Abstract
Introduction: The increased risk of cerebro- and cardiovascular disease in migraineurs may be the consequence of a systemic condition affecting whole body vasculature. At cerebrovascular level, this may be reflected by interictal global or regional cerebral perfusion abnormalities. Whether focal perfusion changes occur during interictal migraine has not been convincingly demonstrated.
Methods: We measured brain perfusion with dynamic susceptibility contrast magnetic resonance imaging (DSC-MRI) in 29 interictal female migraineurs (12 migraine with aura (MA), 17 migraine without aura (MO)), and 16 female controls. Perfusion maps were compared between these groups with a voxelwise (p < 0.001, uncorrected, minimum cluster size 20 voxels) and a region-of-interest approach.
Results: In whole brain voxelwise analyses interictal hyperperfusion was observed in the left medial frontal gyrus in migraineurs and in the inferior and middle temporal gyrus in MO patients, in comparison with controls. Hypoperfusion was seen in the postcentral gyrus and in the inferior temporal gyrus in MA patients and in the inferior frontal gyrus in MO patients. Additional focal sites of hyperperfusion were noted in subgroups based on attack frequency and disease history. Region-of-interest analyses of the pons, hypothalamus, occipital lobe, and cerebellum did not show interictal perfusion differences between migraineurs and controls.
Conclusions: We conclude that interictal migraine is characterized by discrete areas of hyper- and hypoperfusion unspecific for migraine pathophysiology and not explaining the increased vulnerability of particular brain regions for cerebrovascular damage.
Keywords
Introduction
Migraine is a prevalent neurovascular disorder, characterized by recurrent attacks of disabling headache accompanied by dysfunction of the autonomic nervous system. In up to a third of the patients, neurological (mostly visual) aura symptoms precede or accompany the headache phase of migraine attacks (1). Migraine, especially migraine with aura (MA), has been identified as an independent risk factor for clinical and subclinical brain infarction (2,3). The posterior circulation territory, notably the cerebellum, seems to be specifically vulnerable (2,4,5). Furthermore, female migraineurs both with and without aura are at increased risk of deep white matter and brainstem hyperintensities (2,5,6). Repetitive physiological and biochemical changes during migraine attacks, including changes in brain perfusion, are among the mechanisms proposed to explain or contribute to the increased risk of ischemic brain lesions (7).
Besides the increased risk of cerebrovascular complications in migraineurs, there is increasing evidence that migraine is also independently associated with other (ischemic) vascular disorders, including angina pectoris, myocardial infarction, claudication, and retinopathy (7,8). Given that it is unlikely that these conditions are all direct consequences of migraine attacks, or that they induce migraine themselves, a more plausible interpretation of these associations is that both migraine and these vascular disorders are influenced by subtle systemic changes in the vessel wall and its interaction with circulating coagulation factors, platelets, and other substances. The diversity of organs involved suggests that a systemic condition has a role in the pathogenesis of these conditions. Several observations in migraineurs support this concept, including interictal evidence of endothelium (or endothelium-dependent) dysfunction, impaired cerebrovascular reactivity, reports on ictal and interictal generalized or coronary artery vasospasm, impaired brachial artery compliance, increased aortic stiffness, and hypercoagulability, which all seem to be independent of the coincidence of established cardiovascular risk factors (9).
Because several of the observed systemic changes in migraineurs are persistent threats, and because they could result in changes in brain perfusion, detection of changes in brain perfusion in migraineurs could be expected interictally. Earlier, single photon emission tomography (SPECT) was reported to show gross differences in cerebral hemodynamics between interictal migraineurs and control participants, but results were contradictory (10–15). More recent perfusion studies, using positron emission tomography (PET) (16–20) and dynamic susceptibility contrast magnetic resonance imaging (DSC-MRI) (21–24), concentrated on the ictal phenomena, so it has still not been convincingly proven whether differences in cerebral hemodynamics are present or absent in the interictal stage of migraine in comparison with headache-free control participants.
