Abstract
Introduction
Carbon monoxide is an endogenously produced signaling gasotransmitter known to cause headache and vasodilation. We hypothesized that inhalation of carbon monoxide would induce migraine-like attacks in migraine without aura patients.
Methods
In a randomized, double-blind, placebo-controlled crossover design, 12 migraine patients were allocated to inhalation of carbon monoxide (carboxyhemoglobin 22%) or placebo on two separate days. Headache and migraine characteristics were recorded during hospital (0–2 hours) and post-hospital (2–13 hours) phases.
Results
Six patients (50%) developed migraine-like attacks after carbon monoxide compared to two after placebo (16.7%) (p = 0.289). The median time to onset of migraine-like attacks after carbon monoxide inhalation was 7.5 h (range 3–12) compared to 11.5 h (range 11–12) after placebo. Nine out of 12 patients (75%) developed prolonged headache after carbon monoxide. The area under the curve for headache score (0–13 hours) was increased after carbon monoxide compared with placebo (p = 0.033).
Conclusion
Carbon monoxide inhalation did not provoke more migraine-like attacks in migraine patients compared to placebo, but induced more headache in patients compared to placebo. These data suggest that non-toxic concentrations of carbon monoxide had low potency in migraine induction and that the carbon monoxide inhalation model is not suitable to study migraine.
Introduction
Carbon monoxide (CO) is not only a toxic gas, but also an endogenously produced signaling gasotransmitter involved in nociception (1,2), neurotransmission (3) and cerebral hemodynamics (4–8). One well-known effect of CO exposure is headache (9), and several studies have also reported that CO exposure can cause migraine-like headache (10,11). Recently, we reported that inhalation of CO induced mild and prolonged headache in healthy volunteers (12). The pathophysiological mechanisms of CO induced headache are not fully clarified (13). CO has several functional parallels and bi-directional influences with nitric oxide (NO) (14), which is a known trigger of migraine (15,16), cluster headache (17) and tension-type headache (18,19). Both CO and NO are involved in relaxation of cranial vessels via cyclic guanosine monophosphate (cGMP) (20,21) and activation of the cGMP signaling pathway is the likely cause of NO-induced migraine (22). Another well-known effect of CO is hypoxia (9,23), which may trigger migraine attacks with and without aura (12,24). The possible migraine provoking effect of CO has never been investigated in migraine patients. We hypothesized that inhalation of CO would induce migraine-like attacks in migraine without aura patients in a new migraine provocation model useful for further studies of the role of CO in migraine pathophysiology. To test this hypothesis, we conducted a double-blind placebo-controlled crossover study in migraine without aura patients. To assess the vasoactive effect of CO, we measured facial skin blood flow by laser speckle contrast imaging.
Material and methods
We recruited 12 patients with migraine without aura (11 women and one man, mean age 24 years, (range 21–47 years)) via a Danish website for recruitment of volunteers to health research (forsoegsperson.dk). Patients with a minimum of two migraine attacks per month according to the International Classification of Headache Disorders (ICHD-3 beta, 2013) (25) were recruited for the study.
The study was approved by the Ethics Committee of the Capital Region of Denmark (H-16021497) and the Danish Data Protection Agency. The study was registered at Clinicaltrials.gov (ID: NCT03075020). All participants provided their written informed consent to participate in the study after detailed oral and written information in accordance with the Declaration of Helsinki 2013 version.
Experimental design
In a double-blind placebo-controlled crossover design, the participants were randomly allocated to inhalation of CO (carboxyhemoglobin 22%) or placebo (atmospheric air) through a mask on two separate days with a minimum interval of one week and a maximum of two months. The study was carried out from October 2016 to June 2017 at the Danish Headache Center, Rigshospitalet-Glostrup, located in Glostrup. All participants arrived at the clinic 48 h headache and 5 days migraine free without taking any analgesics in 48 h before the study day. Both study days were conducted at the same time of day ±1 h. Pregnancy tests were carried out on all female participants on both study days. All participants were placed in supine position at a temperature of 23–25℃ and light (dimmed) controlled lab.
