Abstract
Background
The aim of this study was to investigate clinical features of a cluster headache cohort in Sweden and to construct and test a new scale for grading severity.
Methods
Subjects were identified by screening medical records for the ICD 10 code G44.0, that is, cluster headache. Five hundred participating research subjects filled in a questionnaire including personal, demographic and medical aspects. We constructed a novel scale for grading cluster headache in this cohort: The Cluster Headache Severity Scale, which included number of attacks per day, attack and period duration. The lowest total score was three and the highest 12, and we used the Cluster Headache Severity Scale to grade subjects suffering from cluster headache. We further implemented the scale by defining a cluster headache maximum severity subgroup with a high Cluster Headache Severity Scale score ≥ 9.
Results
A majority (66.7%) of the patients reported that attacks appear at certain time intervals. In addition, cluster headache patients who were current tobacco users or had a history of tobacco consumption had a later age of disease onset (31.7 years) compared to non-tobacco users (28.5 years). The Cluster Headache Severity Scale score was higher in the patient group reporting sporadic or no alcohol intake than in the groups reporting an alcohol consumption of three to four standard units per week or more. Maximum severity cluster headache patients were characterised by higher age at disease onset, greater use of prophylactic medication, reduced hours of sleep, and lower alcohol consumption compared to the non-cluster headache maximum severity group.
Conclusion
There was a wide variation of severity grade among cluster headache patients, with a very marked impact on daily living for the most profoundly affected.
Introduction
Cluster headache (CH) is one of the most severe primary headaches, characterised by attacks of recurrent, unilateral, excruciating pain with a retro-orbital maximum and which usually appear in periods (clusters). The attack duration varies from 15–180 minutes (1). During an active phase, attacks occur from once every second day to eight times a day. In the episodic CH type (ECH), cluster periods have a duration of seven days up to one year, separated by pain-free periods lasting one month or longer. About 10% of patients have chronic CH (CCH) with a remission period of one month or less per year. However, it is important to note that ECH can develop into CCH and vice versa during the disease time course (2). CH usually appears for the first time in the affected individual between the age of 20–40 years (1). An established estimation of CH prevalence from a meta-analysis of population-based studies is almost 0.1% (3). CH has been reported to be more common in men, and earlier studies have shown a male:female ratio of 5–6:1 (4,5), but over the past decades this dominance has been reported to be less prominent, at 2.5–3.5:1 (6,7).
Our aim was to define the clinical pattern of CH in Sweden and construct and test a novel scale for grading CH in this population in terms of CH attacks per day, CH attack duration in minutes, and CH period duration in months. We aimed for a scale that could be used shortly after diagnosis and without regarding the intermittent ECH and CCH subtypes. Barloese et al. previously developed a CH score system called the CH index, which is calculated by the equation (attacks per day × hours per attack) × (days per cluster ×clusters per year), which is not ideal for recently diagnosed CH patients only having experienced one cluster (8). There is also a recent report on a CH scale in a Korean population focusing on pain and the visual analogue scale (9). In addition, there is a British report on a scale grading CH quality of life: the Cluster Headache Quality of life scale (CHQ), where questionnaire results have been grouped into four subscales labelled “restriction of activities of daily living”, “impact on mood and interpersonal relationships”, “pain and anxiety”, and “lack of vitality” (10).
We wanted to further classify CH sufferers into possible new subgroups related to disease burden and whether there are clinical differences between identified subgroups.
Methods
Study design and population
Subjects were recruited from central Sweden (Stockholm, Linköping, Örebro, and Uppsala). Subjects were identified by screening medical records for the ICD 10 code G44.0, that is, CH, from 2004 onwards. All medical records from G44.0 patients were read by experienced neurologists (the authors AS, CS or EW) to confirm that the diagnosis was fulfilled by the International Headache Society (IHS) criteria for CH (1). Subjects were then contacted by phone and/or postal mail and asked to fill in a questionnaire. For patients attending the outpatient clinics, the questionnaire was completed in relation to the visit. Informed consent was obtained from all study participants. The study was approved by the Regional Ethical Review Board, Stockholm, Sweden.
Questionnaire
The questionnaire was composed of three parts (see Supplementary Data). The first part included personal, demographic and medical aspects, the second included questions designed to assess the CH pattern according to the IHS criteria third edition, and the third part included questions related to lifestyle (1).
