Abstract
Background
Previous studies have demonstrated the analgesic effects of ginger in different conditions, but evidence about its efficacy in migraine treatment is scarce.
Objective
This study aimed to evaluate the potential of ginger to improve acute migraine as an add-on strategy to standard treatment.
Methods
A double-blind placebo-controlled randomized clinical trial in the emergency room of a general hospital was conducted. Patients who sought medical care at the time of migraine attack were enrolled in this study. Only adults with episodic migraine (one to six migraine attacks per month) with or without aura were included. Sixty participants were randomized into two groups in which they received 400 mg of ginger extract (5% active ingredient) or placebo (cellulose), in addition to an intravenous drug (100 mg of ketoprofen) to treat the migraine attack. Patients filled a headache diary before, 0.5 h, 1 h, 1.5 h and 2 h after the medication. Pain severity, functional status, migraine symptoms and treatment satisfaction were also recorded.
Results
Patients treated with ginger showed significantly better clinical response after 1 h (p = 0.04), 1.5 h (p = 0.01) and 2 h (p = 0.04). Furthermore, ginger treatment promoted reduction in pain and improvement on functional status at all times assessed.
Conclusions
The addition of ginger to non-steroidal anti-inflammatory drugs may contribute to the treatment of migraine attack. This trial is registered at ClinicalTrials.gov (NCT02568644)
Introduction
Migraine is a debilitating primary headache with a prevalence rate of 12% in the Western world’s population (1). At least 1% of the general population has one day of migraine attack per week (2). During migraine attacks, approximately 53% of patients report severe impairment or need for bed rest (3).
The disability caused by migraine encourages the search for new therapeutic strategies. Complementary and alternative medical treatment has become an increasingly common practice among patients who have headaches motivated by dissatisfaction with conventional therapies and side effects originating from them (4).
Ginger (Zingiber officinale Rosc.) is a spice consumed worldwide. In China and India, ginger is used as a medicinal plant to treat several conditions such as stomach pain, nausea, diarrhea, respiratory disorders, toothache and arthritis (5,6). Ginger rhizome has several bioactive compounds, which are divided into volatile and non-volatile. Volatile compounds correspond to 1–3% of the ginger rhizome and they are responsible for the aroma and flavor of ginger (7). The non-volatile compounds are the major constituents and include gingerols and shogaols. These compounds are obtained in ginger extracts (7). Several studies have shown the pharmacological activity of ginger non-volatile components, including antiemetic, antioxidant, anti-inflammatory, antithrombotic and neuroprotective effects (6–8). Specifically, ginger seems to present significant analgesic effects, relieving dysmenorrhea, arthritis, and musculoskeletal pain (9,10). Several mechanisms have been proposed to justify the analgesic action of ginger, including the inhibition of arachidonic acid metabolism via the cyclooxygenase (COX) pathways, similar to the non-steroidal anti-inflammatory drugs (5). Ginger also acts to block lipoxygenase (LOX), another enzyme associated with the arachidonic acid pathway (11). The concomitant inhibition of COX and LOX may increase anti-inflammatory action and reduce its side effects (12). Furthermore, shogaols seem to modulate neuroinflammatory response through the down-regulation of inflammatory markers on microglial cells (13), while gingerols may act as agonists of the capsaicin-activated vanilloid receptors (14).
To date, only a few uncontrolled studies and one case report have shown the analgesic effect of ginger in migraine (15–18). In order to provide further evidence that ginger is a potential candidate for the treatment of migraine attacks, we carried out a double-blind placebo-controlled study using ginger extract as an adjuvant strategy to conventional treatment in acute migraine.
Methods
This is a double-blinded placebo-controlled randomized clinical trial conducted following the Guidelines for Controlled Trials of Drugs in Migraine (19). This study was registered at the ‘Plataforma Brasil’ (CAAE: 28236814.3.0000.5149) and the Clinical Trials Registry Platform (NCT02568644), and it was approved by the Ethics Research Committee of the Vera Cruz Hospital and Universidade Federal de Minas Gerais, Brazil. All participants were informed regarding the study protocol before taking part.
Sample size was estimated at 60 participants considering 25% of pain relief with ginger treatment according to previous studies (10,20–22) and a 22% confidence interval range. Calculation of sample size was based on 5% alpha error, 80% statistical power and 2% possible losses (16,23).
