Abstract
Background
Migraine is a well-known feature of mitochondrial disorders (MDs). However, no systematic epidemiological data are available in large populations of patients.
Aims
The aim of this cross-sectional cohort study was to describe the prevalence and migraine characteristics in a large cohort of patients with mitochondrial encephalomyopathies.
Methods
We studied 93 consecutive patients with characterised MDs referred to our Neuromuscular Unit during a 12-month period. All patients (age range = 16–78 years; 31 men; 58 progressive external ophthalmoplegia [PEO], 12 myoclonic epilepsy with ragged red fibres [MERRF], eight mitochondrial encephalomyopathy, lactic acidosis and stroke-like episodes [MELAS], two mitochondrial neurogastrointestinal encephalomyopathy [MNGIE] and 13 other MDs) underwent a structured diagnostic headache interview using an operational diagnostic tool following the IHS criteria. If they met the criteria for migraine, they were included in the ‘Migraine Group’. The other patients were counted in the ‘No Migraine Group’. Patient demographic and migraine characteristics were examined. Clinical, neuroradiological and neurophysiological data were compared between groups.
Results
Migraine was reported in 35.5% of patients. Migraine without aura was the most common headache (81.8%). The migraine group showed younger age (P < 0.01), increased prevalence of epilepsy (P = 0.01), myoclonus (P = 0.03), stroke-like episodes (P = 0.03) and decreased prevalence of muscle weakness (P < 0.01). Multivariate analysis showed that migraine was positively associated with absence of muscle weakness (P = 0.04) and presence of EEG abnormalities (P = 0.02).
Conclusion
Migraine has a higher prevalence in MDs compared with general population-based data, independently from genotype or phenotype. Migraine is not merely a phenotypic aspect of specific MDs but is rather the expression of vulnerability of the central nervous system, probably directly related with defects of the respiratory chain.
Keywords
Introduction
Mitochondrial diseases (MDs) are a heterogeneous group of genetic disorders characterised by primary defects in mitochondrial oxidative phosphorylation, the main source of cellular adenosine triphosphate (ATP), under a dual genetic control with mitochondrial DNA (mtDNA) and nuclear DNA (nDNA) working in concert. Mitochondria are ubiquitous and MDs may present with a multitude of clinical features in different combinations, and tissue with high dependence on oxidative metabolism, including the brain, eye, skeletal muscle and heart, are the most frequently involved (1,2). Particularly, the central nervous system (CNS), metabolically very demanding, is consequently vulnerable to defects of the mitochondrial respiratory chain. As a result of bioenergetics defects, the range of CNS manifestations of MDs is broad, including migraine, stroke-like episodes (SLEs), dementia and epilepsy (3,4). Morphological, biochemical and neuro-radiological studies strongly support a close connection between mitochondria and migraine (5). Furthermore, migraine or migraine-like headaches are a prominent feature of mitochondrial disorders. For example, in the syndrome of mitochondrial encephalomyopathy, lactic acidosis and stroke-like episodes (MELAS), most commonly due to the m.3243A > G point mutation, patients suffering repeated episodes of migraine is one of the most frequent symptoms (6,7). Headache is sometimes the only clinical feature in oligosymptomatic maternal relatives of patients with this pathology (8). In other syndromes, such as myoclonic epilepsy with ragged red fibres (MERRF), usually linked to the m.8344A > G point mutation, the differences in prevalence across studies (from 8.8% to 52%, respectively, in Italian and German cohorts of patients) can be largely explained by the absence of a structured diagnostic headache tool (9–11). In fact, no systematic headache epidemiological data are available in large populations of MDs. In this paper, we present a large-scale cross-sectional cohort study on prevalence and characterisation of migraine in adult patients with genetically defined MDs.
Participants/Materials and methods
Patient population
Clinical symptoms and signs, morphological, biochemical, radiological and neurophysiologic findings in the ‘Migraine Group’ (MIGR+).

