Abstract
Studies on the treatment of migraine in children and adolescents are rare and difficult to design. In particular, the high placebo response in some trials makes it difficult to prove efficacy of a verum drug. We analysed all available placebo-controlled trials on acute and on prophylactic migraine treatment in children and adolescents with respect to different placebo rates (pain free and pain relief at 2 h; rate of responders with ≥ 50% attack frequency decrease). We identified eight crossover and 11 parallel group trials on acute treatment. The placebo response rates were considerably lower in crossover trials than in parallel group trials (19.2% vs. 27.1% for pain free after 2 h and 39.4% vs. 56.9% for pain relief after 2 h). In the 10 prophylactic trials included in this analysis, only a small trend towards a lower placebo rate in crossover trials could be observed. Further significant factors associated with a lower placebo rate in childhood and adolescence trials on the acute treatment of migraine were single-centre (vs. multicentre) trials and small sample size. Age and sex were not associated with the placebo response. Our study suggests that parallel group trials on the acute treatment of migraine in children and adolescents show a very low therapeutic gain due to a high placebo rate. The verum response rates, however, are very similar to those seen in adulthood trials. In conclusion, trial designs on the acute and prophylactic treatment of migraine in children and adolescents should consider the specific findings of this analysis in order to exhibit a higher probability of showing significant differences between placebo and verum drug.
Introduction
The treatment of migraine in childhood and adolescence is based on only very few randomized and placebo-controlled trials. In summary, only for ibuprofen (1–3) and intranasal sumatriptan (4–6) has sufficient and consistent evidence been obtained from more than one placebo-controlled, double-blind study for acute attack treatment. For prophylactic treatment, only flunarizine has shown consistent, although limited, efficacy in childhood and adolescence (7, 8). Therefore, only these substances are recommended for first-line use in different national treatment guidelines for the treatment of migraine in childhood and adolescence (9–11).
The most frequent reason for inefficacy of drugs in both acute and prophylactic migraine treatment trials is a very high placebo response, making it impossible to calculate a statistically significant difference between the drug and placebo (12). The reasons for this high placebo response in children and adolescents are still unknown. Many authors assume that the on average short duration of migraine attacks in children is one, but not the only, reason for this high placebo response. However, a recent meta-analysis of some studies on acute migraine treatment has suggested performing further and larger head-to-head studies with the primary end-point of migraine free (i.e. free of pain and associated symptoms at 2 h) in order to obtain significant results (12).
Children and adolescents with migraine often do not receive sufficient attack treatment (13). More studies on acute drugs are warranted to extend the treatment possibilities. Furthermore, safety of acute migraine drugs, in particular triptans, has to be shown for these age groups. We were interested in the trial design background of acute and prophylactic migraine studies in children and adolescents. Therefore, we performed a literature search on all placebo-controlled, randomized and double-blind trials on the acute and prophylactic treatment of migraine until the age of 18 years. In particular, we were interested in the placebo rate differences between the different trial designs.
Methods
According to a literature search in available public databases (MedLine, Embase, Web of Science), in congress contributions since the year 1990, and in review books on headache, we identified all trials reporting data of double-blind, placebo-controlled, randomized trials on the acute or prophylactic treatment of migraine in children and adolescents. We also included trials that were not published as a full paper but for which sufficient data was available on file or in the abstract publication. However, we did not include combined or single post hoc analyses of such trials. As search terms, we used ‘migraine’, ‘child∗’, ‘adolescen∗’ and ‘trial’. The diagnosis of migraine had to be established according to the criteria of the International Headache Society (or in studies before 1988 according to criteria similar to those) (14, 15).
In studies on the acute treatment of migraine, studies were analysed according to the number of children and adolescents in the intent-to-treat (ITT) population, according to the different substances (and in part also to different doses) tested, according to the 2-h pain relief and 2-h pain-free rates for both placebo and the different verum drugs, and according to the total number of adverse events. For studies on migraine prophylaxis, we analysed the percentage of responders for verum and for placebo for the ITT population, i.e. the percentage of patients with a reduction of attack frequency by ≥ 50%. However, these trials in particular often did not report sufficient data for placebo analysis.
We recorded the response rates as percentage and calculated pooled mean response rates (pooled for studies according to the number of patients). The data were grouped for acute crossover, acute parallel group, and prophylactic trials. We focused on responder rates since this was the most consistent end-point described in most studies and is clearly defined in contrast to the reduction of pain intensity or frequency, which is a very heterogeneous end-point (e.g. intensity rated on visual analogue scale or by another scale, frequency change related to a prospective baseline or a retrospective mean frequency by percentage or in absolute figures).
