Abstract
Premise
In this article we review some lesser known cranial neuralgias that are distinct from trigeminal neuralgia, trigeminal autonomic cephalalgias, or trigeminal neuropathies. Included are occipital neuralgia, superior laryngeal neuralgia, auriculotemporal neuralgia, glossopharyngeal and nervus intermedius neuralgia, and pain from acute herpes zoster and postherpetic neuralgia of the trigeminal and intermedius nerves.
Problem
Facial neuralgias are rare and many physicians do not see such cases in their lifetime, so patients with a suspected diagnosis within this group should be referred to a specialized center where multidisciplinary team diagnosis may be available.
Potential solution
Each facial neuralgia can be identified on the basis of clinical presentation, allowing for precision diagnosis and planning of treatment. Treatment remains conservative with oral or topical medication recommended for neuropathic pain to be tried before more invasive procedures are undertaken. However, evidence for efficacy of current treatments remains weak.
Keywords
Introduction
Clinical features and treatment of facial neuralgias and comparison with trigeminal neuralgia.
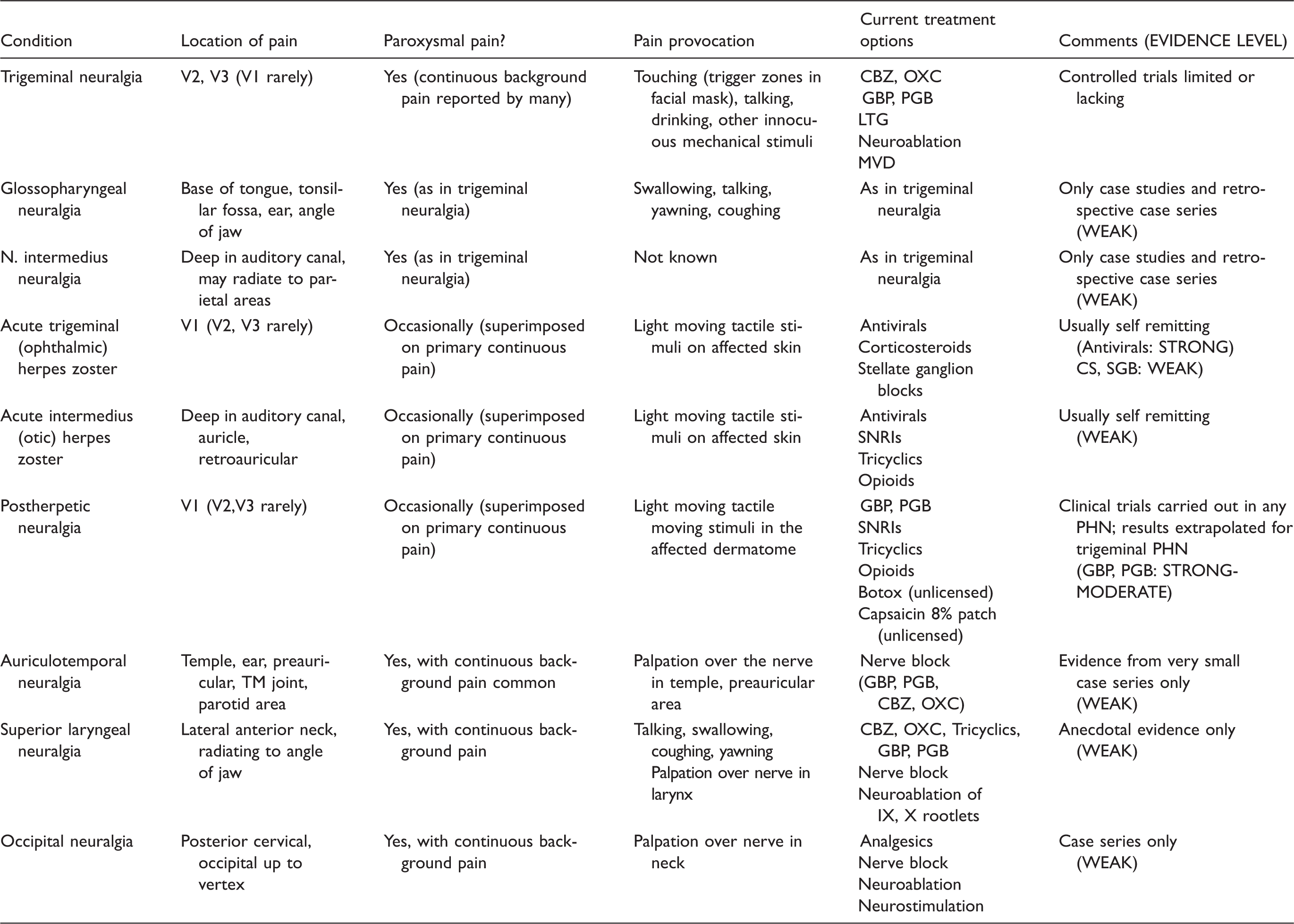
V1, ophthalmic division of trigeminal nerve; V2, maxillary division of trigeminal nerve; V3, mandibular division of trigeminal nerve; IX, glossopharyngeal nerve; X, vagal nerve; CBZ, carbamazepine; OXC, oxcarbazepine; GBP, gabapentin; PGB, pregabalin; LTG, lamotrigine; MVD, microvascular decompression; SNRI, serotonin-noradrenaline reuptake inhibitor; SGB, stellate ganglion block; PHN, postherpetic neuralgia.
Occipital neuralgia
Definition
First described in 1821 by Beruto y Lentijo and Ramos (1) occipital neuralgia is a pain within the distribution of one or more of the greater, lesser or third occipital nerves. It is felt in the upper posterior cervical region and back of the head. It may spread up towards the vertex of the scalp.
Symptomatology
Predominantly paroxysmal or shooting/stabbing in nature; a persistent deep aching between paroxysms may be present. Pain may be provoked by palpation over the occipital nerve and temporarily eased by local anesthetic block.