SLE, stroke-like episodes; WM, white matter abnormalities; Cortical/Cerebellar A., cortical/cerebellar atrophy; abn, abnormalities; CK, creatine kinase.
Clinical symptoms and signs, morphological, biochemical, radiological and neurophysiologic in the ‘No-Migraine Group’ (MIGR–).

SLE, stroke-like episodes; WM, white matter abnormalities; Cortical/Cerebellar A., cortical/cerebellar atrophy; abn, abnormalities; CK, creatine kinase.
Procedure and analysis
All MD patients were interviewed for characteristics of recurrent head pain and accompanying symptoms of headache by means of a structured diagnostic headache questionnaire and underwent a neurological examination using the operational diagnostic tool based on the IHS criteria (III beta) (12). If they met the criteria for migraine (at least five attacks during the lifespan, fulfilling the criteria for ‘migraine without aura’ and at least two attacks fulfilling criteria for ‘migraine with aura’) (12), they were included in the ‘Migraine Group’ (MIGR+). Conversely, the patients without recurrent migraineous attacks were recorded in the ‘No Migraine Group’ (MIGR–). Both neurologists confirmed the diagnosis of migraine, but only the migraine specialist administered the structured diagnostic questionnaire and evaluated the clinical diaries. Migraine was not taken in account when only temporally and exclusively correlated with SLEs. Patients were routinely assessed for muscle weakness (weakness in the neck, upper and lower limbs), according to the Medical Research Council (MRC) criteria (13). Furthermore, brain magnetic resonance imaging (MRI) and EEG were performed in all participants during the interictal migraine period.
Written informed consent was obtained from each participant. The present study was performed in agreement with the Declaration of Helsinki and was approved by the local Ethics Committee.
Accompanying symptoms in the last three months (as reported in patient’s clinical diary), frequency of attacks and drugs used in the ‘Migraine Group’ (MIGR+).

Patients with migraine with aura are reported in bold.
1, present; 0, absent; NSAIDs, non-steroidal anti-inflammatory drugs.
We finally analysed and compared clinical, neurophysiologic and neuroradiological data between the MIGR+ and MIGR– groups.
Statistical analyses
Numeric variables (age, age of onset, lactate at rest, lactate after exercise, creatine kinase [CK]) were compared by means of one-way analysis of variance Mann–Whitney U-test. Nominal variables (sex, muscle weakness, epilepsy, myoclonus, SLEs, white matter lesions, cerebellar atrophy, cortical atrophy, EEG abnormalities) were analysed and compared between MIGR+ and MIGR– by means of 2 × 2 contingency tables using the Chi-square or Fisher’s exact test. Contingency tables were also used to compare the relative frequency of migraine in different phenotypes and genotypes. Multivariate analysis was performed to identify risk factors of migraine. The level of significance was set to P < 0.05. Statistics were performed by means of the SYSTAT 12 software version 12.02.00 for windows (SYSTAT® Software Inc. 2007).
Results
Clinical findings
Clinical, morphological–biochemical and/or genetic study allowed a definite diagnosis of MDs in all cases. Patients were classified in subgroups according to the specific phenotype: eight MELAS, 12 MERRF, 58 PEO, two MNGIE; 13 patients did not meet the clinical criteria to be included in the above categories and were classified as ‘other MDs’.
Laboratory evaluation
Forty-nine patients had increased levels of serum CK, in the range of 2× –10×. Lactic acidosis at rest (lactate levels > 2.2 mmol/L) was observed in 41 patients and abnormal levels of lactate after exercise in 60 patients.
Co-morbidities, neuroradiological and neurophysiologic evaluation
Co-morbidities
The other main CNS manifestations were the following: epilepsy (18/93, 19.4%); myoclonus (15/93, 16.1%); and SLEs (10/93, 10.8%) (Tables 1 and 2).