The data are presented as described in the papers. The mean weighted response rates are presented with the 95% confidence interval (CI). For correlation analyses, we used the Spearman rank correlation coefficient.
Results
We identified a total number of 41 trials fulfilling our inclusion criteria. Several trials did not present responder rates or adequate description of placebo responses and were therefore excluded from analysis (16–27). In addition, two further trials on oral sumatriptan (protocols S2CT37 and S2CT40) were described in one paper but were not published, not even as congress contributions with sufficient data presentation (28).
Table 1 presents all trials on acute migraine treatment with a crossover design. The pooled placebo response for the 2-h end-point was 19.2% (range 0–28%) for pain free and 39.4% (range 13–54%) for pain relief. The pooled placebo response for all adverse events was 8.6% (range 0–14%). The mean difference to the verum response was 20.2% for the pain-free end-point and 24.5% for the pain relief end-point.
Placebo and verum response rates for pain free and pain relief at 2 h after intake in different studies on acute migraine treatment in children and adolescents in double-blind, placebo-controlled, crossover trials

aMelting tablet.
bPreviously therapy-resistant migraine.
+ or − in parentheses denotes whether the study was positive or negative, respectively, in its primary end-point.
The data are based on the intent-to-treat (ITT) population (if possible).
ND, no data; po, oral; in, intranasal.
Table 2 shows all trials on acute migraine treatment with a parallel group design. Here, the pooled placebo response for the 2-h end-point was 27.1% (range 15–34%) for pain free and 56.9% (range 42–69%) for pain relief. The pooled placebo response for all adverse events was 21.3% (range 8–40%). The mean difference to the verum response was 5.8% for the pain-free end-point and 5.6% for the pain relief end-point.
Placebo and verum response rates for pain free and pain relief at 2 h after intake in different studies on acute migraine treatment in children and adolescents in double-blind, placebo-controlled, parallel group trials
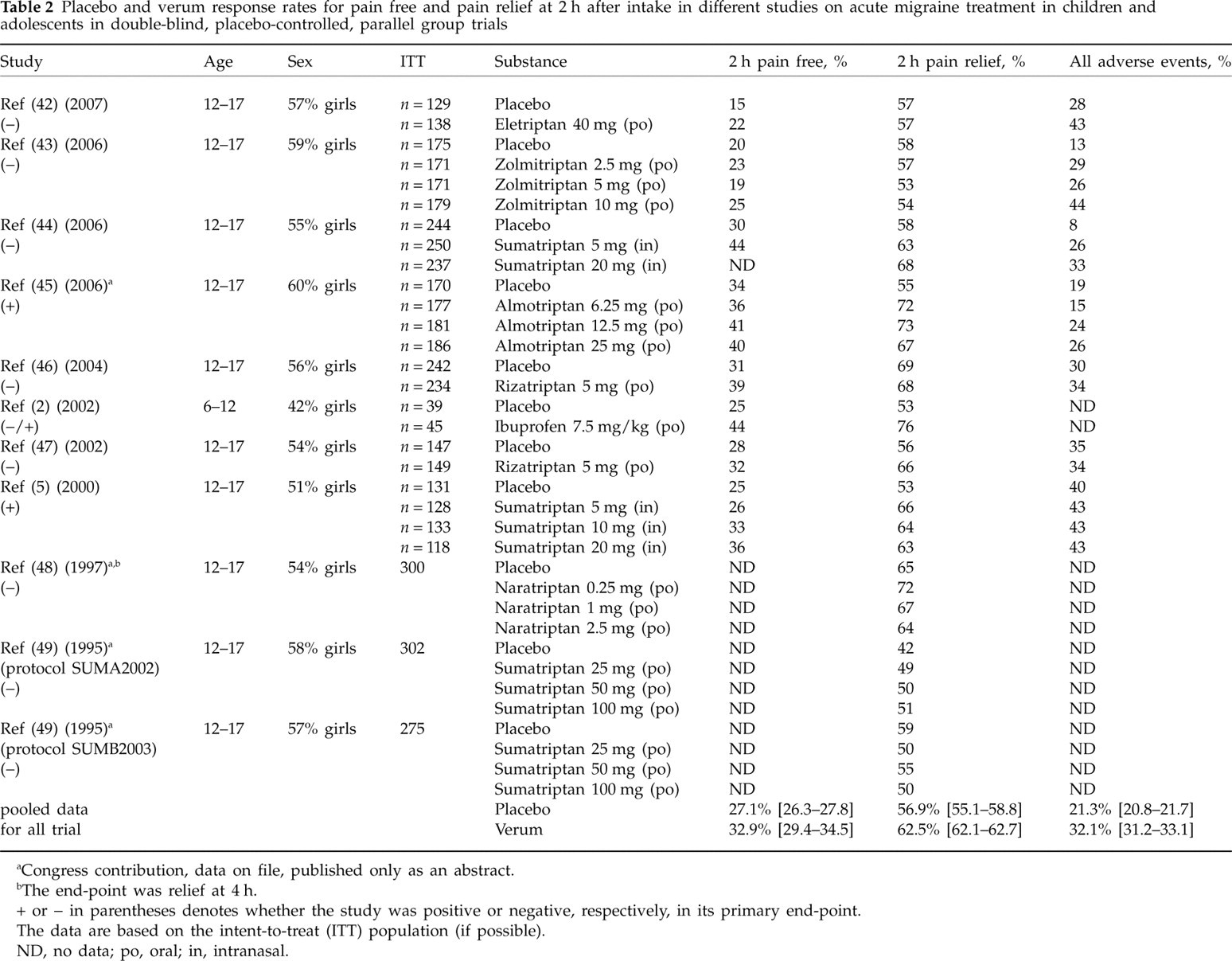
aCongress contribution, data on file, published only as an abstract.