In the current study, we investigated interictal groups of MA and migraine without aura (MO) patients and controls, using DSC-MRI. DSC-MRI has several advantages over earlier performed PET and SPECT studies, including a better spatial resolution with whole brain coverage and a higher signal-to-noise ratio (SNR), allowing voxelwise detection of small perfusion differences (25). We performed unbiased whole brain voxelwise analyses of cerebral blood flow (CBF), cerebral blood volume (CBV), mean transit time (MTT), and time-to-peak (TTP) maps, but also performed region-of-interest (ROI) analyses in predefined regions that have been reported to show perfusion changes during the different stages of migraine, or that have been reported to be specifically at risk for ischemic lesion development, such as the cerebellum.
Methods
Participant population
Thirty female migraine patients (13 MA: mean age 42.9, range 28–52; 17 MO: mean age 47.6, range 38–57) and 17 female control participants (mean age 39.9, range 21–55) participated in this study. Migraine patients were enrolled through advertisements in newspapers and magazines. Migraineurs were diagnosed with MA or MO according to the criteria of the Headache Classification Committee of the International Headache Society (IHS) (1) at the Department of Neurology (by GGS); headache-free, unrelated control participants without a history of migraine attacks or other headaches were recruited by local advertisement and enrolled in the study. A total of 25 migraine patients were able to estimate their average number of attacks and/or duration of headache. None of the migraine patients were taking prophylactic medication. All were headache-free for at least 7 days before and 2 days after the MRI examination (verified by telephonic consultation). The study was approved by the institutional review board and all participants gave written and informed consent.
Neuroimaging protocol
T2-weighted turbo spin echo images (repetition time (TR) 4741 ms, echo time (TE) 80 ms, echo train length 16, acquisition matrix 448 × 392 mm, field of view (FOV) 224 × 180 mm2, 48 slices of 3 mm thickness) and fluid-attenuated inversion recovery (FLAIR) (TR 10128 ms, TE 120 ms, echo train length 36, acquisition matrix 224 × 224, FOV 224 × 180 mm2, 48 slices of 3 mm thickness) were acquired on a 3.0 Tesla MRI system (Achieva, Philips Medical Systems, Best, the Netherlands) and evaluated by an experienced neuroradiologist (MCK). Two cases showing structural brain abnormalities that could have impeded or confounded perfusion image post-processing were excluded from further analyses: one control participant with a frontal cortical infarct and one MA patient with confluent white matter hyperintensities (WMHs). Small, punctuate deep WMHs were not considered an exclusion criterion.
DSC-MR imaging was performed using an 8-channel receive array head coil; 0.2 ml/kg bodyweight gadolinium DTPA (Magnevist®, Schering) was injected intravenously at 5 ml/s followed by a saline chaser of 25 ml (injected at 5 ml/s (20 ml) and 2 ml/s (5 ml)). PRESTO (PRinciples of Echo-Shifting with a Train of Observations, a 3D ultrafast gradient echo sequence combining whole brain coverage with T2*-weighted imaging (26)) was used for the acquisition with the following parameters: TR 17 ms, TE 26 ms, data matrix 64 × 53 mm (zero-filled to 128 × 108 mm), flip angle 5°, FOV 224 × 168 mm2, SENSE factor of 2.4, 48 slices of 3 mm thickness, number of echoes in a echo train 21, and 60 segments per volume resulting in a dynamic scan time of 1.1 sec.
Post-processing
The perfusion maps of CBF, CBV, MTT, and TTP were generated using software developed at the Massachusetts General Hospital using block-circulant singular value decomposition with oscillation index regularization (27). This program requires a manual selection of the arterial input function (AIF, the passage of contrast agent through a major brain-feeding artery). At least eight voxels near the middle cerebral artery were selected to form the global AIF used in the deconvolution.
The signal drop in the T2*-weighted images resulting from the contrast agent passage was converted to the concentration contrast agent (C(t)) using

The deconvolution was performed over the first and second passage of the gadolinium concentration passage with a noise threshold of 0.4 and an oscillation threshold of 0.095.
The CBF maps were used to determine the transformation parameters for coregistration of all perfusion maps spatially to the PET template in SPM5 (Wellcome Trust Centre for Neuroimaging, Institute of Neurology, UCL, London UK (28)). To improve the spatial normalization further, an average CBF map was constructed over all participants and the individual CBF maps were coregistered to that average CBF template together with all other perfusion maps. The relative CBF and relative CBV maps were converted to quantitative CBF and CBV maps by setting the white matter CBF to 22 ml/100 g/min (29). The white matter segmentation for this quantification step was performed in SPM5 on the CBF map; the segmentation mask consisted of all voxels with a 95% or higher certainty that the voxel is white matter.
