Abstract
Background
We report the first literature description of ictal epileptic headaches closely mimicking glossopharyngeal neuralgia and short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing.
Case 1
A 37-year-old man complained of short-lasting, electric-shock like headache, confined to the pharynx. During the episodes, he could not speak because he felt “words blocked at the throat”. An EEG recorded epileptic discharges concomitant with headache; a brain MRI disclosed frontal polymicrogyria.
Case 2
A 66-year-old man complained of short-lasting, right periocular headache, associated with ipsilateral ptosis, conjunctival injection and lacrimation. Some episodes were followed by tonic contraction of the right facial and limb muscles; on one occasion, headache was followed by a generalized seizure. A brain MRI revealed hippocampal abnormalities.
Discussion
These cases highlight the complex relationship between headache and epilepsy, and suggest a possible contribution of cortical structures to the genesis of paroxysmal headaches such as glossopharyngeal neuralgia and short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing.
Background
Headache may be the isolated symptom of a seizure. This condition, also known as “ictal epileptic headache” (1), is very rare, occurring in 3–5% of epileptic patients, and may be misdiagnosed due to the similarities with primary headache disorders, mainly migraine with or without aura (1,2).
Ictal headache must be differentiated from headache occurring after the ictal phenomenon, that is, post-ictal headache, which is common and follows up to 50% of generalized tonic-clonic seizures (2).
The complex and bidirectional association between headache and epilepsy is well reflected within the current International Classification of Headache Disorders 3rd edition (ICHD-3 beta) criteria (3), which recognize that headache can either follow “post-ictal headache” (cod. 7.6.2) or trigger a seizure; the latter phenomenon has been reported only after an attack of migraine with aura (3), and is referred to as migralepsy or migraine aura-triggered seizure (cod.1.4.4).
Finally, the ICHD criteria recognize the possibility of headache occurring simultaneously with the onset of the seizure. The former ICHD-2 criteria (4) designated this condition as hemicrania epileptica (HE) and described it as a headache “secondary to a partial epileptic seizure, ipsilateral to the epileptic discharge, lasting seconds to minutes and with characteristics of migraine”. In the current ICHD-3 beta criteria, this entity, now called “headache attributed to epileptic seizure” (cod. 7.6), has lost more detailed clinical descriptions, including “migrainous” phenotype and short duration. Furthermore, the current diagnostic criteria do not specify the relation between headache side and epileptic focus; this is in accordance with the observation by Belcastro and colleagues (1) that headache can be the symptom not only of a focal seizure but also of a non-convulsive status epilepticus (SE) and that headache can be ipsilateral or contralateral to the ictal epileptiform discharge.
The clinical features of ictal headache have been evaluated in only a few literature studies [1–2,5–6].
Herein we report the first observation to our knowledge of ictal headache cases closely resembling two paroxysmal primary headache disorders: Glossopharyngeal neuralgia (GN) and short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT).
Case 1
A healthy 37-year-old man complained of childhood-onset headache attacks confined to the left hemipharynx, with electric-shock like quality and severe intensity, lasting five to 20 seconds; some attacks were triggered by swallowing. During the episodes, he could not speak because he felt “like words were blocked at the throat”, although he reported that he was fully aware of his surroundings; pain could evoke occasional contraction of the facial muscles, with variable side.
The attacks had daily frequency, and could be isolated or grouped in sequences lasting up to 30 minutes. Pain occurred frequently during sleep, causing awakening.
Throughout the years, he had received different diagnoses, including glossopharyngeal neuralgia, primary stabbing headache, anxiety disorder and epilepsy, and tried various drugs without benefit, including AEDs (carbamazepine up to 800 mg/day, lamotrigine up to 400 mg/day, topiramate up to 200 mg/day, pregabalin up to 600 mg/day, valproate up to 1200 mg/day), benzodiazepines and SSRIs (duloxetine, sertraline).