Associated symptoms of visual impairment/ocular pain, tinnitus, dizziness, and nausea have been described but are probably misdiagnoses of other headache disorders.
Epidemiology
From a Dutch general practitioner electronic patient database Koopman et al. identified the incidence of occipital neuralgia to be 3.2 per 100,000 patient years (95% CI 2.2–4.5) with a slight female predominance (2).
Pathophysiology
Occipital neuralgia has been reportedly caused by irritation or compression of the occipital nerves by muscular entrapment (3), vascular anomalies (4–6), vascular inflammation (7), primary (8–10) or metastatic tumors (11), myelitis (12), multiple sclerosis (13), cervical degenerative disease (14) or callus formation (15) and secondary to scalp biopsy (16), and whiplash injury (17). However, some of these descriptions may be better accounted for under other diagnoses, for instance; persistent headache attributed to whiplash, headache attributed to giant cell arteritis, or nummular headache.
Diagnostic considerations
Clearly, given the variety of etiologies, a priority should be to exclude serious pathology of the cervical structures with either magnetic resonance or radiographic imaging and relevant blood tests. Once excluded, consideration can be given to other differential diagnoses.
As can be seen, there is considerable overlap in symptoms of occipital neuralgia and those in cervicogenic headache, referred muscle pain, and other primary headache disorders. The patient history may prove discriminating but can be vague.
Tenderness over the occipital nerve region is a non-specific sign and probably leads to over diagnosis. The location of palpation may be important as palpation either above or below the superior nuchal line may also evoke pain from tender occipitalis, trapezius, and semispinalis capitis muscles, respectively. Occipital nerve tenderness would also be expected in cases of nerve irritation at the cervical facet joint, which may be more in keeping with cervicogenic pain. Ultrasound may be useful in detecting muscular nerve entrapment (18).
