Abstract
Background
The association between migraine and cognitive performance is unclear. We analyzed whether migraine is associated with cognitive performance among participants of the Brazilian Longitudinal Study of Adult Health, ELSA-Brasil.
Methods
Cross-sectional analysis, including participants with complete information about migraine and aura at baseline. Headache status (no headaches, non-migraine headaches, migraine without aura and migraine with aura), based on the International Headache Society classification, was used as the dependent variable in the multilinear regression models, using the category “no headache” as reference. Cognitive performance was measured with the Consortium to Establish a Registry for Alzheimer’s Disease word list memory test (CERAD-WLMT), the semantic fluency test (SFT), and the Trail Making Test version B (TMTB). Z-scores for each cognitive test and a composite global score were created and analyzed as dependent variables. Multivariate models were adjusted for age, gender, education, race, coronary heart disease, heart failure, hypertension, diabetes, dyslipidemia, body mass index, smoking, alcohol use, physical activity, depression, and anxiety. In women, the models were further adjusted for hormone replacement therapy.
Results
We analyzed 4208 participants. Of these, 19% presented migraine without aura and 10.3% presented migraine with aura. All migraine headaches were associated with poor cognitive performance (linear coefficient β; 95% CI) at TMTB −0.083 (−0.160; −0.008) and poorer global z-score −0.077 (−0.152; −0.002). Also, migraine without aura was associated with poor cognitive performance at TMTB −0.084 (−0.160, −0.008 and global z-score −0.077 (−0.152; −0.002).
Conclusion
In participants of the ELSA-study, all migraine headaches and migraine without aura were significantly and independently associated with poorer cognitive performance.
Introduction
Migraine is the third most prevalent disorder and the seventh-highest specific cause of disability worldwide (1,2). In the Global Burden of Disease, it has been increasingly prevalent in young and middle-aged adults, accounting for almost 33 million disability-adjusted life years (DALYs) in 2015 (3). In Latin America, a multicenter study conducted in Argentina, Brazil, Colombia, Mexico, and Venezuela found a range of 6.1–17.4% of migraine prevalence in the female population, and 2.9–7.8% in the male population (4). Similarly, in the USA, the prevalence of migraine was found to be 17.1% among women and 5.6% among men, in a large representative study that used a self-reporting questionnaire based on the International Headache Society (IHS) criteria (5).
On the other hand, dementia has a long pre-clinical phase, with evidence of neuropathological lesions starting 20 years before the onset of clinical symptoms (6). Studies reporting the association of migraine with cognitive performance show conflicting results. Some studies found no impact of migraine on several cognitive tests. In a report from the population-based Maastricht Aging Study (7), the cognition performance of 99 migraineurs was compared to that of 1768 non-migraineurs (mean age 52 years) and migraine was not associated with cognitive performance. In the Epidemiology of Vascular Ageing-MRI (8) study, 116 migraineurs were compared to 617 non-migraineurs (mean age 68.8 years) and there was no evidence that migraine alone, or in combination with brain lesions, was associated with cognitive impairment. Additionally, in a single-blind, retrospective analysis of cognitive function data comparing 148 case-matched migraineurs and non-migraineurs (mean age 64.4 years), migraine patients both with and without aura did not differ significantly from age- and sex-matched controls, in terms of performance in four cognitive tests (9).
Other studies found that migraine could be associated with poorer cognitive scores. In a cross-sectional analysis of 478 adults in primary care (mean age 50 years) (10), the presence of headaches in late adulthood was related to poorer performance in executive functioning. A controlled retrospective study found that migraineurs with severe headaches showed consistently poorer performance in a series of memory and information-processing tests (11). Interestingly, one recent report from the Rotterdam Study comparing 1021 migraineurs to 5399 non-migraineurs showed better global cognitive performance and executive function in migraineurs compared to non-migraineurs, independently of several well-known vascular risk factors (12).