Cluster Headache Severity Scale (CHSS)
Detailed criteria for the Cluster Headache Severity Scale (CHSS).

Cluster headache maximum severity (CHMS) subgroup
To implement and test the CHSS, we further defined and characterised a subgroup of patients with a high CHSS score, and thereby a presumed particularly severe CH, and comprising at least 10% of the CH patients for comparison with the CCH group, which led us to a CHSS score ≥9.
Statistical analysis
We used Student’s t-test to compare continuous variables and Fisher’s exact test to compare categorical variables. One-way ANOVA and chi-square test were used when there were more than two groups. All tests were two-tailed. An alpha level of <0.05 was considered statistically significant. All analyses were performed using GraphPad Prism v5.04 (GraphPad Softwares Inc, La Jolla, CA, USA).
Results
Summary of cluster headache patient data

based on 484 individuals
based on 475 individuals
Age at onset presented as mean ± standard deviation.
CH: cluster headache.
Age at time of investigation: The mean age at the time of investigation for women was 48.9 ± 15.5 (range 17–83) years and for men 51.6 ± 13.6 (range 17–83) years. For age and gender distribution of all CH patients in relation to episodic versus chronic CH at the time of the investigation see Supplementary Figure 1. The age range of 50–59 years was the most common age group for both genders, including 25.3% of men and 25.6% of women. There was a statistically significant trend towards a younger age distribution among women compared to men (p = 0.0407).
Alcohol intake by gender or cluster headache subtype. Distribution of alcohol intake by gender or cluster headache subtype is divided into the following four groups; never or rarely consume alcohol, 1–2 standard units per week, 3–4 standard units per week and 1–2 standard units per day.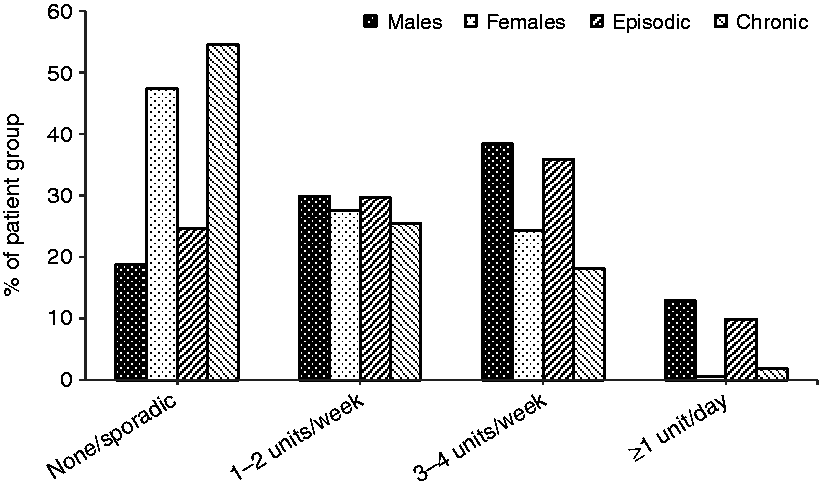
Age at onset: The mean age at onset for women was 30.3 ± 14.8 (range 11–69) years and for men 31.2 ± 12.7 (range 7–69) years. CCH patients had a significantly later mean age at onset, at 35.0 years compared to ECH patients with 30.4 years (p = 0.0205).
Tobacco habits: Smoking habits were known for 498 patients. Overall, 146 (29.3%) CH patients were current smokers, 210 (42.2%) CH patients were previous smokers, and 142 (28.5%) CH patients had never smoked. We compared the number of current smokers with the general Swedish population using data from the Swedish statistics agency SCB (Statistiska centralbyrån), which reported 11.8% of the population over the age of 16 to be current smokers (11). We observed a significantly higher proportion of smokers in the CH population compared to the general population (p < 0.0001). When stratifying the CH population in relation to gender, we additionally observed a higher proportion of smokers among males as well as females, compared to the general Swedish population (p < 0.0001 for both comparisons). As snuff is a very common means of tobacco consumption in Sweden (12.1% of the population over the age of 16 use snuff), we investigated snuff use as well as overall tobacco consumption, combining smoking and/or snuff, among our CH patients. Overall, 18.3% of the patients used snuff, 14.3% had used snuff previously, while 67.4% had never used snuff. There was a significantly higher proportion of patients using snuff in the CH population compared to the general population (p < 0.0001). The difference between CH patients and the Swedish population regarding smoking and snuff use remains highly significant when analysing overall tobacco consumption (p < 0.0001). CH patients who were current or previous tobacco users had a later onset of disease, at 31.7 years compared to non-tobacco users at 28.5 years (p = 0.0260). The difference in age of onset remains after removal of CCH patients, however it was not significant due to the small sample size (31.1 vs. 28.2; p = 0.0526). No difference was found between ECH and CCH patients in any of the three groups.
