Abstract
Aim
To examine the characteristics of visual symptoms during attacks of migraine in children and adolescents.
Method
A qualitative analysis of prospectively collected data over 5 years, on characteristics of visual symptoms during migraine attacks. Diagnosis of migraine and aura was based on the International Classification of Headache Disorders 3rd edition beta version. We also provided the opportunity for patients to illustrate their visual aura symptoms to aid in diagnosis.
Results
Visual symptoms were reported by 387/1079 (36%) of migraineurs. Of these, 172 (16%) patients fulfilled the International Classification of Headache Disorders Criteria A, B, C iv and D, but missed one (n = 75; 43.5%) or two (n = 97; 56.5%) of the remaining items of criteria C as the visual symptoms were of non-gradual spread (n = 35; 20%), appeared in both visual fields (n = 99; 58%), or lasted less than 5 minutes or more than 60 minutes (n = 129; 75%).
Conclusion
The International Classification of Headache Disorders 3rd edition beta version criteria are useful in diagnosis of migraine with visual aura in children and adolescents, but visual symptoms varied considerably in duration, pattern, mobility, location, mode of onset and colours. Providing opportunity for patients to illustrate their symptoms can provide additional diagnostic information. The pathophysiology and the clinical concept of typical MVA is still to an extent an assumption and needs further evaluation.
Approval
The study was approved by the Health Research Authority – London and the local Research and Innovation Department at Barking Havering and Redbridge National Health Service Trust. Formal parental consent was not considered essential for this type of study.
Background
ICHD-3 beta diagnostic criteria for migraine with aura – Code 1.2.

The main objective of this study was to examine the clinical features of visual symptoms during attacks of migraine among children and adolescents with particular emphasis on the ICHD criteria C i, ii, iii and iv (gradual onset, duration, location, accompanied or followed within 60 minutes by headache). A secondary aim was to study the flexibility of MVA by analysing its characterisation in terms of diagnostic eligibility on the basis of the ICHD of typical MVA. Examining characteristics of non-visual aura migraine symptoms is beyond the scope of our study. We also wanted to explore whether providing children with opportunity to illustrate their symptoms would be helpful in the diagnostic process.
Method
This is a qualitative observational hospital-based study on visual symptoms during attacks of headaches among 1079 migraineurs who were referred to our headache clinic between May 2012 and May 2017. Diagnosis of migraine and typical MVA was based on the diagnostic criteria (Table 1) according to the International Classification of Headache Disorders 3rd edition (ICHD-3 beta) (4). First, we sought to determine the characteristics of visual symptoms and then we assessed the entitlement of eligible patients for the diagnosis of typical MVA. All patients were assessed and screened for inclusion and exclusion criteria by one of the authors (MASA). This is an outpatient clinic cohort in which there was heterogeneity in terms of arranged tests. For example, not all patients had EEG or neuro-imaging, as in our study this was arranged according to our local guidelines and national recommendations. We categorised our patients into those who fulfilled all ICHD Criteria A, B, C i, ii, iii, iv and D of typical MVA and those who missed one or two of the ICHD Criteria C i, ii, iii, to allow us to further explore the symptoms that did not meet all the “typical” criteria. The categorization was based on our clinical assessment and strictly concurs with the definition and diagnosis of MVA on the basis of the ICHD-3 beta.
To be included in the study, the patients must have experienced visual symptoms during attacks of migraine, and be otherwise neurologically normal. Their age was between 6 and 17 years, and they had had at least two migraine attacks. Those with visual problems in between attacks of their migraine were not included. Other exclusion criteria were as follows: (1) past diagnosis of epilepsy, syncope, communication problems, and/or psychiatric illnesses; (2) medication overuse headaches; (3) taking medication known to cause visual disturbances; (4) migraine with brainstem aura; (5) difficulty in identifying colour; (6) insufficient information or incomplete medical history.
Visual symptoms
Patients were allowed to talk freely about the experiences of their headaches. Once a focused headache history was completed, each patient was asked “what else happens when you have a headache attack?”. Those who had not spontaneously reported visual symptoms were asked specifically if they had experienced any visual disturbances during attacks of their headaches.