Statistical analyses
All perfusion maps were smoothed using an isotropic Gaussian kernel (full width at half maximum of 8 mm). Whole brain voxelwise comparison was performed, comparing interictal scans of MA, MO, and control participants using one-way analyses of variance implemented in SPM5, correcting for age. Further subanalyses, also corrected for age, were performed comparing controls with migraineurs with a high (HF, > 2 attacks/month, n = 12) and a low (LF, ≤2 attacks/month, n = 13) attack frequency and to migraineurs with a long (LH, > median of 28 years, n = 13) and a short (SH, ≤28 years, n = 12) disease duration. For statistical tests, the probability threshold was set at p <0.001, uncorrected for multiple comparisons, with a cluster extent of minimal 20 neighboring voxels.
Next to voxel-based comparison, ROIs for the occipital lobe and cerebellum (constructed from the corresponding regions from the automated anatomical labeling (AAL) template for SPM5) (30), the pons and the hypothalamus (drawn manually and incorporated into the AAL template) were created to study the average distribution of perfusion parameters in these areas in the normalized, non-smoothed images. The choice of ROIs for pons, hypothalamus, and occipital lobe was based on previous reports of hemodynamic changes during migraine (16,18–20). The cerebellum was analysed specifically because of its suggested increased vulnerability for cerebellar infarction in migraineurs. Distributions of mean CBF, CBV, MTT, and TTP were compared between migraine patients and control participants using multivariate general linear models adjusted for age in SPSS for Windows, release 16.0.2 (Chicago, USA). Subanalyses were also carried out on the ROI data to investigate the influence of disease duration and attack frequency.
Results
After exclusion of the two participants with larger brain abnormalities, 45 female participants remained for analysis: 12 MA patients, 17 MO patients, and 16 control participants. Mean age (MA 42.9 ± 8.2 years, MO 47.6 ± 5.3 years, controls 39.9 ± 12.3, p > 0.05, one-way ANOVA) was not different across the groups. Migraineurs had a mean of 3.1 ± 2.2 attacks per month and a history of migraine of 28 ± 11 years.
The results of the voxelwise whole brain comparison between interictal migraineurs and controls are shown in Table 1 and Figures 1 and 2. We found a higher CBF in a part of the left medial frontal gyrus when analyzing the whole group of migraineurs vs. controls (Figure 1A). This difference also remained when assessing MA and MO groups separately vs. controls (not shown in Figure 1A). CBF was also increased in the right inferior gyrus in MO (Figure 1B). Decreased CBF was observed in parts of the right inferior temporal gyrus (Figure 1C) and the left postcentral gyrus (Figure 1D) in MA patients and in part of the inferior frontal gyrus in MO patients (Figure 1E). CBV was increased in part of the right inferior and middle temporal gyrus in MO (Figure 2). CBV reductions were not identified. In addition, no increases or decreases in MTT and TTP were observed between migraineurs and control participants.
