Abstract
Objective
A child presenting with a first attack of migraine with aura usually undergoes magnetic resonance imaging (MRI) to rule out stroke. The purpose of this study was to report vascular and brain perfusion findings in children suffering from migraine with aura on time-of-flight MR angiography (TOF-MRA) and MR perfusion imaging using arterial spin labelling (ASL).
Methods
We retrospectively included all children who had undergone an emergency MRI examination with ASL and TOF-MRA sequences for acute neurological deficit and were given a final diagnosis of migraine with aura. The ASL perfusion maps and TOF-MRA images were independently assessed by reviewers blinded to clinical data. A mean cerebral blood flow (CBF) value was obtained for each cerebral lobe after automatic data post-processing.
Results
Seventeen children were finally included. Hypoperfusion was identified in one or more cerebral lobes on ASL perfusion maps by visual assessment in 16/17 (94%) children. Vasospasm was noted within the intracranial vasculature on the TOF-MRA images in 12/17 (71%) children. All (100%) of the abnormal TOF-MRA images were associated with homolateral hypoperfusion. Mean CBF values were significantly lower (P < 0.05) in visually hypoperfused lobes than in normally perfused lobes.
Conclusion
ASL and TOF-MRA are two totally non-invasive, easy-to-use MRI sequences for children in emergency settings. Hypoperfusion associated with homolateral vasospasm may suggest a diagnosis of migraine with aura.
The prevalence of migraine in children is in the range of 2.7–17% (1). There are two major subtypes of migraine: with or without aura. Aura migraines are characterised by transient focal neurological symptoms associated with headache. Symptoms can be motor, visual, sensory, aphasia or decreased level of consciousness. They last for 5–60 min and are accompanied, or followed within 60 min, by headache. Most children with migraine experience aura-type migraines during their lifetime (1,2).
Migraine with aura presents as an acute neurological disorder mimicking stroke, especially the initial attacks. It calls for emergency imaging. But strokes are less common than migraine with aura in children (3,4). Magnetic resonance imaging (MRI) combined with diffusion-weighted imaging (DWI) sequences is the most sensitive radiological technique for ruling out stroke. The diagnosis of migraine is generally a clinical diagnosis, with normal findings on conventional imaging (structural imaging and DWI) if undertaken.
The pathophysiology of migraine attacks with aura is still unclear. Hypoperfusion was described in adult populations by Olesen in 1990 using intracarotid xenon-133 injection and single-photon emission computed tomography (SPECT) (5,6). Olesen described a hypoperfusion pattern during the aura phase of the migraine attack followed by a hyperperfusion pattern during the headache phase itself. More recently, two studies demonstrated cerebral hypoperfusion in 14/20 and 18/33 adult patients suffering from aura migraine with neurological deficit mimicking acute stroke by using susceptibility-based MR perfusion-weighted imaging following bolus injection (DSC-PWI) (7,8).
Among the various perfusion imaging techniques available, arterial spin labelling (ASL) is an emerging MRI sequence that can be easily performed during conventional MRI without contrast injection or any side effects (9). The only downside is a longer examination time of approximately 5 min. In children, a single study in the literature has shown the sensitivity of ASL in detecting perfusion abnormalities in a series of ten patients suffering from migraine with aura (10). Time-of-flight MR angiography (TOF-MRA) sequences provide visualisation of the intracranial arteries without contrast injection and are usually used if a stroke is suspected. Literature studies describing TOF-MRA findings in migraine with aura are rare. A reversible vasospasm has been reported in an 11-year-old girl (11) and more recently in a series of eight children suffering from acute hemiplegic migraine (12).
The aim of our study was to report ASL cerebral perfusion imaging and TOF-MRA findings in children with aura migraine.
Materials and methods
Patients
A retrospective analysis was conducted in our institution over a three-year period. We retrospectively included all children who had undergone emergency MRI with ASL and TOF-MRA sequences in our radiology department for acute neurological (visual, language, motor or sensory) deficit and whose final diagnosis was migraine with aura, according to the ICHD-3 beta criteria (13).
Children with a motor deficit were classified as having hemiplegic migraine, and children with a decreased level of consciousness and no motor deficit were classified as having brainstem aura.