Patients with visual symptoms were asked if they could describe what they see during attacks of their headaches. Patients were given the opportunity if they wished to draw a picture and/or write details of what they had seen during attacks of their migraine. Willing patients were asked to sit in a room with a chair and a table, and each provided with a blank piece of white paper and a number of coloured pencils. Patients were allowed an unlimited time to draw and write. No leading questions or additional instructions were given. Elaboration of the drawings and written documentation was not encouraged. After completion of the drawings and/or writing, the examiner asked patients to describe the pictorial features in their drawings and/or to talk more about their visual symptoms. Photographs of the drawings and/or written descriptions were taken by our medical photographer and kept in the medical notes with no form of alteration.
Patients were asked to describe the exact colours (e.g. black, white, red, and blue, green, yellow) of their visual symptoms. Also, we asked each patient if his or her visual symptoms occurred in some, most, or all headache attacks. Additionally, we inquired about the similarities or variation in the characteristics of visual symptoms during attacks of migraine. Data on visual symptoms with particular emphasis on duration (seconds, minutes, hours, days); mode of onset (gradual vs. sudden); colours (achromatic, monochromatic, dichromatic or polychromatic); location (unilateral vs. bilateral); patterns (formed vs. unformed); mobility, and whether the size and/or shape remained the same or changed were collected. Relevant data were also obtained after scheduled or unscheduled clinic visits. Finally, data were kept confidentially in an Excel spreadsheet and analysed using numerical, percentage and simple statistical tools.
Definition
For the purpose of this study, visual symptoms were defined as visual hallucinations (no external stimulus) which occurred before, concurrently or immediately following the headache phase of migraine attacks. In our study, visual symptoms were categorised into formed, unformed and complex visual disturbances. Unformed visual symptoms were defined as simple sparks, zigzags, flickers or flashing lights. Formed visual symptoms were defined as specific shapes or figures (e.g. circles, dots, blobs, triangles, and stars). In our study, the appearance of whole or parts of people or animals or distortion of objects in shape, size, colour, or distance were defined as complex visual symptoms. Duration of visual symptoms was defined as less than 5 minutes; 5 to 60 minutes or more than 60 minutes. Gradual spread was defined as a spread of visual symptoms over 5 minutes or more. The term ‘sudden onset’, in this study, means a development of the visual symptoms of less than 5 minutes. Unilateral was defined as visual symptoms appearing in one eye and remaining in the same visual field. In our study, we avoided using specific terms such as fortification spectrum, scintillating scotoma, photopsias, phosphenes, teichopsia, palinopsia or metamorphopsia, as they often cause confusion or are falsely interpreted by the investigators.
Results
Inclusion and exclusion criteria: Patients (387) who fulfilled the ICHD Diagnostic Criteria C i, ii and iii of MVA.
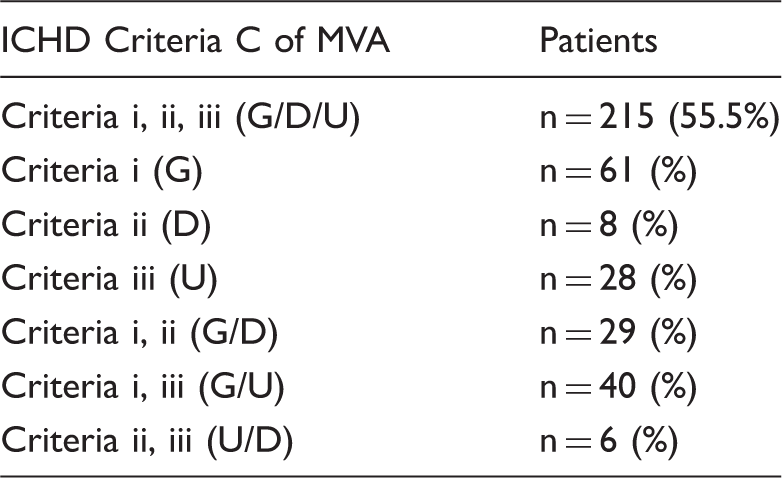
ICHD: international classification of headache disorders; Criteria i (G): gradual spread over 5 minutes or more; Criteria ii (D): duration for 5 to 60 minutes; Criteria iii (U): unilateral or one visual field; MVA: migraine visual aura.
