Abstract
Background
Several previous studies have investigated cortical abnormalities, specifically cortical thickness, in patients with migraine, with variable results. The relatively small sample sizes of most previous studies may partially explain these inconsistencies.
Objective
To investigate differences of cortical thickness between control subjects and migraineurs in a large cohort.
Methods
Three Tesla MRI data of 131 patients (38 with and 93 without aura) and 115 control subjects were analysed. A vertex-wise linear model was applied controlling for age, gender and MRI scanner to investigate differences between groups and determine the impact of clinical factors on cortical thickness measures.
Results
Migraineurs showed areas of thinned cortex compared with controls bilaterally in the central sulcus, in the left middle-frontal gyrus, in left visual cortices and the right occipito-temporal gyrus. Frequency of migraine attacks and the duration of the disorder had a significant impact on cortical thickness in the sensorimotor cortex and middle-frontal gyrus. Patients without aura showed thinner cortex than controls bilaterally in the central sulcus and in the middle frontal gyrus, in the left primary visual cortices, in the left supramarginal gyrus and in the right cuneus. Patients with aura showed clusters of thinner cortex bilaterally in the subparietal sulcus (between the precuneus and posterior cingulate cortex), in the left intraparietal sulcus and in the right anterior cingulate.
Conclusion
These results indicate cortical abnormalities in specific brain regions in migraineurs. Some of the observed abnormalities may reflect a genetic susceptibility towards developing migraine attacks, while others are probably a consequence of repeated head pain attacks.
Introduction
Morphological abnormalities in the brain of patients with migraine have been previously identified in both cortical and subcortical structures (1). However, previous studies on cortical abnormalities showed inconsistent results. Although some studies reported a thicker cortex in patients with migraine compared with control subjects in the somatosensory cortex (2,3), visual areas (4), frontal lobe (3) and temporo-occipital gyrus (5), areas with thinned cortex were also observed, mainly in the frontal lobe (6) and in the somatosensory cortex (5,7). Additionally, a study on a relatively large group of patients with migraine (56 patients and 28 controls) did not show cortical abnormalities compared with control subjects at all (8).
The interpretation of these results is further complicated by the complex relationship between cortical abnormalities and clinical/demographic factors. While some studies did not report any correlation (5,9) between cortical thickness (CTh) and clinical factors, other studies showed either linear (3,6) or non-linear (10,11) relationships. In this regard, Maleki and colleagues (10) reported thicker cortex in the somatosensory cortex, in the anterior cingulate cortex and the inferior temporal gyrus in patients with a high frequency of migraine attacks compared with patients with a low frequency of attacks and control subjects. Interestingly, patients with a low frequency of attacks showed thinner cortex in those areas not only compared with patients with a high frequency, but also compared with control subjects.
Other characteristics of the migraine symptomatology such as headache lateralization may impact the morphology of the respective hemisphere. Indeed, Hougaard and colleagues (6) observed a cluster of thinner cortex in the frontal lobe on the headache side compared with the non-headache side. Moreover, there is evidence that demographic factors may affect the structure of the brain of patients with migraine and control subjects in a differential way. Chong and colleagues (12) showed an accelerated age-related thinning in the somatosensory cortex bilaterally, in the right temporal pole and in the right fusiform gyrus of migraineurs compared with control subjects. In contrast, Maleki and colleagues (9) reported a lack of age-related cortical thinning in the insula of a group of 46 female patients compared to controls.
Overall, these results point towards a complex pattern of morphological abnormalities in migraine influenced by methodological, clinical and demographic factors.
In the present study, we aimed at investigating CTh and its relationship to clinical factors in a large, multicenter cohort of patients with migraine in order to account for population variability.
Methods
Subjects
MRI and clinical data from four academic headache centres were previously pooled to study subcortical changes in migraine (13). The same cohort of patients and control subjects was now investigated to study cortical abnormalities. The participating centres were located in Germany (Munich and Hamburg), Denmark (Glostrup) and the United States (San Francisco). Data analysis took place in Basel (Switzerland). The structural MRI data provided by the centers had originally been acquired in the context of local functional imaging studies to exclude structural abnormalities and for co-registration purposes.
Written informed consent had been obtained from the participants at each centre when the data were originally acquired. The original study protocols were approved by the local ethics committees and conducted in accordance with the Declaration of Helsinki.
