Abstract
Background
Fifty-three percent of adolescent girls report headaches at the onset of menses, suggesting fluctuations of ovarian hormones trigger migraine during puberty.
Aims
To determine if urinary metabolites of estrogen and progesterone are associated with days of headache onset (HO) or severity in girls with migraine.
Methods
This was a pilot study and included 34 girls with migraine balanced across three age strata (pre-pubertal (8–11), pubertal (12–15), and post-pubertal (16–17) years of age). They collected daily urine samples and recorded the occurrence and severity of headache in a daily diary. Urine samples were assayed for estrone glucuronide (E1G) and pregnandiol glucuronide (PdG) and the daily change was calculated (ΔE1G, ΔPdG). Pubertal development was assessed by age, pubertal development score (PDS), and menstrual cycle variance. The primary outcome measures were HO days and headache severity. Generalized linear mixed models were used, and included the hormonal variables and three different representations of pubertal development as covariates.
Results
Models of HO days demonstrate a significant age*PdG interaction (OR 0.85 [95% CI 0.75, 0.97]) for a 1 standard deviation increase in PdG and three-year increase in age. A separate model showed a significant PDS*PdG interaction (OR −0.85 [95% CI; 0.76, 0.95]). ΔPDG was associated with headache severity in unadjusted models (p < 0.017).
Conclusion
Age and pubertal development could moderate the effect of ovarian hormones on days of headache onset in girls with migraine.
Puberty represents a time period of significant changes in reproductive hormones for girls, with three different stages of development: thelarche (breast development), adrenarche (pubic hair growth), and menarche (onset of menses). Each stage of pubertal development is associated with a different milieu of reproductive hormones (1). Adrenarche and thelarche begin at 8–11 years of age, while menarche occurs at 12–13 years of age (2). Puberty has also been associated with the onset of a variety of pain disorders, including headache, temporomandibular joint disorder, and back pain (3–6).
Most studies generally report an increased prevalence of migraine in older adolescent girls compared to younger girls (7–10). The peak incidence of migraine occurs at 12–13 years of age for migraine with aura and 14–17 years of age for migraine without aura (11). While epidemiological studies suggest a relationship between pubertal development and migraine onset, the influence of ovarian hormones in this correlation is unknown.
Menstrually-related migraine (MRM) occurs in 35–50% of adult premenopausal women with migraine (12). The prevalence of MRM in adolescents has not been well established, but 53% of menarchal girls self-report a worsening of migraine at the time of menses (13). MRM has been defined as an attack of migraine without aura that occurs during a five-day time period encompassing the first day of menstrual bleeding and the two days preceding and following it. Attacks of migraine must repeatedly occur in two out of three menstrual cycles, but can also occur at other times of the month (14). Adult studies demonstrate that declining serum estrogen levels during peri-menstrual time periods trigger attacks of MRM (12,13,15–18). These results could suggest that fluctuations in ovarian hormones might trigger attacks of migraine not only in adults, but also in adolescent girls.
Based on the above data, we hypothesized that the stage of pubertal development would moderate the effect of ovarian hormones on attacks of headache in girls with migraine. The primary objective of this study was to determine if the absolute levels and/or the change in urinary estrogen and progesterone metabolites are associated with the headache onset (HO) days or severity of headache in pre-pubertal, pubertal, and post-pubertal girls with migraine before and after controlling for three different representations of pubertal development (age, pubertal development score, and cyclic hormonal variance).
Methods
Participants
Participants with headaches that met International Classification of Headache Disorder-II (ICHD-II) diagnostic criteria for migraine without aura, migraine with aura, or chronic migraine were recruited from the Cincinnati Children’s Headache Center between 10/1/11 through 11/1/12 equally, across three age strata: 8–11, 12–15, and 16–17 years of age, representing pre-puberty, puberty, and post-puberty. Exclusion criteria included: (1) girls currently on hormonal therapy; (2) the presence of medication overuse headaches; (3) females who were sexually active; (4) pregnant females; and (5) continuous daily headaches. Headache management and treatment was not altered due to study participation. The study was approved by the Cincinnati Children’s Hospital Medical Center Institutional Review Board.