Treatment
Multiple treatments have been investigated for occipital neuralgia including conservative treatment, simple analgesia, local anesthetic nerve block with steroid (19), botulinum toxin injection (20,21), radiofrequency ablation (22–24), as well as surgical alternatives such as neurectomy (25), neurolysis (26), rhizotomy (27), and neurostimulation (28). Unfortunately neither the efficacy nor long-term outcomes of these have been established in well-designed clinical trials and therefore the evidence for recommendation is weak.
Obviously in the case of local pathology, such as tumors, surgical removal of the offending lesion is recommended if possible, but if no such pathology is present, treatment should be based on a cascade from least invasive conservative treatment through to more invasive procedures on an individual basis, until good quality evidence becomes available.
Superior laryngeal neuralgia
Definition
A very rare disorder first described by Tobold in 1866 (29) and characterized by severe pain in the lateral aspect of the anterior neck, radiating from the side of the thyroid cartilage or pyriform sinus to the angle of the mandible and occasionally the ear.
Symptomatology
Characteristic evocation of pain by palpation of the superior laryngeal nerve at its point of entrance into the larynx (trigger point) but may also be precipitated by talking, swallowing, coughing, and yawning. Pain on swallowing can affect dietary intake and cause weight loss (30). Hoarseness and cough may be associated (31,32).
Epidemiology
Little robust data exists but may be 1.3% to 3% of cranial neuralgias (33,34), affecting both sexes equally (35).
Pathophysiology
Superior laryngeal neuralgia has been reportedly caused by trauma/previous surgery (31,35,36), inflammation (33), laryngeal diverticulum (34), and deviation of the hyoid bone (37).
Diagnostic considerations
After exclusion of serious pathology (e.g. pharyngeal carcinoma), the differential diagnosis must include other pain conditions that affect this anatomical region, for example, Eagles syndrome, carotidynia, and glossopharyngeal neuralgia. These may be differentiated by the location within which palpation provokes the familiar pain.
Treatment
Treatment is based on case reports but carbamazepine, gabapentin, and amitriptyline have been shown effective (30,32,37–39).
Pain relief for up to 1 year following nerve blocks with 2% lignocaine (33,40) and 5%–10% lignocaine (37) have been reported.
While case reports of peripheral neurectomy have shown initially good results with follow-up between 7 months and 4 years, some cases, however, required subsequent intracranial section of the glossopharyngeal and upper vagal rootlets for recurrence of pain. Follow up of these secondary procedures has not been reported (31,41–43). Surgical removal of contributing local pathology has also been reportedly successful (34,44).
Auriculotemporal neuralgia
Definition
Auriculotemporal neuralgia is a pain of nerve origin felt in the temple, ear, preauricular, temporomandibular joint, and parotid area.
Symptomatology
It is almost always a unilateral, moderate to severe pain, which can be stabbing, aching or throbbing with associated paresthesias, tenderness or trigger points in the preauricular/temporal area. Episodes last several seconds to 30 minutes and a background pain may be present.
Epidemiology
Two studies suggest a prevalence of 0.2–0.4% being more common in middle-aged females (45,46).
Pathophysiology
Entrapment of the nerve may occur as it runs through the lateral pterygoid muscle (47,48), local effects of compression may accompany inflammation/myositis in the area as it traverses the roof of the infratemporal fossa (49,50) or be caused by synovial cysts (51), malformation or aneurysm of the middle meningeal artery (50), and fracture to the mandibular condyle (52). Perineurial spread of tumor affecting the auriculotemporal nerve has also been reported (53).
Diagnostic considerations
A much more common source of discomfort in this area is pain from the temporomandibular joint. Dysfunction of this structure must be excluded by careful functional examination of the joint and the masticatory muscles. Sensation from this joint is mostly carried by the auriculotemporal nerve and thus local anesthetic blockade is not a discriminatory diagnostic procedure. As auriculotemporal neuralgia can result from pathology of the joint or infratemporal fossa (see references above), it would be advisable to perform a CT or MRI scan of the region to exclude this pathology. As a branch of the trigeminal nerve, pain in this distribution could represent a true trigeminal neuralgia and in these cases treatment would be the same as for trigeminal neuralgia.