Two hundred and forty six subjects (131 patients with migraine, 38 with aura and 93 without aura, and 115 control subjects) with an age range of 18–55 years were included in the study. All patients met the criteria for episodic migraine as defined by the International Classification of Headache Disorders, second edition (14). Patients were studied interictally and none of them had medication overuse headache. Patients with a previous history of cardiovascular or neurological disease other than migraine were excluded. Infrequent episodic tension-type headache was allowed in both groups. Demographic and clinical characteristics of the study participants have been previously reported (13). In brief, control subjects had an average age of 29.1 years (range 19–52) and migraineurs were on average 30.8 years old (range 19–53). Eighty three percent of migraineurs and 70% of control subjects were female. Patients with migraine had an average duration of the disorder of 14.1 ± 8.5 years (range:1–41); the average number of migraine days per month was 3.3 ± 2.5 (range: 0.75–15). Fifty patients had a lateralized headache (left: 23, right: 27), 68 had bilateral headache and in 10 patients the side was not recorded.
MRI acquisition
All MRI data were acquired on 3 Tesla systems using eight or 12 channel head coils. The four centres used different MRI systems and vendors (two Siemens TrioTim, one General Electric Sigma and two Philips Achieva). Details of the scanners used, and the applied sequences, have been published (13). In brief, high-resolution T1-weighted data with 156–170 slices and an isotropic spatial resolution of 1 mm were acquired in each subject (TE 1.5–4.6 ms, TR 2300–9000 ms).
MRI analysis
Cortical thickness was estimated using FreeSurfer (Version 5.3; http://freesurfer.net/), a well-established and extensively described pipeline (15). High reliability for CTh estimated by FreeSurfer has been previously reported across MRI scanners and vendors in both cross-sectional and longitudinal studies (16,17).
The quality assessment of the tissue segmentation was performed by an experienced user. The procedure needs a first check after skull stripping and a second check after the tissue segmentation. In the majority of the MRIs (80%), the parameters for skull stripping were slightly adjusted to completely remove non-brain structures. The white matter segmentation was corrected in only 10 cases, using the standard procedure implemented in FreeSurfer. The final segmentation was then reviewed and finally accepted by a second expert blinded with respect to group allocation (patients and controls).
The anatomical characterization of the significant clusters of CTh abnormalities was based on the Destrieux cortical parcellation (18). The Destrieux parcellation applies a surface-based labelling of sulco-gyral structures based on the Duvernoy nomenclature (19). Specifically, the gyral cortex was defined as the one seen on a 3D reconstruction before inflation and the hidden part was labelled as sulcus (18).
Statistical analysis
CTh was compared between groups using a vertex-wise ANCOVA model including age, gender and MRI scanner as covariates. The results were corrected for multiple comparisons using the false discovery rate (FDR) approach with an FDR-adjusted p-value of 0.05. In patients with migraine, a hierarchical multiple linear regression (MLR) analysis was performed on each significant cluster to investigate the relationship between mean CTh and clinical variables. Specifically, the MLR model included age, gender and MRI scanner in the first block and the clinical variables in the second block (duration of the disorder and frequency of migraine attacks per month).
The ANCOVA model described above was also used to perform an exploratory analysis to compare patients with migraine with aura (MwA) to patients with migraine without aura (MwoA) and both subgroups to control subjects. As the overall MwoA group differed from the MwA group in terms of demographic characteristics, a pseudo randomly selected subgroup of patients with MwoA matched for age, gender and MRI scanner (mean age: 32.5 ± 10.1 years, range 19–53 years; 31 women and seven men; mean disease duration: 15.8 ± 9.8 years, mean days with headache per month: 3.5 ± 2.8), was compared with patients with MwA (mean age: 32.8 ± 8.7, range: 21–50; 31 women and seven men; mean disease duration: 13.3 ± 8.1; mean days with headache per month: 3.2 ± 1.9). Moreover, a subgroup of control subjects matched for age, gender and MRI scanner (mean age 31.7 ± 8.7, range: 22–48; 32 women and seven men) was compared with patients with MwA. The subgroup of patients with MwoA (mean age: 28.5 ± 9; 78 women and 15 men; mean disease duration: 14.4 ± 8.8; mean days with headache per month: 3.4 ± 2.7) was compared to the whole sample of control subjects.