Study procedures
At study initiation, subjects completed a standardized, semi-structured, headache interview and the Pubertal Development Scale, which is a five-item questionnaire on the characteristics of pubertal development. The pubertal development score (PDS) has been correlated with physician ratings of the Tanner scale (r = 0.61–0.67) and with drawings of pubertal development (r = 0.72–0.80) (19,20).
Each participant completed a daily headache diary, for at least 90 days, that included headache severity rated on a 0–10 point numerical pain scale and the presence or absence of nausea, vomiting, photophobia, and phonophobia. The daily headache diary was initiated irrespective of the first day of menstruation, because vaginal bleeding did not occur in all participants in the study and even when present was irregular in 46%. Daily urine samples were collected from their first morning void, frozen and stored in the participant’s freezer, and then returned “on ice” in a cooler. Parents of subjects confirmed that daily headache diaries and urine samples were completed.
Urinary levels of estrone-3-glucuronide (E1G; Estrone Glucuronide EIA kit) and pregnanediol-3-glucuronide (PdG; Pregnandiol Glucuronide EIA kit) were obtained through direct enzyme immunoassays using ELISA (EIA) kits (Immunometrics, UK). Urinary levels of these hormones closely parallel changes in serum levels (21). The kits contain 96 well plates that are coated with respective antibodies. The diluted urinary samples, standards or controls were added in duplicate into the appropriate wells on the coated plates, where E1G or PdG in the urine sample compete with a conjugate for antibody. The intraassay coefficient of variation was 6%, while the interassay coefficient of variation was 12%. Final results were corrected for creatinine excretion and reported as mcg/dl per ng/ml creatinine for E1G and mcg/cl per ng/ml creatinine for PdG.
Covariates and moderators
The independent covariates were: (1) daily measurements of E1G and PdG corrected with creatinine concentration and (2) E1G and PdG change scores (ΔE1G, ΔPdG), which were defined as the day n + 1 hormone values subtracted by the day n hormone values. All the hormone variables were phase shifted back one day, since the morning urinary collection represented hormonal events from the day before.
To examine the hypothesis that the association between the ovarian hormone predictors and headache activity were moderated by pubertal development, three different moderator candidates were examined (age, pubertal development using PDS, and menstrual cycle variance). The age of each participant at baseline was used as a continuous variable in the models. Menstrual cycle variance was defined as the percentage of variance in urine hormone values that could be attributed to regular menstrual cycle variation. To quantify this, we summed the spectral density of the variance that could be accounted for by cyclic fluctuations of urine hormone values with a period frequency of 24–35 days per cycle. This period frequency was chosen as it represents the typical duration of menstrual cycles encountered in adolescent girls (22). Higher percentages of menstrual cycle variance reflect more regular cycling, which is indicative of a mature menstrual cycle. Menstrual cycle variance was divided into the following three categories: <5%, 5–10% and >10%. For example, if the menstrual cycle variance is >10%, it significantly increases the probability of cyclic ovulatory menstrual cycles. If <5% of the variance of a participant’s hormonal measurements is associated with a menstrual cycle pattern, then 95% of the variance in this hormone is unaccounted for by regular cycling, and may simply be randomly varying day-to-day fluctuations.
Outcome measures
The primary outcome measures in the models were headache onset (HO) days, defined as days with an onset of a headache after observing a day without a headache, and headache severity. Outcome measures related to “headache” as opposed to “migraine” were chosen as the primary outcome measures, as treatment of an acute headache was allowed, thus leading to headache resolution prior to the development of migraine characteristics.
Data analysis/statistical analysis
No a priori sample size calculation was performed to guide sample size for this pilot study. To assure an adequate sample size, 40 subjects were recruited in order to have 30 subjects (10 per age strata) complete >80% of diary data and urine collections, providing at least 90 samples per subject for assessment of fluctuations in hormonal levels.