Epidemiological and clinical characteristics of patients.

FH: positive family history of migraine in first degree relatives.
Shapes and patterns of visual symptoms.

Colours of visual symptoms.

Onset and spread of visual symptoms.

NS: not specified; VS: visual symptoms.
Three colours or more.
Location of visual symptoms.

VS: visual symptoms; NS: not specified.
Three colours or more.
Characteristics according to the duration of visual symptoms.

VS: visual symptoms; NS: not specified.
Three colours or more.
Characteristics according to the mobility of visual symptoms.

VS: visual symptoms; NS: not specified.
Three colours or more.
Characteristics according to the patterns and shapes of visual symptoms.
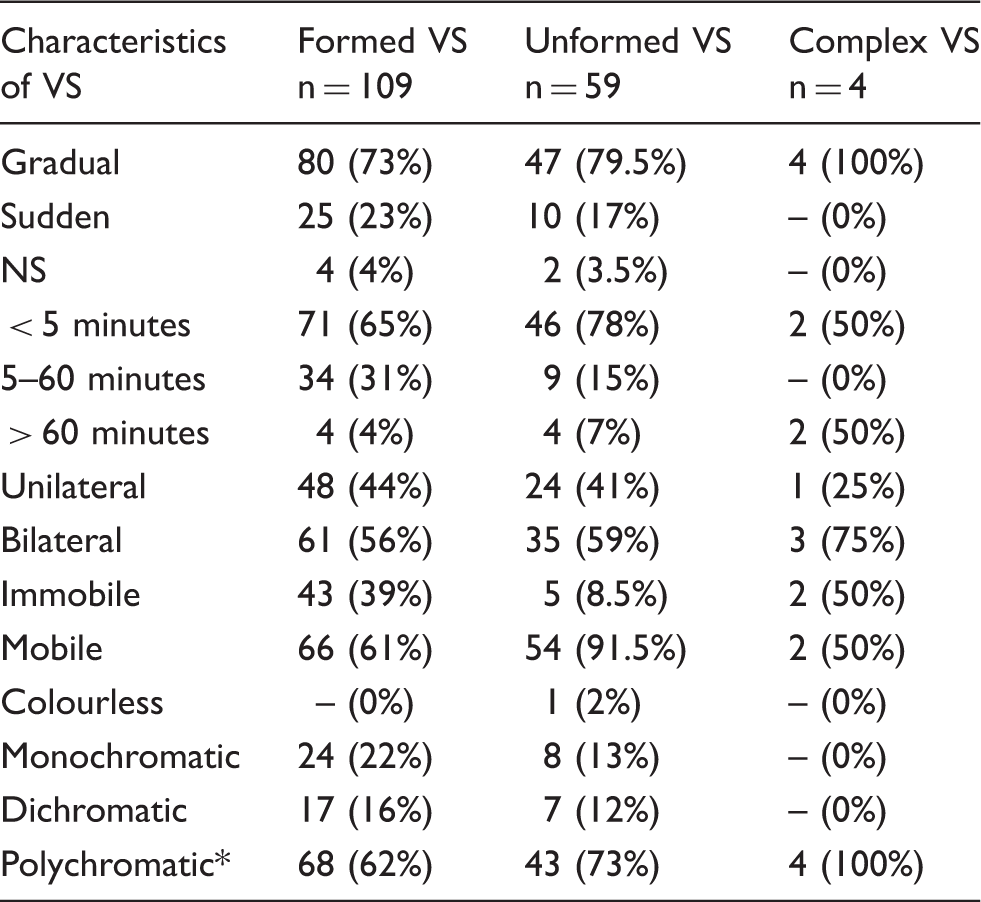
VS: visual symptoms; NS: not specified.
Three colours or more.
