Abstract
Background
The World Health Organization Essential Medicines List (WHO EML) contains two analgesics for treatment of acute migraine attacks in children, ibuprofen and paracetamol.
Methods
The Embase, CDSR, CENTRAL, DARE and MEDLINE databases were searched up to 18 April 2017. We analyzed randomized controlled trials (RCTs) and systematic reviews (SRs) that investigate the efficacy and safety of ibuprofen or paracetamol for treatment of acute migraine attacks in children. We conducted meta-analysis and assessments of evidence with GRADE, Cochrane risk of bias tool, and AMSTAR.
Results
Three RCTs (201 children) and 10 SRs on ibuprofen and/or paracetamol for acute migraine attacks in children were included. Meta-analysis indicated that ibuprofen was superior to placebo for pain-free at 2 h or pain relief at 2 h, without difference in adverse events. There were no differences between paracetamol and placebo, or ibuprofen and paracetamol. Ten SRs that analyzed various therapies for migraine in children were published between 2004 and 2016, with discordant conclusions.
Conclusion
Limited data from poor quality RCTs indicate that ibuprofen and paracetamol might be effective analgesics for treating migraine attacks in children. Inclusion of ibuprofen and paracetamol as antimigraine medicines for children in the WHO EML is supported by indirect evidence from studies in adults.
Keywords
Introduction
Over the last two decades, the prevalence of migraine in children has been estimated to reach 10%, increasing with age up to puberty (1,2). According to the International Headache Society (IHS), migraine is a common disabling primary headache disorder (3). In young children, diagnosis of migraine may be difficult as the migraine is defined by subjective symptoms, is highly variable, and it is often susceptible to rapid improvement (4).
It has been indicated recently that there are few reliable trials about pharmacological interventions to relieve the pain of migraine in children, and that using paracetamol or NSAIDs as first-line agents is “widely accepted good clinical practice during acute attacks unless contraindicated” (5).
The World Health Organization Model List of Essential Medicines (WHO EML) contains medicines that are defined by the WHO as those that satisfy priority health care needs of the population (6). The WHO indicates that medicines for inclusion into the WHO EML are identified through an evidence-based process in which quality, safety, efficacy, and cost-effectiveness are the key selection criteria (7). The idea behind the WHO EML is that the use of a limited number of well-known and cost-effective medications may improve long-term medicine supply, lower the costs, and enable better health care provision (8).
The master WHO EML contains a limited number of analgesics for treatment of acute attacks of migraine: Acetylsalicylic acid, ibuprofen, and paracetamol (also known as acetaminophen) (7). The master list has a sub-list, which specifies medicines for children. Only ibuprofen and paracetamol are included as analgesics for acute migraine attacks in the children list. They are important therapeutic options in acute attacks, based on accepted good clinical practice and indirect evidence derived from studies in adults (9,10). Nevertheless, it is important to assess whether direct evidence is available, as this would increase the confidence in these medicines.
The aim of this study was to systematically analyze randomized controlled trials (RCTs) and systematic reviews (SRs) about analgesics listed on the WHO EML for the treatment of acute migraine attack in children, assessing the extent of experimental evidence that supports their inclusion in the WHO EML for this indication. We also evaluated how this evidence was synthesized in SRs, and if there are discordances on the therapeutic value of these medicines.
Methods
Study design
We conducted a SR of the literature, followed by an overview of SRs. We reported the study according to the PRISMA guidelines (11).
Antimigraine analgesics on the WHO EML for children
Firstly, we determined which analgesics are listed as antimigraine medications on the WHO EML for children and accompanying corrigenda. All drugs classified in the N02A, N02B, M01A and M02A groups of the Anatomical Therapeutic Chemical (ATC) classification system of active substances (13) were searched for their presence in the WHO EML for children under indication 7. Antimigraine medicines. The only analgesics found with this indication were ibuprofen and paracetamol, and they were found under the subheading “7.1 For treatment of acute attack” (5). Subsequently,
Information sources and literature search
Search strategy for MEDLINE.

Screening and data extraction
Bibliographic records retrieved by database search, consisting of titles and abstracts, were screened by two authors each (AJK, MJ, NS) independently to assess their eligibility. Disagreements were resolved by discussion and involvement of the third author (LP). Full texts of all potentially-includible studies were retrieved and analyzed by two authors independently (MJ, NS), whereas disagreements were resolved by discussion and involvement of the third author (LP). References and citations of included studies were downloaded from Web of Science and screened by two authors independently (MJ, LP) to identify additional citations that may have been missed through electronic database searching methods.