Despite the facts that migraine and dementia are both associated with higher vascular risk, and the two conditions have been linked with structural brain lesions, evidence of the association between migraine and cognitive performance is controversial. We therefore analyzed the cross-sectional association between migraine and cognitive performance in middle-aged participants from the ELSA-Brasil study (13).
Methods
Study population
ELSA-Brasil is a multicenter prospective cohort, which enrolled 15,105 civil servants aged 35 to 74 years (54% women) from six Brazilian cities (Belo Horizonte, Porto Alegre, Rio de Janeiro, Salvador, São Paulo, and Vitória) (14). The aims of the study were to investigate factors associated with the development and progression of cardiovascular disease (CVD), diabetes, and other chronic conditions (15). The baseline assessment consisted of an evaluation lasting approximately seven hours, which included in-person interviews conducted by trained personnel (16). These standardized interviews focused on sociodemographics, health, medical history, use of medication, and clinical and laboratory measurements (17). Approvals were obtained from the institutional review boards of all the centers, and the participants signed an informed consent form.
Of the 15,105 participants of the ELSA-Brasil, 5061 participants from the São Paulo center were evaluated for migraine status with aura and are therefore included in this analysis. In this study, we excluded subjects with a prior history of stroke, those who were taking medications that could interfere with performance in the cognitive tests (carbidopa, levodopa, pramipexole, selegiline, entacapone, amantadine, biperiden, phenobarbital, phenytoin, lamotrigine, primidone, risperidone, chlorpromazine, lithium, clozapine, fluphenazine, trifluoperazine, quetiapine, memantine, levomepromazine, olanzapine, penfluridol, periciazine, pimozide, agomelatine and benzodiazepines, valproate acid, carbamazepine, gabapentin, oxycarbamazepine, and topiramate), and those with incomplete data in the cognitive tests, migraine status, or other covariates (Figure 1). However, even after considering these exclusions, no one was removed from the sample because nobody was using these medications at baseline of study.
Participants included in the study.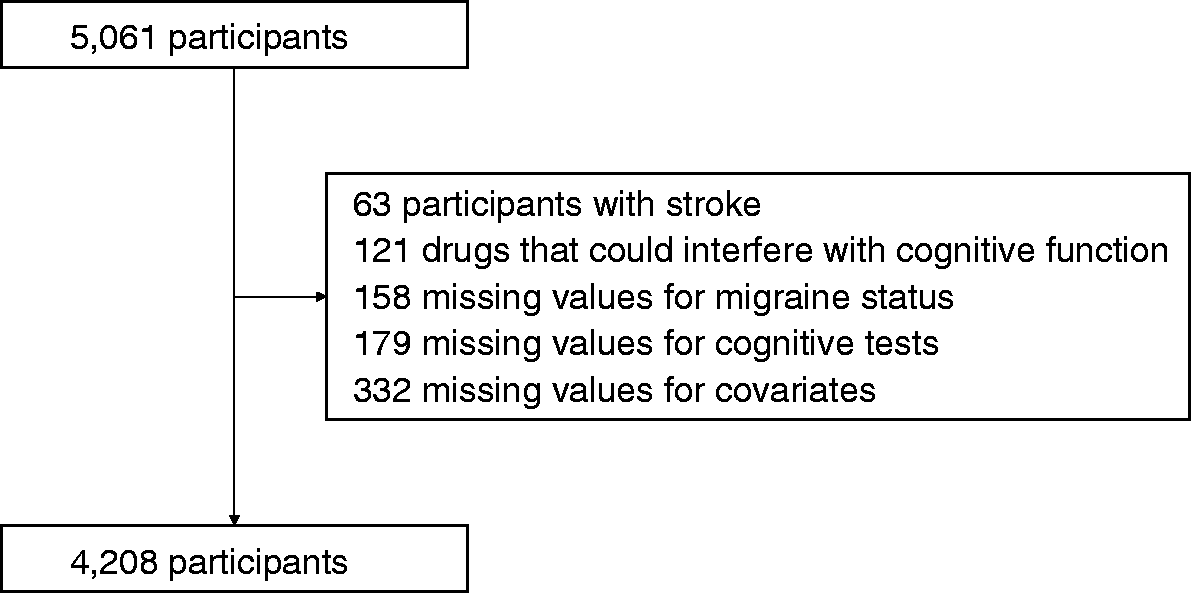
Migraine status assessment
All participants who answered “yes” to the question “In the last 12 months, did you have a headache?” at the ELSA-Brasil baseline evaluation were invited to fill out a detailed headache questionnaire based on the International Headache Society criteria (IHS-2004) (18) that was adapted, validated and previously used in Brazil (19). Briefly, the questionnaire investigates pain frequency, duration, quality, location, intensity, triggering and accompanying symptoms, such as nausea or vomiting and the presence of aura. We classified individuals as having definite migraine if they answered “yes” to the question about headache and fulfilled all the criteria for migraine. Based on this questionnaire, we classified participants as having definite migraine if they fulfilled the criteria for IHS codes 1.1 (migraine without aura) or 1.2 (migraine with aura). We classified individuals as having probable migraine if they answered “yes” to the question about headache and fulfilled all but one criteria for migraine. For the purposes of this analysis, definite and probable migraine are considered together. Participants with other headaches, or without headache, were considered as having no migraine.