Treatment
Due to the rarity of this condition no high quality studies exist. Evidence for treatment is based on case series showing response to local anesthetic block of the auriculotemporal nerve with or without steroid, however, the use of concomitant steroids has been associated with cutaneous atrophy in this region (54). Response to single or multiple injections has been reported to last 1–2 years after which repeat injections may be necessary (45,46,55). Subjects may also be responsive to pharmacological therapies such as carbamazepine and gabapentin (45,46).
Glossopharyngeal neuralgia
Definition
The term glossopharyngeal neuralgia was proposed by Harris in 1921 (56). Glossopharyngeal neuralgia is defined as severe transient stabbing pain perceived in the distribution of the glossopharyngeal nerve, including the ear, the base of the tongue, the tonsillar fossa, or the angle of the lower jaw.
Symptomatology
Attacks come in clusters of sharp, stabbing pain felt in the ear, posterior part of the tongue, tonsillar fossa, or in the angle of the mandible. They are provoked by swallowing, talking, yawning or coughing and may remit and relapse for months. The pain can be severe enough for patients not to eat and to lose weight. Spontaneous remissions and recurrences are common. Glossopharyngeal neuralgia can be divided into an oropharyngeal and an otalgic form, according to the predominant pain localization (57). In up to 10% pain attacks are associated with vagal symptoms such as bradycardia, syncope or even cardiac arrest, which is then called vagoglossopharyngeal neuralgia, see (58) for review. Mild sensory deficits may be present. Prominent sensory deficits or a missing gag reflex raise the suspicion of a glossopharyngeal neuropathy and the underlying cause should be looked for.
Epidemiology
The disorder is very rare with an estimated incidence of 0.8 per 100.000 (57). A retrospective review from one institution described 217 patients seen over a period of 27 years (59).
Pathophysiology
Afferents from the carotid sinus as well as the involvement of vagal afferents have been implicated as the cause of associated bradycardia and syncope. Vascular compression of the nerve root entry zone has been shown similarly to trigeminal neuralgia, with precise localization by 3D MRI imaging (60,61). Rare familial cases have been described, hypothesized to be caused by mutations in a voltage gated sodium channel gene (62).
Diagnostic considerations
MR-imaging may be used to show neurovascular compression of the glossopharyngeal nerve.
Secondary (symptomatic) glossopharyngeal neuralgia may be caused by a variety of disorders like neck trauma, multiple sclerosis, regional tumors, and Chiari I-malformation. In most of these cases, the disorder should be named glossopharyngeal neuropathy, since the pain is usually continuous and there is damage to the nerve or its nucleus through the underlying disease. Here, the pain is often described as burning, squeezing, or like pins and needles, with superimposed paroxysms. Hints toward a neuropathy as opposed to a neuralgia are sensory deficits in the ipsilateral posterior part of the tongue and tonsillar fossa and a weak or missing gag reflex.
Treatment
Glossopharyngeal neuralgia is usually responsive to the sodium channel anticonvulsants as used in trigeminal neuralgia. Cases responding to gabapentin and pregabalin have also been reported. Response rates may decline over time. When pharmacological treatment fails, microvascular decompression is considered a safe and effective form of therapy (59). A recent review reports long-term pain freedom in 84.7% after microvascular decompression (63).
Percutaneous radiofrequency (thermal) rhizotomy of the 9th and 10th nerves has been used, but frequently causes severe and persistent dysphonia and dysphagia (64). It has mostly been used to treat patients with pain caused by malignancy, i.e. glossopharyngeal neuropathy rather than neuralgia. Surgical rhizotomy is also performed in this indication (65), as is stereotactic radiosurgery (66). Vagal involvement may necessitate cardiac pacemaker placement.
