Abstract
Background
In cluster headache, neuromodulation is offered when patients are refractory to pharmacological prophylaxis. Non-invasive peripheral neuromodulatory approaches are of interest. We will focus on these and particularly on nociception specific, transcutaneous supraorbital nerve stimulation.
Methods
In a study using the nociception specific blink reflex, we made a serendipitous discovery, notably the potential prophylactic effect of bilateral, time contingent, nociception specific, transcutaneous stimulation of the supraorbital nerve.
Results
We report on a case series of seven cluster headache patients, in whom attacks seemed to disappear during repeated stimulation of the supraorbital nerves. Three patients stopped experiencing attacks since study participation.
Conclusions
Bilateral, time contingent, nociception specific, transcutaneous supraorbital nerve stimulation may have a prophylactic effect in episodic and chronic cluster headache. Given its limited side effects and its non-invasive nature, further studies to investigate this potential peripheral neuromodulatory approach for both episodic and chronic cluster headache are warranted.
Keywords
Introduction
When cluster headache patients are refractory to or do not tolerate pharmacological prophylaxis, peripheral nerve blocks may be effective, but attacks may recur within weeks (1). The next line of treatment comprises neuromodulation and peripheral as well as central approaches are available. Deep brain stimulation of the posterior hypothalamus (2), however, may bring on even fatal side effects. Less invasive methods are being studied (3) and occipital nerve stimulation is considered the first-line neuromodulatory approach in refractory chronic cluster headache, although randomized controlled trials are still awaited (4,5). Because even peripheral invasive neuromodulatory approaches carry a certain risk of device- and surgery-related side effects, non-invasive peripheral neuromodulatory approaches are of particular interest (4).
Study of the blink reflex is of interest in cluster headache. A concentric planar stimulating electrode with a central cathode and external anode ring selectively stimulates superficial nociceptive A-delta fibres of the supraorbital nerve in the nociception specific blink reflex (6). In order to further unravel cluster headache pathophysiology, we are investigating the effect of oxygen on medullary interneurons. The study uses a repeated measures design, in which the nociception specific blink reflex is measured every 2 hours before a spontaneous attack, shortly after onset of an attack and 12–15 minutes after start of oxygen treatment. At present, eight patients have enrolled and none of them experienced a spontaneous attack during the clinical study time, reason to report on this series of patients.
Methods
Study population
Patients diagnosed by their neurologist with active cluster headache according to the International Classification of Headache Disorders, second edition (ICHD-II) criteria (7) were included. Patients were excluded in the case of secondary cluster headache, other headache diagnoses or painful conditions, pregnancy or lactation, intolerability of the oxygen face mask and incapacitation to understand and sign for informed consent. Chronic obstructive pulmonary disease and previous thermolesion of the sphenopalatine ganglion were among the relative exclusion criteria. Patients were not allowed to use nitroglycerin, subcutaneous sumatriptan and alcohol in the preceding 2.5, 12 and 24 hours respectively and other triptans in the preceding 6 days. Dosages of prophylactic medication were not allowed to be changed in the preceding 2 weeks.
Eligible patients were scheduled for study participation when the chance of occurrence of a spontaneous cluster headache attack was expected to be ‘moderate’ to ‘high’ on that single day in the afternoon or evening (and one night), based on the individual patient’s attack frequency and pattern during previous days and weeks.
Time contingent, nociception specific blink reflex
The supraorbital nerves were electrically and separately stimulated with a 200 pulse per second train of three 0.5 ms pulses by the special concentric planar stimulating electrode with central cathode and external anode ring (6), placed on the skin 10 mm cranial of the supraorbital notch on both the affected and non-affected side. The current intensity was adjusted with increasing 0.3 mA steps, delivered at unpredictable intervals, until 2.1 mA (once 2.4 mA), until 1.2 times the intensity of acquired stable R2 responses or until the patient's tolerance limit was reached. Four well-displayed nociception specific blink reflexes were obtained for each side (using the stimulation sequence two at the affected side – four at the non-affected side – two at the affected side), with different intervals of at least 15 seconds to minimize habituation. To exclude diurnal variance, the nociception specific blink reflex without oxygen treatment was elicited every 2 hours before the expected occurrence of a spontaneous attack. As a control, the nociception specific blink reflex was measured once outside an attack during 4 minutes of inhalation of 100% oxygen with a flow rate of 12 L/min using a non-rebreathing facial mask.
Questionnaires
Before study participation, patients filled in a questionnaire to double-check the diagnosis and preceding medication use.
At variable times following study participation, we contacted the patients to inquire retrospectively more specifically about medication use and attack frequency and pattern in the week before and following study participation. A diary was kept by two patients in the week before and three patients in the week following study participation.
Ethics
All patients gave written informed consent for participation in the original study, which was approved by the local ethics committee.