Study population
Migraine patients were recruited at the emergency department of the Vera Cruz Hospital, Belo Horizonte, MG, Brazil, during a migraine attack with moderate or severe pain intensity. Individuals of both genders were enrolled in this study. Inclusion criteria included: (i) being aged between 18 and 60 years old, (ii) a migraine diagnosis with or without aura for at least one year, (iii) a frequency of one to six migraine attacks per month. Exclusion criteria were: (i) headaches not characterized as migraine, (ii) pregnancy or lactation, (iii) use of any analgesics more than two times per week, alcohol or other drugs, (iv) hypersensitivity to ginger, (v) other neurological diseases, and (vi) use of anticoagulant drugs, as ginger exhibits an antiplatelet action and may increase the risk of bleeding. Migraine diagnosis was performed by a neurologist according to the criteria of the International Headache Society-2nd edition (ICHD-II) (24).
Clinical and demographic measures
Demographic data (age and marital status) and headache characteristics (length of disease, age of onset of the disease and headache impact), depressive symptoms and clinical data (weight, blood pressure and heart rate) were recorded. Headache impact was evaluated through the Headache Impact Test, version 6 (HIT-6) (25) and the Migraine Disability Test (MIDAS) (26) questionnaires. Depressive symptoms were evaluated through the Beck Depression Inventory (BDI) (27). All data were collected during the period the patient stayed at the emergency department.
Study design
As the Brazilian legislation does not allow the evaluation of an experimental strategy alone when there is an approved treatment for the condition (i.e. non-steroidal anti-inflammatories or triptans for migraine), we designed a trial with ginger as an adjunctive treatment to ketoprofen. Ketoprofen was chosen as this anti-inflammatory was the drug of choice to treat moderate to severe pain in the hospital where the study was carried out.
Patients who reported migraine attack with moderate or severe (≥2 on a four-point scale) pain intensity received 400 mg of ginger extract divided into two capsules (containing 5% active gingerols or 20 mg) (K + G) or placebo (cellulose) (K + P), in addition to an intravenous drug (100 mg of ketoprofen) to treat the migraine attack.
The capsules of placebo and ginger extract were made of gelatin, were odorless, and had the same color and format. Clinicians and patients could not discriminate between placebo and ginger capsules. Ginger extract was obtained at Amphora (Belo Horizonte, Brazil), and the amount of gingerol in the extract (>5% gingerols) was confirmed by an independent analysis (Ideal Pharma, Sao Paulo). The allocation for each group was done according to a double-blind (participants and care providers were blinded) and randomized design. The randomization sequence was created by the Randperm function of Matlab Mathworks software (Matrix Laboratory) considering the ratio of 1:1. Only after the end of the study was the allocation of each participant revealed to the clinicians involved in data collection. No change to trial outcomes occurred after the trial start. Gingerol dose was chosen following a previous study that showed that doses of less than 2 g of ginger extract containing 5% of gingerols were not toxic and highly tolerable (28).
Patients filled out a paper-and-pencil report form before the medication and 0.5 h, 1 h, 1.5 h and 2 h after taking the medication. In this form, patients reported pain intensity, functional capacity, migraine symptoms (nausea, vomiting, photophobia and phonophobia) at each time point, as well as side effects and satisfaction with treatment.
Intensity of pain was evaluated through a four-point scale (i.e. 0 = no pain, 1 = mild pain, 2 = moderate pain, 3 = severe pain), a visual numeric scale and a faces pain scale. Functional capacity and migraine symptoms were assessed using ordinal scales as well. Since we used ginger extract as an adjunct treatment, we expected that all patients would improve to a certain extent as they were receiving an established treatment, that is, intravenous ketoprofen. Based on that, we chose to use three pain scales to increase the sensitivity of the analyses.
Response to treatment was measured through the percentage of patients that check 0 (no pain) or 1 (mild pain) after taking the medication in the four-point scale as recommended in the Guidelines for Controlled Trials of Drugs in Migraine (19).
Rescue medication 2 h after the intake of the ginger extract or placebo was also analyzed as an additional parameter to verify the efficacy of the treatment. Furthermore, patients were advised to observe and register migraine recurrence in a 48-h period after taking the medication. We could reach all the patients by telephone to obtain this latter information.