Within this cohort, visual symptoms occurred in all (n = 103; 60%) or most migraine attacks (n = 69; 40%). Regarding succession of visual symptoms, headache started after onset of visual symptoms (n = 101; 59%), before (n = 36; 21%), or the succession was not specified (n = 35; 20%). Regarding the presence of other migraine auras, visual symptoms occurred without a run of other symptoms in 136 (79%). The remaining 36 (21%) patients reported non-visual aura (29 somatosensory aura; five somatosensory/language, one somatosensory/motor; one language). Each of these patients reported auras before onset of visual symptoms and none simultaneously experienced such aura at or before onset of visual symptoms.
78/172 (45%) patients had an interictal EEG that was normal (n = 75) or showed non-specific changes (n = 3). Two of these three patients had a normal sleep-deprived sleep EEG. 122/172 (71%) patients had brain imaging (three CTs and 119 MRI brain-epilepsy protocol) that was normal (n = 113), or showed non-specific abnormalities (n = 9) that required no intervention and were unlikely to cause headaches or visual symptoms. In all, 26/172 (15%) patients depicted visual symptoms during attacks of their migraine (see Supplementary figures, available online).
Regarding location, we were able to identify a number of our patients (42%) with unilateral visual symptoms, but the remaining (57.5%) patients reported to us visual symptoms that exclusively appeared and remained in both visual fields. In our cohort, 50 (29%) patients reported immobile visual symptoms. The remaining 122 (70%) patients with mobile visual symptoms were eligible for the diagnosis of typical MVA as their symptoms were unilateral (49%) or gradually spread (75%) or lasted 5 to 60 minutes (20%).
Discussion
To the best of our knowledge, this is the first study to examine in depth the characteristics of visual phenomena in a clinic population of children and adolescents with migraine. The most striking finding in our study is the huge variation in visual aura features that occur in children and adolescents who fulfil the ICHD criteria, with 44% of those diagnosed with MVA in our study missing one or two of the criteria for typical MVA. Although a large proportion of our patients (56%) reported a homogenous phenotype of visual disturbances and fulfilled all the elements of the ICHD criteria of typical MVA, in the next section we shall explore in detail the visual aura features that were outside the typical criteria to understand further the spectrum of symptoms that may occur in this population.
Development of visual symptoms
One of the most distinguishing features of MVA is the gradual spread of visual symptoms over at least 5 minutes. A study by Mattsson et al. (1999) reported visual disturbances indicative of migraine visual aura, and revealed that a gradual onset of five or more minutes was stated by as few as 45% of migraineurs (19). In our study, the majority of our patients (76%) reported visual symptoms that developed over a period of at least 5 minutes, but interestingly 20% reported to us that their visual symptoms spread and developed fully over a shorter period and certainly for less than 5 minutes.
Duration of visual symptoms
The ICHD-3 states that the duration of each migraine aura symptom has to be no longer than 60 minutes, while recent data shows that single aura symptoms may last longer than 60 minutes (3). In our cohort, 10 patients reported prolonged visual symptoms. Our data provide strong evidence for the current views confirming single migraine aura symptoms may last for longer than an hour (10–18). Literature on prolonged migraine non-motor aura in children is sparse (11,13). We previously reported prolonged non-motor migraine aura in 4.5% of 267 children and adolescents with migraine (11).
Interestingly, in our cohort, 69% of patients reported symptoms lasting for less than 5 minutes. All of these patients were qualified for the diagnosis of typical MVA, as their visual symptoms were either unilateral (54%) and/or gradually spread (78%).
Formation of visual symptoms
Typical MVA are often present as uncoloured, unformed patterns (zigzags, flickering) appearing in the centre of the visual field, and gradually progressing toward the periphery, often leaving scotoma. In our cohort, 34% of patients reported unformed patterns consisting mainly of flickering lights or zigzags. Interestingly, visual symptoms, among these patients, were predominantly polychromatic (73%) dominated by colours such as red, blue, green, yellow, brown, purple and orange, and only seven (12%) patients reported pure white and/or pure black patterns. In our cohort, complex visual symptoms of distorted animals or faces of people during attacks of migraine were reported by only four (2%) patients.