Exclusion criteria were children with a complex neurological history (e.g. coma or epilepsy), an uncertain diagnosis of migraine with aura, an incomplete MR examination (no TOF-MRA or ASL) or uninterpretable TOF-MRA or ASL sequences.
Four of our patients underwent a control MRI examination.
Our study was approved by the local Ethics Committee. Parental consent was obtained.
Clinical data
The following demographic and clinical data were retrospectively collected from medical files: age; sex; past medical history including personal or family history of migraine; ongoing medical treatment; symptom onset; and duration. The neurological examination was assessed by a senior emergency paediatrician.
Imaging study
All children were imaged using a Siemens 1.5T Magnetom Aera scanner and a 16-channel head coil (Siemens; Erlangen, Germany). The brain MRI protocol included routine sequences as implemented in our standard protocol: axial fluid-attenuated inversion recovery (FLAIR); axial T2 gradient-echo; three-dimensional (3D)-T1; diffusion-weighted MRI; and TOF-MRA.
As the TOF-MRA sequence is performed to rule out stenosis or occlusion, standard TOF-MRA acquisition box placement is usually over the circle of Willis. We noticed two different acquisition box positions in our population due to practice variability: the standard position (stroke protocol) and a higher position where the distal arterial branches are best visualised.
The non-contrast perfusion two-dimensional (2D) pulsed ASL MRI sequence used was PICORE Q2TIPS (14). Imaging parameters were as follows: TR/TE/TI1/TI2 = 3500/12/700/1800 ms; voxel size = 4 × 4 × 8 mm; gap = 2 mm; 30 label/control pairs; total scan time = 3 min 20 s. One M0 reference image was acquired. Multi-slice single-shot echo-planar imaging (EPI) was used as readout. ASL perfusion images were automatically generated by the Siemens station and used for visual/qualitative analysis.
Additionally, post-processing of both 3D-T1 and ASL data was performed using AutoASL, a custom-built ASL processing tool based on SPM software (Wellcome Trust Centre for Neuroimaging, University College of London, UK) and Matlab® (The MathWorks, Inc.) to create a quantitative map of cerebral blood flow (CBF). Furthermore, brain segmentation was automatically performed using templates. Regions of interest (ROI) were defined using the Automated Anatomical Labeling (AAL) atlas (15) to obtain a quantitative CBF value (mL/100 g/min) for each lobe.
Imaging analysis
All MR images were retrospectively analysed by two paediatric radiologists in consensus. The TOF-MRA and ASL images were evaluated separately. The reviewers were blinded to clinical data, including the side affected by the neurological symptoms.
First, the perfusion maps were analysed visually with the readers blinded to all other sequences, including MRA. Perfusion maps were classified as showing a normal perfusion pattern or abnormal perfusion pattern. Perfusion symmetry was also assessed. Perfusion abnormalities were classified as areas of hypoperfusion or hyperperfusion in each cerebral lobe: frontal (F); temporal (T); parietal (P); and occipital (O). Second, a mean CBF value (mL/100 g/min) was obtained for each lobe after data post-processing. Finally, the mean CBF and standard deviation (SD) were determined in the lobes that were visually hypoperfused and those that were visually normally perfused.
Syngo.via® (Siemens) was used to analyse the circle of Willis vessels on the 3D TOF stack. TOF-MRA images were analysed for each hemisphere and arterial territory of the circle of Willis: anterior cerebral artery (ACA); middle cerebral artery (MCA); and posterior cerebral artery (PCA). TOF-MRA findings were classified as follows: normal; vasoconstriction of the peripheral branches; vasodilation of the peripheral branches; or stenosis.
Statistical analysis
Quantitative variables were described by a mean ± SD or median (range). After verification of normality, the comparison of CBF values was analysed using Student’s t-test. All statistics were performed with a 0.05 level of significance.
Results
Clinical and laboratory findings
Between January 2014 and November 2016, we finally included 17 children aged 11–16 years (mean age = 13.6 years ± 1.5). There were eight girls and nine boys.
Six children had a personal history of migraine, four of which also had a family history. For the 11 remaining children, there was either no history of migraine or no information available. Five of them (30%) had a history of CT scan performed for ‘brain trauma’ between six months and nine years previously (median time = 5 years).