For each ANCOVA model, the following assumptions were tested: Normality was tested using the Shapiro–Wilk test (20), linearity and homoscedasticity using the Levene’s test (21), and homogeneity of the regression slopes was verified by testing the significance of the interaction terms. For each MLR model, the following assumptions were tested: Linearity and homoscedasticity were tested by plotting the regression standardized predicted value. The assumption of normality of the residuals was tested using the Shapiro–Wilk test and the probability-probability plot. Autocorrelations were tested by using Durbin–Watson and collinearity by computing the variance inflated factor. All analyses were performed using R (http://www.r-project.org/).
Results
Compared to control subjects, patients with migraine had a significantly thinner cortex bilaterally in the central sulcus, the left middle frontal gyrus, the left primary visual cortex (V1), the left secondary visual cortex (V2) and in the right occipito-temporal area (see Table 1 and Figure 1). The regression analysis showed a significant impact of clinical factors on CTh in the left middle frontal gyrus (adjusted-R2 = 0.38, F(8, 118) = 10.82, p < 0.0001) and in the left central sulcus (adjusted-R2 = 0.32, F(8, 118) = 8.39, p < 0.0001) after regressing out the effects of age, gender and MRI scanner. Specifically, the frequency of migraine attacks was a significant predictor for CTh in the left middle frontal gyrus (beta = −0.1, p < 0.05) and in the left central sulcus (beta = −0.14, p < 0.001). The duration of the disorder was a significant predictor for CTh in the left middle frontal gyrus (beta = −0.01, p < 0.05) and showed a trend in the left central sulcus (beta = −0.01, p < 0.1).
Significant clusters of thinner cortex in the patients with migraine compared with control subjects. Data were corrected for multiple comparison using false discovery rate. FDR adjusted p-values of 0.05 and 0.1 are reported. Brain regions with significant differences in cortical thickness for the comparisons between control subjects and patients.

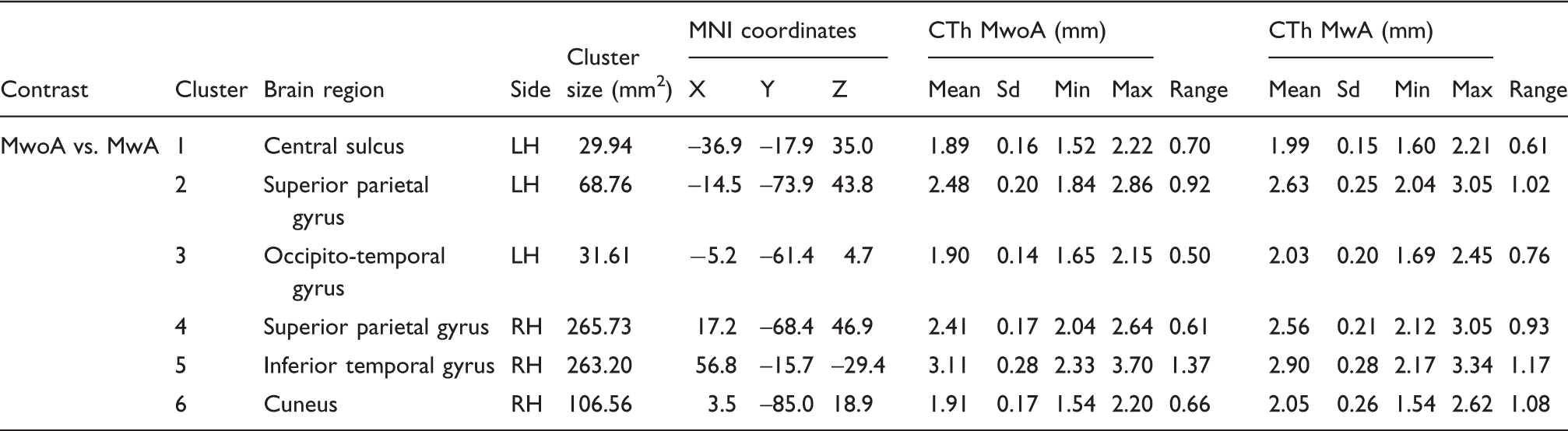
Patients with MwoA showed thinner cortex bilaterally than control subjects in the central sulcus and in the middle frontal gyrus, in the left V1 and V2 areas, in the left supramarginal gyrus and in the right cuneus (Table 1). Patients with MwA showed clusters of thinner cortex bilaterally in the subparietal sulcus (between the precuneus and the posterior cingulate cortex), in the left intraparietal sulcus and in the right anterior cingulate cortex. Moreover, patients with MwA showed an area of thicker cortex compared with control subjects in the posterior portion of the right middle temporal gyrus.