Inclusion criteria
We included randomized controlled trials (RCTs) and SRs analyzing ibuprofen and/or paracetamol as a pharmacological intervention for the treatment of acute attacks of migraine in children < 18 years. A SR was defined as a review with a clearly formulated question that used systematic and explicit methods to identify, select and critically appraise relevant research and to collect and analyze data from the studies that were included in the review (14). As such, the review had to describe a detailed search of the literature for relevant studies and provide a synthesis of the results. For reviews with updates (e.g. Cochrane reviews), only the latest version was included.
Exclusion criteria
Reviews by a single author were excluded even if they had certain other elements of a systematic review methodology (i.e. searching specific databases) because abstract screening, data extraction and quality assessment need to be performed in duplicate to reduce potential errors.
Data items and data extraction process
Data were extracted into the
The following data were extracted: First author, year of publication, type of study, language of publication, number of participants, population included in the study, type of study, study setting and country. Additionally, the following outcome data were extracted from RCTs: Pain-free at 2 h, pain relief at 2 h, adverse events, and the following data from SRs: Studied topic, outcomes, and conclusions from abstracts, or from the text if there was no conclusion in the abstract.
Risk of bias assessment in included trials
We assessed RCT quality using the Cochrane risk of bias tool, which includes seven dimensions: Sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessors, incomplete outcome data, selective outcome reporting, and other potential threats to study validity. Quality of SRs was analyzed using a measurement tool to assess the methodological quality of systematic reviews (AMSTAR) (15).
Data synthesis
Meta-analysis for the three comparisons (ibuprofen versus placebo, paracetamol versus placebo, and paracetamol versus ibuprofen) was conducted using RevMan software version 5.3 (Cochrane Review Manager Version 5.3; Nordic Cochrane Centre, Cochrane Collaboration, Copenhagen, Denmark). Random-effects meta-analysis was conducted and results shown as odds ratio (OR) and 95% confidence interval (95% CI). Heterogeneity was analyzed and reported. For meta-analyses, trials adopting a cross-over design were combined with those using parallel groups. The data in the cross-over trials were presented per treatment group, as if the trials had parallel group design. This approach ignores cross-over design, giving the same point estimate as if cross-over was taken into account, but resulting in larger confidence intervals (16). This also has influence on the overall meta-analysis estimates, again producing slightly more conservative estimates.
Conclusiveness of evidence
Conclusiveness of evidence from SRs was analyzed by two authors independently (AJK, LP), whereas evidence for each medicine was rated as either positive, negative, or inconclusive. Evidence was rated as positive if the conclusion was in favor of an intervention, negative if an intervention was inferior compared to the comparator, and inconclusive if the authors indicated that there was not enough evidence to enable conclusions.
Quality of evidence assessment for analyzed outcomes
Strength of evidence for the key outcomes was assessed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology (17). The GRADE system uses the following assessment criteria for levels of evidence: High, moderate, low, or very low. High level indicates that we are very confident that the true effect lies close to that of the estimate of the effect. Moderate level is defined as being moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. With low quality level, our confidence in the effect level is limited; the true effect may be substantially different from the estimate of effect. Very low quality level indicates that we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect (18). The grade for an outcome was decreased if we found serious (−1) or very serious (−2) limitations to study design; important inconsistency(ies) (−1); some (−1) or major (−2) uncertainty about directness; imprecise or sparse data (−1) and high probability of reporting bias (−1).
GRADE assessment was conducted for the following outcomes: pain-free at 2 h, pain relief at 2 h and adverse events. We selected these outcomes because they are primary outcomes frequently used in migraine trials (19). The assessment is presented in the Summary of findings (SoF) tables for relevant comparisons, including paracetamol versus placebo, ibuprofen versus placebo and paracetamol versus ibuprofen. The SoF tables include key information about the quality of evidence, the magnitude of effect of the interventions examined, as well as the sum of available data on the main outcomes. SoFs were created using the online GRADE software (GRADEpro 2015, GRADE Working Group, McMaster University, Hamilton, ON, Canada).
Results
The database search yielded 2692 bibliographic records. After excluding duplicates, screening and excluding irrelevant studies, 84 studies were analysed in full text; 71 studies were excluded and 13 were included (Figure 1). The list and characteristics of excluded studies are available on request. Among included studies there were three RCTs and 10 SRs. Characteristics of RCTs and their risk of bias are summarized in Tables 2 and 3, respectively; SRs are summarized in Table 4.