The median time between arrival at the emergency room and the MRI examination was 1 h 51 min (range = 7 min–5 h 32 min). The median onset-to-MRI time was 6 h (range = 1 h 36 min–2 days) for onset of the aura and 6 h 15 min (range = 2 h 30 min–3 days) for headache. All children presented with headache before or during the MRI examination.
Symptoms included visual disturbance (n = 7), aphasia/dysarthria (n = 12), sensory deficit (n = 10), motor deficit (n = 2), sensory and motor deficit (n = 3) and decreased level of consciousness (n = 3). Some children had several symptoms simultaneously.
Population characteristics.

Three patients also underwent electroencephalography (EEG) examinations. Two of them had abnormal EEG results showing hemispheric slowing contralateral to the side of hemiplegia (patients 9 and 13). One patient had a normal EEG (patient 10).
Four children underwent follow-up MRI between 1 day and 3 h (27 h) and 3 days and 2 h (74 h) after the acute neurological episode. Symptoms had disappeared when follow-up MRI was performed and the neurological examination was normal. The same MRI protocol including TOF-MRA and ASL was used.
Neuroimaging findings
Standard conventional MR sequences were normal for every patient, particularly the DWI and FLAIR sequences.
Perfusion maps
In 16 patients (16/17 [94%]), alteration of cerebral perfusion was found by visual assessment. It was always hypoperfusion (100%), which was unilateral in 13/16 (81%) and bilateral in 3/16 (19%) patients. Among the patients with unilateral hypoperfusion, left hemisphere involvement was predominant (10/13 patients, 77%) compared with right hemisphere involvement (3/13 patients, 23%).
Bilateral and symmetrical hypoperfusion was found in two patients (patients 13 and 15). Bilateral and asymmetrical hypoperfusion with left-side predominance was found in one case (patient 6).
When hypoperfusion involved the left hemisphere, four (FTOP; n = 7) and three (TPO; n = 3) lobes were affected. When hypoperfusion involved the right hemisphere, four (FTOP; n = 1), two (TO; n = 1) and one (P; n = 1) lobes were affected.
CBF values
The mean CBF value in the visually hypoperfused lobes was 40.3 ± 6.4, 44.9 ± 8.4, 45.0 ± 9.5 and 35.9 ± 6.5 mL/100g/min in the frontal, temporal, occipital and parietal lobes, respectively. The mean CBF value in the normally perfused lobes was 53.6 ± 13.1, 59.5 ± 12.1, 59.9 ± 9.5 and 51.9 ± 9.5 mL/100g/min in the frontal, temporal, occipital and parietal lobes, respectively. There was a significant difference between all the lobes with P = 0.036, P = 0.008, P = 0.005 and P < 0.001, respectively (Figure 1).
Box plot of CBF values in the frontal, temporal, parietal and occipital lobes. There is a significant difference (P < 0.05) between mean CBF in hypoperfused and normally perfused lobes assessed visually on ASL perfusion maps.
The mean difference in CBF values when asymmetrical cerebral perfusion was observed by visual assessment was 32.7 ± 8.6, 30.2 ± 9.2, 28.8 ± 12.7 and 28.4 ± 11.9% in the frontal, temporal, occipital and parietal lobes, respectively. The mean difference in CBF values when there was a symmetrical perfusion map on visual assessment was 6.8 ± 2.7; 7.0 ± 4.2; 11.6 ± 10.3 and 7.4 ± 3.8% in the frontal, temporal, occipital and parietal lobes, respectively. There was a significant difference between all the lobes with P < 0.001, P = 0.001, P = 0.044 and P = 0.003, respectively.
TOF-MRA
Finally 9/17 (53%) MRI examinations had a higher acquisition box and the findings were all abnormal. All normal TOF-MRA (5/17, 30%) images had standard acquisition box placement which did not allow visualisation of the distal arterial branches.
Twelve TOF-MRA images displayed abnormalities (12/17, 71%). These were vasospasm with decreased visualisation of the peripheral branches in all cases. Neither stenosis nor occlusion was found.
The arteries affected were both MCA and PCA in seven children (7/12, 58%) and MCA or PCA alone in three children (3/12, 25%). There was bilateral involvement (PCA or/and MCA) in two children (2/12, 17%).
ASL/TOF-MRA mapping
Perfusion map abnormality (16/17) was greater than TOF-MRA abnormality (12/17). All vascular abnormalities found on the TOF-MRA sequence matched the homolateral hypoperfusion on the ASL sequence (12/16, 75%).