Brain regions with significantly different cortical thickness for the comparisons between patients with migraine with and without aura.
All ANCOVA and MLR models met the assumptions. Hence, they were accurate for the sample and generalizable to the population level.
Discussion
In the present study, we investigated cortical thickness changes in a large cohort of patients with episodic migraine. The analysis on the whole dataset showed a significantly thinner cortex in patients compared with control subjects in the bilateral central sulcus, in the left middle frontal gyrus, in the left visual cortices (primary and secondary) and in the right occipito-temporal gyrus. The frequency of migraine attacks and the duration of the disorder were significant predictors showing an inverse relationship with the CTh in the left middle frontal gyrus and in the left central sulcus in its motor and somatosensory portion. There is evidence that all these brain regions are part of a brain network involved in different aspects of pain processing (22).
The somatosensory cortex is particularly relevant for pain processing. Indeed, it is thought to play an important role in the processing of sensory aspects of pain such as localization and intensity (23,24). Structural abnormalities in the somatosensory cortex of patients with migraine have been reported in previous studies; however, in an opposite direction. Indeed, DaSilva and colleagues (2), using a region of interest (ROI) approach, reported a thicker somatosensory cortex in patients with migraine compared with control subjects. Thicker somatosensory cortex has also been reported using a vertex-wise analysis by Kim and colleagues (3) in a group of patients with MwoA. Interestingly, structural abnormalities in the somatosensory cortex seem to be related to clinical (10) and demographic (12) factors.
Areas within the frontal lobe are also part of the brain network involved in pain processing. In particular, the middle frontal gyrus, as part of the prefrontal cortex, is thought to be involved in the cognitive evaluation and modulation of pain (25). We observed a thinned cortex in the middle frontal gyrus of patients with migraine compared with control subjects, while previous studies reported a thicker cortex (3,5).
Another region of thinned cortex was observed in the right occipito-temporal gyrus (fusiform gyrus). The fusiform gyrus is involved in the integration of higher-order visual information such as colour, body and face recognition (26,27). However, it has also been associated with pain perception and anticipation (28,29). Previous studies on migraine reported CTh abnormalities in this brain region (5), and a non-linear relationship between CTh in the occipital-temporal area and the frequency of migraine attacks was also reported (10). Moreover, increased grey matter (GM) volume in the fusiform gyrus of patients with MwA and reduced GM volume in MwoA was observed in a paediatric population (11). Interestingly, altered age-related cortical thinning in the fusiform gyrus of migraineurs compared with control subject was also reported (12).
In the present study, we also observed CTh differences between patients and control subjects in primary and secondary visual cortices. Altered CTh in visual brain regions was previously described by Granziera and colleagues (4). These authors, using a ROI approach, reported a thicker cortex in V3a and MT+ of patients with migraine. Moreover, a recent study that investigated patients with MwA, their twins discordant for MwA, and migraine-free control subjects showed thicker cortex in V2 and V3a in patients with MwA compared to control subjects and in V2 in MwA compared to the co-twins without aura (30). Chong and colleagues reported a thicker cortex of visual areas in migraineurs with interictal photophobia compared to those without interictal photophobia (31). Unfortunately, we were unable to confirm this interesting finding due to the lack of individual clinical data on this specific symptom in the present study.
The analysis performed on the migraine subgroup revealed clusters of thinner cortex in the somatosensory cortex, middle frontal gyrus, primary and secondary visual areas in patients with MwoA compared with control subjects, as observed in the analysis of the whole sample. Additional small clusters of thinned cortex were observed in the left supramarginal gyrus and in the right cuneus. The cuneus is part of the occipital lobe and involved in visual processing, including the monitoring of changes in ocular position (32). The supramarginal gyrus is involved in a variety of cognitive functions such as visual attention and imagery (33,34). Structural (11) and functional (35) abnormalities in the supramarginal gyrus have also been reported in other studies in both patients with MwoA and MwA. Interestingly, a reduced pain-related activity of the supramarginal gyrus has been reported in medication overuse headache patients (36).