Headache intensity and characteristics, electrocardiogram (ECG) (Cardiofax V, Nihon-Cohden, Shinjuku-ku, Tokyo, Japan), blood flow in the facial microvasculature (Speckle contrast imager, moorFLPI (Moor Instruments, Devon UK), vital signs, mean arterial blood pressure (MAP), heart rate (HR), respiration frequency (RF) and end-tidal CO2 (PETCO2) (ProPaq Encore®, Beaverton, OR, USA) were recorded at baseline and then every 10 min until 120 min, in the same sequence. Two capillary blood samples were collected at baseline and every 10 min from 0 to 30 min, and every 30 min from 30 to 120 min.
Capillary samples were analysed at the site immediately to determine concentration of COHb, haemoglobin (ctHb), potassium, sodium and glucose (blood gas analyser, Radiometer, Copenhagen, Denmark). After 120 min, the participants were discharged from the hospital and asked to complete a headache diary every hour until 13 h after the first inhalation.
Migraine-like attack criteria
Human migraine provocation models cannot fulfil strictly the ICHD-3 beta criteria for migraine without aura, which require the migraine attack to be spontaneous. Most spontaneous migraine attacks develop over hours, with an initiation phase that fulfils only the criteria for tension-type headache. Often, patients can predict the onset of migraine in the early stage, and in an experimental setting migraine treatment cannot be denied. Therefore, the following migraine-like attack criteria were used, as previously described (26).
Migraine-like attack fulfilling either [1] or [2]:
[1] Headache fulfilling criteria C and D for migraine without aura according to the ICHD-3 beta (25): C. Headache has at least two of the following characteristics: Unilateral location; pulsating quality; moderate or severe pain intensity (moderate to severe pain intensity is considered ≥ 4 on verbal rating scale); aggravation by cough (in-hospital phase) or causing avoidance of routine physical activity (out-hospital phase). D. During headache at least one of the following: Nausea and/or vomiting; photophobia and phonophobia. [2] Headache described as mimicking the patient’s usual migraine attack and treated with acute migraine medication (rescue medication).
Carbon monoxide
Each participant inhaled CO/placebo (atmospheric air) through a cushioned full-face mask (Hans Rudolph) connected to a tube through which CO or placebo was administrated. A COHb concentration of 22% was chosen based on a previous study in healthy volunteers showing there were numerically higher headache scores and headache of longer duration after COHb 22% compared to COHb 10% (12). A specific amount of CO (99.997% purity, Strandmoellen, Denmark) was added to the tube through a 100 ml syringe. COHb from capillary blood samples was determined and the volume of CO (ml) necessary to increase COHb by 1 mM was calculated (200 ml CO/(COHb after inhalation – baseline COHb)). Using the calculated value, the volume of CO needed for the next inhalation was calculated. In total, three inhalations (at time 0, 10 and 20 min) were performed to reach COHb 22%. Each inhalation took approximately three minutes, and under the procedure the participants were instructed to take deep breaths and exhale slowly. The CO inhalation method was previously described in healthy volunteers (12).
Headache and adverse events
The headache intensity and associated symptoms were recorded on a numerical rating scale (NRS) from 0–10 (0: No headache; 1: A very mild headache (including a feeling of pressing or throbbing pre-pain); 10: Worst imaginable headache) (27). Headache localization, quality, aggravation by physical activity, associated symptoms (nausea, photo-phonophobia) and premonitory symptoms (unusual fatigue, yawning, thirst, craving, mood swings, flushing and concentration deficit) were recorded.
Facial skin blood flow
Laser speckle contrast technique (moorFLPI, Moor Instruments, Devon, UK) was used to measure the facial skin blood flow and intensity fluctuations as previously described (12,28,29). The instrument was positioned 30 cm straight above the face of the participant and focus was optimized. The instrument was calibrated using a reference flux signal generated by the laser light scattered from a suspension of polystyrene microspheres in water undergoing thermal or Brownian motion. Participants were instructed to lie completely still with eyes closed during measurements. The images were processed manually to produce a scaled color-coded live flux image (red equaled high perfusion, blue equaled low perfusion), which correlated with the blood flow velocity in the face skin. The mean perfusion of the entire face was calculated at each time point based on the flux images.
Data analysis and statistics
All values are presented as mean ± standard deviation (SD), unless otherwise stated. Percentage changes are presented with a 95% confidence interval (CI) and headache scores are presented as median and individual values. We defined baseline as t0 before the start of the experiment. Calculation of sample size was based on the difference between CO and placebo in incidence of headache at a 5% significance (two-tailed) level and 90% power. Twelve migraine without aura patients were included, assuming that CO would induce migraine attacks in 75% of the patients versus 10% after placebo.