Statistical parametric maps demonstrating increases (yellow) and decreases (blue) in DSC-MRI measured cerebral blood flow (CBF). Increases in CBF between migraineurs and controls in the medial frontal gyrus (A) and between MO patients and controls in the inferior temporal gyrus (B) and decreases in CBF between MA patients and controls in the inferior temporal gyrus (C) and postcentral gyrus (D) and between MO patients and controls in the inferior frontal gyrus (E) are superimposed on a single subject T1 template coregistered to the standard SPM5 PET template. Color bars represent Z-values. The left side of each picture is the right side of the brain. Statistical parametric maps demonstrating increases in DSC-MRI-measured cerebral blood volume (CBV) in MO patients compared with headache-free control participants in the middle temporal gyrus (A) and inferior temporal gyrus (B, C), superimposed on the single subject T1 template coregistered to the standard SPM5 PET template. The color bar represents Z-values. The left side of each picture is the right side of the brain. Significant differences in cerebral blood flow (CBF), cerebral blood volume (CBV), mean transit time (MTT), and time to peak (TTP) at the interictal stage between migraineurs (n = 29; MA n = 12, MO n = 17) and controls (n = 16) MA: migraine with aura, MO: migraine without aura, MNI: Montreal Neurological Institute; all significant differences p < 0.001, uncorrected, cluster extent threshold of 20 voxels; figures in parentheses are the mean [SD] CBF, CBV, MTT, and TTP values of corresponding clusters across participants per group.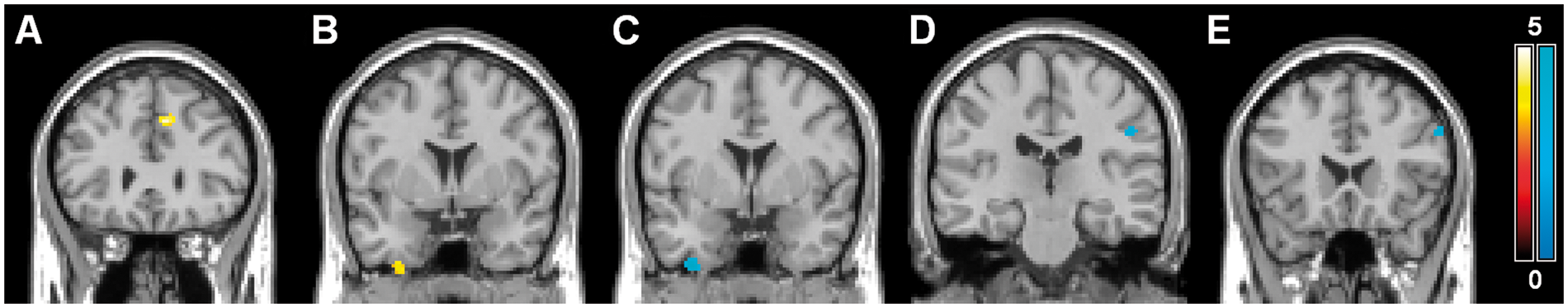


Significant differences in cerebral blood flow (CBF), cerebral blood volume (CBV), mean transit time (MTT), and time to peak (TTP) at interictal stage between high (HF, >2 attacks, n = 12) and low (LF, ≤2 attacks per month, n = 13) frequency migraine and controls (n = 16)

MNI: Montreal Neurological Institute; all significant differences p < 0.001, uncorrected, cluster extent threshold of 20 voxels; figures in parentheses are the mean [SD] CBF, CBV, MTT, and TTP values of corresponding clusters across participants per group.
Significant differences in cerebral blood flow (CBF), cerebral blood volume (CBV), mean transit time (MTT), and time to peak (TTP) at interictal stage between long (LH, >28 years, n = 13) and short (SH, ≤28 years of headache, n = 12) migraine history and controls (n = 16)

MNI: Montreal Neurological Institute; all significant differences p < 0.001, uncorrected, cluster extent threshold of 20 voxels; figures in parentheses are the mean [SD] CBF, CBV, MTT, and TTP values of corresponding clusters across participants per group.
The ROI analyses of CBF, CBV, MTT, and TTP in the pons, hypothalamus, occipital lobe, and cerebellum showed no differences between migraineurs and controls, nor between migraine subgroups and controls (Supplementary Table 1).
Discussion
To the best of our knowledge, this is the first explorative DSC-MRI study assessing brain perfusion characteristics and patterns in female migraine patients in an interictal state vs. headache-free control participants. In this relatively large sample of migraineurs (MA and MO) and control participants, our voxelwise comparison of perfusion maps identified some small areas of perfusion differences between migraineurs and controls, including both hyper- and hypoperfusion in frontal, parietal, and temporal regions in the interictal migraine brain. Similarly, a variety of additional areas of perfusion differences appeared in voxelwise subanalyses based on attack frequency and disease duration, including several focal areas in the frontal and parietal lobes, the putamen and the cerebellum. In these data, there seems to be a tendency towards areas with higher CBF values in migraineurs with higher attack frequencies, and for areas with lower CBV and MTT values in migraineurs with shorter disease history. Analyses assessing regional perfusion differences in the pons, hypothalamus, occipital lobe, and cerebellum between subgroups of interictal migraineurs and controls did not show significant differences.