Nervus intermedius neuralgia
Definition
This is a rare disorder, also known as geniculate neuralgia, characterized by brief paroxysms of pain felt deeply in the auditory canal, sometimes radiating to the parieto-occipital region (67).
Symptomatology
Patients complain of intermittent stabbing pain, like electric shocks, deep in the ear. The syndrome is always unilateral and patients are mostly elderly. The pain may be accompanied by disorders of lacrimation, salivation or taste.
Epidemiology
This entity is extremely rare. Less than 150 cases were included into a systematic review spanning a literature search between 1932 and 2012 (67).
Pathophysiology
It is presumed that the etiology of nervus intermedius neuralgia is analogous to that of trigeminal neuralgia, i.e. that there is microvascular compression syndrome.
Diagnostic considerations
To make the diagnosis of nervus intermedius neuralgia, other causes of otalgia must be excluded by neurological, dental, and ENT examinations. There may be an overlap with glossopharyngeal neuralgia. Nervus intermedius neuralgia may be secondary to herpes zoster (see below). There are rare familial cases associated with occipital neuralgia (62,68).
Treatment
The main drugs used are carbamazepine, oxcarbazepine, and lamotrigine. Gabapentin has also been used. Surgical options include microvascular decompression and transection of the nervus intermedius and the geniculate ganglion (67). The evidence for efficacy of any of the procedures is poor.
Acute herpes zoster of the trigeminal and intermedius nerves
Definition
Unilateral pain of less than three months’ duration in the distribution of one or more branches of the trigeminal nerve or the intermedius branch of the facial nerve, caused by and associated with clinical signs of acute herpes zoster (HZ).
Symptomatology
The hallmark sign of HZ is maculopapular or vesicular rash in the involved dermatomes. In the face it is confined to a single dermatome innervated by one the three branches of the trigeminal nerve (69–71). The ophthalmic branch is affected most commonly of the three. When the geniculate ganglion of the facial nerve is involved, the rash appears on the tympanic membrane, auditory canal, auricle, and skin overlying the mastoid process, i.e., areas supplied by the intermedius nerve. Vesicles may also be seen in the anterior third of the tongue, which the infection has reached via chorda tympani, or on the hard palate, supplied by a vestigial remnant branch of the facial nerve (72,73). When the motor branches of the facial nerve are affected to cause facial palsy, the condition by definition is called Ramsay Hunt syndrome (73).
Pain and other sensory symptoms, such as tingling, itch and numbness are present in 90–95% (70,74,75). They may develop days, weeks or occasionally months, before the eruption (70,76). Whether localized in the face or in the ear, it is mostly continuous, and described as burning, throbbing, smarting or aching in quality (70,74). Nearly one-half of patients describe stabbing or shooting pains superimposed (70,74). Mechanical allodynia, usually of the dynamic type, is found diffusely over the involved dermatome, and is present in one-half with trigeminal HZ (70,71). Distinct from trigeminal neuralgia, it is rare to see small trigger zones from which shooting pain are elicited by innocuous stimuli.
Due to viral spread, other cranial nerves may become affected. In trigeminal HZ, cranial nerves III, IV, and VI may be affected to cause ophthalmoplegia, while in in Ramsay Hunt syndrome cranial nerves VIII, IX, X, and XI may be affected in addition to the facial nerve and cause tinnitus, hearing loss, vertigo, nausea, hoarseness, and dysphagia (77). In patients with ophthalmic HZ, 25–50% develop serious eye complications, such as keratitis, iritis, and glaucoma and, rarely, necrotizing retinitis and optic neuritis (78,79). A rare complication is stroke caused by the virus travelling along afferent fibers from the trigeminal ganglion to cerebral arteries to infect the middle cerebral artery.
