Abstract
Background
Previous functional and structural imaging studies have revealed that subcortical structures play a key a role in pain processing. The recurring painful episodes might trigger maladaptive plasticity or alternatively degenerative processes that might be detected by MRI as changes in size or microstructure. In the current investigation, we aimed to identify the macro- and microstructural alterations of the subcortical structures in episodic cluster headache.
Methods
High-resolution T1-weighted and diffusion-weighted MRI images with 60 gradient directions were acquired from 22 patients with cluster headache and 94 healthy controls. Surface-based segmentation analysis was used to measure the volume of the subcortical nuclei, and mean diffusion parameters (fractional anisotropy, mean, radial and axial diffusivity) were determined for these structures. In order to understand whether the size and diffusion parameters could be investigated in a headache lateralised manner, first the asymmetry of the size and diffusion parameters of the subcortical structures was analysed. Volumes and diffusion parameters were compared between groups and correlated with the cumulative number of headache days. To account for the different size of the patient and control group, a bootstrap approach was used to investigate the stability of the findings.
Results
A significant lateralisation of the size (caudate, putamen and thalamus) and the diffusion parameters of the subcortical structures were found in normal controls. In cluster headache patients, the mean fractional anisotropy of the right amygdalae, the mean axial and mean diffusivity of the right caudate nucleus and the radial diffusivity of the right pallidum were higher. The mean anisotropy of the right pallidum was lower in patients.
Conclusion
The analysis of the pathology in the subcortical structures in episodic cluster headache reveals important features of the disease, which might allow a deeper insight into the pathomechanism of the pain processing in this headache condition.
Introduction
Episodic cluster headache (CH) is a primary headache disorder with the prominent feature of extremely severe unilateral, periorbital headache attacks accompanied by ipsilateral autonomic symptoms, and occurs in clusters usually lasting some weeks, followed by much longer headache-free periods (1,2).
The basal ganglia and generally the subcortical structures were recently proposed to have a central role in nociception. The facts that they receive input directly from the spinal cord and also via the thalamus, and exhibit connections with various cortical regions involved in pain processing, suggest that this system is ideal for the integration of various aspects of pain-related information (for an in-depth review, see (3) and (4)). Furthermore, it has been shown that basal ganglia structures are activated during painful stimuli (5), and putaminal and pallidal neurons are able to encode the intensity of noxious thermal stimuli (6). There are opioid receptors in the striatum, and nociceptive neurones have also been found (7). The nociceptive neurones of the spinal cord project directly to the globus pallidus, amygdala and hypothalamus (8). Moreover, the activation of the striatum correlates with the variability in pain sensation (9). Putaminal lesions have been shown to alter nociceptive information processing (10). Neuroimaging studies have demonstrated structural alterations in the striatum in chronic pain syndromes (11). In migraine, a larger caudate nucleus was observed in patients who displayed a high attack frequency (12). Imaging studies have also indicated the involvement of the subcortical structures in CH. Atrophy of the thalamus and caudate nucleus has been reported in CH (13).
Disease related structural alterations could possibly be captured at the macrostructural level as size changes, and the highly organized microstructure of the subcortical nuclei makes them readily available to study with diffusion weighted MRI. The two features might show parallel alterations, but since the histological background of the diffusion alterations in the grey matter is not well understood, the micro- and macrostructural alterations may also occur independently.
In the current study, we set out to investigate volumetric changes in CH by an approach highly tuned to the detection of subcortical volume changes (FIRST (14)). Furthermore, we made use of the highly organized microstructure of subcortical nuclei to investigate diffusion in the segmented nuclei. We also hypothesised that the disease did not affect all subcortical structures at the same rate, and we also wanted to analyse the co-occurrence of the micro- and macrostructural alterations in CH.
We expected two possible outcomes: (i) Repetitive painful attacks might induce maladaptive plasticity in pain related cortical and subcortical structures, and these changes might present in a volume augmentation and possibly in a more organized microstructure, as detected by diffusion tensor imaging. (ii) Alternatively, the recurrent severe pain and concurrent pathological processes might induce degenerative changes, which might present in the form of microstructural disintegration and atrophy.