Alcohol intake: Alcohol habits were known for 470 patients (Figure 1). We used the following standard unit to measure alcohol consumption: One standard unit represented 12 g of alcohol equivalent to 15 cl table wine, 33 cl beer with alcohol strength >4.5%, 50 cl beer >3.5%, 8 cl dessert wine, or 4 cl spirits. Overall, 28.1% patients never or rarely consumed alcohol, 29.1% had a consumption of one to two standard units per week, 33.8% consumed three to four standard units per week and 8.9% had a consumption of one to two standard units per day. When comparing alcohol habits of ECH and CCH patients, we found that a significantly larger proportion of episodic patients (35.9%) compared to chronic patients (18.2%) had a regular alcohol intake of three to four standard units per week (p = 0.0090). In addition, there was a significantly larger proportion of chronic patients (54.5%) compared to episodic patients (24.6%) who never or rarely consumed alcohol (p < 0.0001). Similarly, we observed that female patients consumed less alcohol than male patients, which agrees with the general Swedish population (12). In the general Swedish population, 18% of women and 10% of men report no alcohol consumption. In addition, 15% of men and 8% of women report a high alcohol consumption that corresponds to 2.5 bottles of wine or more per week for men and two bottles of wine or more per week for women, and can be compared to the patient group reporting one to two standard units per day. There was no significant difference with respect to age at onset between patients with high alcohol intake of one to two standard units per day or more compared to those with a lower consumption (p = 0.4456).
CHSS score: The most common CHSS score was 5, representing 27.6% of the patients (Figure 2). The most uncommon score was 12, representing 0.9% of the patients. A significant difference was seen between ECH and CCH regarding mean CHSS (p < 0.0001). ECH had a mean score of 6.2, while CCH had a mean score of 8.4. The CHSS score also differed significantly between men and women (6.3 vs. 6.7; p = 0.0248). The CHSS score mean ranged from 4.6 to 7.0 per age decade, see Supplementary Figure 2 for additional information on the CHSS distribution in episodic and chronic CH. Chronic CH patients generally had a higher CHSS score in each age decade compared to episodic CH, except for the <20 group and the 80–89 group, which consisted of few individuals. There was no significant difference in mean CHSS score regarding late age at onset (≥50) compared to earlier age at onset (<50) (6.5 vs. 6.4; p = 0.69). However, the severity score was higher in the patient group reporting sporadic or no alcohol intake than in the group reporting three to four standard units of alcohol per week or a higher consumption, (6.9 vs. 6.3; p = 0.0017), see Supplementary Figure 3.
Distribution of Cluster Headache Severity Scale score. Graph illustrates the distribution of the Cluster Headache Severity Scale (CHSS) score between episodic (n = 412) and chronic (n = 51) cluster headache (CH).
Triggers: One or more trigger factors were reported in 53.8% of the 481 CH patients who answered the question (Figure 3a). Alcohol was the most prominent: 27.9% of CH patients reported alcohol as a trigger factor. However, when patients were asked specifically about alcohol as a trigger factor, 55.0% answered positively.
Trigger factors and attack distribution for cluster headache attacks. (a) Different trigger factors reported for cluster headache attacks; 53.8% of all patients (n = 500) reported at least one trigger factor. (b) Attack distribution per 24 hours in two-hour intervals in episodic (n = 421) and chronic (n = 54) cluster headache (CH) patients, separately and combined.