Results
At present, eight of the targeted 20 cluster headache patients have participated in the study. Despite a double-check of the neurologist’s ICHD-II diagnosis of cluster headache (7), we questioned the diagnosis of one patient during follow-up. We therefore excluded this patient and present the characteristics of seven patients (Table 1; Figures 1–2).
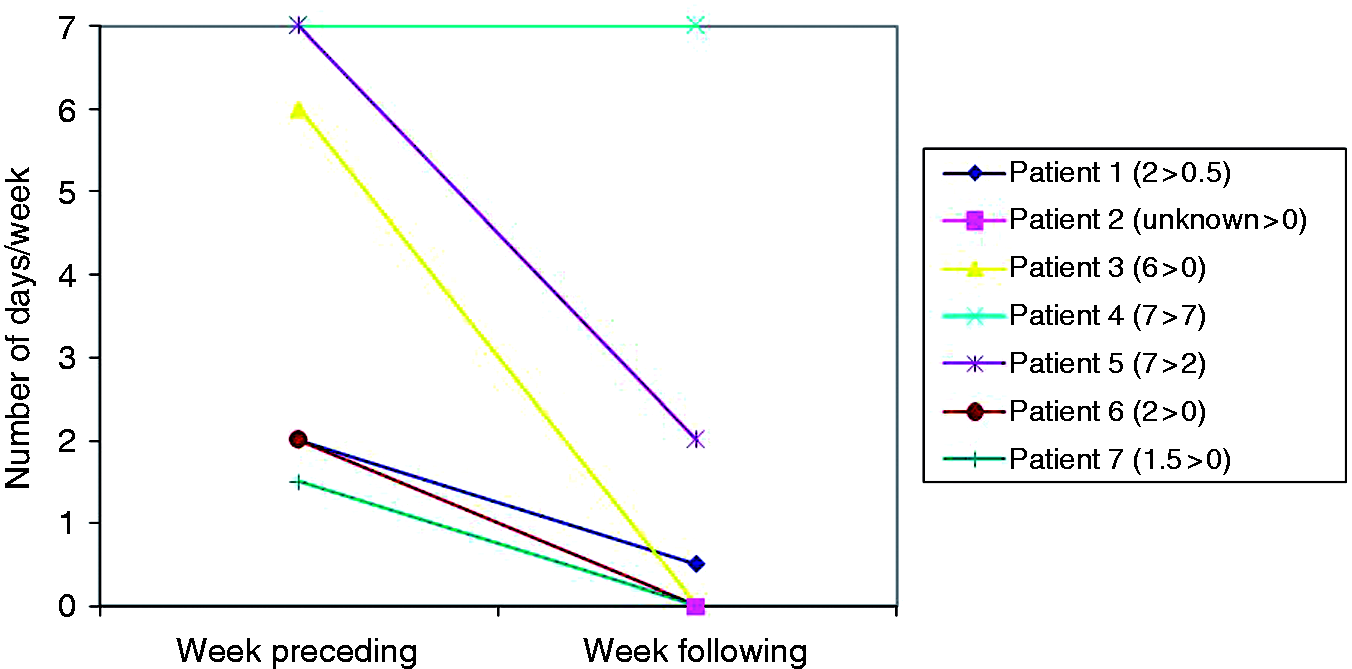
Estimated mean number of cluster headache attacks/day in week preceding and following study participation. Estimated number of days/week in which the patient experienced a cluster headache attached in the afternoon/evening (for patient 3: at night). Patient and cluster headache characteristics. CCH: chronic cluster headache; CH: cluster headache; ECH: episodic cluster headache; first: first cluster; ADHD: attention deficit hyperactivity disorder; TIA: transient ischaemic attack; CEA: carotid endarterectomy; DM: diabetes mellitus; COPD: chronic obstructive pulmonary disease; M: man; sc: subcutaneous; od: once daily; b.i.d.: twice daily; t.i.d.: three times per day; q.d.s.: four times per day; tab: tablet. All retrospective answers presented in this table were given by the patients at given number of days following study participation. The patient has not taken sumatriptan tablets in the 6 days before study participation, according to the study protocol. The patient uses oxygen at a flow rate of 12 L/min during 15 minutes as CH attack treatment. The patient had a maximal period of CH attack freedom in 5 years before study participation of 1 week.

Despite questioning on exclusion criteria and instructions on medication intake beforehand, four patients satisfied at least one exclusion criterion at time of study participation. We chose for continued study participation with complete mention of the satisfied exclusion criteria. All patients located their pain at least in the first trigeminal division.
The pinprick-like pain of the nociception specific stimulation was tolerated by all patients, although in one patient (no. 2) a low current of 0.9 mA had to be used to retain tolerability. Patients underwent a median of six stimulation sequences and, except for one patient, who underwent seven stimulation sequences, the first, third and higher numbered sequences were given at intervals of 2 hours. None of the patients experienced a spontaneous attack during the clinical study time of median 8.4 hours.