Differences across the three age groups were examined using the Kruskal-Wallis test for continuous data with skewed distributions, or the chi-squared test for categorical data. To examine the hypotheses, generalized linear mixed models were conducted with subjects as the unit of random effects. Random intercepts were utilized to account for the repeated measures (i.e., daily diaries) within subjects. Each model was assessed for best fit with the addition of random slopes (i.e., varying slopes across subjects) using likelihood ratio tests, but no model fit was improved so fixed slopes were employed. Daily pain reports from subjects who experience episodic migraine have several distinct properties. The within-person pain reports exhibit distributions with clumping at zero (reflecting no headache on most observation occasions), with a skewed distribution of non-zero pain reports (reflecting the less frequent occurrence of a migraine). For this reason, the existence of HO days using a binomial distribution (and logit link) was utilized, and the intensity of an attack (when present) was used with a normal distribution (with log link, if necessary) in two separate models for each predictor. To examine the moderator hypotheses, each of the three moderator candidates were entered in the model as main effects and interactions (e.g., moderator x PdG). A statistically significant interaction effect is interpreted as evidence that the levels of the moderator altered the relationship between the ovarian hormone and headache activity (e.g., older participants have stronger associations).
To facilitate interpretation of the models, the predictor candidates were re-coded to reflect meaningful units. For example, the ovarian hormone predictors were each coded so that the model estimates reflect a 1 SD change in hormonal value. For example, daily PdG levels varied across the entire sample by 19.9 mcg/cl, so the odds ratio for HO days is in relation to a day when PdG was 19.9 points higher than a reference day. The moderator candidates were also coded to reflect a 1 SD difference in the moderator candidates that corresponded to a three-year difference in age, four points on the PDS, and 3–4% on menstrual cycle variance. All analyses were performed using the r statistical package (R Core Team, Vienna, Austria). Where appropriate, all analyses were two-sided with no corrections made for multiple comparisons, p < 0.05. There were no imputations of missing data for any of the models because of the high completion rates for diary data and daily urine samples (see results section).
Results
Sample characteristics
Thirty-nine participants were initially randomized into the study. Five participants withdrew, leaving 34 participants that completed the study (12 in the 9–11 age stratum, 11 in the 12–15 age stratum and 11 in the 16–17 age stratum). A flow diagram of the study is shown in Figure 1. The participants contributed 3485 diary entries (mean: 102, range: 92–121 days), with no missing diary entries. We were able to collect and later assay E1G in the first void urine samples from 3353/3485 (96.2%) of the study days, while PdG was assayed on 3355/3485 study days (96.3%).
Flow diagram for the study.
Demographic characteristics.

E1G: absolute values of urinary estrone glucuronide (mcg/dl); PdG: absolute values of pregnandiol glucuronide (mcg/cl); ΔE1G: daily change of urinary estrone glucuronide (mcg/dl); ΔPdG: daily change of urinary pregnandiol glucuronide (mcg/l); NS: non-significant. Menstruation: the percentage of girls with one or more days of vaginal bleeding; Menstrual cycle variability for E1G and PdG: variance series accounted for by cyclic fluctuations of urine hormone metabolites ranging from 24–35 days; max: maximum; min: minimum. Differences across the three age groups were examined using the Kruskal-Wallis test for continuous data with skewed distributions, or the chi-squared test for categorical data.
E1G, PdG, and headache activity
Univariate associations for E1G and headache activity
A small, statistically non-significant association was found for E1G and HO days, OR: 0.97 (95% CI: 0.85–1.12), p = 0.70. Similarly, a small, statistically non-significant association was found for ΔE1G, OR: 0.93 (95% CI: 0.81–1.04), p = 0.19.
When examining the association between E1G and headache severity, a very small, statistically non-significant association was found for E1G and headache severity, B: 0.09 (95% CI: −0.06–0.23), p = 0.24, as well as ΔE1G, B: 0.09 (95% CI: −0.03–0.20), p = 0.14.