Areas with altered interictal perfusion may reflect local interictal differences in neuronal metabolic activity or density. This could reflect a preexisting difference in local brain structure and/or function or be the consequence of repetitive migraine attacks or chronic adaptive mechanisms, leading to neuroplastic changes in cortical and subcortical structures. Shorter-term explanations include changes related to, for instance, the migraine cycle, the presence of some degree of interictal cerebrovascular dysregulation in migraineurs, or a (migraine disease-related) systemic condition altering vessel wall characteristics that influence regional blood flow. Changes could also be reactive; for example, episodic regional hypoperfusion might be followed by compensatory hyperperfusion. The findings in the subgroups with higher attack frequency and shorter migraine duration may relate to any of these potential mechanisms, although we were not able to ascertain the direction of such potential associations. It thus remains difficult to assess the primary nature of the perfusion changes, partly because the changes in individual perfusion parameters are inter-related (CBF = CBV/MTT, where CBF is depended on perfusion pressure and cerebrovascular resistance).
Most if not all currently identified areas of altered perfusion have earlier been reported in some way in structural imaging studies in migraine or other pain conditions. For instance, the area of higher CBF that we identified in the left medial frontal gyrus seems to colocalize with a reported area of gray matter reductions in migraine patients (31). Gray matter changes in the medial frontal gyrus have been reported to correlate with pain scores in another chronic pain condition, fibromyalgia (32), suggesting a correlation between morphology and function. Similarly, it is tempting to relate our findings in HF migraine patients of increased CBF in the postcentral gyrus to thickening of the somatosensory cortex in migraineurs, as was described in one earlier morphometric study and that was suggested to be an adaptation to repetitive migraine attacks (33).
Although thus seemingly altered interictal perfusion in certain brain areas in migraineurs might be co-localized with earlier reported structural or hemodynamic changes in the brains of patients with migraine and other pain conditions, we want to stress that the changes measured by DSC-MRI are small, a direct correlation between functional and structural changes is still lacking, and the nature of any of such potential associations remains unclear and difficult to interpret. For instance, higher or lower perfusion did not consistently relate to specific structural changes: hypoperfusion seems in some areas to be related to reported cortical thickening and in other areas with gray matter reduction. The interpretation of the hemodynamic differences is further hampered by the lack of a histopathological correlate for the described structural changes in migraine brains: gray matter volume or ‘density’ on MRI scans is influenced by neuron number and size, the amount of extracellular or microvascular fluid, gliosis, and atrophy. In this context, we should also consider that cell density is not necessarily directly related to the neuronal metabolic activity. Future studies may overcome some of these major interpretative limitations of our study by directly comparing (voxelwise) structural imaging measures with (voxelwise) perfusion imaging approaches.
We found no interictal differences in CBF, CBV, MTT, or TTP in the regional maps of the pons, hypothalamus, and occipital lobe, although perfusion changes have previously been identified in these areas during migraine attacks (16,18–20,22). Similarly, we found no interictal perfusion abnormalities in the cerebellum in the group of all migraineurs. These negative interictal findings may be explained for example by high inter- and intra-individual variance in cerebral hemodynamics, and of course they do not exclude the possibility of ictal perfusion changes in these areas. In the voxelwise analyses, however, we found focal increases in CBV in the cerebellum of SH migraineurs, accompanied by a longer MTT in the same area, compared with both controls and LH migraineurs. Further studies should assess whether these perfusion changes relate to the known increased vulnerability of the cerebellum to infarction.
All migraineurs in our study were scanned at least 7 days after the last migraine attack and none of them suffered their next attack within 2 days after scanning. Inter-individual variance in hemodynamics may, however, still have been influenced by the specific moment in the migraine cycle in which each individual participant has been examined; we regard this as a difficult-to-overcome limitation of our study. Future studies should assess the reproducibility of and/or variation in regional cerebral perfusion changes during different (interictal and ictal) time-points in the migraine cycle.
We are not aware of any PET or DSC-MRI studies that report perfusion differences in migraineurs during the interictal stage in comparison with control participants. There are, however, several reports on interictal CBF changes in regular migraine compared with controls, measured with SPECT, but results are contradictory. Lauritzen and Olesen did not find interictal CBF changes when comparing 11 migraineurs to 20 controls (14). Levine et al. reported general hypoperfusion in 15 MA and 12 MO patients in the posterior circulation territory, compared with 20 controls (15). Other studies reported single or multiple foci of interictal hypoperfusion in 43–67% of migraineurs (10,11). However, yet another SPECT study reported interictal global hyperperfusion in 50 MO patients (mainly located in frontal regions) and global hypoperfusion in 20 MA patients (mainly located in posterior regions), compared with control participants (12). These contradictory results may be due to the unreliability of visual evaluation of SPECT-derived CBF images in terms of picking up abnormalities in interictal migraineurs (13). The small focal areas of hyper- and hypoperfusion we observed in frontal, temporal, and parietal regions in migraine patients compared with controls seem to show overlap with some of the regions found in the aforementioned SPECT studies, but comparison between these different techniques remains complicated because of differences in post-processing, which is discussed further below.