The chance of occurrence of an attack during study time could be considered ‘high’ in patients no. 3–5 (Figure 2). However, patient no. 5 had a dose increase of verapamil 2 days before study participation.
The fact that patients no. 2, 3 and 7 completely stopped experiencing cluster headache attacks since study participation was unexpected and of even more interest. Patients no. 2 and 3 were studied while they were 6 and 8.5 months in their first cluster and their sustained attack freedom lasted for at least 133 and 128 days (i.e. time until retrospective inquiring) respectively. Patient no. 7 had chronic cluster headache and was retrospectively questioned only 13 days following study participation. He was experiencing his longest time of attack freedom since onset of chronic cluster headache 5 years before. In contrast to these striking frequency decreases, one chronic cluster headache patient (no. 4) described an increase in attack frequency in the week following study participation. This increase, however, was not sustained, as this patient experienced 6 days of attack freedom 3 months following study participation. We have no data of the period in between.
Discussion
In this study on cluster headache pathophysiology, we may have found a serendipitous discovery that is of importance for future prophylactic treatment studies. All our seven cluster headache patients did not experience any attack during study participation using 2-hourly, nociception specific, transcutaneous stimulation (NSTS) sequences on the bilateral supraorbital nerves to elicit nociception specific blink reflexes. Furthermore, three of seven cluster headache patients have not experienced any attack following study participation.
Non-invasive supraorbital nerve stimulation
The supraorbital nerve can easily be stimulated in a non-invasive way, transcutaneously. In a recent double-blind trial (PREMICE) in migraine patients, bilateral, daily transcutaneous stimulation of the supraorbital and supratrochlear nerves was proven effective. No adverse or side effects were reported (8). A prophylactic effect of punctual transcutaneous electrical nerve stimulation in migraine patients, after more than 6 months, was shown in 1988, although methodological concerns may be raised in that study (9).
NSTS as possible prophylactic therapy offers advantages over invasive stimulation and pharmacological therapy. First, side effects are few. It is known to produce a pinprick-like pain at intensities of more than 0.6 mA (6). Furthermore, there is a possible counter effect of an increase in cluster headache attack frequency, as experienced by patient no. 4. A second advantage of NSTS is the non-invasiveness of the procedure. It gets around the known side effects of invasive peripheral neuromodulatory approaches, which include electrode migration, local infections and depletion of the implantable battery (4). Furthermore, as there are no fixed electrodes, stimulation can be easily applied on both sides and for a certain period of time, preventing the problem of side shift and allowing episodic cluster headache patients to be treated by neuromodulation, respectively.
Neurostimulation and cluster headache pathophysiology
At present, little is known about cluster headache pathophysiology and mechanism of action of neurostimulation. Peripheral nerve stimulation is traditionally thought to modulate the intrinsic electrical, afferent impulses travelling to the brainstem and higher. The ‘gate- control theory’ of Melzack and Wall deals with the influence of a competition between nociceptive and innocuous signals on second-order neurons (10,11), the latter signals transmitted by A-beta fibres. One may question the applicability of this theory to NSTS of the supraorbital nerve, in which nociception specific stimulation of trigeminal A-delta afferents seems to suppress the transmission of the other nociceptive (i.e. headache) signal on a segmental level. Furthermore, mediation in the analgesic effect of descending pain inhibitory pathways through stimulation of ascending tracts by peripheral nerve stimulation has been suggested (11). In addition to a central influence, Reed and colleagues speculated on a possible relationship with the only partial convergence of trigeminal and occipital neurons on the unilateral trigeminocervical complex (12). In that way, direct stimulation of the terminal branches of the trigeminal nerve in particular could play an additional role.
It is obvious that our findings need to be interpreted carefully and that further studies are required. There are several limitations in our present case series, including the absence of a control group, some violations to the study protocol, the heterogeneity in clinical study time, stimulation characteristics, group of patients and data collection, and the small sample size.
In conclusion, bilateral, time contingent, nociception specific, transcutaneous supraorbital nerve stimulation could have a prophylactic effect in episodic and chronic cluster headache. The suggested effect emerged as serendipity in a study using the nociception specific blink reflex to investigate the effect of oxygen treatment on medullary interneurons in cluster headache. Given its limited side effects and its non-invasive nature, further studies are required.
Clinical implications
Because even peripheral invasive neuromodulatory approaches carry a certain risk of device- and surgery-related side effects, non-invasive/transcutaneous nerve stimulation is of particular interest. Bilateral, time contingent, nociception specific, transcutaneous supraorbital nerve stimulation may have a prophylactic effect in episodic and chronic cluster headache, and further studies are required.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
Acknowledgements
The authors wish to thank M.P.W.A. Houben, J. Haan and M.D. Ferrari for their advice on the protocol of the study ‘Blink reflex changes following oxygen therapy in active cluster headache’.