Univariate associations for PdG and headache activity
A very small, statistically non-significant univariate association was found for PdG and HO days, OR: 0.97 (95% CI: 0.86–1.09), p = 0.62, and ΔPdG and HO days, OR: 0.97 (95% CI: 0.87–1.07), p = 0.50.
When examining the univariate association between PdG and headache severity, a very small, statistically non-significant association was found for PdG and headache severity, B: 0.03 (95% CI: −0.10–0.17), p = 0.65. However, a statistically significant association was observed for ΔPdG, B: 0.14 (95% CI: 0.02–0.25), p = 0.017. The latter association corresponds to dubious clinical significance, for an increase/decrease of 7.1 SD of ΔPdG is associated with a ∼1 point on a 0 to 10 pain scale.
Variance in moderators of ovarian hormones and headache activity
The three moderator candidates were all associated with each other. Scores on the PDS scale increased with increasing age group (p < 0.0001), with six points separating the 8–11 year-olds and 12–15 year-olds, while the latter group exhibited only one point less than the 16–17 year-olds. The proportion of variance in the hormonal measurements that could be attributed to a 24–35 day menstrual cycle, which also increased steadily with increasing age (p < 0.001) (Table 1).
As can be seen in Figure 2, the younger participants exhibited variability in the hormonal cycles that was much less related to menstrual cycle variance, but with increasing age the variance in the hormones began to more closely resemble a 24–35-day menstrual cycle, especially for PdG.
Ovarian hormone levels during the observation period. The x-axis (study day) displays the 92–121 days of diary data contributed by the N = 34 participants in the study. The top panel is E1G levels and the bottom panel is PdG (y-axes). Each of the three age groups is displayed in rows, with the columns stratified by the proportion of variance in the cycle that could be accounted for by menstrual cycle variance (i.e., cycles that varied between 24 and 35 days). As expected, for PdG and E1G levels, age group was associated with increasing variance in the cycles attributable to menstrual cycle variation. The number of participants is shown in the upper left hand corner of each graph.
Examination of the moderators of ovarian hormones and headache activity
The moderating effect of age and PDS on the relationship between ovarian hormones and headache.

E1G: estrone glucuronide; PdG: pregnandiol glucuronide; PDS: pubertal development scale; ΔE1G: daily change in E1G; ΔPdG: daily change in PDG.
Generalized linear mixed models were used for the statistical analyses. The odds ratio represents the interaction between the hormonal and moderator covariates. The ORs were calculated based on a one standard deviation increase of the hormonal variables, a 3-year increase in age, four point increase in the pubertal development scale, and a 3–4% increase in menstrual cycle variance.
For HO models, 11/12 models exhibited an effect size in the direction that supported our initial hypothesis (all except E1G × menstrual cycle variance). It should be noted however that only two of these 12 models reached statistical significance. Specifically, as the maturity of the participant increased, the relationship between each of the four hormonal predictors and HO days was enhanced. The effect sizes ranged from OR: 0.85 to 0.97 reductions in the odds of experiencing a headache for each SD increase in the moderator candidates. For Age × PdG (OR: 0.85 [95% CI: 0.75, 0.97], p = 0.016), and PDS × PdG (OR: 0.85 [95% CI: 0.76, 0.95], p = 0.005), increases in PDS scores and age of the participant were both associated with higher levels of PdG being protective of HO days.