MRI findings
Fifty-four of the 93 (58.1%) patients enrolled in the study had white matter lesions, 14 (15.1%) had cerebellar atrophy and 35 (37.6%) had cortical atrophy (see Table 1 for details).
Neurophysiologic findings
In 47 out 93 patients (50.5%) EEG was abnormal, mainly consisting of diffuse slowing of background activity (Tables 1 and 2).
Migraine prevalence and characteristics
Ninety-three patients (31 men) were enrolled in the present study. The mean age was 51.3 ± 16.2 years (age range = 16–78 years). Clinical data are summarised in Tables 1 and 2. The distribution of the migraine in all specific phenotype is reported in Figure 1a. The distribution of the migraine in all specific genotypes is reported in Figure 1b. Of the 93 patients enrolled, we identified 33 participants (35.5%) with migraine (MIGR+) and 21 out of 33 (63.6%) were women. When we evaluated the migraine prevalence estimated by sex, we found similar values in men (12/31; 38.7%) and women (21/62; 33.9%).
(a) Histograms showing the distribution of the migraine in all phenotypic groups of MDs. (b) Histograms showing the distribution of migraine between different genotypic groups of MDs; multiple mtDNA deletions include patients with POLG1 and TYMP mutations. sDel mtDNA, single mtDNA deletion; mDel mtDNA, multiple mtDNA deletions.
Most MIGR+ patients fulfilled criteria for migraine without aura (81.8%; 27/33) and presented unilateral pain (n = 22, 66.7%), frontal localisation of pain (n = 19, 57.6%), pulsating or throbbing pain (n = 18, 54.5%). Migraine was worse by physical activity in most patients (60.6%; 20/33). Vomiting was experienced by 18.2% (6/33), nausea by 48.5% (16/33) and photophobia or phonophobia (or both) by 69.7% (23/33) of patients. Twenty patients (60.1%) reported habitual severe attacks. Twenty-five out of 33 MIGR+ (75.8%) had a relevant impairment due to migraine in the last three months. The mean monthly frequency was 3.9 ± 6.3 attacks per month.
Nonsteroidal anti-inflammatory drugs (NSAIDs) were given in 45.5% (n = 15) of the MIGR+ group. The other symptomatic drugs were prescribed as follows: Acetaminophen in 10 (30.3%); Triptans in three (9.1%); other (i.e. corticosteroids, other analgesics, combined drugs, etc) in two (6.1%); no medication in eight patients (24.2%).
Characteristics of migraine attacks and accompanying symptoms are summarised in Table 3.
Comparison between groups
Patients fulfilling criteria for migraine (MIGR+ group) were younger (MIGR+ group = 45.5 ± 17.2 years; MIGR– group = 54.5 ± 14.8 years; P = 0.007).
As far as ‘severity measures’ for mitochondrial impairment ‘lactate at rest’, ‘lactate after exercise’ and ‘CK’ tend to be higher in the MIGR+ group (lactate at rest: MIGR+ group = 2.8 ± 2.6; MIGR– group = 1.9 ± 0.9 years; P = 0.332; lactate after exercise: MIGR+ group = 6.7 ± 3.6; MIGR– group = 6.2 ± 3.3; P = 0.486; CK: MIGR+ group = 389.5 ± 516.7; MIGR– group = 228.3 ± 194.6; P = 0.438), but none of these parameters reaches statistical significance.
In terms of co-existent medical conditions, in the MIGR+ group the number of patients with epilepsy (MIGR+ group = 11/33; MIGR– group = 7/60; P =0.0103), myoclonus (MIGR+ group = 9/33; MIGR– group = 6/60; P = 0.0406) and SLE (MIGR+ group =7/33; MIGR– group = 3/60; P = 0.0290) was significantly higher than in MIGR– group. Furthermore, abnormal EEG findings were significantly more frequent in the MIGR+ group than in the MIGR– group (MIGR+ group = 20/33; MIGR– group = 22/60; P = 0.0136), in particular epileptic focal abnormalities (MIGR+ group = 12/30; MIGR– group = 11/58; P = 0.0425). By contrast, the proportion of patients without muscle weakness was significantly higher in the MIGR+ group than in the MIGR– group (MIGR+ group = 11/33; MIGR– group = 9/60; P < 0.0001).