Attack duration: The attack duration was 15–30 minutes in 21.2% of the patients, 30–120 minutes in 55.3% of the patients, 120–180 minutes in 17.7%, and more than 180 minutes in 14.6% of the patients (multiple answers were possible). In addition, 11.6% reported solely an attack length with a duration of more than 180 minutes. The distribution of attack duration was similar between the two CH subtypes.
Number of attacks: A majority of the CH patients reported one to two attacks per day in active periods (45.5%) while a minority specified <1 attack per day (7.4%) or ≥ 6 attacks per day (13.3%). When analysed separately in relation to number of attacks in the CH subtypes, a majority of the ECH patients (47.6%) reported one to two attacks per day compared to CCH patients where a majority (50.9%) reported three to five attacks per day.
Timing of attacks: 66.7% of the patients reported that attacks appear at certain time intervals, while 33.3% could not specify any typical time interval. Almost half of the patients (47.6%) reported that attacks could appear at a random time of the day in addition to time intervals that could be more common. Night time was the most commonly reported time interval for attacks (Figure 3(b)). In addition, patients with a diurnal rhythmicity have a lower CHSS mean score compared to patients without a diurnal rhythmicity (6.3 vs. 6.7; p = 0.0195).
Duration of periods: Regarding the length of the CH periods, a majority of patients (35.2%) reported this as one to two months, while a minority (5.7%) reported seven to 12 months, and some reported more than one length of period. The rest of the distribution was as follows: <1 month = 22.6%; 2–4 months = 24.3%; 4–7 months = 7.1%, and >12 months = 10.5%.
Attack medication: The majority of CH patients (52.6%) used sumatriptan injections, while a nasal spray of sumatriptan or zolmitriptan was the second most common acute medication (38.4%). Sumatriptan injections were in addition used by 53.7% of the ECH patients compared to 43.9% of the CCH patients (see Figure 4(a) for details).
Attack and prophylactic medication with regard to episodic and chronic cluster headache. (a) Distribution of reported acute treatments for the chronic cluster headache (CH) patients (n = 57) and episodic CH patients (n = 443). (b) Distribution of reported prophylactic treatment for chronic CH patients (n = 57) and episodic CH patients (n = 443).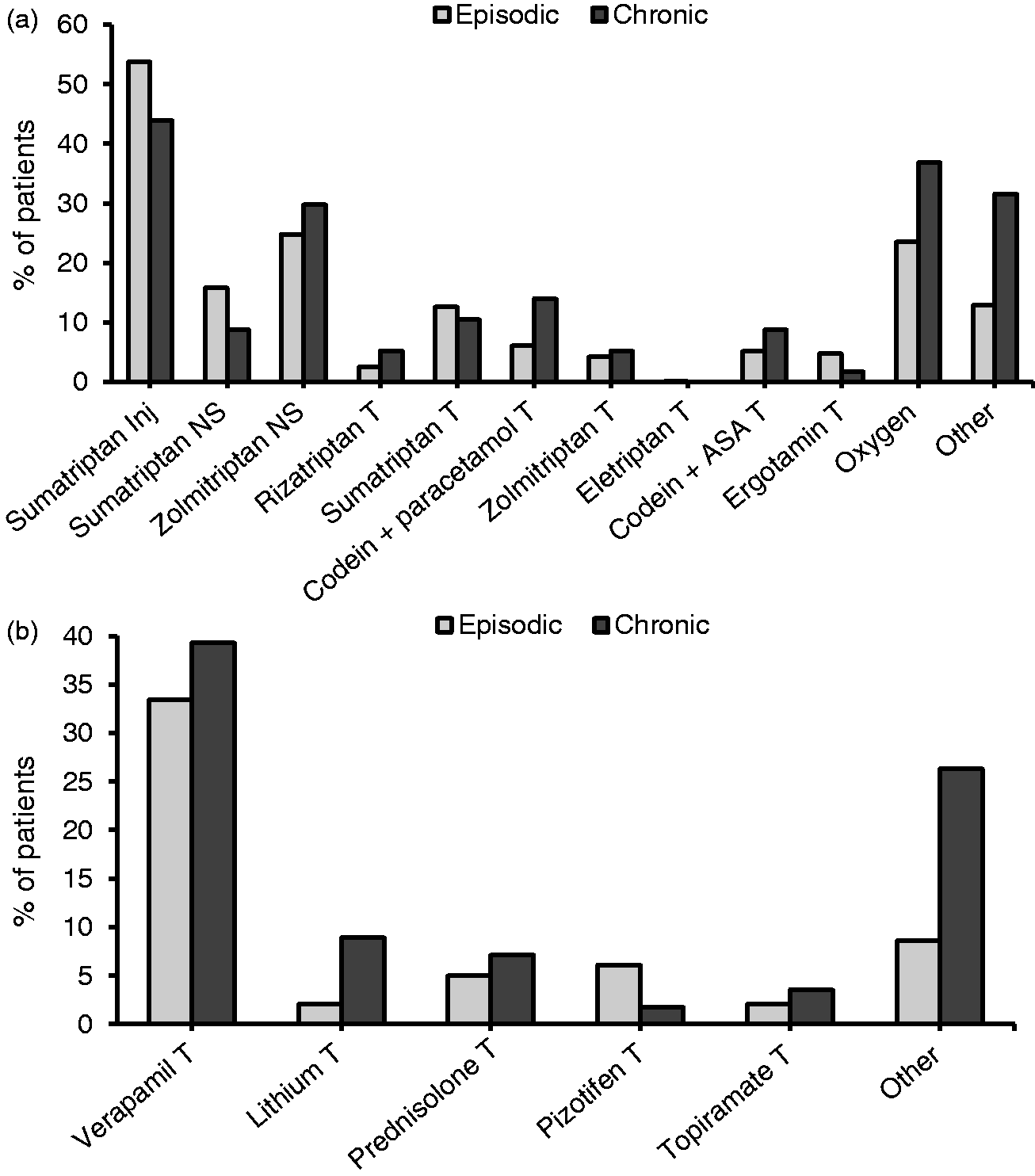