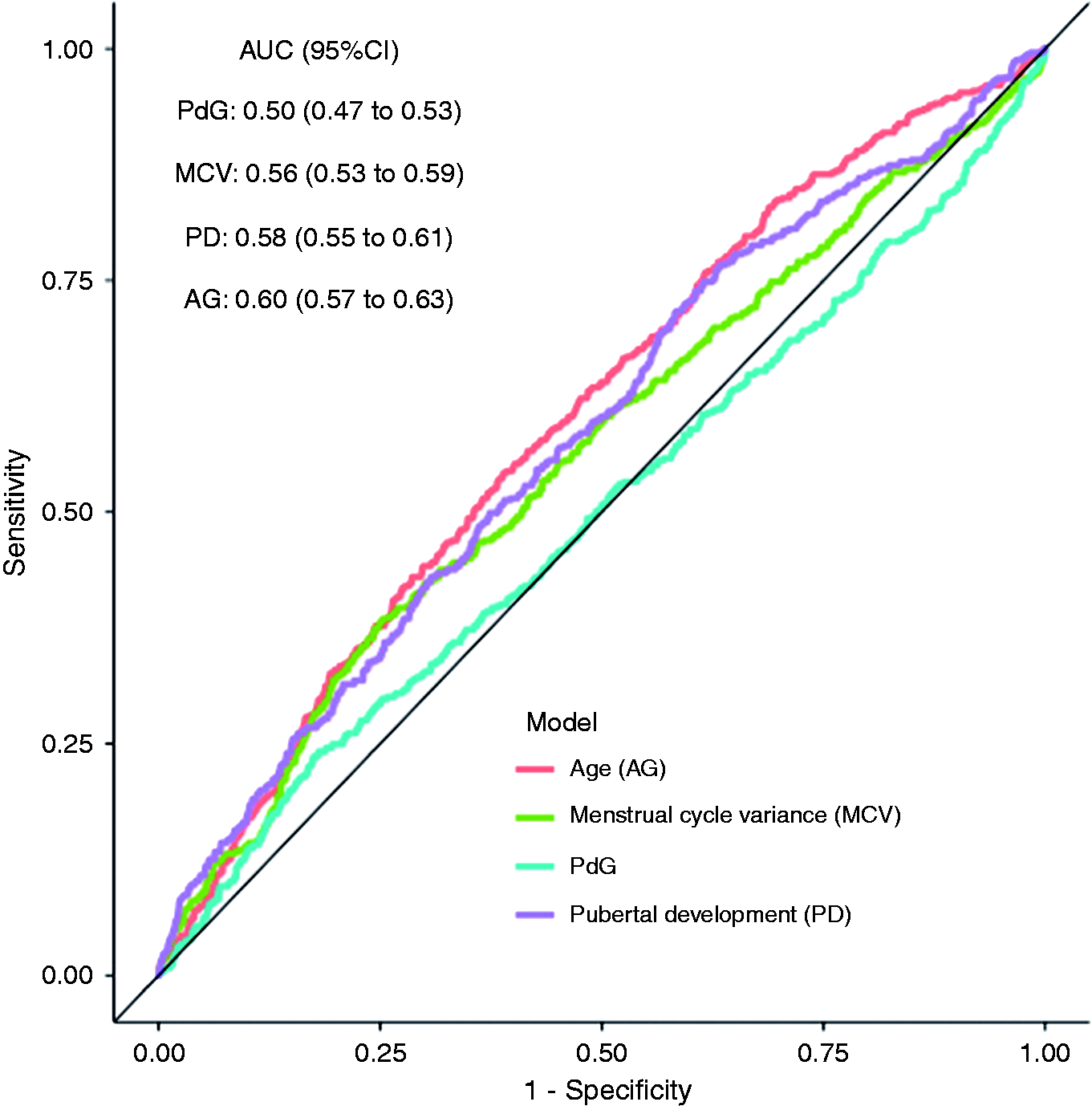
Figure 3 displays the associations between the four ovarian hormone predictors and HO days as a function of age group. Note that hormonal variables had their greatest effect in the 16–17 age group and little to no effect in the 8–11 and 12–15 age groups. Figure 3 displays the predictive power of the PdG models in the area under the curve (AUC) for each of the PdG models with each of the moderator candidates. As can be seen in Figure 4, each of the moderator candidates improves the predictive accuracy of PdG to a modest degree.
The association between ovarian hormones and probability of headache onset days. The x-axis (ovarian hormones) and the y-axis (predicted probability of headache) by age group. Each panel represents a different form of ovarian hormones, with the relationship between PdG and headache being significantly different by age level. The units for E1G were mcg/dl and for PdG were mcg/cl. Predictive accuracy of the PdG predictors using ROC curves. Each of the predictors had modest predictive utility, with the three moderator candidates each providing superior (p < 0.05) prediction to the PdG series alone, which did not exhibit better than chance prediction. The data support the hypothesis that the association between PdG and onset headache days increases with increasing maturity.