Epidemiology
The global annual incidence of zoster estimated from over 60 studies is between 3 and 5 per 1,000 person-years (80). The incidence increases steeply with age. Immunocompromised patients and those with autoimmune disease, chronic pulmonary or kidney disease, or depression are at an increased risk for HZ (81). Some 10–15% of all zoster infections involve the ophthalmic division of the trigeminal nerve (78). Other divisions and the intermedius nerve are rarely affected (69). One half of new facial palsy cases are due to varicella zoster virus (VZV) reactivation, and one-sixth fulfills criteria for Ramsay Hunt syndrome (82). The incidence levels may change significantly in the future as VZV vaccinations of children and elderly become more widespread, although in which direction it is difficult to predict (83).
Pathophysiology
HZ is caused by reactivation of the varicella virus which has remained dormant in the cranial nerve sensory ganglia since the primary infection, varicella, contracted in childhood. Once reactivated, the virus migrates distally to the skin causing local inflammation and proximally to the nerve root and the central nervous system (84,85). HZ most frequently occurs in the elderly as cell-mediated immunity to VZV declines. Other groups at risk for zoster are people on immunosuppressive drugs and patients with autoimmune diseases.
Histopathological studies show varying degrees of inflammatory changes in the skin, nerve, ganglion, and spinal cord, which offer a natural explanation for the pain and other symptoms. Inflammatory changes in the skin may induce sensitization of the nociceptors while the virus activation within the nerve causes direct damage to the cell bodies and their axons and converts surviving nociceptors into actively firing ones (86). The viral inflammation commonly extends into the central nervous system, as shown by neuroimaging studies, which may add to the pain pathophysiology (84,85,87).
Diagnostic considerations
The diagnosis is usually easy because of the cutaneous manifestations. Patients seeking advice during the prodromal non-rash phase, or never develop rash (zoster sine herpete) pose a diagnostic difficulty. In these cases, laboratory tests such as direct immunofluorescence assay for VZV antigen or a polymerase-chain-reaction (PCR) assay for VZV DNA in cells from the base of lesions or from CSF samples are helpful (88, 89). Testing for both herpes simplex and zoster may be needed if the clinical picture is unclear.
Treatment
In the vast majority HZ runs a self-limiting course. Antivirals shorten the duration of the infection and reduce pain but they do not prevent the development of postherpetic neuralgia (PHN) (90). Recent guidelines recommend their commencement in any HZ involving the head and neck. Optimally they should be commenced within 72 hours after the appearance of the rash (90). In immunocompromised patients and those with a complicated form of HZ, especially if they are elderly, intravenous acyclovir is recommended (40). Corticosteroids may be useful in reducing pain in the acute phase but do not reduce the total duration of pain (91,89). Symptomatic treatment with analgesic medication may include those with antineuralgic effect (gabapentinoids, SNRIs, and nonopioid analgesics), while oxycodone has shown greater efficacy than gabapentin in severe HZ related pain (92,40). In the case of trigeminal HZ, stellate ganglion blocks may reduce pain but no adequately powered controlled studies have been carried out to determine if they prevent PHN (93).
Postherpetic neuralgia
Definition
Unilateral facial pain persisting or recurring for at least three months after the appearance of herpes zoster rash in the innervation area of the trigeminal or intermedius nerves.
Symptomatology
Irrespective of the localization of PHN, it is customary to divide pain complaints of patients with PHN into three different categories: spontaneous continuous pain, paroxysmal pain, and evoked pain. The continuous pain is described as burning, smarting, throbbing or gnawing and localized within the affected dermatome, and only rarely radiating outside it. In trigeminal PHN, evoked pain is characteristically dynamic mechanical allodynia, shown by approximately one-half, while some have pinprick hyperalgesia and heat or cold allodynia (94,95). Itch is found in one out of six patients and may last longer than pain (75). Subjective numbness and paresthesias are common. Clinical examination shows sensory loss (increased detection thresholds) of all sensory modalities although sensory profiles are variable (94,96). It has been suggested that somatosensory phenotyping may reveal subcategories of PHN, including (i) irritable nociceptor (characterized by thermal hyperalgesia and limited sensory deficits), (ii) deafferentation (with major sensory deficits across all modalities), with some patients showing both deafferentation and allodynia (97). Long-term follow up studies suggest that in over 95% of those diagnosed with PHN the pain will eventually disappear (98,99). After spontaneous resolution, pain may rarely recur (99,100).