The area under the curve (AUC) was used as a summary measure for analyzing the difference in response between CO and placebo in accordance with the trapezium rule (30). The primary endpoint was the difference between incidence of migraine headache and AUC for headache score (0–13 h) between CO and placebo. The secondary endpoints were the differences between CO and placebo in the AUC (0–120 min) for HR, MAP, RF, PetCO2 and capillary blood samples (COHb, hemoglobin, sodium, potassium and glucose). AUC was calculated by subtracting the baseline to reduce within-participant variation between sessions. AUC and baseline differences of all variables between CO and placebo were compared using the non-parametric Wilcoxon signed rank test. We tested for period and carry-over effects for all baseline variables with the Mann-Whitney test.
All statistical analyses were performed with SPSS version 23.0 (Chicago, IL, USA). We made no adjustment for multiple analyses. Five percent (p < 0.05) was accepted as the level of significance.
Results
Incidence of adverse events 0 to 13 h after inhalation of CO or placebo.

NA: not applicable.
The following adverse events were reported by a few participants: After CO, yawning urge (n = 2), urination urge (n = 1), hunger (n = 3); after placebo urination urge (n = 1).
Migraine-like attacks and headache
Characteristics of headache and associated symptoms in migraine without aura patients after CO and placebo (0–13 h).

Localization/intensity/quality (throb = throbbing; pres = pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
Nausea/photophobia/phonophobia.
Migraine-like attacks are defined according to criteria described in methods.
Pain freedom or pain relief (≥50% decrease of intensity) within 2 h. ND: not determined.
Excedrin contains 250 mg paracetamol, 250 mg aspirin and 65 mg caffeine.
Treo contains 500 mg acetylsalicylic acid and 50 mg caffeine.
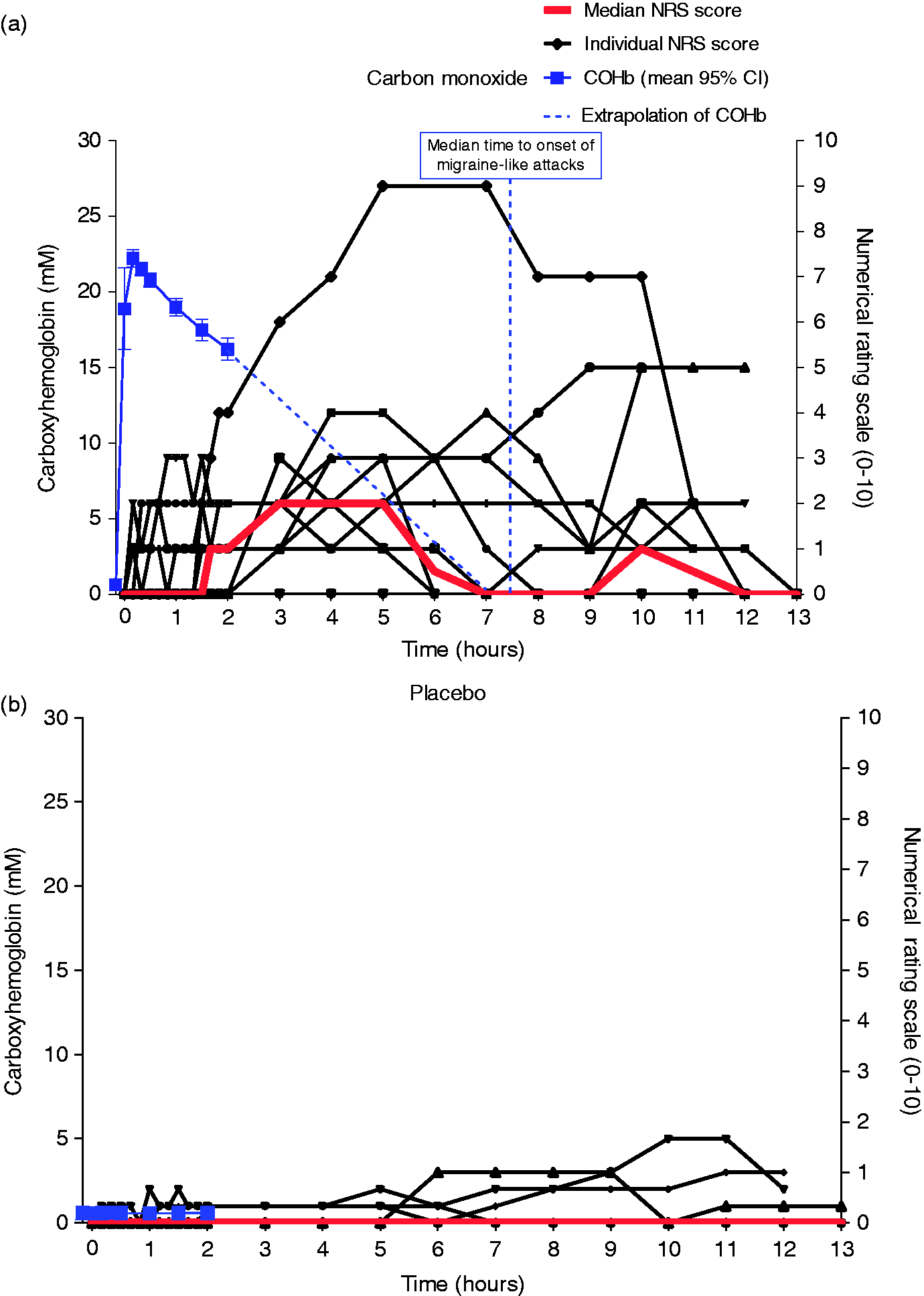