Differential diagnoses
Retinal migraine, as a cause of visual disturbances in our cohort, is unlikely as it would cause visual loss of the entire visual field of one eye, not both eyes and not in association with visual hallucination, which was not described by any of our patients. None of our patients reported a history suggestive of dysarthria, vertigo, tinnitus, hyperacusis, ataxia or reduced level of consciousness and therefore, based on the ICHD-3 beta criteria, migraine with brainstem aura is unlikely as a cause of their symptoms.
In view of the fact that some of our patients reported short lasting (< 5 minutes) symptoms, one may argue that this could have been misdiagnosed and other differential diagnoses such as epilepsy should be considered. In epilepsy, particularly idiopathic occipital lobe epilepsy, which is uncommon in healthy children and adolescents, visual symptoms usually consist of small, brightly coloured spots or shapes that flash, and the content may be distorted in size or it may suddenly change shape, moving from lateral toward the centre of the field of vision. More specifically, the epileptic elementary visual hallucinations frequently consist of hundreds of multi-coloured small balls occurring almost on a daily basis, and their onset is mostly unilateral (21,22). Visual symptoms, in particular in occipital lobe (area 17) epilepsy, are typically simple, elementary unformed symptoms that lack depth. Such symptoms tend to start more abruptly and remain immobile, while seizures in the visual association areas (areas 18 and 19) tend to have more decorative form, depth, colour, and movement. In addition, visual symptoms in occipital lobe seizures last seconds, rarely minutes, and tend to occur daily. However, visual symptoms of occipital epileptic seizures may be followed by no noticeable alteration in awareness or other seizure manifestations and may not raise suspicion of seizure, and therefore differentiation of epileptic visual aura from MVA is not always straightforward.
Our patients provided a definite description of visual symptoms that only occurred during attacks of migraine, associated with moderate to severe headaches, accompanied with other symptoms (e.g. nausea) and were followed by full normalisation of vision. Moreover, their visual symptoms were either unilateral and/or gradually spread. None of our patients presented with other clues to occipital seizures such as sensation of ocular movement, tinnitus, eye deviation, head turning or tonic clonic seizures. According to our assessment and on the basis of the ICHD-3 beta, our patients fulfilled the diagnosis of typical MVA and we believe epilepsy as a cause of the visual aura among these patients is unlikely.
In our cohort, complex visual symptoms of distorted animals or faces of people during attacks of migraine were reported by four patients. In these patients, some may consider epilepsy and in particular temporal lobe epilepsy as an alternative diagnosis; however, in our patients, visual symptoms never progressed to other motor seizures or impaired awareness and occurred exclusively during attacks of migraine. None of these patients reported other seizure types or were known to have epilepsy, and all four patients had a normal interictal EEG and MRI brain. To this end, we believe migraine is the most likely cause of the visual hallucination amongst this patient group also.
Pathophysiology
Visual hallucination is defined as the perception of an event or an object in the absence of an external stimulus. Visual symptoms in our patients were episodic visual hallucinations (not based on external environmental images) that were mostly coloured, formed or unformed patterns, or specific shapes. Numerous hypotheses have been suggested to explain the pathogenesis of visual hallucination, including psycho-biochemical, psycho-physiologic and psychodynamics.
Migraine is the most commonly observed paroxysmal neurologic disease in childhood. Migraine with aura is a condition with various subtypes that likely differ in pathophysiological mechanisms. Cortical spreading depression (CSD) is thought to be the substrate of the migraine aura (23–31). The distance to which CSD spreads rests on the steadiness between factors that predispose or inhibit the brain to CSD, and the recovery from CSD depends largely on intact neurovascular coupling to match the increased energy demand and to restore ion gradients. One may argue that the structures CSD reaches during migraine attacks might determine the appearances and arrangement of the aura. It is also known that the thalamus is a key structure in migraine pathogenesis, and its direct connection to the visual pathway and the cerebral cortex provides a vital interaction between these structures (32). Investigators have demonstrated that CSD markedly alters neuronal firing of ipsilateral third order thalamic neurons, independent of peripheral trigeminal inputs (31). It is well recognised that the thalamus processes signals from the retina to create images and plays a key role in coordinating complex sensory and motor input to and from the cortex. In our study, the precise mechanism of visual phenomena during attacks of migraine among our patients is unclear to us, although we simply understand that such symptoms were generated somewhere in the visual system rather than the globe or eye itself. Also, assuming the interplay between neuroanatomy, neurochemistry and psychodynamics, our data guide us to think that the characteristics of MVA might depend on the balance between CSD and other factors that could inhibit or predispose the visual system to CSD. Our findings clearly illustrated clinical heterogeneity of migraine visual accompaniments that is reflected by a variability of patterns, shapes, colours, duration, mobility, and location, suggesting that typical MVA could have a spectrum of presentations rather than explicit portrayal or a specific description.