All the participants with a diagnosis of migraine completed a telephone interview performed by a medical doctor with great expertise in headache classification, including aura symptoms, based on the IHS criteria (20). According to the IHS criteria, typical aura consists of visual and/or sensory and/or speech complaints that occur gradually over 5 to 20 minutes and last no longer than 1 hour. Finally, we collected information about the use of prophylactic medications for migraine based on evidence level A (established as effective for migraine prevention and that should be offered to patients with migraine to reduce the frequency and severity of the migraine attack) or level B (possibly effective for migraine prevention and that should be considered for migraine prevention), according to the 2012 guidelines of the American Academy of Neurology (21).
Participants were oriented to cancel the visit to ELSA-Brasil research center if they presented symptoms such as any kind of pain, fever, fatigue, dizziness, and/or nausea in the 24 h before the visit to the center. Participants were also oriented to not practice physical activities in the 24 h before the visit. Morehover, a trained nurse screened participants at the reception deks about their symptoms in the last 24 h and, if necessary, the visit was rescheduled. Cognitive tests were applied at the beginning of the questionnaire, when participants were more relaxed and less tired.
Cognitive function evaluation
Three cognitive tests were used in this study: (a) the Consortium to Establish a Registry for Alzheimer’s Disease word list memory test (CERAD-WLMT) (22), (b) the semantic fluency test (SFT) (23), and (c) the Trail Making Test version B (TMTB) (24). The protocols were standardized, and trained examiners administered the tests in a fixed order in a single session, in a quiet room, with good lighting and low levels of noise or other distracting stimulations (25).
We used the Brazilian version of the CERAD-WLMT (26), which includes a list of 10 unrelated words that were presented at a rate of one word every two seconds, over three learning trials. After each trial, the participants were asked to recall as many words as they could (immediate recall). After a 5-minute filled delay, they had 60 seconds to record the words that had been presented previously (delayed recall). The scores in the immediate and delayed recalls were equal to the number of recalled words. Retention was also tested in the recognition test, when 10 previous words were presented together with 10 distractor words. The score in the retention test was equal to the number of correctly-recognized words minus the number of words wrongly recognized.
The SFT is a test of language and executive function. The participants were asked to generate as many categories of animals as possible for one minute. The score was equal to the total number of generated words. The TMTB is a test of executive function, speed of processing, and attention. The participants were instructed to draw lines connecting letters and numbers in an order that alternated between increasing numerical value and alphabetic order. The test score was the total time taken, in seconds, to complete the task. Therefore, in the TMTB, higher total times were classified as poorer performance.