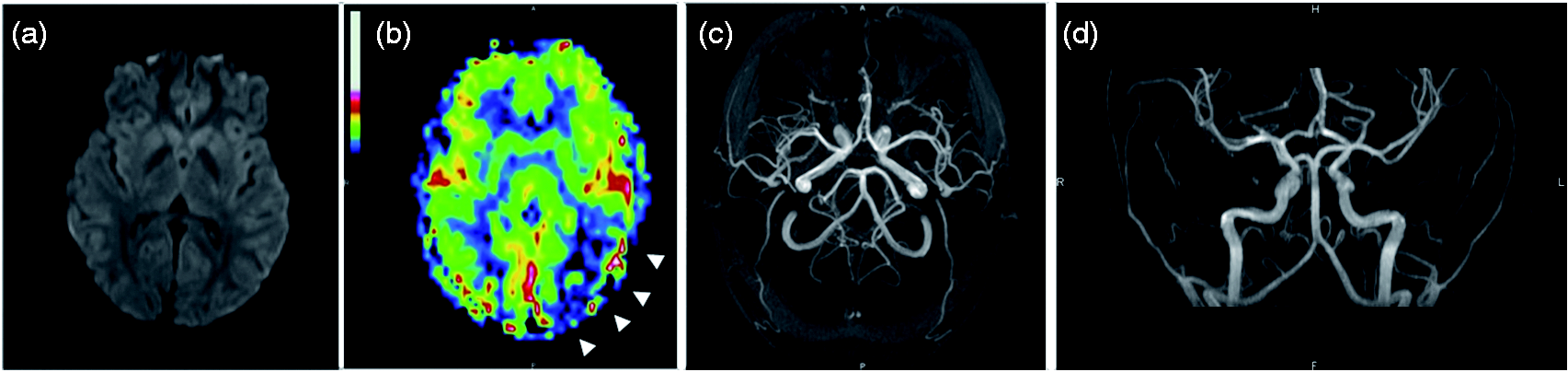
Representative hypoperfusion and homolateral vasospasm are shown in Figure 2. An example of an abnormal perfusion map and normal TOF-MRA image is shown in Figure 3.
A 13-year-old girl presenting with acute right sensory deficit, dysarthria and decreased level of consciousness. Initial MRI (a–d) performed 9 h 16 min after the onset of aura shows no ischemia on DWI (a), left hypoperfusion in the frontal, temporal, parietal and occipital lobes (arrow heads) on the ASL perfusion map (b), and vasospasm of the distal branches of the left middle cerebral arteries and posterior cerebral arteries (ellipse) on axial (c) and coronal (d) 3D TOF-MRA. Follow-up MRI (e–h) performed one day later shows no ischemia on DWI (e), normalisation of the ASL perfusion map (f), and resolution of vasospasm (ellipse) on axial (g) and coronal (h) 3D TOF-MRA. A 14-year-old boy presenting with acute dysarthria and right sensory deficit. Initial emergency MRI shows no ischemia on DWI (a), left temporo-occipital hypoperfusion (arrow heads) on the ASL perfusion map (b), and no vascular abnormalities on axial (c) and coronal (d) 3D TOF-MRA. Note that the low position of the TOF-MRA box does not allow visualisation of the distal intracranial arteries, meaning that distal vasospasm may go underdiagnosed in this case.
MRI/clinical mapping
Among the children presenting with focal neurological deficit (15/17), contralateral hypoperfusion was seen on the perfusion maps in 14/15 cases. One child had a normal perfusion map (patient 7), but we note that for this patient’s MRI was performed two days after the onset of symptoms. One patient (patient 13) had bilateral temporo-occipital hypoperfusion but presented with left sensory-motor deficit.
Among the two patients with no focal neurological deficit, one (patient 8) had visual symptoms and right hemisphere hypoperfusion, and the other (patient 15) had a decreased level of consciousness with a Glasgow Coma Scale score of 13 and diffuse symmetrical cerebral hypoperfusion with diffuse vasoconstriction on TOF-MRA.