Contingency analysis showed no differences between different MD phenotypes (P = 0.259) (Figure 1a) or genotypes (P = 0.548) (Figure 1b) in terms of prevalence of migraine.
Multivariate analysis
Multivariate analysis showed that the absence of muscle weakness (confidence interval [CI] = 1.030–12.629; P = 0.045) and the presence of EEG abnormalities (CI = 1.183–11.478; P = 0.024) were significantly associated with the presence of migraine (Figure 2).
Forest plot showing the odds ratio and direction of association between migraine and other clinical conditions. Multivariate analysis showed that the absence of muscle weakness and the presence of EEG abnormalities were significantly associated with the presence of migraine.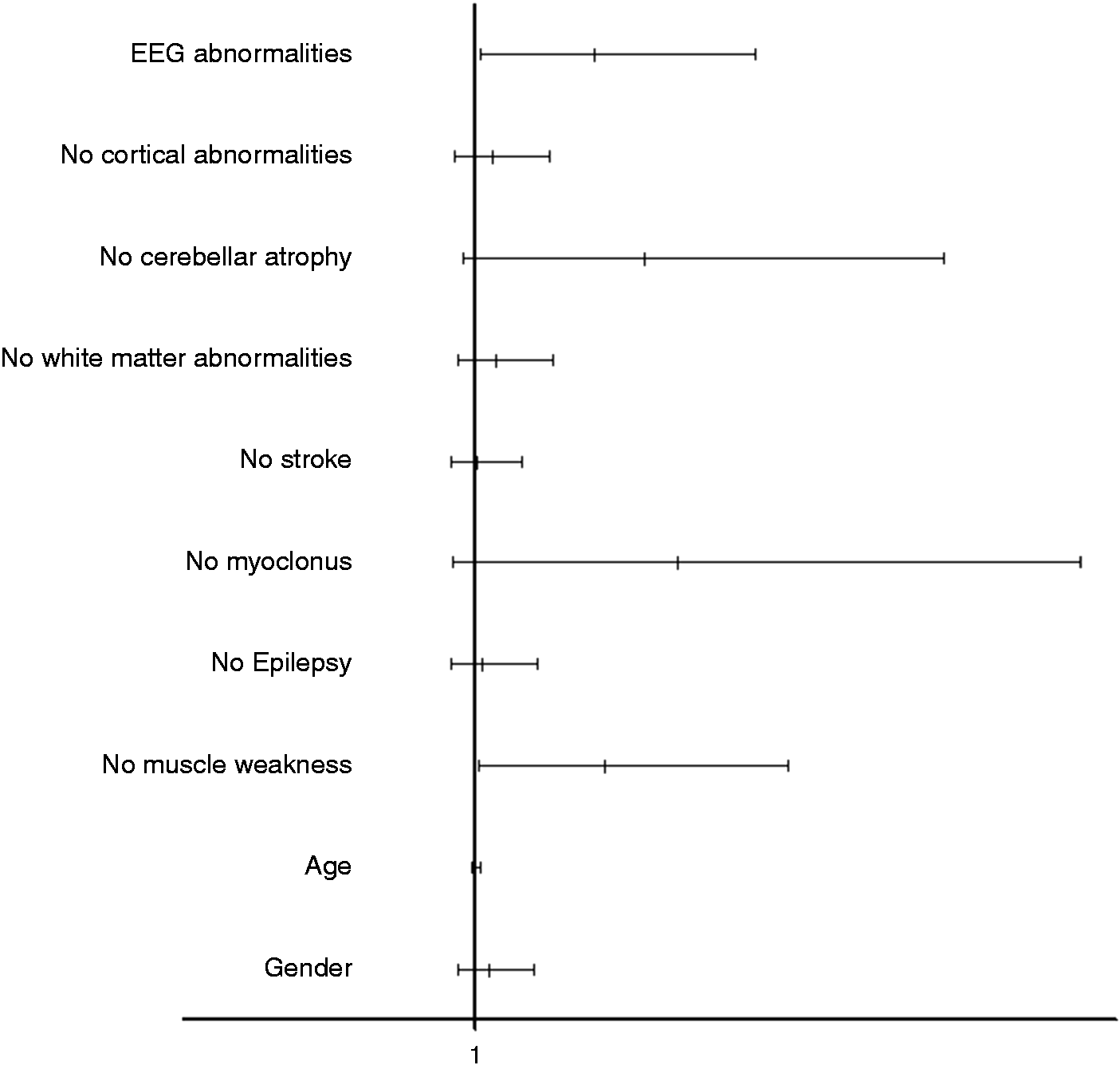
Discussion
We present a large-scale cohort study on prevalence and characterisation of migraine in adults with genetically defined MDs. A mitochondrial dysfunction is often associated with pathology affecting the CNS. Neurons have special metabolic needs and require ATP for a number of energy-consuming processes: control of membrane potential by the Na+/K+ ATPase pump, regulation of intracellular Ca2+ and exocytosis/recycling of synaptic vesicles (14). The range of CNS manifestations of MDs is broad and migraine is the most frequent CNS-related clinical feature in our cohort. Much biochemical, histological, molecular and neuroradiological evidence strongly supports the hypothesis that mitochondrial dysfunction plays a significant role in the pathogenesis of migraine (15). In current literature, the prevalence of migraine in patients affected by MDs varies considerably. This discrepancy may be partly due to the non-use of specific questionnaires, interviews and clinical examination. In the present study, instead, the diagnosis of migraine relied on two evaluators one of which conducted a structured diagnostic headache interview using an operational diagnostic tool following the IHS criteria.
The overall prevalence of migraine in our population is 35.5% that is approximately double the prevalence reported in general population-based studies (16).