Study flow diagram. Characteristics of included randomized controlled trials. Risk of bias in the included RCTs. Characteristics of included systematic reviews. Conclusiveness of evidence about paracetamol and ibuprofen in included systematic reviews (conclusions from the text if not found in the abstract).




Randomized controlled trials
The three RCTs on ibuprofen and paracetamol were small – a total of 201 children completed the three studies. The age range of the children was somewhat different between the studies: 4−16 years in one study (20), 6−12 in the second (21) and 6−18 in the third (22). The only studied dose of paracetamol was 15 mg/kg. Additionally, the three studies used different doses of ibuprofen, whereas the first two indicated doses per kilogram – 10 mg/kg (20) or 7.5 mg/kg (21), and the third indicated that children under 12 years received 200 mg while older children received 400 mg (22) (Table 2).
Assessment of RCT quality using the Cochrane risk of bias tool indicated that all three trials had unclear or high risk of bias on most of the analyzed domains (Table 3). Two RCTs had high attrition, with 17% attrition in one (20) and 39% in the other (21), without description of attrition per group. This might have consequences, unbalancing the groups for important prognostic factors.
Meta-analyses of randomized controlled trials
Analyses were conducted for three outcomes: Pain-free at 2 h, pain relief at 2 h and adverse events. Different definitions of pain relief were used in each RCT. In Hamalainen et al., pain relief was defined as “Reduction in severe or moderate headache (grades ≥ 3 on a scale of 1 to 6) by two grades” (20). In Lewis et al., pain relief was “those subjects whose headache severity decreases from moderate or severe at baseline to none or mild” (21). In Evers et al., pain relief was defined as “no or mild headache after moderate or severe headache” (22).
Ibuprofen versus placebo in children and adolescents
Outcome 1.1: Ibuprofen was superior to placebo for pain-free at 2 h in the pooled analysis of three studies (Figure 2(a)). The OR was 3.96 (95% CI 1.78 to 8.82; Comparisons of ibuprofen versus placebo for migraine in children for outcomes pain-free at 2 h, pain relief at 2 h and adverse events.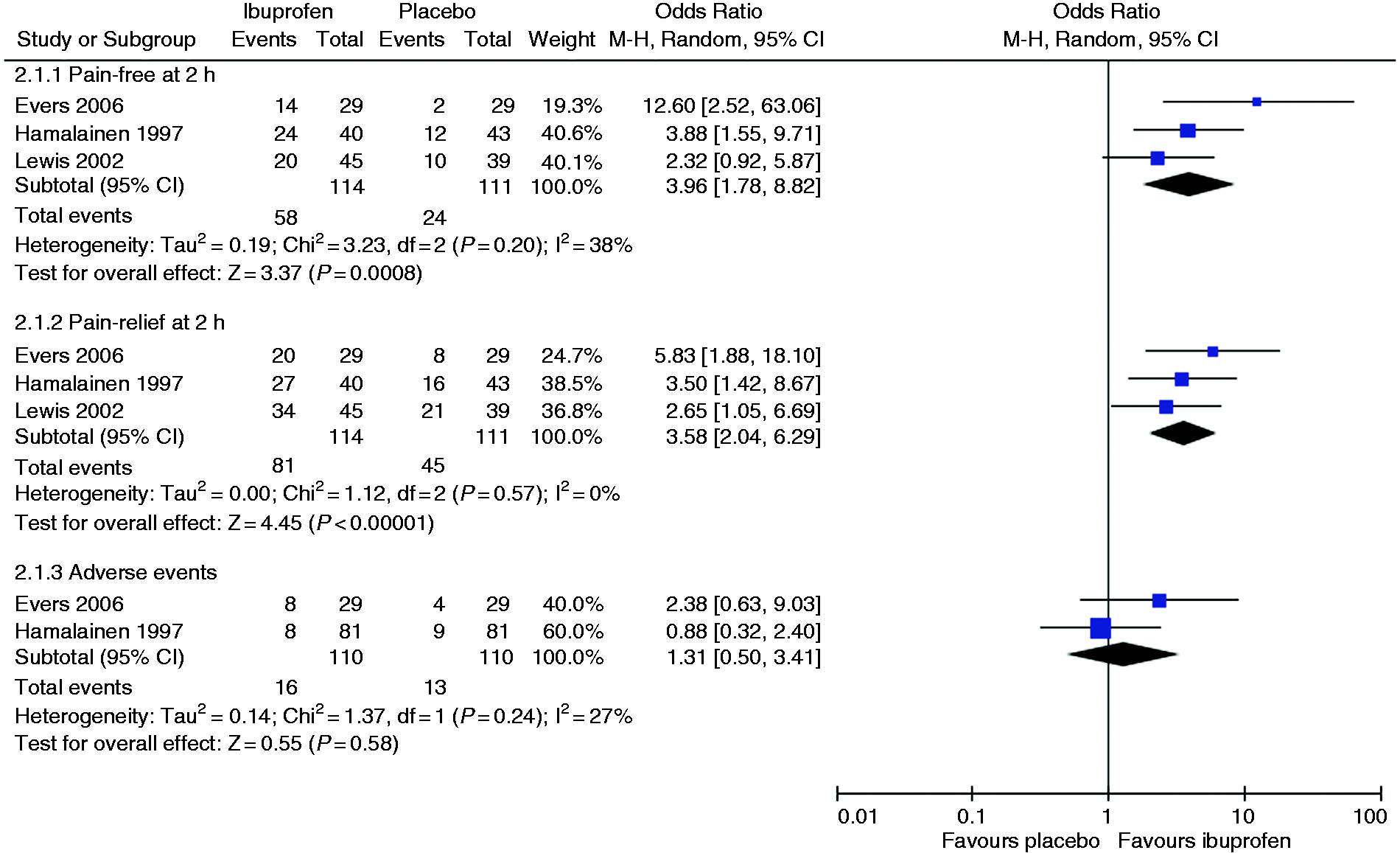
Outcome 1.2. Ibuprofen was superior to placebo for pain relief at 2 h in the pooled analysis of three studies (Figure 2(b)). The OR was 3.58 (95% CI 2.04 to 6.29;
Outcome 1.3. There was no difference between ibuprofen and placebo in the number of patients with adverse events in the pooled analysis of two studies (Figure 2(c)). The OR was 1.31 (95% CI 0.50–3.41;
Paracetamol versus placebo in children and adolescents