The comparison between MwA and control subjects showed a different pattern of cortical abnormalities compared with that observed in the entire patient sample. Patients with MwA had a thinner cortex in the subparietal sulcus, the intraparietal sulcus and in the anterior cingulate cortex compared with control subjects. Moreover, a cluster of thickened cortex was observed in the middle temporal gyrus. The subparietal gyrus is located between the precuneus and the posterior cingulate cortex. These brain regions are densely connected with the rest of the brain modulating the activity of various brain networks involved in higher order cognitive functions such as memory, learning, arousal and awareness (37). A thinner cortex in the precuneus of patients was previously reported by Messina and collaborators (5). The intraparietal sulcus is involved in cognitive functions such as visual attention, saccades and visual control of reaching and grasping. Structural and functional abnormalities in the intraparietal sulcus of patients with migraine were reported by previous studies as well (5,38). The anterior cingulate cortex is thought to be a key region of the brain network involved in the processing of pain and, specifically, in the processing of the affective component of pain (39). Reduced GM in the anterior cingulate cortex has also been reported by previous studies that investigated morphological changes using voxel brain morphometry (VBM; (40)).
The exploratory analysis aimed at comparing patients with and without aura showed that patients with MwoA had a thinner cortex in somatosensory areas, the occipito-temporal gyrus and the cuneus compared to patients with MwA, regions also observed in the whole sample analysis. Interestingly, patients with MwoA also showed a cluster of thinner cortex compared to MwA in the bilateral superior parietal gyrus. Due to its connection with the somatosensory cortex and insula, the superior parietal gyrus is thought to be involved in the attentional modulation of pain (41).
Although the localization of cortical thickness abnormalities observed in the whole sample analysis is consistent with previous reported results, the direction of these changes is only partially in agreement. Indeed, previous studies reported both thinner and thicker cortex in patients with migraine compared with controls. The thinner cortex observed in the present study is consistent with previous results from VBM studies, which reported a reduction of GM density in patients. A recent meta-analysis on VBM results in patients with migraine showed a significant GM reduction in the middle and inferior frontal gyrus, insula and inferior temporal gyrus (42).
The pathophysiological mechanisms underlying the observed cortical thinning of migraineurs compared with control subjects, as well as the difference between patients with and without aura, are still unclear. One may speculate that the differences observed at the cortical level relate to brain plasticity induced by attacks (e.g. with repeated pain stimulation) with changes of dendritic complexity or numbers of synapses. The genetic background may also play a role and cause such changes. Cortical thinning as the result of repetitive pain has been described in other chronic or episodic pain conditions (e.g. trigeminal neuralgia and cluster headache (30,43)). This view is supported by the inverse relationship between CTh and clinical factors such as disease duration and frequency of migraine attacks observed in the present study. However, this relationship was statistically significant only for the middle frontal gyrus and the somatosensory cortex. Therefore, it could be that repeated attacks and the genetic background impact brain morphology in distinct brain networks. In this regard, a study on twins with and without MwA showed that the differences observed between the two groups of twins were only partially overlapping with the cortical abnormalities observed in the comparison between twins with MwA and control subjects (30). Moreover, differences between patients with and without aura may also be related to other factors such as altered metabolism in specific brain regions related to the aura phenotypes (44).
The observed variability among studies in terms of involved brain regions and decreased or increased cortical thickness could relate to both physiological and methodological reasons. There is some evidence suggesting that demographic factors such as age may have a differential impact on the brain morphology of migraineurs compared to healthy subjects. Indeed, Chong and colleagues (12) reported a different age-related cortical thinning pattern in the precentral gyrus and in the temporal lobe of patients with migraine compared to control subjects. Changes in the brain of migraineurs may relate to the migraine cycle as well. Interestingly, Coppola and colleagues (45) reported an opposite direction of morphological changes according to the migraine cycle, with a lower GM density in patients compared to controls during the interictal phase and an increased GM density in the ictal phase. In the present study patients were scanned interictally, but, the time between the MRI scan and the next/previous migraine attack was not recorded.
The combination of these sources of variability with the small sample size enrolled in the majority of previously published studies limits the generalizability of the obtained results to the population of patients with migraine, rendering them valid only at the group level. These considerations point out important sources of variability, which are difficult to take into account in small groups of patients, emphasizing the need for larger studies. Moreover, longitudinal studies are needed to clarify the direction and the role of CTh abnormalities in the pathophysiology of migraine.
Clinical implications
Patients with migraine showed cortical thinning in the central sulcus, the left middle frontal gyrus, the left visual cortex and in the right occipito-temporal area compared with control subjects. Cortical abnormalities may serve in the future as a biomarker of the disorder and/or treatment efficacy. Differences of cortical thickness in the parietal lobe between patients with migraine with and without aura observed in the present study may help to classify different phenotypes of the disease.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