The neurologic examination was normal. A long-term video-EEG recorded epileptic discharges with left-sided emphasis, concomitant with “typical” episodes of pain. Headache attacks were associated with oral automatisms and slight contractions of facial muscles with inconstant laterality; during the attacks, he was not able to speak, it not being possible to distinguish between aphasia and anarthria due to the short duration of the event.
A 3-tesla brain MRI protocol for epilepsy disclosed left frontal polymicrogyria involving the insular cortex and the frontal operculum (Figure 1). The final diagnosis was pharmacoresistant focal epilepsy secondary to left frontal polymicrogyria.
Brain magnetic resonance images show focal cortical thickening, due to small gyri with shallow sulci, involving left insular cortex and adjacent frontal operculum. The two top images are axial (a) and coronal (b) T2 turbo spin echo images; the bottom images are axial (c) and coronal (d) multiplanar reconstructions of volumetric T1 images.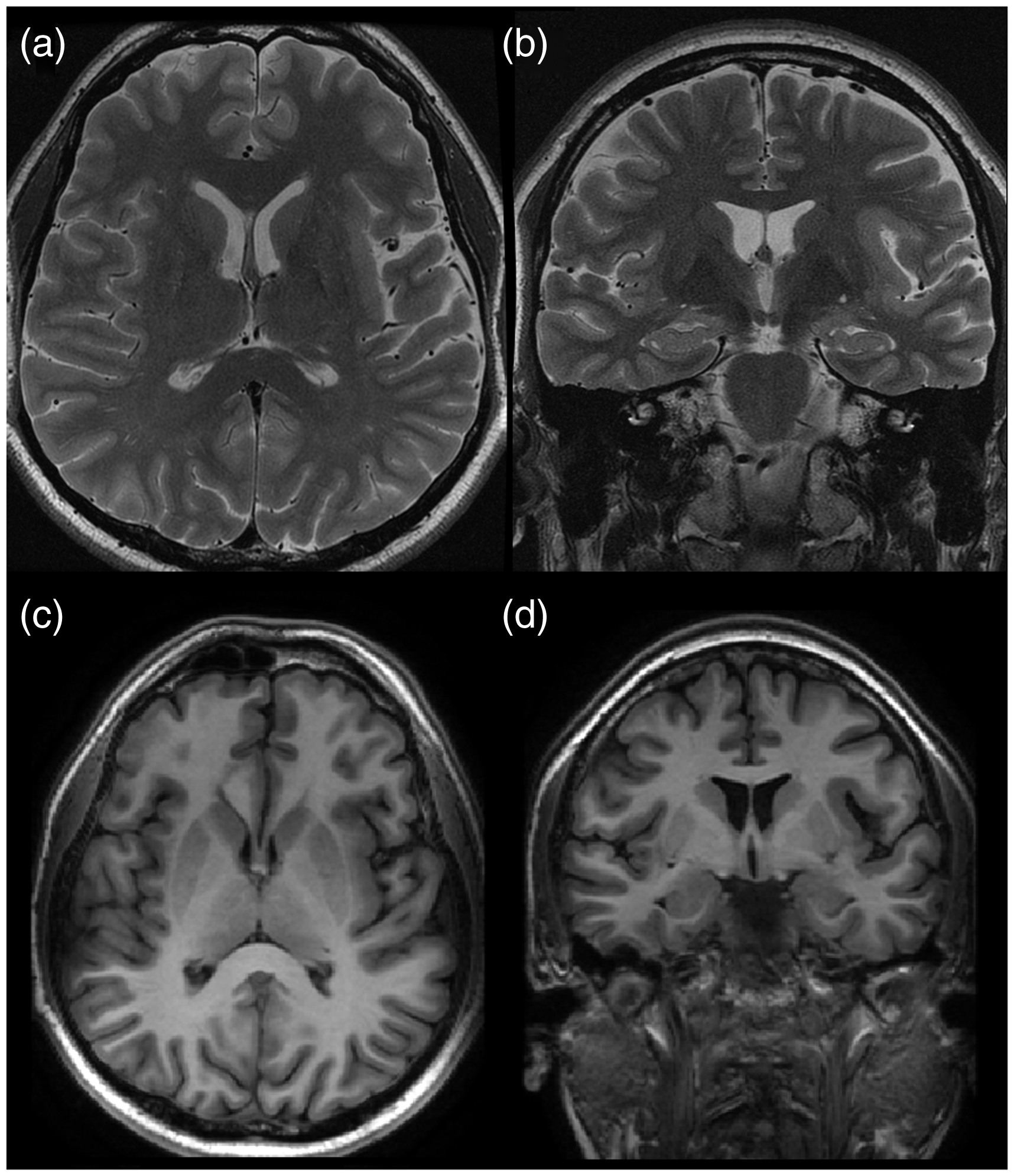
The patient was offered a pre-surgical evaluation with stereo-EEG recording, but he declined.