Only one study provided data for the comparison (20). When paracetamol and placebo were compared, there was no statistically significant difference in any of the three outcomes: The number of children that were pain-free at 2 h (OR 1.65, 95% CI 0.66–4.13), the number of children with pain relief at 2 h (OR 1.95, 95% CI 0.82–4.67) and the number of adverse events (OR 0.41, 95% CI 0.12–1.37) (Figure 3). The limited number of patients is likely to reduce the power of this comparison.
Comparisons of paracetamol versus placebo for migraine in children for outcomes pain-free at 2 h, pain relief at 2 h and adverse events.
Ibuprofen versus paracetamol in children
Likewise, when direct comparison between ibuprofen and paracetamol was made, there was no difference in any of the three outcomes: The number of children that were pain-free at 2 h (OR 2.34, 95% CI 0.96–5.71), the number of children with pain relief at 2 h (OR 1.79, 95% CI 0.73–4.42) and the number of adverse events (OR 2.16, 95% CI 0.63–7.49) (Figure 4). Again, only one study provided data (20).
Comparisons of ibuprofen versus paracetamol for migraine in children for outcomes pain-free at 2 h, pain relief at 2 h and adverse events.
Included systematic reviews
The 10 included SRs (19,24–32) were published between 2004 and 2016. None of them exclusively addressed ibuprofen and paracetamol for migraine in children; their scope was broader (Table 4).
The majority were narrative reviews, while only two SRs meta-analyzed the results of the primary studies (19,30). Regarding evidence on paracetamol, nine reviews included Hamalainen et al.; one excluded it, providing reasons. Regarding evidence on ibuprofen, only two SRs (19,24) included all three RCTs.
Conclusions about the efficacy of ibuprofen and paracetamol were discordant. For ibuprofen, nine SRs had positive conclusions and one SR concluded that there was insufficient evidence to draw a conclusion (Table 5). Regarding paracetamol, six SRs reported positive conclusive evidence, two reported positive inconclusive evidence, and one reported negative conclusive evidence (favoring placebo over paracetamol). One SR did not have any conclusion at all regarding paracetamol (Table 5).
Risk of bias in included reviews
Methodological quality of included systematic reviews judged with the AMSTAR tool.

AMSTAR scoring: each item is scored 1 if the specific criterion is met, or 0 if the criterion is not met, is unclear or is not applicable. An overall score refers to the sum of individual item scores. Quality ratings are as follows: 8–11 is high quality, 4–7 is medium quality and 0–3 is low quality.
Quality of evidence
Summary of findings table for Comparison 1: Ibuprofen versus placebo.