Investigating the brain structures by pooling data as being contralateral or ipsilateral to the headache side is a frequently used approach in the headache literature to boost the number of observations. However, this approach can only be used if no lateralisation exists in healthy subjects. The lateralisation in healthy subjects was investigated by processing the MRIs of a large group of volunteers, measured with the same parameters as those of the CH patients. The same large control group was used in all further comparisons, since any selection of a subgroup could cause a statistical bias. In order to account for the different size of the groups compared, a bootstrap approach was used to confirm stability of the findings (15). The bootstrap approach takes random samples from each subgroup, of the same size. By repeating the random sampling with replacement a large number of times, the bootstrap approach not only gives a statistical estimator of the model, but the stability of that estimator can also be evaluated.
Methods
Participants
Twenty-seven patients were recruited into the study. Five patients were excluded due to comorbidity (depression, benign tumour), cessation of pain after tooth extraction (e.g. secondary cluster headache), and structural abnormality on the MRI scans. Finally, 22 patients were included (mean age: 38.10 ± 11.33, male: 19). Inclusion criteria for the CH patients were: 18–80 years of age, primary CH according to The International Headache Society diagnostic criteria (1), no interval therapy for the CH, no accompanying neurological (including other primary headache disorders and pain conditions) or psychiatric disease, no regular neuro-psychiatric medication, negative routine MRI scan. Special attention was paid to the exclusion of depression, for which the Hamilton questionnaire was used (>16 points was the exclusion criterion) (16). The MRI acquisitions were carried out in the interictal period (at least one month after the last headache episode). There were 12 left-headache-sided (LHS-CH) and 10 right-headache-sided patients (RHS-CH) in the CH group. All the participants were right handed. Clinical variables, such as disease duration, time between bouts and average length of bouts were recorded for all patients. Furthermore, the cumulative number of headache days – that is, the total number of days the patient had experienced cluster headache over his/her entire life – was estimated for all the patients.
Demographic data of the participants.

Not significant while bootstrapping. Bold value highlights only significant (p < 0.05) results. CH (all): RHS-CH and RHS-CH patient together. RHS-CH: cluster headache patients with pain on the right side. LHS-CH: cluster headache patients with pain on the left side.
Statistics (p-values) of the demographic data.
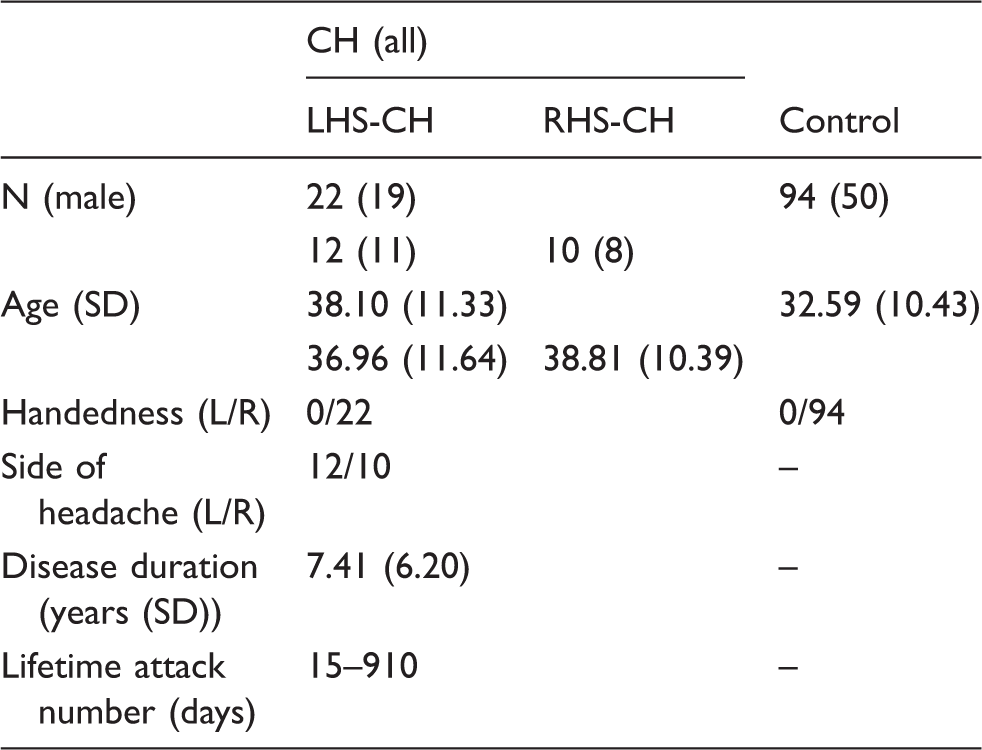
CH (all): RHS-CH and RHS-CH patient together. RHS-CH: cluster headache patients with pain on the right side. LHS-CH: cluster headache patients with pain on the left side.