Clinical and laboratory variables
After fasting overnight, a blood sample was collected from each participant. Blood glucose was determined enzymatically by the hexokinase method. Total cholesterol, high-density cholesterol (HDL) and triglycerides were determined by the enzymatic colorimetric method. LDL-cholesterol was calculated using the Friedewald equation (27).
Demographic and clinical variables included age, sex, education (below high school, high school, college or above), self-reported race (White, Mixed race, Black, Asian, or Native), and smoking status (never, former, and current). Alcohol use was defined as never, former or present. Height and weight were measured with light clothes and without shoes, and body-mass index was calculated by dividing body weight by the squared height in meters (kg/m2).
Blood pressure was obtained in the sitting position after a minimum rest period of 5 minutes. Three consecutive readings were obtained for each participant, with a one-minute interval between each. The mean of the two last measurements was defined as the office blood pressure. Diabetes was defined as the presence of one of the following: Medical history of diabetes, use of medication to treat diabetes, fasting plasma glucose ≥126 mg/dL, 2-h glucose post load test ≥200 mg/dL, or HbA1C ≥ 6.5%. Coronary heart disease was defined as prior history of myocardial infarction, angina, or cardiac revascularization. Dyslipidemia was defined as LDL ≥ 130 mg/dL or use of lipid-lowering medication.
Mental diagnoses were assessed by trained interviewers, using the validated, Portuguese version of the Clinical Interview Schedule – Revised (CIS-R) (28). The CIS-R is a structured questionnaire for the measurement and diagnosis of non-psychotic psychiatric morbidity in the community. It was developed by Lewis et al. (29) specifically for use in community and primary care, and is a short, straightforward questionnaire. Importantly, lay interviewers are as reliable as psychiatrists in using CIS-R to perform mental diagnosis, making it a suitable instrument for use in our cohort. The complete CIS-R version was applied, allowing us to perform the diagnoses of anxiety disorders that correspond to the 10th version of the International Classification of Diseases (ICD-10) (30) categories: General anxiety disorder (GAD, F41.1); panic disorder (F41.0); social anxiety disorder (F40.1); phobias (F40.2); obsessive-compulsive disorder (F42); and depressive disorder (F32.xx).
Statistical analysis
The categorical variables were reported as absolute and relative frequencies, and the continuous variables as means and standard deviations (SD). Proportions and mean values of migraineurs were compared across headache (no headaches, non-migraine headaches, migraine without aura and migraine with aura) using the Chi-Square (X2) and ANOVA respectively. Z-scores for each cognitive score were created by subtracting the participant’s score in each test from the sample mean score, and dividing the difference by the sample SD (31). Thus, a z-score of −1 describes a cognitive performance that is 1 SD below the mean sample score. Finally, we averaged and standardized the Z-scores of all tests (after inverting the sign of the TMTB, since a higher number of seconds represents lower performance) as a measure of composite global cognitive score. Dependent variables were the z-scores for cognitive tests, and independent variables were headache status categories. Multilinear models were built considering the “no headaches” category as the reference. We first ran a crude analysis (Model 1) and then adjusted for age, gender, education and race (Model 2). Further adjustments were made for coronary heart disease, heart failure, hypertension, diabetes, dyslipidemia, body-mass index, smoking, alcohol use, physical activity, depression and anxiety. As hormone replacement therapy (HRT) has been linked with neuroprotective effects in some observational studies (Model 3) (32), we further explored confounders by introducing HRT to the model in women. We included a final model (Model 4) considering all migraine (migraine with and without aura together) including adjustments in Model 3 plus presence of aura (yes or no) in the final model.
The statistical analyses were performed using SPSS version 22 (Armonk, NY: IBM Corp). All reported
Results
Characteristics of studied population according to headache status (n = 4208).