Until now, DSC-MRI has been performed on only a few occasions in regular migraine with and without aura (21–24); DSC-MRI has most often been used for studying perfusion changes during attacks of rare subtypes of migraine, such as familial hemiplegic migraine or persistent migraine aura, most often in only a single case (34–41). The DSC-MRI acquisition and analysis in the current study was applied with more advanced acquisition and post-processing techniques than previously used in DSC-MRI studies in regular migraine (24). First, the 3D PRESTO acquisition in combination with parallel imaging covered the entire brain at a temporal resolution of 1.1 s. A low dynamic scan time is important because the concentration of the contrast agent is monitored dynamically, and a simulation study by Knutsson et al. showed that the dynamic scan time should be below 1.5 s (42). Second, delay-insensitive deconvolution was used. This improves the quality of the perfusion estimates, especially given that delay effects are more present when a global AIF is used for the deconvolution (43,44). In our analysis, a global AIF was selected close to but outside the middle cerebral arteries (45). These two brain-feeding arteries supply the majority of but not the entire cortex. Third, spatial normalization was performed in two steps, first to the PET template and second to an average CBF map. This improves the accuracy of the voxelwise comparison analysis.
Apart from an increased spatial resolution allowing voxelwise comparisons, DSC-MRI has several advantages over other techniques used for measuring perfusion in migraine. DSC-MRI does not expose the participants to ionizing radiation and is therefore better suited for measuring cerebral perfusion in large samples of both migraineurs and controls than PET or SPECT. Further, DSC-MRI is more widely available and less time consuming. SPECT only measures CBF and some SPECT techniques provide only relative values. Furthermore, the spatial resolution of PET and SPECT is lower than of DSC-MRI, making it harder to detect local perfusion changes. In the SPECT studies that compared interictal migraineurs with controls, images were acquired with varying SPECT-contrast agents and were often evaluated visually or analysed semiquantitatively leading to a substantial degree of subjectivity. To overcome this problem, our results of local hyper- and hypoperfused areas between migraine attacks were generated by an unbiased whole brain voxelwise comparison of DSC-MRI images. Besides the aforementioned advantages, DSC-MRI also has a major disadvantage. Recent studies showed that the (repetitive) use of DSC-MRI contrast agents in patients with renal failure increases the risk of developing nephrogenic systemic fibrosis (46,47), although this seems to be limited to certain specific agents. Arterial spin-labeling (ASL) is probably a good alternative technique for both DSC-MRI and PET. ASL, a relatively new MR technique, is better for perfusion estimation and enables repeated measurements to increase the sensitivity (48). Although this technique allows absolute quantification only of CBF (and not CBV or MTT), intravenous administration of contrast agents or radioactive tracers is not necessary. The first results of ASL during migraine attacks show results comparable to those in PET studies (49,50).
In summary, our study shows in an unbiased whole brain voxelwise approach that interictal migraineurs have discrete areas of cerebral hyper- and hypoperfusion, measured with DSC-MRI. Their specificity for migraine pathophysiology is thought to be small, and we did not find changes in hemodynamics of interictal migraineurs that could account for subclinical cerebellar lesions found previously.
Footnotes
Funding
This work was supported by the Dutch Brain Foundation (Nederlandse Hersenstichting, HsN H04/08) and the Dutch Technology Foundation (STW VENI 7291) and in part by grant R01 NS059775 (OW) from the National Institutes of Health.
Declaration of conflicting interests
OW has a patent on ‘Delay-compensated calculation of tissue blood flow,’ US Patent 7,512,435, 31 March 2009, and the patent has been licensed to General Electric, Siemens, and Olea Medical. The remaining authors declare that there is no conflict of interest regarding the subject matter of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