Epidemiology
The incidence and prevalence of trigeminal PHN vary depending on the definition used for the transition from trigeminal herpes zoster. A recent population based study suggests an incidence rate of 3.9 per 100,000 person years (2) increasing with age, from 2.0 among the 40–49 age group, to 11.9 among the 70–79 age group, and 44.2 among those over 80. No prevalence rates for trigeminal PHN have been published; those on any PHN show it to be rare at a population level (0.09% of the general population or 0.7 per 1000 in GP surgery attendees (101). Extrapolating from these figures the prevalence of trigeminal PHN (assuming they represent some 10–15% of the total PHN population) will yield very low levels, making this condition much less common than, for example, trigeminal neuralgia or cluster headache. Vaccination against herpes zoster, known to reduce the incidence of not only zoster but also PHN may influence these figures in the future, although there has also been an overall tendency in the last two decades toward increasing incidence of HZ in the general population (83).
Ophthalmic HZ carries twice the risk of development of PHN compared to ‘non-ophthalmic’ PHN (81). Other established risk factors include prodromal pain, severe acute pain, and severe rash, and old age (81).
Pathophysiology
There is no evidence that PHN is caused directly by viral replication in the trigeminal, geniculate or any other ganglion. By contrast, post-mortem studies have shown extensive virus-induced damage in the peripheral nerve, trigeminal ganglion, and even the brainstem trigeminal complex (85,102). Epidermal nerve fiber density (ENFD) measured from skin punch biopsies is significantly more reduced in patients with PHN compared to controls and those who do not develop PHN after the rash has healed (103). Interestingly, an ENFD decrease has been shown also on the contralateral side, corresponding to the severity of pain, although the mechanism is unclear (104). In a small study involving patients with trigeminal PHN it was shown that the epidermal unmyelinated afferents were primarily affected while autonomic fibers were mostly preserved (95).
Diagnostic considerations
With preceding herpes zoster infection, and clinical signs and symptoms, the diagnosis of PHN is usually straightforward. When acute HZ is not associated with rash, the diagnosis may be more challenging. However, detection of VZV DNA in cerebrospinal fluid may help to establish the etiology if the infection is suspected during the early stages of HZ. Paroxysmal pains are usually spontaneous rather than provoked by innocuous touch, differentiating this condition from trigeminal neuralgia (9). Combined vascular compression of the trigeminal nerve and PHN have been reported; such cases appear to represent genuine trigeminal neuralgia that responds to microvascular decompression (105).
Treatment
Once PHN is established, antiviral medication and corticosteroids are not effective. Management of pain should follow the guidelines drawn for neuropathic pain in general, with gabapentinoids, SNRIs, and tricyclics used initially, and opioids reserved for severe cases only (106). Capsaicin 8% patch, useful in managing cervical, thoracic, and lumbar PHN, is not licensed for trigeminal or intermedius nerve PHN. Botulinum toxin has been shown to reduce PHN related pain in two small controlled trials; however the follow up was only up to three months (107,108). Surprisingly, neurostimulation studies are lacking, apart from case reports, despite a number of technological developments that offer significant potential. Nerve blocks, removal of affected skin, or neuroablation are not to be recommended. Patients benefit from advice of the high likelihood of eventual resolution of pain, even if it takes years.
Expert opinion: Open questions and burning desires
Many problems with this group of disorders are related to their rarity. For example, many occipital neuralgia studies have been conducted in mixed patient groups including patients with migraine or cervicogenic headache. This may in part be due to the difficulty discriminating between these conditions diagnostically. In future, adjuncts such as diagnostic ultrasound and ultrasound with EMG guided local anesthetic nerve block may help to distinguish between muscle origin or nerve origin pain or the site of nerve compromise.