Evaluation of the treatment efficacy
The primary end point was the proportion of patients who responded to treatment in the measured periods, mainly at 2 h after drug intake. We considered response to treatment when patients’ pain intensity decreased from severe or moderate to none or mild (Figure 1) and when at least two of the three scales showed a statistically significant difference (Table 3). Secondary end points were:
Improvement of the functional capacity after taking the medication, measured by the percentage of patients that reported no disability on a four-point scale: 0 = no disability (i.e. able to function normally); 1 = mild disability (i.e. able to perform all activities of daily living but with some difficulty); 2 = moderate disability (i.e. unable to perform certain activities of daily living); 3 = severe disability (i.e. requiring bed rest). Migraine symptom relief (nausea, vomiting, phonophobia and photophobia) after taking the medication measured by a four-point scale (i.e. 0 = none, 1 = mild, 2 = moderate, 3 = severe). Drug rescue use 2 h after taking the medication. Pain relief sustained for 48 h after treatment. Side effects reported. Satisfaction with treatment measured by a seven-point scale (i.e. 0 = fully satisfied, could not be better; 1 = very satisfied; 2 = somewhat satisfied; 3 = neither satisfied nor dissatisfied; 4 = somewhat dissatisfied; 5 = very dissatisfied and 6 = fully dissatisfied, could not be worse). Flow diagram of the study.

Statistical analyses
Statistical analyses were performed with SPSS, version 20.0 (IBM Corp., Armonk, NY, USA). The Kolmogorov–Smirnov test was used to test normality. The simple t-test and Mann–Whitney test were used for the mean and median comparisons, respectively, to evaluate the differences between the two groups. The paired samples t-test and Wilcoxon test were used for the mean and median comparisons, respectively, to evaluate the differences between time points. Categorical variables were compared using chi-square analyses. Continuous variables taken at different time intervals were compared by Generalized Estimating Equation (GEE) Model to evaluate the effect of group allocation, adjusting for time effect (group × time). Crude Odds Ratios (OR) and 95% confidence intervals were measured to assess response to treatment. A p ≤ 0.05 was considered statistically significant.
Results
Demographic and clinical characteristics
Between July and October 2014, a total of 120 individuals that arrived at the emergency room with a headache attack were evaluated by a neurologist, of whom 60 subjects were included in this study: 30 in a group that received placebo and intravenous 100 mg of ketoprofen, and 30 in a group that received ginger extract in addition to intravenous 100 mg of ketoprofen. Figure 1 shows the reasons why patients with headache attack were not enrolled in the study.
Demographic and clinical characteristics in individuals with migraine.

K + P: group that received intravenous ketoprofen and oral placebo; K + G: group that received intravenous ketoprofen and oral ginger extract; MIDAS: Migraine Disability Test; HIT-6: Headache Impact Test – version 6; BDI: Beck Depression Inventory. Values presented as mean ± standard deviation.
Simple t-test.
Mann-Whitney test.
Chi-square test.
Clinical parameters of patients with migraine at baseline.

K + P: group that received intravenous ketoprofen and oral placebo; K + G: group that received intravenous ketoprofen and oral ginger extract. Values presented as mean ± standard deviation and frequency.
Paired t-test.
Chi-square test.
Pain intensity related by migraine patients according to three different scales.

BL: Baseline.
Statistical differences between groups.
Statistical differences intra groups.
Treatment efficacy
Primary end-point: After one hour of medication, patients that received ginger extract related lower pain intensity on all scales. This difference remained after 2 h of medication in the faces pain and visual numeric scales (Table 3).
Patients that received ginger extract also had better response to the treatment (i.e. reduction in headache intensity from severe/moderate to mild/no pain) 1 h (p = 0.04), 1.5 h (p = 0.01) and 2 h (p = 0.04) after medication (Figure 2(a)). The odds ratio for response to treatment with ginger extract versus placebo in measured times were 5.09 (CI 95% 0.98–26.43; p = 0.05), 10.54 (CI 95% 1.23–90 66; p = 0.03) and 7.25 (CI 95% 0.81–64.45; p = 0.07), respectively. There was an evident trend towards greater pain relief in patients who received ginger in contrast to those receiving placebo. Corroborating this assumption, the proportion of patients who were pain free (i.e. reporting 0 on the scales) after 2 h was higher among patients that received ginger extract (56.7 vs. 33.3; p = 0.03) (Figure 2(b)).
