programmed with an adjustable pulse frequency of 1–200 Hz (48). eTNS delivers electrical impulses at a rate of 60 Hz. This was based on precedent set by average frequency rates used in earlier trials of ONS for headache treatment (49,50). In a double-blind, sham-controlled crossover study, high frequency stimulation with supraorbital eTNS for 20 minutes had a sedative effect not observed with low frequency stimulation (51).
This was the first indication of a central mechanism of efficacy. Magis et al. used 18-fluorodeoxyglucose positron emission tomography (FDG-PET) to measure cerebral metabolic activity before and after 3 months of daily eTNS in episodic migraineurs compared to control subjects. Hypometabolism was identified at baseline in pain modulating fronto-temporal regions of the individuals with migraine. The authors found a significant metabolic normalization in the migraineurs following 3 months of treatment. These findings suggest a possible supraspinal mechanism of eTNS neuromodulation in migraine prevention (52).
Using whole-brain BOLD-fMRI, functional changes in the anterior cingulate cortex were observed following eTNS in migraine patients without aura. Migraineurs had greater BOLD response to noxious thermal trigeminal heat stimulation in the anterior cingulate cortex at baseline compared to controls. Following a 60-day treatment period with eTNS, migraineurs had a significantly decreased BOLD response in the same area (53).
Pulse amplitude
The goal of stimulation is to excite or recruit the maximal percentage of nerve fibers. As amplitude increases the amount of charge delivered to tissue, the percentage of fibers recruited increases as well. However, if excess charge is delivered to tissue, energy will penetrate deeper and stimulate high-threshold noxious fibers. Optimal pulse amplitudes for eTNS were measured in healthy controls without allodynia by slowly increasing stimulation and recording the subjective intensity of paresthesia, a standard procedure to measure sensory fiber recruitment (54). When subjective paresthesia peaked without any sensation of pain, pulse amplitude levels were recorded. These levels reflect near maximal A-beta fiber recruitment without recruitment of higher threshold A-delta nociceptive fibers. The average value was 16 mA, a relatively low value considering a conventional TENS device functions in the range of 1–60 mA (see Figure 9) (48).
Pulse amplitude determination using healthy controls.
Session duration
Factors determining session duration include the time needed to reach and maintain the optimal amplitude of 16 mA. If pulse amplitude is increased too quickly, excess charge is delivered and high threshold nociceptors are activated, which results in pain. During sensory fiber electrical stimulation, acute habituation occurs. Sensory nerve fibers become less excitable; that is, tolerant to a constant level of pulse amplitude. In order to maximize acute analgesic effects, the amplitude must be increased incrementally over the course of the session (55). This adaptation effect can be exploited to maximize stimulation without causing pain.
Determining the maximal rate of increase was achieved with bench testing in healthy controls. The maximal rate of non-painful amplitude increase in controls was 30 µA per second. Considering the pulse duration of 250 µS and maximum amplitude increase of 30 µA/sec, the time necessary to comfortably reach 16 mA is 14 minutes. The device then maintains an intensity of 16 mA throughout the duration of the 20-minute session.
Session frequency and analgesic tolerance
Some studies have demonstrated increased efficacy of TENS with repeated use, possibly by a reversal of peripheral and central sensitization (56). There is also evidence that repeated TENS use results in analgesic tolerance mediated by cholecystokinin (CCK). Blocking CCK and n-methyl-d-aspartate (NMDA) receptors prevents analgesic tolerance (57,58). Varying stimulation intensity also prevents tolerance (59). eTNS may benefit from this, as the intensity level builds over the duration of each preprogrammed session.
Summary
Migraine is a tremendous source of disability worldwide despite currently available treatment options. Side effects, poor compliance, and contraindications often limit medication use. Dysregulation of peripheral and central nociceptive pathways involving the trigeminal nerve occurs in migraine. In contrast to a standard TENS device, Cefaly® was designed specifically for trigeminal nerve stimulation for the treatment of migraine. The electrical parameters were optimized specifically for external stimulation of the trigeminal nerve, in order to maximize efficacy and safety. eTNS is generally well tolerated in the absence of cutaneous allodynia. eTNS is FDA approved for the acute and preventive treatment of migraine. Use of eTNS may result in normalization of dysregulated central and peripheral pain pathways. Further studies are required to assess the efficacy of eTNS in the most disabled of migraine patients including those with chronic migraine. The cost of the device ($349–499 USD) and the presence of cutaneous allodynia may be limiting factors to use.
By sharing the rationale behind the determination of eTNS electrical parameters and reviewing pathophysiological mechanisms in migraine, we hope to stimulate discussion of neuromodulation for migraine and encourage further development of existing and novel treatments.
Clinical implications
Limits to medical treatment of migraine include medication adverse events, drug interactions, and contraindications based on medical comorbidities. eTNS is safe, effective and FDA-approved for the acute and preventive treatment of migraine. eTNS may result in normalization of dysregulated peripheral and central pain pathways. By providing the rationale behind the determination of eTNS electrical parameters and reviewing pathophysiological mechanisms in migraine, we hope to stimulate discussion of neuromodulation for migraine and encourage further development of novel and existing treatments.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Clinton Lauritsen has received honoraria from Cefaly Technology. Stephen Silberstein has received honoraria from Alder Biopharmaceuticals; Allergan, Inc.; Amgen; Avanir Pharmaceuticals, Inc.; Cefaly Technology (unrelated to this manuscript); Depomed; Dr. Reddy’s Laboratories; eNeura Inc.; electroCore Medical, LLC; Ipsen Biopharmaceuticals; Medscape, LLC; Medtronic, Inc.; Mitsubishi Tanabe Pharma America, Inc.; NINDS; St. Jude Medical; Supernus Pharmaceuticals, Inc.; Teva Pharmaceuticals and Trigemina, Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Clinton Lauritsen received funding from Cefaly Technology to investigate eTNS electrical mechanisms for the purpose of creating this manuscript.