bThe end-point was relief at 4 h.
+ or − in parentheses denotes whether the study was positive or negative, respectively, in its primary end-point.
The data are based on the intent-to-treat (ITT) population (if possible).
ND, no data; po, oral; in, intranasal.
Table 3 lists the trials on the prophylaxis of migraine. We do not present separate tables for crossover and parallel group trials since only very few trials with sufficient data could be detected. The pooled mean for the responder rates in the placebo groups was 42.2% (95% CI 35.7, 50.1) for the crossover trials and 44.0% (95% CI 38.5, 50.0) for the parallel group trials. The corresponding mean responder rate in the verum groups was 68.3% (95% CI 56.0, 83.1) for the crossover trials and 63.8% (95% CI 55.6, 71.4) for the parallel group trials.
Placebo rates for 10 different studies on prophylactic migraine treatment in children and adolescents in double-blind and placebo-controlled trials

aOnly adverse events leading to withdrawal from the study.
bResponse defined as attack frequency reduction by ≥75%.
+ or − in parentheses denotes whether the study was positive or negative, respectively, in its primary end-point.
The data were analysed according to the responder rates in month 3 and are based on the intent-to-treat population (if possible).
ND, no data; po, oral; in, intranasal; pg, parallel group; co, crossover.
For the trials on acute treatment, the correlation between the placebo and verum responses was also calculated. In the crossover trials, there was a significant correlation for the pain relief end-point (r = 0.76; P = 0.006) but not for the pain-free end-point (r = 0.39; P = 0.232). In the parallel group trials, a converse result could be seen. Here, the pain relief end-point did not show a significant correlation (r = 0.14; P = 0.520), but the pain-free end-point was significantly correlated between placebo and verum response (r = 0.76; P = 0.002). Finally, we calculated the correlation between the sample size and the different placebo rates. For the crossover trials, a significant positive correlation was obtained between sample size and pain-free placebo response (r = 0.75; P = 0.008) as well as between sample size and pain relief placebo response (r = 0.74; P = 0.009). For the parallel group trials, these correlations were not significant.
Discussion
Our analysis clearly shows that the placebo responses in children and adolescents both for pain free and pain relief are higher in parallel group trials than in crossover trials. The verum responses were not very different, resulting in a high difference between placebo and verum response (i.e. therapeutic gain) in crossover and almost complete lack of such a difference in parallel group trials. Also, the placebo rate for all adverse events was lower in crossover trials than in parallel group trials. This has a direct impact on the study outcome in children and adolescents. Seven out of eight crossover trials on acute migraine treatment were positive, whereas only two out of 11 parallel group trials were positive in their primary efficacy end-point.
In adults, the placebo response in trials of analgesics in migraine ranges between 7% and 50% with a mean of about 30% for headache response after 2 h and a resulting therapeutic gain of 30%. The mean respective placebo response for pain free after 2 h is 9% with a resulting therapeutic gain of 15–20% (29, 30). The therapeutic gain in crossover trials on childhood and adolescence migraine is about the same. Interestingly, it has been shown that the placebo response in adulthood trials on migraine prophylaxis is also significantly smaller in crossover than in parallel group trials (31), similar to our results in childhood and adolescence.
The analysis of placebo and verum response within one trial shows that there is a significant correlation between the two responses, suggesting that the magnitude of both placebo and verum response is explained by shared factors, perhaps the study population or the study design. The lower placebo response in crossover trials compared with parallel group trials, which our results show for children both for headache response and pain free, can also be observed in trials on analgesics in adulthood migraine for the headache response after 2 h (16% vs. 33%) (29), but not for the pain-free rate after 2 h (30). However, in adults the verum response is also lower in crossover than in parallel group trials (44% vs. 62%), resulting in a very similar therapeutic gain of 28 and 29%, respectively (29, 30).