Most of our population was comprised of women (53.9%). Frequency of migraine without aura and with aura was higher in women, and the mean age decreased linearly across migraine status categories. BMI was slightly higher in the migraine with aura group. Current smoking, depression and anxiety were more common in the migraine with aura group and the proportion of those physically active was lower in the migraine without aura and migraine with aura groups.
Association between headache status and performance on cognitive tests using z-scores (n = 4,208).

Hierarchical linear regression models: aModel 1: crude analysis; bModel 2: adjusted for age, gender, education and race; cModel 3: adjusted for age, gender, education, race, previous history of coronary heart disease, heart failure, hypertension, diabetes, dyslipidemia, body mass index, smoking, alcohol use, physical activity, depression and anxiety (in women, further adjustment for TRH). dModel 4: All migraine headaches (migraine with aura and migraine without aura considered together) adjusted for all variables in Model 3 further adjustment for aura; CI: confidence interval.
Use of medication in the sample during migraine episodes included analgesics (51.3%), NSAID (6.5%), ergot (30.8%), triptans (2.7%) and opioids (0.6%). Approximately 60% used more than one medication. The number of migraineurs using prophylaxis is low.
Discussion
In our study of a large and well-defined non-clinic based population, migraine without aura and all migraine headaches were associated with poorer cognitive performance, measured by executive function (TMTB). This association was also significant for the global cognitive score, and remained significant after multivariate adjustment for important confounders.
Several studies have tested the association between migraine and cognitive performance, with heterogeneous results. In a cross-sectional analysis of 478 individuals (aged over 50 years) (10) recruited in a primary care center, 13% presented with migraine, and headaches in late adulthood were related to poorer performance in several measures of executive functioning. In a retrospective single blinded study comparing 148 case-matched migraineurs and non-migraineurs (mean age 64.4 years), migraine patients both with and without aura did not differ significantly from age- and sex-matched controls in terms of performance in four cognitive tests (9). In another case-control study (31) of 60 migraine patients from a single clinic center and 30 healthy control subjects (15 to 68 years old), poorer performance in two tests, measuring verbal and visual memory, respectively, was found in migraine patients experiencing higher frequency of attacks and in those with a long history of migraine. Interestingly, the Rotterdam Study (12) found that migraineurs tended to score higher in cognition tests than non-migraineurs. In that population-based study of 1021 migraineurs (mean age 63.8 years) compared to 5399 non-migraineurs (mean age 65.9 years), cognition was measured by the MMSE and a global cognition score composed of several tests involving immediate and recall memory, processing speed, and attention.
Some well-designed prospective studies focused on cognitive decline rather than cognitive performance and found that migraine was not associated with cognitive decline. In the Maastricht Study (7), 99 migraineurs were compared to 1768 non-migraineurs (mean age 52 years), and it was found that migraine did not influence information processing speed and memory. They also tested the interaction between age and migraine and found no significant results. Aligned with those findings in the EVA study (32), with a population of 1170 participants (mean age 69 years), migraine was tested with a wide variety of cognitive evaluations; no differences were found at baseline, and people with migraine or non-migraine headache did not have a greater rate of cognitive decline than those without headache. In that study, the authors adjusted their analysis for socio-demographics, biological parameters, comorbidities, and the APOE4 allele.
A possible explanation for this discrepancy is that cognitive test protocols varied among those studies. The evaluation of cognitive performance tests using the Mini Mental State Examination (MMSE) may not be sensitive to small cognitive changes (33). Other possible explanations are the different forms of migraine assessment (e.g. self-report, physician diagnosed, questionnaires, not based on the IHS criteria), the source populations (clinically-based, population-based), sample size, and study design. Selection bias and mis-classification in studies looking at the potential link between migraine and cognition have been thoroughly described in a topical review (32). We could add to those limitations the fact that controlling for confounders was poor in some studies.