The study was approved by the Ethics Committee of the University of Szeged (authority number: 87/2009), and all the subjects provided their written informed consent.
Image acquisition
MR image acquisition took place at least one month after the end of the last headache bout. Imaging was carried out with a 1.5 T GE Signa Excite MRI scanner. High-resolution T1-weighted images (3D IR-FSPGR: TR/TE/TI: 10.3/4.2/450 ms, flip angle: 15°, ASSET: 2, FOV: 25 × 25 cm, matrix: 256 × 256, slice thickness: 1 mm) and 60 direction diffusion-weighted images with six non-diffusion-weighted reference volumes (TE: 93.8 ms, TR: 13500 ms, matrix: 96 × 96, FOV: 23 × 23 cm, flip angle: 90°, in-plane resolution: 2.4 × 2.4 mm, which was resampled to 0.89 × 0.89 mm by the scanner, slice thickness: 2.4 mm, b: 1000 s/m2, NEX: 2, ASSET) were recorded.
Image processing
Data were processed by means of the tools from the FMRIB Software Library (FSL, version 5.0; Oxford Centre for Functional MRI of the Brain (FMRIB), UK; www.fmrib.ox.ac.uk/fsl).
Partial brain volumes of the brain and subcortical structures
For comparisons of the volumes of the subcortical structures between the groups, FIRST analysis was used (14). FIRST is a model-based segmentation/registration tool. It uses deformable surface meshes specific to subcortical structures (the amygdala, caudate nucleus, hippocampus, pallidum, putamen and thalamus). Based on learned models, FIRST searches through linear combinations of shape modes of variation for the most probable shape instance given the observed intensities in a T1-weighted image. The nucleus accumbens was not investigated because of the inappropriate segmentation.
Partial brain volumes (total brain volume, total grey matter, white matter, peripheral grey matter [cortex] and ventricular cerebrospinal fluid), with and without normalization for the intracranial volume, were compared between the two groups. The intracranial volume and the grey and white matter volumes were estimated by SIENAX (17,18), part of FSL (19). SIENAX starts by extracting brain and skull images from the single whole-head input data (20). The brain image is then affine-registered to MNI152 space (21,22) (using the skull image to determine the registration scaling); this is primarily carried out in order to obtain the volumetric scaling factor (vscale) to be used as normalization for the head size. Next, tissue-type segmentation with partial volume calculation is carried out (23) in order to calculate the total volume of brain tissue (including separate estimates of the volumes of the grey and white matter), and the grey and white matter partial volumes.
Diffusion parameters of the basal ganglia
In order to evaluate the internal microstructure of the subcortical structures, diffusion parameters were estimated for each of the segmented subcortical structures and compared between the two groups. The diffusion data were corrected for eddy currents and movement artefacts by 12 degrees of freedom affine linear registration to the first non-diffusion-weighted reference image (22). Diffusion tensors at each voxel were fitted by the algorithm included in the Diffusion Toolbox (FDT) of the FMRIB’s Software Library (FSL v. 4.0, www.fmrib.ox.ac.uk/fsl; (19)). Fractional anisotropy (FA), mean diffusivity (MD), and diffusivity parallel (AD, axial) and perpendicular (RD, radial) to the principal diffusion direction were computed for the whole brain. The binary masks of subcortical structures segmented by FIRST analysis of each subject were registered (FLIRT) to the subjects’ own DWI images with 6 degrees of freedom. The transformed masks were thresholded at 0.5 and binarised again. All registered images were then checked visually and corrected so they did not contain parts of ventricles or white matter tracts close to the subcortical structures. Mean diffusion parameters were calculated under the masked areas.
Furthermore, the trigeminal nerves were identified in the axial plane using colour orientation maps created by overlaying the principal eigenvector image (V1) over the FA map. Then, 15 voxel sized ROIs were manually drawn on the root entry zone of the trigeminal nerves. The maximum and the mean of the diffusion parameters were estimated.
Statistical analysis
Lateralisation in healthy subjects
The left-right ratio of the size and diffusion parameters of the subcortical structures was estimated as the ratio of the left and right side parameters (1 meaning no lateralisation, numbers higher than 1 indicating higher parameters on the left and numbers lower than 1 indicating higher parameters on the right). One sample t-test was used to test lateralisation.