Prophylactic medication: 51.0% of all CH patients reported using prophylactic treatment, and it was significantly more common among CCH patients than ECH patients (66.7% vs. 49.0%; p = 0.0161). Verapamil was the most common reported prophylactic treatment (34.1%) and was used by 39.3% of the CCH patients and by 33.4% of the ECH patients. Lithium was used by 8.9% of the patients in the CCH group and by 2.0% of the ECH patients. Verapamil and pizotifen have been reported to reduce frequency of attacks, while lithium, prednisolone, and topiramate reduce the number of periods and topiramate additionally reduce period duration (13,14). For an illustration of this data see Figure 4(b).
Heredity: In the total group of CH cases, 11.2% reported at least one first-, second- or third-degree relative with CH. The distribution between ECH cases and CCH cases was 10.8% compared to 14.0% (p = 0.4719). However, when comparing heredity among female and male CH patients, a significantly larger proportion of the women (17.5%) had a relative with CH diagnosis compared to the men (8.2%), p = 0.0021.
Migraine: Migraine with or without aura occurred in 16.7% of the CH cases, and the distribution between genders was 25.2% of the women and 12.8% of the men. Furthermore, 16.0% of the ECH and 22.2% of the CCH patients reported that they suffered from migraine, with no significant difference between groups (p = 0.2561).
Associated symptoms: We analysed the occurrence of the six standard symptoms usually associated with a CH attack. Sixty percent of all patients reported conjunctival injection (eye redness), 75.8% lacrimation (tearing), 49.0% ptosis (drooping eyelid), 50% rhinorrhea (runny nose), 46.8% nasal congestion, and 47.8% reported a sense of restlessness (see Supplementary Figure 4). At least one of the associated symptoms occurred in 93.2% of the patients, while 6.8% of the patients stated that they had no autonomic symptoms.
Chronotypes and sleep: When comparing the two different chronotypes, morning people (larks) and evening people (owls), we found that CCH patients (47.4%) were more commonly larks compared to ECH patients (32.5%; p = 0.026). In contrast, 40.4% of the ECH patients were owls compared to 24.6% of the CCH patients (p = 0.0207). Regarding sleep duration, 21.1% of the CCH patients slept for less than five hours per night compared to 8.1% of the ECH patients (p = 0.0018). We found no significant differences between females and males. We additionally analysed sleeping patterns and attack as well as period duration in patients, and a significant association with short sleep duration (<5 hours per night) was observed for both attack durations of more than 120 minutes (p = 0.019), and periods of more than two months (p = 0.038).