Clinical symptoms and MR imaging findings. Frontal (F), Temporal (T), Parietal (P), Occipital (O), Left (L), Right (R), middle cerebral artery (MCA), posterior cerebral artery (PCA), distal branches (distal), vasoconstriction (VC), nausea (N), vomiting (V).

Follow-up MRI
Four patients underwent follow-up MRI 27–74 h after the onset of neurological symptoms. No perfusion abnormality was detected on imaging in three out of four cases and the TOF-MRA images were normal in all four cases. Representative normal follow-up MRI is shown in Figure 2.
For one child (patient 3, time = 28 h), visual hyperperfusion was observed in the cerebral regions that were initially hypoperfused (four lobes).
Discussion
The main finding of our study was that all but one (94%) child presented with regional cerebral hypoperfusion on MR perfusion imaging with ASL, which was frequently associated (75%) with a minor vasospasm of the intracranial arteries on the TOF-MRA images. To our knowledge, this is the first study in the literature to report both perfusion abnormalities and angiography findings in children with atypical aura.
ASL is a non-invasive perfusion technique which does not require injection of contrast material, unlike DSC PWI. In recent years, the technique has therefore become more widely used for paediatric patients. In addition, it offers the capability to quantify absolute CBF. In our study, the mean CBF values were significantly different between the visually hypoperfused and normally perfused lobes.
The pathophysiology of migraine with aura is still poorly understood. The neuronal theory, introduced by Lashley and Leao in 1946 (16,17) describes a phenomenon known as cortical spreading depression (CSD), triggered by initial cortical hyperexcitability and moving from the posterior (occipital) regions to the anterior regions. Vascular phenomena have been shown with different perfusion imaging techniques in adults, in connection with CSD (6,18). In 1990, Olesen demonstrated parieto-occipital oligemia using an intra-carotid xenon-133 injection triggering migraine aura and similar to cortical spreading depression. More recently, blood-oxygenation-level-dependent (BOLD) functional imaging confirmed the association of a BOLD signal decrease initiated in the occipital cortex contralateral to the aura but without reaching the ischemic threshold (19). This phenomenon is characterised by anterior propagation limited by fissures, not confined to vascular territories, and similar to the cortical spreading depression described by Leao. Perfusion changes vary over time and hypoperfusion tends to occur during the aura phase and hyperperfusion during headache, although the headache phase can begin during hypoperfusion. These abnormalities can vary greatly from one patient and generation to another.
In our study, MRI was performed very early on (median time of 6 h after the onset of neurological symptoms) which probably explains why the abnormalities observed were always hypoperfusion. The only MRI examination with no perfusion abnormality was performed two days after the onset of aura, and we may assume that the abnormalities had already regressed. Hyperperfusion observed on the control scan of one child was consistent with the literature data. This late hyperperfusion was shown in several studies within 17–72 h after the onset of symptoms (10,20,21).
Moreover, we observed that the perfusion abnormalities were predominant in the posterior (parieto-occipital) regions, without being confined to a particular vascular territory, and were mostly asymmetrical. The data are similar to the findings for DSC PWI in adults (7,8) and concordant with the theory that CSD begins in the posterior (occipital) region with anterior propagation of the cortical depression broken up by fissures. This distribution of perfusion abnormalities is also consistent with the clinical data reported for adults (22), in that the symptoms are most often unilateral, with a visual impact in the majority of cases. Lastly, in our study we noted that the perfusion abnormalities mostly affected the left hemisphere, and that when the right hemisphere was affected it was to a lesser extent. To our knowledge, left predominance of symptoms is not reported in the literature.
Cerebral perfusion abnormalities are generally due to a vascular abnormality. A study on adult patients with migraine without aura revealed a discreet intracranial vasodilation (23). These data concord with the hyperperfusion phenomenon described during the headache phase. Data regarding MRA findings in migraine with aura are sparse and limited (8,12). Safier et al. report the largest series in the literature which includes eight children with suspected acute hemiplegic migraine. They found vasospasm within the middle cerebral artery branches during attacks in seven patients which had resolved on follow-up imaging. No concomitant perfusion imaging was performed in the study. Our findings corroborate this study and show that the vasospasm observed is associated with homolateral hypoperfusion in ASL, in line with the clinical findings. To our knowledge, there are only two literature reports describing concomitant perfusion and angiographic abnormalities. Forster et al. (8) reported side-to-side differences in peripheral branch visualisation in seven of 33 adult patients with migraine with aura. The patients in their study had undergone MRI for suspected stroke. We may therefore assume that TOF box placement was slightly low as in the standard stroke protocol, and that abnormal TOF findings had been underestimated. In their case report, in addition to cerebral hypoperfusion Kim et al. described homolateral vasoconstriction of the distal branches of the MCA and PCA (24).