A possible explanation for the association of migraine headaches with a worse performance in TMTB, as we observed in the present analysis, is that poorer performance in executive function is the earliest symptom of vascular causes of dementia. Migraine headaches are also associated with vascular alterations in endothelial function, which may help to explain our results. Despite previous conflicting results demonstrating positive (34,35,36) and negative (37) association of migraine headaches and endothelial function, we may hypothesize a common vascular mechanism linking migraine headaches and poorer performance at TMTB. White matter hyperintensities have been described not only as independent predictors of dementia and mild cognitive impairment (38) in pre-clinical phases (39), but also with migraine headaches (40). A prospective study also confirmed that migraine was independently associated with deep white matter hyperintensities progression in women (41). A prospective study has also confirmed that migraine was independently associated with deep white matter hyperintensities progression in women (41), who present a higher prevalence of migraine headaches compared to men. It is possible that migraine may affect cognitive decline through these vascular lesions.
Information on aura has also been studied to help clarify the association between migraine and vascular risk. In a recent review (42), possible mechanisms have been described. Along with some known hypotheses for vascular events in migraineurs, such as the pharmacologic agents used to treat migraine (e.g. NSAIDS, triptans and ergotamine), anxiety, depression, prothrombotic factors, decreased platelet hemostasis time, clotting and collagenous-induced thrombus formation, patent foramen ovale and some polymorphysms, the authors describe the trigeminovascular pain pathway with cortical spreading depression as being responsible for aura induced by vasoconstriction, and hypoperfusion and a cascade of inflammatory events as potential explanations for the high risk of stroke in migraineurs, especially in those with aura. However, to clarify the risk of vascular events outside the brain among migraineurs, another review from the same group (43) has described conflicting results concerning altered endothelial function in migraineurs, with low evidence of risk of atherothrombosis and a more consistent group of studies showing altered arterial function in migraineurs measured by pulse wave velocity, augmentation index and distensibility.
Although migraine with aura has been associated with higher vascular risk, which in turn could be associated with poorer cognitive performance, we cannot confirm at this point that aura is associated with poorer cognitive performance, since our results showed a significant association for Trail Making Test B and global score only for the migraine without aura group. One possible explanation for that is that our sample is relatively young, and the exposure to migraine with aura is still not sufficient for cognitive implications. However future prospective studies in our populations could help clarify that. Another potential explanation is that we lack power in the migraine with aura group.
At this point, some limitations of our study should be considered. The cross-sectional nature of our analysis enables us to study association but not causality. As the main objective of ELSA-Brasil is the study of cardiovascular diseases and diabetes, we did not include individuals aged under 35 years, which is adequate for cognitive performance but may limit the study of migraine in our sample. We do not have any information on medication overuse or migraine associated with medication overuse. Moreover, it is possible that we do not have power to evaluate the association of migraine with aura and cognitive performance. Although our results are speculative, we were able to examine a large and well-defined sample of adults with information about migraine and aura according to the IHS criteria, and also a validated battery of cognitive tests, after adjusting for several important confounders.
Our findings suggest an association between migraine and poor cognitive performance compared to non-migraineurs. Future prospective analysis of ELSA-Brasil should help clarify whether this association is causal or not. In conclusion, migraine was associated with poorer cognitive performance in a cross-sectional analysis of the ELSA-Brasil.
Clinical implications
Migraine without aura and all migraine headaches were associated with poorer cognitive performance compared to no headaches. Executive function was poorer in migraineurs compared to no headaches. The association remained significant after controlling for important confounders. No association was found when migraine with aura was compared to no headaches.
Footnotes
Acknowledgments
The authors would also like to acknowledge the 15,105 individuals recruited for this study for their participation, without which this study, and those based on the ELSA-Brasil cohort, would not have been possible.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The ELSA-Brasil baseline study was supported by the Brazilian Ministry of Health (Science and Technology Department) and the Brazilian Ministry of Science and Technology and CNPq National Research Council (grants # 01 06 0010.00 RS, 01 06 0212.00 BA, 01 06 0300.00 ES, 0106 0278.00 MG, 01 06 0115.00 SP, 01 06 0071.00 RJ).