Given that nerve entrapment within the lateral pterygoid muscle is a potential etiology of auriculotemporal neuralgia, surprisingly few papers have described the extent of examination of the masticatory system. This description may help understand the contribution to this etiology in the diagnosis but also to confirm that temporomandibular joint pathology has been excluded satisfactorily. The use of EMG guided local anesthetic blocks could be explored as a potentially useful adjunct in diagnosis.
PHN remains an unfortunate long-term complication of HZ; only vaccination has been shown to reduce its incidence in prospective studies. Much remains to be discovered of the early pathophysiological changes that engender pain and lead to the development of PHN. Prevention of PHN in those at high risk of developing it using pharmacological agents known to reduce peripheral and/or central sensitization remains an important target even if it requires large clinical trials.
All types of PHN remain difficult to treat. Ultimately one of the problems is the heterogeneity of the pathophysiological mechanisms involved, which warrant further research. Somatosensory phenotyping is emerging as a method of identifying patients who could respond to specific medication (e.g., those with an ‘irritable nociceptor’ type sensory profile to sodium channel blockers). It is encouraging to see intensive research in this field. Neuromodulation treatments are undervalued; mini-invasive stimulation methods that have been successful in occipital neuralgia or cluster headache could be adopted for trigeminal PHN.
Expert opinion: Where the field would need to go
Clearly there is a need for well-designed prospective clinical trials, but given the relative rarity of most of these neuralgias, achieving adequate samples sizes would undoubtedly require more multicenter collaborative studies. In contrast to other types of neuropathic pain, destructive procedures are still quite frequently used in these neuralgias, and critical follow-up of these patients is warranted.
Many cases of superior laryngeal neuralgia described follow surgery and it is unclear what proportion is as a direct result of damage to the nerve. Given this potential etiology, long-term follow up of outcomes from further destructive surgical interventions used as treatment would give useful insight into whether these procedures are warranted.
Phenotyping using sensory testing and taking advantage of the unique somatic and autonomic innervation of the head could add significantly to our understanding of what maintains the pain or makes it chronic. This might be complemented with novel neurophysiological and neuroimaging techniques. Genetic profiling offers another avenue to pursue in the search for factors leading to postherpetic neuralgia. The role of VZV in causing idiopathic trigeminal and other cranial neuralgias should be reassessed.
Accumulation of epidemiological data on PHN will be important to assess whether there is a significant reduction of PHN in the post-vaccination era. Further studies are needed to improve our understanding of the pathophysiological mechanisms which in the first instance could involve combined histopathological, neurophysiological, and neuroimaging methodology. For established PHN, mini-invasive neuromodulation methods should be trialed.
Clinical implications
The diagnosis of each cranial neuralgia is clinical and critically dependent on the location of the reported pain. Because of overlapping innervation territories, nerve anastomoses and occasional involvement of vestigial nerves, this is not always straightforward. Most of these neuralgias are rare and many physicians do not see such cases in their lifetime, so patients with a suspected diagnosis within this group should be referred to a specialized center where multidisciplinary team diagnosis may be available. The pharmacological treatment of postherpetic neuralgia and acute herpes zoster should follow published guidelines, since data from randomized clinical trials are available for these conditions. In spite of the scarce evidence, the mainstay of treatment remains conservative (with oral or topical medication recommended for neuropathic pain) to be tried before more invasive procedures are undertaken.
Footnotes
Acknowledgments
CS receives grant support for studies on neuropathic pain from the European Union’s Seventh Framework Programme (FP7/2007-2013) under grant agreement 602133.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FO’N has no conflicts of interest to declare. TN has received honoraria for speaking and serving on the advisory board of Astellas, research support from Pfizer and Pain Relief Foundation, UK. CS has given educational talks for pharmaceutical companies that produce drugs to treat neuropathic pain: Astellas, Pfizer.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