Illustration
Meticulous artistic impressions of migraine had been well documented in literature (33–35). In our cohort, 26 patients depicted illustrations of their visual symptoms during attacks of their migraine. We found simply asking patients to draw what they interpreted as their visual symptoms provided extremely valuable data and has widened our knowledge of migraine. Artwork should be encouraged as part of the day-to-day clinical practice, as it could aid in the diagnosis of paroxysmal disorders such as migraine.
Limitations
Our study has key limitations. Firstly, the study is hospital-based, meaning only clinic-visit patients were included and therefore the study could be biased by the selection of patients and this might in turn limit its applicability. Secondly, one could argue that the developmental age, lack of vocabulary, difficulties in articulation, and in particular the difficulty with the concept of time in some children, might hinder the accuracy of our data. Also, due to the short nature of visual symptoms in most of our patients, it was difficult to obtain an EEG during an episode.
Our study has major strengths, briefly summarised as follows: (a) prospectively collected data; (b) homogenous sample of patients; (c) migraine aura diagnosis is based on the ICHD-3 beta; (d) many patients provided clear illustrations of their visual experiences and (e) strict study eligibility. Interestingly, in our cohort, the frequency of MVA (35%) was similar to previously reported prevalence, suggesting that the clinic population was not considerably dissimilar to previous studies (1,2). We believe the diagnostic stability of MVA among our patients is substantial and remained solid. Even with such limitations, we believe that our data provide a reliable description of the visual aura experienced by a large number of children.
Conclusion
We established that a wealth of data collected in condition-specific clinics can contribute to our knowledge of specific disorders such as migraine. We also demonstrated the use of illustration as a supplementary method for children to express their symptoms. Our data validates the use of the ICHD criteria in children and adolescents. Our findings also show that visual phenomena occurring during attacks of migraine with full recovery of vision and no other identified cause, might be considered as MVA irrespective of duration, patterns, location, colours or mobility, and it is right that the ICHD criteria provide this flexibility to take account of this heterogeneity. Future collaboration in recruiting children and young people with visual aura across many centres would confirm whether the conclusion described here is supported over other patients with migraine. This could have a significant implication on day-to-day clinical practice and migraine research.
Supplemental Material
Supplementary figures -Supplemental material for Visual symptoms and childhood migraine: Qualitative analysis of duration, location, spread, mobility, colour and pattern
Supplemental material, Supplementary figures for Visual symptoms and childhood migraine: Qualitative analysis of duration, location, spread, mobility, colour and pattern by Mas Ahmed, Catriona Boyd, Radhika Vavilikolanu and Bushra Rafique in Cephalalgia
Footnotes
Clinical implications
The ICHD-3 beta criteria provided a useful basis for diagnosis of migraine with visual aura in this large cohort of children and adolescents with migraine.
Only 56% of participants in this study fulfilled all ICHD criteria for typical migraine visual aura. The remaining patients, though fulfilling the diagnosis of typical MVA, reported a heterogeneous phenotype of visual disturbances during attacks of migraine.
Visual phenomena occurring during attacks of migraine with full recovery of vision and no other identified cause might be considered as migraine visual aura irrespective of duration, patterns, location, colours or mobility.
Allowing children to illustrate their visual symptoms may add valuable contribution to the consultation by providing additional information that they may not be able to articulate verbally.
Acknowledgment
The authors are indebted to the patients and their parents who gave their time to participate in the study. We also extend our gratitude to Nigel Collister and Charlotte Cook for their help with the development of photographs and illustrations.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