Case 2
An otherwise healthy 66-year-old man complained of a 10-year history of headache, first experienced after an injury with a high-voltage electrical discharge. Pain had right periocular localisation, severe intensity, stabbing quality, and was associated with ipsilateral ptosis, conjunctival injection, and lacrimation. Attacks lasted about two minutes; frequency was one to eight attacks per day. Some episodes were followed by tonic contraction of facial and limb muscles with right-sided prevalence, lasting a few minutes; during these events, the patient was fully aware of his surroundings. On a single occasion, an otherwise typical attack was followed by loss of consciousness and tongue biting. Furthermore, since the electrical injury, he complained of slowly progressive memory impairment.
Neurological examination disclosed only mild memory impairment. A 3-tesla brain MRI revealed signal abnormality involving the hippocampus bilaterally (Figure 2). An EEG recorded after a typical headache attack showed post-ictal abnormalities involving the temporal recordings, with left-sided emphasis. Long-term video-EEG monitoring could not be performed, while an inter-ictal EEG did not show significant abnormalities.
Axial T2-weighted brain magnetic resonance images showing bilateral subtle hyperintensity of hippocampus and dilatation of the temporal horns of lateral ventricles.
Treatment with lamotrigine up to 100 mg po bid was completely and persistently effective on headache.
Discussion
To date, no reports have been published on headaches closely resembling GN and SUNCT as the sole manifestation of epilepsy.
There are few literature descriptions of short lasting ictal headaches. Saitowitz et al. (2) described a patient with ictal headache resembling primary stabbing headache, even if some features (i.e. urinary incontinence, post-ictal confusion, and lack of response to indomethacin) were atypical and suggestive of an ictal aetiology. Charlesworth et al. (5) reported a case of ictal paroxysmal headache resembling trigeminal neuralgia, but in the absence of sensitive triggers and of shock-like pain quality; besides, the patient subsequently developed attacks with relevant epileptic features.
Other short-lasting ictal headaches reported in the literature (6) are not comparable to any primary headache disorder.
As demonstrated by our cases and previous literature reports, the clinical differentiation between ictal headaches and primary headache disorders can be challenging, yet crucial. Saitowitz et al. (2) stated that ictal headache lasts between 5 and 10 minutes, has a stabbing or pulsating quality and some accompanying features that may suggest an ictal aetiology, including: Transient loss of contact, ictal and/or post-ictal confusion, incontinence, and coloured visual auras. The nocturnal occurrence is typical of epileptic genesis, while the response to therapy (benzodiazepines and AEDs) may not differentiate an ictal headache from a primary headache disorder. Ictal EEG remains the gold standard for diagnosis (1).
The patient we described in case 1 fulfils the ICHD-3 beta (3) criteria for GN, except for criterion E (“not better accounted for by another ICHD-3 diagnosis”). A major clinical clue for an ictal aetiology could be the presence of language disturbance and facial muscle contractions, which, however, could also be interpreted as a tic douloureux (7). Although mainly manifesting during the day, attacks of GN can also disturb the sleep, awakening the patient at night (7). Some other features, including the absence of triggers other than swallowing, young age and male gender, are atypical for GN, even if possible.