Headache intensity and carboxyhemoglobin levels. Individual and median headache intensity shown on the right y axis after carbon monoxide (CO) (a) and placebo (b) in 12 migraine without aura patients. Mean COHb concentrations (95% CI) is shown on the left y axis. The COHb time course after 2 h has been extrapolated to 0%. The median peak headache score was 2 (range 0–9) after CO inhalation compared to 0 (range 0–5) after placebo.
Carboxyhemoglobin levels
After inhalation of a mean CO volume of 364 ml (SD = 69 ml), the participants reached a mean COHb of 22.3% (SD 0.8%). COHb was estimated to reach baseline levels again after 8 h by extrapolation of the COHb curve (Figure 1(a)). The COHb (p = 0.002) was significantly different after CO inhalation compared to placebo, and we found no difference in the other vital blood variables (Figure 2(a)).
Capillary blood samples and vital variables. Mean ± SD values of capillary blood samples (a) and vital variables (b) 0 to 120 min after CO inhalation and placebo.
Facial skin blood flow and vital signs
CO significantly increased the facial skin blood flow compared to placebo (AUC0–120 min, p = 0.004) (Figure 3). AUC0–120 min for heart rate was larger after CO inhalation compared to placebo (p = 0.010). There was no difference in AUC0–120 min for RF (p = 0.010), MAP (p = 0.638) and PetCO2 between CO and placebo (p = 0.894), (Figure 2(b)).
Facial skin blood flow changes after CO inhalation and placebo (0–120 min) shown in mean perceptual (95% CI).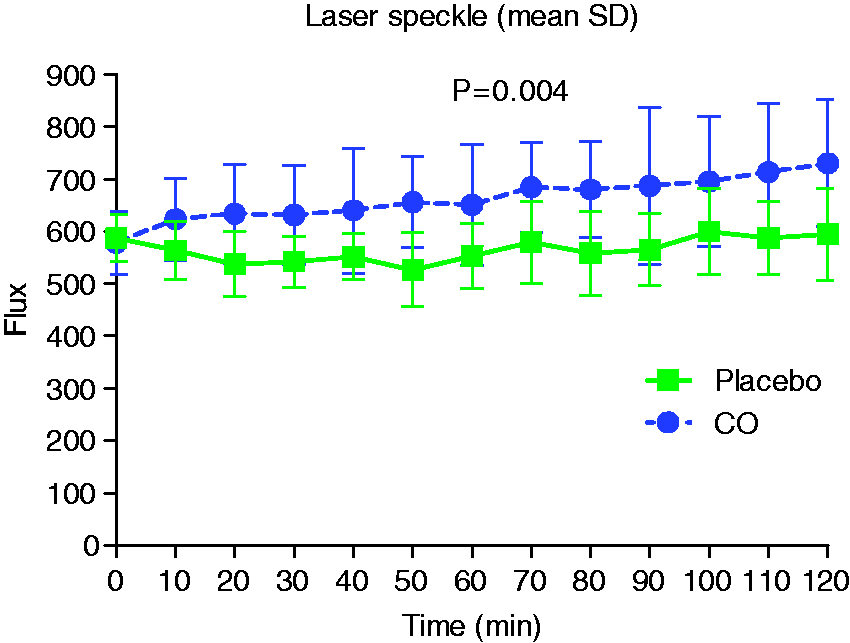
Discussion
The main outcome of the study was that CO inhalation did not provoke more migraine–like attacks in migraine patients compared to placebo. CO induced more headache in patients compared to placebo. Compared to previously published data in healthy volunteers, CO induced more migraine-like attacks in migraine patients compared to healthy volunteers (Figure 4(a) and (b)). In the following, we will discuss mechanisms underlying CO induced headache and the low potency in migraine induction.