Comparison of the clinical features between CHMS and non-CHMS patients.

t: t-test; df: degrees of freedom; OR: Odds ratio for Fisher’s exact test; CI: Confidence interval; n.a: not applicable; CHMS: Cluster headache maximum severity.
Age at onset presented as mean ± standard deviation.
Discussion
In this study we aimed at investigating important clinical features of a CH population in Sweden. To evaluate the disease burden in this population, a new scale for CH status, CHSS, was constructed. Barloese et al. had previously developed a CH score system called the CH index, which is calculated by the equation (attacks per day × hours per attack) × (days per cluster ×clusters per year) (8). In line with our results, a higher CH index was reported in CCH patients compared to ECH. Barloese et al. additionally reported a higher CH index in patients without diurnal rhythmicity, although not significant, which agrees with our findings. In contrast to the CH index, CHSS takes no account of clusters per year; however, the advantage with the CHSS is the 12-step gradient, which enables us to further characterise CH into subgroups. In addition, the advantage of the CHSS scale is that it could be a useful tool for clinicians to further classify CH patients with a recent diagnosis, for example immediately after the end of the first period. It can also be regarded as a valuable tool for intra-individual comparison over time and between different periods. CHSS can possibly be a future indicator on, for example, which acute or prophylactic medication should be used for the most successful outcome in a newly diagnosed CH patient according to scale classification number as well as an indicator of disease progress.
A subgroup of patients with a high CHSS score ≥9, including 14% of the study population, was further characterised to evaluate how the scale can be utilised. Not unexpectedly, the proportion of CCH was larger in this seriously-affected subgroup than in the less-affected group. Data indicated that the disease, for several aspects of their daily living, significantly affected these patients.
Earlier studies have shown a negative prognostic factor for late disease onset, ≥50 years of age (15,16). We could not replicate these findings in our study population for CHSS, which could be due to a small sample size for the late age at onset group with known CHSS (n = 39). We did, however, observe that CHMS patients had a significantly higher mean age at onset compared to non-CHMS patients. We additionally reported a higher age at onset in CCH compared to ECH, which agrees with previous studies (17).
Many studies have described an association between CH and smoking (5,18,19). We compared smoking habits in the CH population with the general Swedish population and could confirm a higher proportion of smokers among CH patients. In addition, we observed a higher proportion of patients using snuff in the CH population compared to the general population. Importantly, we found a significant correlation between the age at onset and tobacco habits in our study: CH patients who were current tobacco users or had a history of tobacco consumption had a later onset of disease (31.7 years) compared to non-tobacco users (28.5 years). In a previous study from one of the co-authors, no consistent change in tobacco consumption could be seen during cluster periods (18).
A broad range of trigger factors for CH attacks has been reported earlier, of which alcohol stands out being reported in more than 50% of CH patients in several populations worldwide (18–21). In our study, 55% reported alcohol as a trigger factor. We have shown a significant difference in severity grade on the CHSS in relation to alcohol intake. CHSS score was inversely proportional to alcohol consumption, that is, patients with no or sporadic alcohol intake had the highest CHSS mean score while those with an intake of one to two standard units of alcohol per day or higher had the lowest of the four groups. In summary, higher disease burden appears to be connected with lower alcohol intake. This is in line with earlier reports of CH patients reducing their alcohol consumption during CH periods (18). This may explain why fewer patients reported alcohol as a trigger factor when asked to list specific trigger factors than when specifically asked if alcohol is a trigger factor for attacks.
A significant difference was seen between CHSS scores in episodic and chronic patients, as CCH was associated with higher mean scores. These results reveal the suffering of CCH patients, who apparently, and not surprisingly, have a heavier disease burden in a multifactorial way.
We reported an 11.2% frequency of CH cases with heredity, and this frequency was slightly higher in CCH patients (14.0%) than in the total cohort, although not significant. Interestingly, more female CH patients had a relative with a CH diagnosis compared to males. The general frequency agreed with previous studies, which reported that 7–20% of the patients had a positive family history (22,23).
We reported that 11.4% of the CH patients suffered from CCH, which agrees with the IHC classification. However, there are small studies from Eastern countries that report CCH to be more rare (3.5% in Japan) or even absent (China and Taiwan) (21,24,25).