Explanations.
For all outcomes, we judged the impact of missing data to be relevant: We decreased the overall quality for limitations (−2).
For all outcomes, we judged the impact of different ages of children and doses of medicines to be unclear: We did not decrease the overall quality for inconsistency.
Summary of findings table for Comparison 2: Paracetamol versus placebo.

Explanations
For all outcomes, we judged the impact of missing data to be relevant: we decreased the overall quality for limitations (−2)
Wide 95% confidence interval considering relative risk and/or absolute events and small sample size. We did decrease the overall quality for imprecision (−1)
Summary of findings table for Comparison 3: Ibuprofen versus paracetamol.

Explanations
For all outcomes, we judged the impact of missing data to be relevant: we decreased the overall quality for limitations (−2)
Wide 95% confidence interval considering relative risk and/or absolute events and small sample size. We decreased the overall quality for imprecision (−1)
Discussion
The WHO EML recommends two analgesics for the treatment of acute migraine attacks in children – ibuprofen and paracetamol. Only three small RCTs about the efficacy and safety of ibuprofen and paracetamol were found. The summarized point estimates indicate a potential large beneficial effect of ibuprofen to reduce pain in children. The results were less compelling for paracetamol, as the reduction of pain was not significant in the analyses. However, the power of this comparison is limited by the inclusion of a single small study. Using GRADE methodology, we are led to conclude that the quality of the evidence is low for ibuprofen and very low for paracetamol.
All included RCTs had high risk of bias, as well as a number of potential problems in the design, conduct and interpretation of their results. In the study by Hamalainen at al., multiple deviations from the original protocol were described (20). In the study by Lewis et al., some patients were allowed to take other prophylactic drugs, introducing a strong confounder in an experimental design (21). Furthermore, the same study concluded that ibuprofen is effective for children and adolescents, particularly boys, whereas efficacy for girls was not established at all. Though there is substantial interest in sex differences in medicine effects, and stratification by sex is common in clinical trials, there is not a strong biological or pharmacological rationale to justify differences in the effectiveness of these analgesics between boys and girls. If subgroup differences are such that only boys or girls deserve treatment with some medicine, this would have major implications for clinical practice, and conclusions should be based on solid ground. Furthermore, the different dose schemes studied in those trials might not be considered equivalent. For example, a 10-year-old boy who weighs 35 kg would be treated with three different doses of ibuprofen: 350 mg, 262.5 mg or 200 mg.
Ten SRs that covered pharmacological treatments of migraine in children varied in terms of their methods and included studies. Conclusions about ibuprofen and paracetamol were discordant. Although the overall conclusion is that ibuprofen and paracetamol are effective for acute migraine attacks in children, very few reviews included all primary studies, meta-analyzed the results of primary studies, or considered the results of quality assessment when drawing conclusions. Evidence about safety was scarce. The methodological quality of the majority of included SRs, judged by the AMSTAR tool, was low.
A limitation of this study is the inability to separate data from primary studies for prepubertal and pubertal children (children and adolescents) because two trials that included both age ranges did not provide separate data for those populations.
The majority of evidence about efficacy and safety of medicines in children comes from studies in adults (33). Evidence related to children is usually derived from pharmacokinetic queries on single drugs in a limited number of subjects (34). However, safety and efficacy profiles of medicines may be significantly different between children and adults because of differences in developmental physiology, disease pathophysiology, or developmental pharmacokinetics and pharmacodynamics (35,36).
Effects of ibuprofen on the pain associated with acute migraine attacks in children were large and consistent across studies. Despite the studies showing some methodological shortcomings, the strong benefit associated with ibuprofen increases our confidence in the therapeutic value of this medicine. Evidence for paracetamol is less straightforward, as direct evidence is limited to one small, inconclusive trial. Nevertheless, this evidence has to be considered in the larger context of evidence derived from pharmacokinetic studies and RCTs conducted in adults.
Public health relevance
Ibuprofen and paracetamol are the only analgesics for migraine in children listed on World Health Organization's Essential Medicines list. Evidence for efficacy and safety of these drugs relies only on three small trials with a total of 201 children. Ten systematic reviews have been published about this topic, but no new trials. We need new trials on this topic to get high-quality direct evidence about efficacy and safety of ibuprofen and paracetamol for migraine in children.
Footnotes
Acknowledgements
We are grateful to Dr Lorenzo Moja and Dr Nicola Magrini of the World Health Organization for participating in formation of the study idea, study conduct and reporting.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