This study therefore contributes to a positive diagnosis of migraine with aura. The normalisation of follow-up MRI (perfusion and TOF-MRA) performed on some of our patients confirmed the transient nature of these alterations related to migraine aura. The rest of the morphological imaging should of course be normal in the acute phase, notably the diffusion imaging. In addition, clinical data are also essential for ruling out a different diagnosis. Particularly with vascular abnormalities in a patient with headaches, the main differential diagnosis is reversible cerebral vasoconstriction syndrome (RCVS). The onset of headaches is generally sudden (peak intensity within 1 min) and they can persist for one to four weeks. Besides the clinical data, the angiographic data are also different. Angiography generally shows diffuse bilateral vasoconstriction, with clear irregularities in size presenting as segmental narrowing and dilation (‘strings of beads’ or ‘sausage-string pattern’) (25,26).
On retrospective review of the clinical history of all our patients, we noticed that 30% of children had suffered a head injury between six months and nine years previously, requiring admission to the paediatric emergency department of our institution. A brain scan had been indicated and the result was normal for all children. This may be a coincidence but the literature reports a link between migraine and children with juvenile head trauma syndrome (JHTS) (27). CSD shows similar involvement in the JHTS and migraine mechanisms according to Haas (28). To our knowledge, no previous study has revealed MRI abnormalities in children suffering from JHTS during the acute phase to be similar to those observed during migraine aura, thereby confirming the theory of a common CSD mechanism.
Our study has certain limitations. The first is its retrospective, single-centre design and small sample size (17 children). This relates to the fact that MRI is not systematically indicated in the diagnostic workup of migraine patients in an emergency setting. It was the first attack for 11 children, and/or aura symptoms were particularly intense, which is one reason why MRI was performed. The ICHD criteria were therefore not fully met as regards the number of attacks. However, the final diagnosis was probable migraine.
TOF-MRA acquisition box position variability is a limiting factor. The five TOF-MRA sequences described as normal were systematically TOF-MRA sequences with a too low standard position not allowing adequate visualisation of the distal arterial branches. It is therefore likely that distal vascular abnormalities were underestimated in these children. This observation helped us to adapt our practice, however, and for investigation of headache in particular, we now use an acquisition box in an intermediate position to allow correct visualisation of the circle of Willis as well as the distal arterial branches.
The absolute CBF values cannot be interpreted alone due to inter-subject variations, which depend on age and brain regions (29), preventing a normal or abnormal perfusion cutoff value from being defined. In addition, there may be bilateral perfusion abnormalities that prevent intra-subject comparison. Visual analysis of perfusion mapping is therefore essential. Besides, this is the analysis method used in daily clinical practice.
Conclusion
The association of perfusion abnormalities on ASL in brain regions not corresponding to vascular territories, and vascular vasospasm in TOF-MRA, can aid diagnosis of migraine with aura. These two sequences are totally non-invasive and easy to perform in routine practice in children and can provide additional information for understanding the pathophysiology of this complicated disorder.
Clinical implications
ASL is a totally non-invasive cerebral perfusion technique that can be easily used in addition to conventional MRI to establish a positive diagnosis of migraine with aura in children. Migraine with aura is typically characterised by cerebral hypoperfusion that is not limited to a specific vascular territory, is often asymmetrical and preferentially affects the posterior brain regions. TOF-MRA can show slightly reduced visualisation of the distal arterial branches relating to a vasospasm. In routine practice, when investigating neurological deficit with headache, the TOF acquisition box can be positioned slightly higher than the standard position to improve visualisation of the distal arterial branches. The association of hypoperfusion on ASL and vasospasm in the distal branches of the intracranial arteries on TOF-MRA can aid MRI diagnosis of migraine and supports the neurovascular theory of migraine with aura.
Footnotes
Acknowledgements
The authors thank Mrs Tracey Westcott for her editorial assistance, and Mr Geoffrey Groussard for his statistical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