(a) Percentage of patients who responded to treatment or still in pain evaluated through four-point scale and (b) percentage of patients who reported pain free, mild pain, moderate pain and severe pain in four-point scale (c) percentage of patients who reported normal functional capacity 0.5 h, 1.0 h, 1.5 h and 2.0 h after the medication in both groups. K + P: group that received intravenous ketoprofen and oral placebo; K + G: group that received intravenous ketoprofen and oral ginger extract. # indicate a statistically significant difference (P < 0.05) between the groups.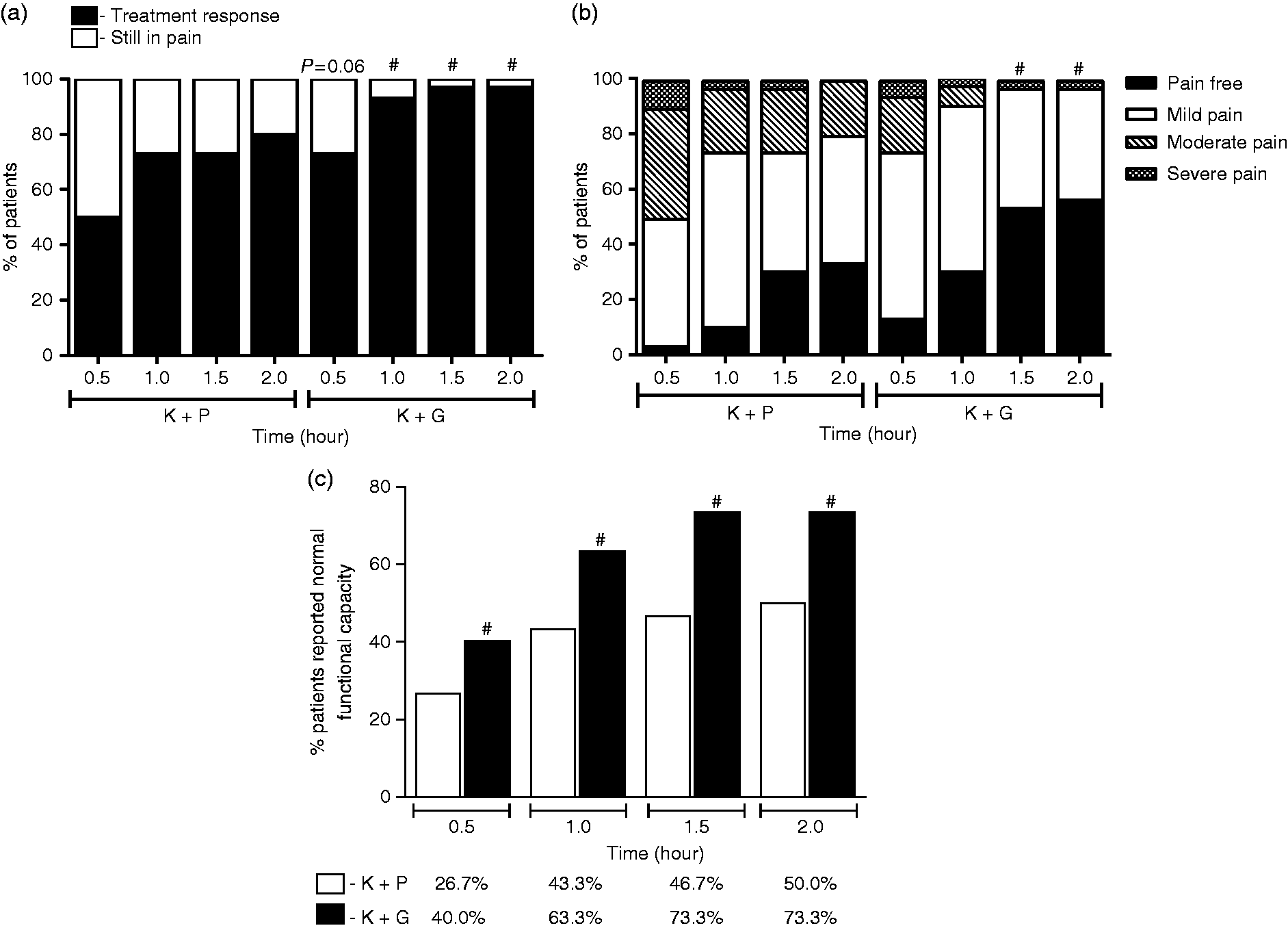
Secondary end-points: Pain decrease or remission was accompanied by improvement of functional capacity. More patients reported “no disability” in the group that received ginger extract compared to the group that received placebo (0.5 h: p = 0.05; 1 h, 1.5 h and 2 h: p < 0.01) (Figure 2(c)).
Presence of symptoms associated with migraine attack at different times after medication.

K + P: group that received intravenous ketoprofen and oral placebo; K + G: group that received intravenous ketoprofen and oral ginger extract. Values presented as frequency.
Chi-square test.
The frequency of rescue medication use was 13.3% in patients that received placebo versus 8.7% of patients that received ginger extract (p = 0.39). Recurrence of pain 48 h after medication was reported by 33.3% of patients that received placebo and 30.0% of patients that received ginger extract (p = 1.0).