Discussion
The current study found that urinary levels of PdG are associated with HO days in pre-pubescent, pubescent, and post-pubescent girls with migraine. This association became evident after controlling for either age or pubertal development scores (PDS). Since age and the PDS were highly correlated (R = 0.88) these results suggest that the association between PdG with HO days is dependent upon the stage of pubertal development.
We chose headache onset days as our primary outcome measure as opposed to migraine onset days. Thus, it is likely that our primary outcome measure included attacks of both migraine and tension-type headache. This distinction, however, may be less important as some researchers believe that migraine and tension headache represent a continuum of migraine when they co-occur in the same individual. This is substantiated by the fact that attacks of migraine and tension headache both respond to triptans when they coexist in the same person (23).
Our interaction models predict an increased probability of HO days with higher levels of progesterone in girls aged 8–11 years of age, little to no effect at 12–15 years of age, and a decreased probability with higher levels in those 16–17 years of age. This hormone effect on HO days reverses from 12–15 years of age, representing the average age of menarche in the United States (e.g. 12.8 years of age) (24). These results suggest that higher levels of PDG are associated with a greater frequency of new onset headache prior to menarche, and a lower frequency after menarche.
The effect size of PdG association is best depicted in Figure 2 at two very specific hormonal levels. In the 16–17 age group, the predicted probability of HO days was 42% at a PdG level of 0 mcg/cl and 24% at 50 mcg/cl, while in the 8–11 age group it was 15% at 0 mcg/cl and 20% at 50 mcg/cl. There was little change noted in the 12–15 age group. These two levels of PdG were chosen to depict the effect size because lower levels of 0–10 ng/ml are typically encountered around menses, while higher levels of 50–100 ng/ml commonly occur during the mid-luteal time period of the menstrual cycle.
The finding that higher levels of progesterone are associated with a lower incidence of NOH in our older adolescent girls is similar to that observed in adult women. Martin and colleagues (25) found that the headache index was lowest in adult female migraineurs during the mid-luteal phase of the menstrual cycle, which is a time period of higher progesterone levels, compared with the follicular phase. In addition, progestins, which are synthetic derivatives of progesterone, have been reported to decrease the frequency of migraine in adult women with migraine (26–28). Therefore, older adolescent girls (post-pubertal) may be developing a more “adult” pattern of response to progesterone.
Conversely, there was a greater incidence of NOH in 8–11 year-old girls with higher levels of progesterone, and little effect of progesterone at any level in 12–15 year old girls. There seems to be a graded effect to progesterone that is dependent on age. This effect might be mediated by allopregnanolone, which a metabolite of progesterone and a potent agonist of γ- aminobutyric acid-A (GABA-A) receptors. Animal studies have found alterations in the subunit composition of GABA-A receptors at the time of puberty, and such alterations change their response to allopregnanolone (29). If such changes occurred in humans, it might explain the differing effects of progesterone noted in our study.
It is interesting that urine progesterone levels were more predictive than urine estrogen levels for headache onset days. This is surprising, given that declines in serum estrogen levels at the time of menstruation have been postulated to trigger attacks of menstrual migraine in adult women (16). It is possible that menstruating adolescent girls may not attain a sufficient magnitude or rate of decline of serum estrogen levels around menses to generate attacks of menstrual migraine. Alternatively, it may require repeated exposures of declining estrogen levels over many menstrual periods to establish attacks of menstrual migraine. A third possibility is that we had an inadequate sample size to detect an interaction for this variable.
Models did show a significant univariate association between ΔPdG and headache severity. However, the effect size was quite small with a sevenfold increase in the standard deviation of ΔPdG leading to less than 1 unit change in headache severity. Such a large increase in PdG only occurred on one day in the entire sample. Therefore, it is unlikely ovarian hormones moderate headache severity to any significant degree.