The clinical picture of case 2 closely resembles SUNCT (3). Some clinical clues helped us in diagnosing a secondary headache disorder, including: The presence of memory impairment, the temporal relation to the electrical injury, the presence of tonic muscle contraction during the episodes, and the occurrence of a seizure following a typical headache attack. Interestingly, this condition responded absolutely to lamotrigine, which is effective in SUNCT too.
Epileptic activity causing ictal headache could occur in any lobe, consistently with multiple representations of pain in the human cerebral cortex (8,9); however, according to some authors (10), parietal lobes are the most common localisation. It has been stated (10) that ictal focus is usually contralateral to head pain in parietal lobe epilepsy, while it is ipsilateral in temporal lobe epilepsy. However, as evident from previous ictal headache cases (1) and from the current ICHD-3 beta criteria, the correlation between seizure onset zone and pain side is not straightforward, and headache could be ipsilateral or contralateral to the ictal epileptiform discharge; interestingly, case 2 was a temporal lobe epilepsy with left-sided emphasis, but pain was localized in the right side.
The likely epileptic focus in our cases, that is, the insular cortex and the frontal operculum in case 1 and the mesial temporal lobes in case 2, confirms pre-clinical data on the role of these areas in pain processing (8,9). The MRI appearance in case 2 was noteworthy, and we did not find any previous literature report on similar brain damages after an electrical injury.
Our cases suggest that a cortical dysfunction, which was evident from a clinical (i.e. seizures), electrophysiological (EEG abnormalities) and morphologic (MRI abnormalities) perspective, may generate paroxysmal headaches with the features of GN and SUNCT. This eventuality is in contrast with the current pathogenic model of GN and SUNCT, which points toward a role of subcortical or peripheral structures, that is, neurovascular conflict or central sensitization of the spinal trigeminal nucleus and hypothalamic dysregulation, respectively (7,11). Only a few literature studies (12) have hypothesized a pathogenic role of cortical areas, including the insula and temporal lobes, based on MRI evidence of cortical thickness changes, either increase or reduction, in patients with classical trigeminal neuralgia. However, it is not clear whether these changes are the expression of a neuronal plastic adaptation to daily attacks of headache, or represent a structural predisposition to pain.
On the other hand, the common cortical genesis of our cases further highlights the similarities and the possible overlap between cranial neuralgias and short lasting unilateral neuralgiform headaches (11).
A pathophysiological explanation of our cases could be that the activation of cortical areas involved in pain processing due to the epileptic discharge may disrupt their descending connections to the spinal trigeminal nucleus and the hypothalamus, which in turn can generate head pain with the features of GN and SUNCT. Alternatively, as suggested by Bernasconi et al (10), an increase of blood flow in brain areas affected by the seizure and the subsequent stretching of dural vessels may provoke stimulation of dural trigeminal afferents, which may cause the activation of the trigeminovascular system and therefore headache.
Clinical implications
This is the first literature report of headache attacks closely resembling glossopharyngeal neuralgia and SUNCT as the sole manifestation of epileptic seizures. The clinical differentiation between ictal headache and primary headache disorders can be challenging, yet crucial. The likely epileptic focus in our cases (i.e. the insular cortex, the frontal operculum, and the mesial temporal cortex) confirms pre-clinical data on the role of these areas in pain processing. Our cases suggest that a cortical dysfunction may generate paroxysmal headaches with the features of glossopharyngeal neuralgia and SUNCT in contrast with the current pathogenic model, which points toward a role of subcortical or peripheral structures. The common cortical genesis of our cases highlights the similarities and the possible overlap between cranial neuralgias and short lasting unilateral neuralgiform headaches.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