Alteration of the size and diffusion parameters of the subcortical structures in CH
The group differences (t0, p0) were evaluated by using a standard general linear model, where the model was encoding group membership. While our analysis in healthy controls found no gender or age effect, these two variables were included in the analysis to account for the theoretical effect. The solution of the regression model was estimated by the ordinary least squares approach. Since the number of subjects in the control and patient groups differed significantly a bootstrap approach was used to confirm the stability of the findings (15). Five thousand bootstrap samples were randomly drawn from each group by the size of the patient population (N = 22), ensuring that the size of the two compared groups was always the same. The parameters (tB) of the GLM model were estimated for every bootstrap sample (tB1 – tB5000), then the mean of it was calculated (
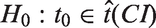
The null hypothesis was to be rejected, and hence the original, non-bootstrapped parameter (t0) was not from the bootstrapped distribution at the significance level of 0.05, if t0 lay outside the two-tailed (1-α) confidence interval
A similar approach was used for the correlations between clinical variables, volumes and diffusion parameters (the correlation coefficient r was used in the bootstrap resampling).
Results
Asymmetry of the size and diffusion parameters of the subcortical structures in healthy subjects
The laterality of the size and the diffusion parameters of the subcortical structures were investigated in 94 healthy control subjects.
Head size normalized volumes of the right caudate nucleus, left putamen and left thalamus were significantly higher than the contralateral pair of these structures.
Laterality in healthy subjects: The volume and diffusion parameters of the subcortical gray matter structures.

Italicized values highlight only close to be significant (p > 0.05 but p < 0.1) results. (+) highlights only close to be significant (p > 0.05 but p <0.1) results. p0 = p value with 1-sample T-test. Mean0 = mean Left/Right ratio. MeanBoot = mean Left/Right ratio of the bootstrapped sample. 95% CI(MeanBoot) = two-tailed (1-alpha) confidence interval of the Left/Right ratio values from the bootstrapped sample.
These results indicate that there is a significant lateralisation of the size and diffusion parameters in healthy subjects. Consequently, pooled parameters according to the headache side cannot be used in the group comparisons.
Alteration of the size and diffusion parameters of the subcortical structures in CH
The GLM analysis showed that the FA of the right amygdala was significantly higher in CH and LHS-CH patients than in healthy subjects. A similar tendency was found in case of the left amygdala in RHS-CH patients. The MD and RD of the right amygdala were higher in healthy subjects compared to the CH and LHS-CH patients. A similar tendency was found in RHS-CH patients.
The AD of the right caudate nucleus was higher in CH, RHS-CH and LHS-CH patients compared with healthy subjects. A similar tendency was found with MD and RD in CH and LHS-CH patients.
In the case of the right pallidum, FA was lower in CH and LHS-CH patients. The RD of the right pallidum showed a tendency to be higher in CH and LHS-CH patients than in healthy subjects. The head size normalized volume of the right pallidum was lower in RHS-CH patients than in healthy subjects.
According to the diffusion parameters of the trigeminal root entry zone, there were no significant differences between patients or controls, or between the ipsilateral and contralateral measures. Total brain volume and grey and white matter volumes did not differ between groups either.
The volume and diffusion parameters of the subcortical gray matter structures compared to healthy controls.

• Higher values in the CH group. Bold values highlight significant and italics highlight close to be significant results. CH (all): RHS-CH and RHS-CH patient together; RHS-CH: cluster headache patients with pain on the right side. LHS-CH: cluster headache patients with pain on the left side. p0: p value from the original GLM. t0: t value from the original GLM. tBoot: mean t value of the bootstrapped sample. 95% CI(tBoot): two-tailed (1-alpha) confidence interval of the t values from the bootstrapped sample.
Correlation of the volumes and diffusion parameters with clinical parameters
The head size normalized volume of the total brain and cortical grey matter showed positive correlation with the cumulative number of headache days in the CH patients. Similar correlation was found in the LHS-CH patients. Also total grey matter and white matter showed similar, but not significant tendency in the CH patients.
The head size normalized volume of the left and right hippocampus and right caudatus showed positive correlation, and the AD of the left and right thalamus, the MD and RD of the left hippocampus showed negative correlation with the cumulative number of headache days in the CH patients. The head size normalized volume of the left pallidum, left and right thalamus showed positive correlation, but the AD of the left hippocampus and the MD of the left pallidum showed negative correlation with the cumulative number of headache days in the RHS-CH patients.