Headache intensity and carboxyhemoglobin levels (n = 12). (a) CO headache score for MO and healthy volunteers. (b) Placebo headache score for MO and healthy volunteers.
CO is endogenously produced as a byproduct of heme catabolism by enzymatic activity of heme-oxygenase (HO) (31–33). Eighty-five percent of CO in the body binds to hemoglobin, the rest binds to extravascular heme-proteins (34), exerting different cellular mechanisms. HO is co-localized with nitric oxide synthase (NOS) in the vascular endothelium, adventitial nerves of blood vessels, autonomic nervous system (35) and trigeminal ganglion (36,37). HO activities in neurons, endothelial cells and smooth muscle cells of cerebral vessels are involved in the modulation of cerebral blood flow (CBF) (38–40). Several studies implicated HO/CO in nociception or modulation of nociception (13,41,42). Endogenously produced CO is a putative neural messenger and a weak activator of cGMP (3). In contrast, NO activates the soluble guanylate cyclase (sGC) more than 100-fold (43). Both CO and NO can be generated from the vascular wall and possess many similarities (14). In vivo studies reported that CO dilated cerebral arteries and arterioles via cGMP and activation of calcium-gated-potassium channels (5,6,44,45). In humans, exposure to CO (mean COHb 20–22%) increased CBF by 26% (46) and the velocity of the middle cerebral artery by 31% (12). In the present study, CO increased facial skin blood flow in migraine patients, similar to previously reported flow increase in healthy volunteers (12). To date, the CO effect on the diameter of cerebral arteries has not been investigated in vivo in humans. Topical application of CO dilates pial arterioles in newborn pigs in a dose-dependent manner (44), and the vascular effect of CO may be blocked by NOS inhibitors (47). In the present study, 50% of patients reported migraine-like attacks after CO inhalation compared to 80% after administration of the NO donor glyceryl trinitrate (GTN) (15). Thus, CO is not as potent as NO in migraine induction (15,18).There are several important factors that determine the difference in potency between CO and NO in migraine induction: a) NO is a stronger activator of cGMP than CO and diffuses freely into blood vessels and brain (43); b) NO dilates extracerebral and intracerebral arteries in patients with migraine more than in healthy volunteers (21), the CO effect on human cerebral arteries is unknown; c) CO is highly bound to hemoglobin, which leaves very small amounts of free CO that can bind to sGC; d) because of toxicity it is impossible to use higher concentrations of CO.
CO-induced hypoxia may be the main mechanism behind the migraine or headache-inducing effects of CO inhalation. CO binds to hemoglobin with an affinity 200–250 times higher than oxygen (48) and it also displaces the O2 dissociation curve (46,49). Thus, CO impairs the oxygen carrying capacity of hemoglobin and oxygen release to the tissue. Hence, CO-Hb induces a hypoxic environment in high oxygen demanding tissues (50). Experimentally induced hypoxia (SpO2 72%) induced migraine without aura-like attacks in 53% of migraine with aura patients and headache in 79% of healthy volunteers (51). This is similar to 50% migraine induction reported in the present study and 83% headache in healthy volunteers exposed to CO inhalation (12). Mild hypoxia (SpO2 75-80%) only induced migraine without aura in 43% in a mixed group of migraine patients (52). In the current study, CO induced relatively mild hypoxia (SpO2 78%) by reducing the oxygen binding capacity of hemoglobin. This may explain the low potency in migraine induction after CO. To achieve severe hypoxia (SpO2 72%) after CO exposure would require using a mean COHb of 28%, which is impossible because of the high risk of toxicity. A short supraphysiological CO concentration was used to mimic the effects of small amounts of endogenous produced CO (53). To distinguish between the direct cellular effects of CO and the hypoxic effects, a more prolonged exposure to CO in a lower dose may be useful. This would likely allow a higher amount of free CO to reach the brain and exert direct cellular effects by binding to different heme-proteins. However, a slower and prolonged exposure may cause accumulation of CO in all tissues in the body and increase the risk of adverse events (34).