Three patients who received ginger extract reported dyspepsia 0.5 h after the medication. Only one of these patients still had this symptom 2 h after medication. One participant that received placebo reported dizziness 1 h after medication and two patients complained of nausea 1.5 h and 2 h after medication. Accordingly, the presence of side effects did not differ between groups in the measured periods: 0.5 h (p = 0.12), 1 h (p = 0.75), 1.5 h (p = 0.55) and 2 h (p = 1.00).
Most patients (73.1%) that received ginger extract reported being fully satisfied with treatment versus 28.1% of patients that received placebo (p < 0.01) (Figure 3). No one reported being very dissatisfied or fully dissatisfied with the treatment.
Percentage of patients who reported “fully satisfied”, “very satisfied”, “somewhat satisfied”, “neither satisfied nor dissatisfied” or “somewhat dissatisfied” with the treatment in both groups. K + P: group that received intravenous ketoprofen and oral placebo; K + G: group that received intravenous ketoprofen and oral ginger extract. # indicate a statistically significant difference (P < 0.05) between the groups.
Discussion
Despite advances in the treatment of migraine, including the development of triptans, side effects and low satisfaction with current therapeutics are still challenging. Furthermore, migraine chronicity and its impact on quality of life have been associated with an increasing demand for alternative therapeutic strategies (18). The current study shows that addition of ginger extract to conventional treatment for acute migraine may improve pain and functional capacity of patients faster and more significantly than conventional therapy alone.
During migraine attacks, trigeminal nerve fibers are activated releasing neuropeptides that trigger the production of inflammatory mediators such as cytokines, bradykinin and prostaglandins, which activate nociceptive pathways (29,30). Drugs used in the treatment of pain usually act by altering the transduction and/or modulation of nociception (31). Previous studies have shown that ginger components, mainly gingerols and shogaols, are able to decrease prostaglandin production through COX-2 inhibition, inducing pain relief (6,31,32). Leukotriene production is also decreased by ginger components through 5-lipoxygenase (5-LOX) inhibition (11). Concomitant blocking of COX-2 and 5-LOX potentiates anti-inflammatory effects and reduces the side effects observed when only COX-2 is inhibited (12). Furthermore, 6-shogaol modulates neuroinflammation by inhibition of pro-inflammatory cytokines expression in microglial cells (13). Together, the proposed mechanisms and the available evidence suggest a potential role of ginger in acute migraine.
Previous uncontrolled studies suggested a role for ginger in migraine treatment. For instance, Mustafa & Srivastava (1990) described a case report in which the consumption of ginger powder mixed in water decreased the pain intensity in migraine attack (17). Cady et al. (2005 and 2011) showed that administration of sublingual supplements of ginger in combination with feverfew, a medicinal herb, reduced pain intensity in migraine attack (15,16). In a recent study, Maghbooli et al. (2014) observed that 250 mg of powdered ginger showed similar efficacy to sumatriptan in relieving migraine pain 2 h after ingestion, while ginger was associated with fewer side effects (18).
Symptoms associated with migraine attack can be activated by nociceptive signals, therefore, the blocking of nociceptive pathways may improve these symptoms (33). In the current study, as all patients evolved with improvement of nausea, vomiting and photophobia, with no clear differences between groups, it was not possible to evaluate the potential antiemetic effect of ginger. Indeed, the antiemetic action of ginger is probably one of the most common indications of ginger in the clinical practice (34). Previous studies reported the efficacy of ginger to reduce nausea and vomiting during pregnancy, post-operative or induced by chemotherapy (34,35).
The limitations of our study include the absence of groups receiving only ginger extract or placebo, i.e. without taking an analgesic drug. However, all patients received the same conventional drug in addition to the treatment in evaluation. The size effect of the add-on treatment was small but was consistent across different measures. Furthermore, we only had information on the need of rescue medications in the first 2 hours of the treatment not beyond this time point. On the other hand, this study stands out because a known dose of gingerol (20 mg) in the ginger extract was used. The quantity of an active component of food is important information as it can help to design future studies.
Conclusion
Herein we showed that the addition of ginger to a non-steroidal anti-inflammatory drug strategy (i.e. intravenous ketoprofen) may contribute to the treatment of migraine attack.
Clinical implications
The association of ginger extract with ketoprofen may be beneficial for the acute treatment of migraine. The addition of ginger does not seem to decrease migraine-associated nausea.
Footnotes
Acknowledgements
The authors would like to thank the unrestricted support from the Vera Cruz Hospital medical and nursing staff.
The authors thank Isabel Myrian Guimarães Campos, Tamara Carneiro Medeiros de Souza and Luciana Alves de Andrade for the help during data collection and care with the participants.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Brazilian government funding agency Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