Correlation of the volume and diffusion parameters of the subcortical gray matter structures with lifetime attack number in in CH.

Bold values are higher values in the CH group. Bold values highlight significant and italics highlight close to be significant results. CH (all): RHS-CH and RHS-CH patient together. RHS-CH: cluster headache patients with pain on the right side. LHS-CH: cluster headache patients with pain on the left side. p0: p value from the original Pearson-correlation. t0: t value from the original Pearson-correlation. RBoot: mean correlation coefficient of the bootstrapped sample 95% CI(RBoot): two-tailed (1-alpha) confidence interval of the correlation coefficients from the bootstrapped sample.
Discussion
In this MRI study, we demonstrated an altered interictal microstructure of the subcortical structures in CH. Some of these microstructural changes were dependent on the lifetime disease burden. Demonstrating the lateralisation of diffusion parameters of the subcortical structures in a large cohort of healthy subjects, we showed that headache side dependent pooling of the data to boost the number of observations is not recommended.
In neuroimaging studies investigating unilateral processes, such as in CH and migraine, to boost the number of observations it is common to flip the data about the midsaggital axis to have hemispheres/structures aligned according to the headache side (24–26). This approach is used despite the fact that it was shown that there is a normal asymmetry in the white matter diffusion parameters (27). The most consistent asymmetry was found in the arcuate fasciculus and in the cingulum (27–29). Similarly, diffusion parameters of the subcortical structures were reported to be asymmetric in healthy subjects (30). While Fabiano’s findings are important and consistent with our findings, in their study the mean of diffusivity from only three directions were examined in a small cohort of healthy subjects, and circular regions of interests were used in some of the structures (30). In our study, we used a larger cohort of healthy subjects and found asymmetries of various diffusion parameters estimated from diffusion measurements from 60 different directions. Based on these results, we concluded that we cannot pool data from the different hemispheres of CH patients. In our analysis, we treated CH patients as one group regardless of the side of the headache. Only as a secondary test, we compared the RSH-CH and LSH-CH patients to healthy subjects. Because of the small size of the groups, the results of these later comparisons have to be handled cautiously even though the stability of the findings were evaluated by a bootstrap sampling. Furthermore, it has to be pointed out that the size of the subgroups of CH patients in our analysis does not allow us to investigate headache side dependent alterations of parameters or alterations of the lateralisation of these parameters.
Macro- and microstructural alterations are well-known features of pain-related disorders (24,31). Regarding CH, the seminal paper by May et al. (32) described an increased grey matter volume in the inferior and posterior hypothalamus in CH, but no other changes in grey matter density in either the cortex or the subcortical structures. In contrast, another VBM analysis indicated a decreased grey matter volume in the right thalamus, in the head of the right caudate nucleus and in several pain-processing-related cortical structures, such as the right precentral gyrus, right posterior cingulate cortex, bilateral middle frontal gyrus, right middle temporal gyrus, left inferior parietal lobule and left insula (13). Importantly, the decrease in the grey matter volume did not depend on the side of the pain. While slightly smaller subcortical structures were found, no subcortical volume alteration was identified with our surface-based analysis approach (except the right pallidum, which was smaller in RHS-CH than in healthy subjects). A recent VBM style investigation also called attention to the fact that the cortical and subcortical volumetric alterations are dynamic in relation to the pain state (e.g. in and out of bouts and in chronic CH) (25).
Alterations in the diffusion parameters in the white matter in primary headache disorders such as migraine and CH are already known (24,31), but the microstructural alterations in the subcortical structures described here have not been investigated previously in CH. Only a few studies have dealt with the pain-related changes in the diffusion parameters in the subcortical structures. An increased fractional anisotropy has been reported in the thalamus contralateral to the affected body side in multiple sclerosis patients with extremity pain (33). In irritable bowel syndrome, decreased FA has been found in the thalamic regions, and a reduced MD in the globus pallidus and a higher MD were described in the thalamus (34). The apparent diffusion coefficient (ADC) in the red nucleus of patients with migraine has been shown to be increased relative to healthy subjects (35). In migraineurs without aura, FA was reported to be higher and MD to be lower in the bilateral thalami compared to controls in the interictal phase (36). The diffusivity parameters have also been found to be similar to those in controls in the ictal phase (36). While the diffusion parameters of the trigeminal nerve or the root entry zone have been investigated in trigeminal neuralgia (37), there have been no prior investigations published in CH. Our results did not find CH-specific alterations of the diffusion parameters of the trigeminal root entry zone, but one possible source of this negative result might be the high variability in the data. On average, the neural and vascular structures of the prepontine fossa range between 0.5 and 3.0 mm in diameter. This small diameter has to be considered in diffusion-tensor imaging studies in terms of the limited spatial resolution of the sequences used (38).