We found comorbidity with migraine in 16.7% of the CH cases: 25.2% of the women and 12.8% of the men. These results are close to the reported migraine prevalence in the general Swedish population (13.8–18%) (26–28). Several studies have been performed regarding the prevalence of migraine in patients suffering from CH, and the reported occurrence of migraine has varied markedly (20,29,30).
Untreated CH attacks have a duration of 15–180 minutes as per the definition; longer or shorter attacks may occur, but over less than half of the time-course of CH (1). We found in our material a range of attack durations, where 30–120 minute attacks occurred in 55.3% of the study population. We also found that 11.6% reported exclusively attack lengths of more than 180 minutes. This could possibly be an effect of the background pain reported by CH patients during active periods, making it difficult to distinguish specific time intervals for attack duration (31).
There was a nightly dominance of attacks in our material, which is in line with previous reports (8,32,33). Interestingly, the Rozen and Fishman study found three troughs during the day (08:00, 11:00 and 17:00), as did Barloese et al. (09:00, 12:00 and 18:00), while we observed a trough between 10:00 and 16:00. In contrast to these studies, patients in an Italian population reported a majority of attacks at 14:00 (30). These data suggest that the diurnal rhythms might be influenced by the culture of the specific region, for example, taking a short rest or nap in the early afternoon (siesta or riposo).
Significantly more patients in the seriously affected subgroup reported sleeping less than five hours per night. Whether this is caused by disturbed sleep due to nocturnal attacks for a long period of time, or a particular trait of vulnerability in these patients, cannot be answered from our questionnaire. Interestingly, Ofte et al. found that 40% of the CH patients in their study reported insomnia (33). Patients with insomnia had significantly longer cluster periods and there was a non-significant trend towards longer cluster attacks, which is in agreement with our results, comparing the patients with the shortest sleep duration to the rest of the CH population. Since Ofte et al. also noted a higher proportion of shift work in patients with CH, they speculated whether shift work and/or insomnia increase the risk of CH.
Some limitations of our study need to be addressed. We acquired 500 questionnaires out of 892 contacted patients with CH in Sweden. In all, 36.1% did not respond and 7.4% did not want to participate, which agrees with previous studies reported on CH (8). Possible selection bias might have occurred: Patients with more severe symptoms may have a higher motivation to seek attention from specialists. Patients who have been in remission for a long time, for example one year or more, at the time of the study might have more difficulty remembering specific cluster periods in detail than patients that had experienced a more recent or current cluster period. This potential recall bias has not been accounted for in this study and may have been avoided by using headache diaries (34). Additionally, the data from the questionnaire is self-reported.
Conclusion
In conclusion, our data regarding clinical findings in a Swedish CH patient cohort agrees with earlier reported studies. Importantly, we constructed a new scale for CH, CHSS, and this scale could be a useful tool for describing symptom severity and ultimately be a score of disease burden. Not surprisingly, CCH appears to be a significantly more severe condition compared to ECH. In addition, a larger proportion of the patients were males (2:1), on the other hand female CH patients reported more severe CH with a larger proportion of CCH, and elevated CHSS scores as well as higher heredity. Alcohol was the most common trigger factor reported by patients in our material. Considering the grave severity described in this report, more studies on the clinical patterns of CH are required regarding the socioeconomic impact of the disease.
Article highlights
A novel scale for grading cluster headache; the Cluster Headache Severity Scale (CHSS) was constructed. Cluster headache patients who are current tobacco users or have a history of tobacco consumption have a later age of disease onset compared to non-tobacco users. A subgroup with particularly severe CH was defined as patients with a CHSS ≥ 9, characterised by higher age at disease onset, greater use of prophylactic medication, reduced hours of sleep, and lower alcohol consumption compared to CH patients with a CHSS ≤ 9.
Footnotes
Acknowledgements
We thank Ann-Christin Karlsson, Fengqing Xiang and the neurology clinics and private practices in Stockholm, Örebro, Linköping, and Uppsala for help with the recruitment of patients. This work was supported by the Swedish Brain Foundation [FO2013-0213], Swedish Research Foundation [2012-02211], Åke Wiberg Foundation [ID:756194137K2012], Swedish Migraine Society, and Karolinska Institutet Research funds [2016fobi48906].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