It is worth noting that migraine patients tended to report a longer duration and intensity of headache compared to healthy volunteers (Figure 5). Moreover, CO inhalation provoked migraine-like attack in one healthy volunteer (12) compared to six migraine patients in the present study. Several cases reported in the literature that prolonged exposure to non-lethal concentrations of CO mimicked migraine-like attacks (10,11,54). Furthermore, methylene chloride, which upon exposure is metabolized to CO, induced severe headache with migraine-like symptoms (54). Accidental CO exposure in divers causes headache and nausea (55). Moreover, subcutaneous sumatriptan injection completely aborted migraine-like headaches following CO exposure (10). Interestingly, migraine-like attacks disappeared after identifying and eliminating the source of exposure to CO (10,11). Furthermore, CO produced by combustion processes is highly toxic due to exposure to various other toxic molecules like carbon particles, benzo-α-pyrene, nitrogen oxide, nitrogen dioxide, polycyclic aromatic hydrocarbons and exhaust emissions formaldehyde (13,56,57). These other toxic molecules may play a role in accidental CO-induced migraine and headache. Interestingly, an association between daily clinic visits for migraine and ambient air pollution (fine particle levels including CO) has been reported (58,59). In the present study, we did not measure for air pollution. However, no major air pollution warnings were issued by the Danish Meteorological Institute during the study period. Furthermore, all participants arrived at the study center with COHB < 1%.
Median headache intensity on verbal rating scale (VRS) after CO inhalation in 12 migraine patients in the present study and in 12 healthy volunteers reported in a previous study.
Future perspectives
Different mechanisms of CO-induced headache and vascular effect should be considered. Several studies showed that CO-induced hypoxia alone could not explain the cerebral hemodynamic changes (4,51,60,61). Therefore, different hemodynamic effects of CO and pure hypoxia are suggested (12). CO-induced headache may be due to direct cellular effects, because of CO’s ability to escape the scavenging properties of hemoglobin and bind to other intracellular hemoproteins. Future studies may explore the possibility of using CO-releasing molecules (53) as pharmacological approaches in animal and human migraine models. Biomarkers of CO exposure in peripheral blood may be useful to understand the headache inducing mechanism of CO (56,62). Furthermore, we cannot rule out that endogenously produced CO in intra- and extracerebral blood vessels and sensory neurons may play a role in spontaneous migraine attacks. Interestingly, recent studies suggest interactions between the signaling molecule hydrogen sulfide (H2S), CO and NO (63,65). Furthermore, H2S exerts a multitude of beneficial effects in the vessel wall, including suppression of oxidative stress, inhibition of inflammation, and enhancement of vasodilation (66,67). The present study suggests that the CO inhalation model is not suitable to study migraine, and future human studies are needed to further explore the pain mechanisms behind CO-induced headache using alternative CO delivery systems.
Key findings
CO inhalation induced prolonged moderate headache in migraine without aura patients. CO inhalation in non-toxic concentrations failed to induce more migraine compared to placebo.
Footnotes
Acknowledgement
We thank lab technicians Lene Elkjær and Winnie Grønning for expert assistance, medical student Mustafa Ghanizada for help with figures, and all patients participating in the study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Messoud Ashina is a consultant, speaker or scientific advisor for Allergan, Amgen, Alder, ATI, Eli Lilly, Novartis, and Teva, primary investigator for Amgen 20120178 (Phase 2), 20120295 (Phase 2), 20130255 (OLE), 20120297 (Phase 3), Alder ALD403-CLIN-001 (Phase 3), Amgen PAC1 20150308 (Phase 2 a), and GM-11 gamma-Core-R trials.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from Lundbeck Foundation (R155-2014-171) and Candys Foundation, Liechtenstein.