The FA of the bilateral amygdala was found to be increased in our study. The amygdala is known for being an important centre of emotional and affective aspects of pain. It is an important hub in the processing of noxious stimuli, and it has been shown to have structural and functional connections to cortical and subcortical structures involved in pain processing (39,40). It has been shown by several studies that there is a significant pain-related plasticity in the amygdala in chronic pain conditions, and amygdalar plasticity was suggested to be the key factor in the establishment of fear memory (41,42). We propose that the increased FA in CH patients might have a similar background. Interestingly, the pain related activation of the amygdala is heavily lateralised to the right (43), but no such lateralisation of diffusion parameters or size was found in our patients. While it is known that basal ganglia structures – such as the pallidum and caudate nucleus, which was found to have altered diffusion parameters in CH in our study – are activated in response to painful stimuli (5), and structural alterations are related to chronic pain conditions (11), the exact role of these structures in the processing of noxious stimuli is not well understood yet.
In a prior study, we showed diffusion parameter alterations in the white matter of CH patients (31). In the white matter, the pattern of diffusion parameter alterations may be related to the underlying pathological microstructural changes. In mouse models (44), the changes observed in AD and RD have been suggested to relate to axon or myelin damage, respectively. However, the background of the diffusion parameter changes in the subcortical grey matter is not so clear. Nevertheless, the subcortical grey nuclei have a well-defined microstructure, which – as we have shown – is altered by CH. There can be a few processes leading to the alteration of the diffusion parameters of the subcortical structures. The contribution of the extracellular and intracellular water fraction to the diffusion signal is not even. A combination of diffusion parameter changes could be explained by the change in the ratio of the extra and intracellular water fractions (e.g. global shrinkage of the structure or intracellular oedema) or, for example, protein deposits limiting the water diffusion. Alterations can also be caused by the change of the myelin or axon content or fibre orientation.
Recent studies suggest that neurodegeneration could result in diffusion changes in related basal ganglia. In movement disorders, in which the basal ganglia play a crucial role in the pathomechanism, altered diffusivity parameters were detected (in Huntington’s disease (45); in Parkinson’s disease (46); and in blepharospasm (47)). We consider that this pattern points to the co-occurrence of degenerative changes, presenting in the form of microstructural disintegration and compensatory or pain-related maladaptive plastic changes in CH.
Limitations
This analysis has some essential drawbacks. First of all, it is a cross-sectional assessment of the focal shrinkage of the subcortical and cortical volumes; also, we do not have exact information about the time elapsed from the last attack. A further drawback is the relatively low power of the secondary analyses (RHS-CH and LHS-CH) due to the limited number of patients. Finally, while only patients with no history of other neurological conditions were recruited/involved, we did not have any information about additional risk factors (e.g. diabetes mellitus) that the patients may have had. In order to acquire an in-depth understanding of the dynamics of brain atrophy and diffusion parameters, longitudinal studies are needed. Given the central role of the hypothalamus in CH, it would be crucial to investigate its size and microstructure. Unfortunately, our approach in its current form is not able to segment the hypothalamus, segmentation of the structure being limited by the low difference in intensity from the surrounding structures.
Key findings
The diffusion parameters of the subcortical structures are lateralised in healthy subjects. Headache side dependent pooling of the imaging data to boost the number of observations is not recommended. The diffusion parameters of some subcortical structures are altered in cluster headache. The diffusion parameter changes of the subcortical structures in cluster headache are related to the lifetime disease burden.
Footnotes
Acknowledgements
The study was supported by the Neuroscience Research Group of the Hungarian Academy of Sciences and University of Szeged, the core facility MAFIL of CEITEC supported by the MEYS CR (LM2015062 Czech-BioImaging), the project FNUSA-ICRC (no. CZ.1.05/1.1.00/02.0123) from the European Regional Development Fund, by European Union project ICRC-ERA-HumanBridge (No. 316345), the National Brain Research Program (Grant No. KTIA_13_NAP-A-II/20) and an OTKA (PD 104715) grant. Dr. Szabó was supported by the Bolyai Scholarship Program of the Hungarian Academy of Sciences.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