Another interesting observation in our cohort is the prevalence of migraine when the patients are divided by sex: in fact, in men it is 38.7% (12/31), much higher than in all studies that analysed the prevalence for sex (6%) (17). Moreover, the prevalence is almost the same for men and women. This is very different from the general adult population in which 18% of women and 6% of men had at least one migraine attack with a typical ratio of 3:1 in favour of women (17).
More relevant and unexpected data are that the prevalence of migraine is high not only in clinical conditions classically considered associated with migraine (i.e. MELAS) but also in MERRF and PEO as well as in all other mitochondrial patients. Although the present study is relatively under-powered to find statistical differences by phenotype and our data must be confirmed in a larger population study, in our cohort the prevalence is in fact >30% in all MD groups without major differences in terms of distribution, considering both phenotypes or genotypes (Figure 1).
Based on these findings, it is possible to hypothesize that migraine is not merely a phenotypic aspect of specific forms of MDs but is a common expression of vulnerability of the CNS to defects of the mitochondrial respiratory chain.
Further, we analysed the association of migraine with other clinical manifestations of CNS involvement, such as epilepsy, myoclonus and SLE.
Some studies report a higher prevalence of migraine in epileptics and a higher prevalence of epilepsy in migraine sufferers. However, a recent controlled cross-sectional study (18) did not confirm the notion of an association between primary forms of these two conditions. Therefore, the finding of a higher prevalence of epilepsy in the population of mitochondrial migraineurs (33.33%; 11/33) compared with non-migraineurs (11.66%; 7/60) presumably means that the association between migraine and epilepsy is not fortuitous in these patients, but could underlie common pathophysiological mechanisms. In fact, the mitochondrial oxidative phosphorylation has a pivotal role in the generation of the ATP needed for synaptic transmission and firing of action potentials, and many of the genetically determined epileptic syndromes associated with mitochondrial dysfunction originate from pathogenic mutations in the mitochondrial genome (19).