Potential mechanisms
There are several potential mechanisms through which age and pubertal development might moderate the effect of ovarian hormones on migraine. First, attainment of more regular menstrual cycles in older and more mature adolescents might explain the findings in this study. We think this is less likely, given that there was no significant interaction between the hormonal variables and menstrual cycle variance, which should be a marker for cyclic patterns of ovarian hormones. Second, there are structural changes in the brain that occur before, during, and after puberty, and these changes might confer a difference in response to ovarian hormones (30). Third, animal studies have shown an increase in the density of estrogen and progesterone receptors in the hypothalamus during puberty, which could modulate the action of ovarian hormones if similar changes occur in women (31). Fourth, pubertal and post pubertal girls have an exaggerated stress response compared to pre-pubertal girls, which could trigger more frequent migraine attacks (32). Fifth, as mentioned above, changes in the subunit composition of GABA-A receptors that occur during puberty might explain the differing effects of progesterone noted in this study (29).
Strengths and limitations
The strengths of the study included a well characterized group of prepubertal, pubertal, and postpubertal girls with migraine where diagnosis of migraine was established by headache physicians based on ICHD-II criteria. In addition, the subjects were very dedicated in diary completion and sample collection. The assays of urinary hormones have been validated, and parallel serum levels of estrogen and progesterone (21). This represents one of the only available and practical methods to measure estrogen and progesterone levels on a daily basis.
This study was limited by small sample size and consequently may lack sufficient power to detect significance for subtle hormonal variables. There were no adjustments for multiple comparisons in our analyses, and therefore these results should be considered exploratory until replicated in future studies. Additionally, the Tanner stage of the children was not specifically assessed, relying on the validated surrogate of pubertal development scale, which has a moderate correlation with the Tanner stage (19,20). These results may generalize to girls with more frequent attacks of migraine headache, as the participants in this study were recruited from a headache clinic. Given the small sample size, it was necessary to include those with more frequent headaches in order to have any chance of detecting different hormonal responses within the three age strata. There was wide variability in the frequency of headache in our study participants that may have influenced our results, but our use of regression models with random intercepts should account for differences between participants. Participants were allowed to remain on preventative therapies during the study. We do not think that these co-therapies influenced our results, as to our knowledge there is no evidence that pharmacological or biobehavioral treatments change levels of ovarian hormones in girls with migraine, or their response to them. Also, preventative medications have little effect in adolescents, as evidenced by a recent large randomized controlled study demonstrating that topiramate and amitriptyline were no better than placebo in the prevention of migraine headache (33).
Conclusions
Our results suggest that age and pubertal development could moderate the effect of reproductive hormones on days of headache onset in pre-pubertal, pubertal, and post-pubertal girls with migraine. Higher levels of PdG were associated with less NOH in the 16–17 age group, had little effect in the 12–15 age group, and higher NOH in the 8–11 age group. The mechanisms through which this occurs are unknown, but may include varying hormonal milieus, structural changes to the brain, alterations in hormonal or other receptors or changes in the response to stress. These results, however, are preliminary and should be interpreted cautiously given the small sample size and variability of hormonal measurements within and between groups.
Footnotes
Article highlights
Age and pubertal development could influence the effect of ovarian hormones on days of headache onset during puberty in girls with migraine.
Higher levels of urinary hormone metabolites of progesterone were associated with a lower incidence of NOH in older girls and a higher incidence in younger girls.
A differing response of migraine to ovarian hormones might be explained by varying hormonal milieus, differences in neuroreceptors or structural changes in the brain that occur with pubertal development.
Abbreviations
Confidence interval (CI), estrone glucuronide (E1G), pregnandiol glucuronide (PgG), E1G and PdG change scores (ΔE1G, ΔPdG), headache onset (HO), pubertal development score (PDS), standard deviation (SD); gamma aminobutyric acid-A (GABA-A); gonadotropin-releasing hormone (GnRH).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research study was funded by a grant from the National Headache Association. We would also like to acknowledge the Driskill Foundation for their generous financial support of this research project.