We cannot explain the underlying mechanisms of the observed difference between crossover and parallel group trials. It is possible that when the patient knows that they will receive placebo at some time during the study the expectations of the efficacy of the treatment will be lower. This might also be true for parents who usually administer the study drug to their children or at least give some advice to their children when and how to take the study drug. The behaviour and the expectancy of parents might also influence the placebo rates in children. It has been shown that parents who volunteer their children to take part in a controlled trial are more vulnerable and socially disadvantaged (32) and fear the randomization to ineffective treatment (33); this might contribute to a higher placebo response expectancy in these parents and subsequently in their children.
Interestingly, the age of the children/adolescents does not play a major role in the placebo response. All but one crossover trials included also children under the age of 12 years, whereas all but one parallel group trials included only adolescents from the age of 12 years upwards. However, the placebo responses were higher in the parallel group trials. If age played a major role (i.e. younger children show a higher placebo response), we would not have obtained this difference between crossover and parallel group trials. Another reason for the low placebo rate in some trials might be that these trials included children and adolescents with severe migraine who were admitted to a tertiary headache clinic. Compared with the clinical features of childhood and adolescence migraine in other triptan trials, the duration and frequency of attacks in these trials were higher, resembling the features of adulthood migraine (28).
In one study on acute treatment (2), there was a clear difference in the placebo response between boys (43%) and girls (67%). Most of the other studies did not evaluate sex differences in placebo or verum response. In summary, there is no evidence for consistently different placebo rates between boys and girls. Therefore, sex should not be regarded as a major factor influencing placebo responses.
In adult studies on acute migraine treatment, a mean of about 23% reported at least one adverse event after placebo and 33% after verum (30). These are nearly the same figures as for parallel group trials in childhood and adolescence. The respective figures for crossover trials are much lower. In adults, the majority of all acute migraine treatment trials have a parallel group design. This might be the reason for this similarity. The number and intensity of adverse events were low and mild in all studies.
The response rates in trials on migraine prophylaxis in childhood are much more difficult to assess, since there is a lack of recent, well-designed trials (7). We focused on the percentage of responders (i.e. reduction of migraine frequency by > 50%), which is one of the recommended primary efficacy parameters in migraine prophylaxis trials (34). As regards this end-point, the response rates in crossover and parallel group trials are similar, but also show the trend to lower placebo rates in crossover trials. This has also been observed in adults (31).
When we analysed the acute treatment trials according to other different subgroups (single-centre vs. multicentre trials; US American vs. European trials; small vs. large sample size; trials including children vs. only adolescents), we made the interesting observation that there were two major subgroups of trials. On the one hand, the US American trials all had a large sample size, all excluded children < 12 years old, all but one had a parallel group design. On the other hand, the European studies all had a small sample size, all included children < 12 years old, and all were designed as crossover trials. The only design parameter which could not be related to one of these two subgroups was single-centre vs. multicentre trials with a pooled pain-free placebo rate of 16.6% for single-centre and 26.0% for multicentre trials (40.1 and 56.3% are the respective figures for the pain relief rates). The latter finding suggests that multicentre trials are more liable to a high placebo response than single-centre trials.
There has long been debate on whether or not triptans are efficacious in the treatment of migraine attacks in children and adolescents. This is obviously more a methodological problem than a matter of response. The response rates to active drugs in double-blind, placebo-controlled trials in children and adolescents are barely lower than those observed in adults. This has even been shown in open studies on subcutaneous sumatriptan (35, 36) and on oral zolmitriptan (37).
Our study has some limitations. First, we did not evaluate all available placebo responses (i.e. efficacy at other time points, recurrence rate, rescue medication, etc.). It might be that the difference between crossover and parallel group trials cannot be observed for these end-points. Second, the calculation of the end-points might be slightly different between the trials. This is in particular true for trials on prophylaxis. Third, most of the studies were performed at tertiary headache centres with a selection bias of the children and adolescents and thus not representative for placebo response in children in general.
In conclusion, our analysis suggests that crossover trials on migraine in children and adolescents have a higher probability of revealing significant differences between verum and placebo than parallel group trials. Age and sex do not play a role in the placebo response and are therefore not important factors for the trial design. A larger sample size and multicentre (vs. single-centre) trials are further, however minor, factors associated with a higher placebo response. Future studies considering these findings can expect similar response rates and subsequent similar therapeutic gains as are observed in adulthood migraine trials.
Footnotes
Competing interests
None to declare.