There are no comparative data on the prevalence of myoclonus in patients with migraine or vice versa, but the highest prevalence of this clinical sign in mitochondrial migraineurs may be justified in the higher prevalence of epilepsy and of all phenomena of increased excitability of the CNS. Accordingly, the incidence of EEG abnormalities in the MIGR+ group is higher than in the MIGR– group. Interestingly Purkinje cells, which lead the inhibitory GABA transmission, seem to have a special vulnerability in MERRF patients with the m.A8344G mutation (20), the phenotype most frequently associated with migraine in our cohort.
It is well-known that headache is strongly associated with SLEs often preceding these events (21). An interesting theory proposed to explain headache attacks associated with the SLEs is that pericapillary plasma extravasations due to mitochondrial angiopathy lead to the activation of the first division of the trigeminal nerve fibres innervating the small vessels around the stroke-like lesion (22). Other studies highlighted that plasma protein extravasation is no longer considered a likely source of pain in migraine but that neurogenic vasodilatation, the other physiological component of neurogenic inflammation, could play a role in migraine pathophysiology (23).
Some studies report a higher prevalence of cerebrovascular disease in migraineurs participants, but usually only migraine with aura is considered a risk factor and is predominantly in women (24). In this regard, the very high percentage of male migraineurs compared with the general population is very surprising (17).
Finally, to better define the differences between mitochondrial patients with and without migraine, we performed a multivariate analysis showing that migraine is statistically associated with the presence of EEG abnormalities and the absence of muscle weakness (Figure 2). Moreover, the patients in the migraine group are younger compared with patients without migraine (MIGR+ group = 45.5 ± 17.2 years; MIGR– group = 54.5 ± 14.8 years; P = 0.007) and have lower muscle impairment (MIGR+ group = 22/33; MIGR– group = 9/60; P < 0.0001). These findings could suggest that migraine may be an early manifestation of the MDs forms characterised by prevalent CNS involvement consistent with an altered brain excitability due to mitochondrial dysfunction. On the other hand, we have also to take into account that migraine prevalence decreases with age in people who do not have clear MD. In fact, it has been speculated that the migraine response might be impaired by decreased capacity for vasodilation due to endothelial dysfunction and atherosclerosis (25) or by decreased functioning of brainstem dopaminergic circuits (26).
Endothelial dysfunction and extrapyramidal involvement, however, may be present in mitochondrial disorders due to the mitochondrial impairment independently from age, both in young and old patients. Thus, it is unlikely that these factors may significantly contribute to the age difference in the two groups.
Conclusion
There is a higher incidence of migraine in MDs when compared with the general population. The high prevalence is independent from sex, phenotype or genotype. Epilepsy, myoclonus and SLEs are more frequent in MD patients with migraine than in patients without migraine. Moreover, in our cohort, migraine is prevalent in younger patients, suggesting that it could represent an early manifestation of the disease. Our study supports the hypothesis that mitochondrial dysfunction may play a role in the pathophysiology of migraine. Assessment and complete characterisation of the headaches in mitochondrial disorders may orientate treatments to the needs of this fragile patient’s population.
Article highlights
The high prevalence of migraine in MDs is independent from sex, phenotype or genotype. Epilepsy, myoclonus and SLEs are more frequent in MDs patients with migraine than in patients without migraine. Migraine is not merely a phenotypic aspect of specific MDs but is rather the early expression of vulnerability of the CNS to dysfunction of the mitochondrial respiratory chain. Our study supports the hypothesis that mitochondrial dysfunction could play a role in the pathophysiology of migraine. Assessment and complete characterisation of the headaches in mitochondrial disorders may orientate treatments to the needs of this fragile patient’s population.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
