Abstract
As migraine is associated with an increased risk for ischaemic stroke and peripheral vasospastic disorders, it was hypothesized that interictal vascular changes may be present in migraine patients. Using ultrasound and applanation tonometry, the cardiovascular properties of migraine patients were compared with those of matched control subjects. Vascular parameters of the carotid arteries, cardiac output and systemic vascular resistance did not differ between both groups. Right temporal artery diameter was larger in migraine patients (mean difference 101 μm; 95% confidence interval (CI) 9/194 μm; P = 0.033). At the brachial artery, migraine patients displayed a smaller distension (difference -24 μm; 95% CI -45/-4 μm; P = 0.021) and a decreased compliance (difference -0.025 mm2/kPa; 95% CI -0.047/-0.003 mm2/kPa; P = 0.024). Thus, migraine patients display an increased peripheral arterial stiffness. The presence of these interictal vascular changes suggests that migraine might be part of a more generalized vascular disorder.
Introduction
The simple concept of migraine being due to a vasodilatation of cranial blood vessels as postulated by Graham and Wolff (1) has shifted towards a complex theory regarding migraine as a neurovascular disorder of the trigeminovascular system (2). Although this theory highlights the activation of pain-producing perivascular sensory nerves, the concept of vascular changes taking place during a migraine attack is generally accepted (3, 4). These vascular changes comprise cranial vasoconstriction and cerebral hypoperfusion during the aura phase followed by vasodilatation and hyperperfusion during the headache phase.
Studies of the pathophysiology of migraine have focused on the dilatation and increased pulsations of the external carotid artery and its branches, including the temporal artery (5, 6). However, during a migraine attack vascular changes are not limited to the cranial circulation. Iversen et al. (5) reported that a migraine attack is characterized by a generalized peripheral vasoconstriction and concluded that migraine is accompanied by abnormal reactions of the arterial system.
The association of migraine with angina pectoris due to coronary artery vasospasm (i.e. Prinzmetal's angina) (7–9) and the increased frequency of Raynaud's syndrome in migraine patients (10) suggest that migraine might be part of a generalized vasospastic disorder (7). In addition, migraine patients have repeatedly been reported to be at increased risk for the development of ischaemic stroke (11–13). As each of these conditions might be the result of a common vascular abnormality, it cannot be excluded that migraine is accompanied by vascular changes which extend beyond the cranial circulation and are present outside the headache phase.
Based on these observations, the present study was designed to compare the cardiovascular properties of migraine patients with those of a matched group of non-migraineurs. To this end, cranial (i.e. common carotid and superficial temporal artery), peripheral (i.e. brachial and common femoral artery) and cardiac measurements were performed during the interictal period.
Subjects and methods
Subjects and subject selection
The cardiovascular parameters of 50 migraine patients (as defined by the International Headache Society migraine criteria (14)) were compared with those of 50 healthy control subjects. Both populations were matched for those parameters known to be associated with vascular changes, i.e. gender, age, body mass index, blood pressure, cholesterol and smoking habits. Migraine patients were recruited from the neurology out-patient clinic and control subjects from among hospital and laboratory staff. Based on medical history, physical examination and routine laboratory tests, all participants were in good health. Major exclusion criteria were: history of cerebrovascular or cardiovascular disease, arterial hypertension (blood pressure>95/160 mmHg), diabetes mellitus, hyperlipidaemia (total cholesterol>6.5 mmol/l), pregnancy or lactation. The regular use of vasoactive drugs was not allowed. After approval by the ethics committee of the academic hospital of Maastricht this research was carried out in accordance with the Declaration of Helsinki (1989). All subjects gave written informed consent to participate.
Study protocol
After screening, eligible subjects were invited to return to the hospital for cardiovascular assessments after a headache-free period of at least 3 days. Before examination subjects were asked not to use any drugs for at least 3 days and to abstain from caffeine or alcohol-containing beverages and from smoking for at least 12 h. All measurements were performed by the same investigator (J.d.H.) in a quiet, temperature-controlled room with the subject lying comfortably in the supine position. The observer was unblinded with respect to the diagnosis of migraine.
Measurements started after 15 min of supine rest and lasted approximately 1.5 h. Systolic blood pressure (SBP), diastolic blood pressure (DBP) and heart rate were recorded at the left upper arm using a semiautomated oscillometric device (Dinamap® 950; Critikon Inc., FL, USA). Blood pressure and heart rate were measured every 3 min for at least 15 min. The mean of these readings was used as the subject's haemodynamic parameters. Using an ultrasound device (Scanner 350; Pie Medical, Maastricht The Netherlands) and an applanation tonometer (Micro-tip® transducer; Millar Instruments Inc., TX, USA), vascular measurements were consecutively performed at the common carotid artery (bilateral), superficial temporal artery (bilateral) and at the right brachial artery and common femoral artery. Cardiac measurements were performed using echocardiography (Vingmed CFM 750; Sonotron, Horten, Norway).
Measurements
Pressure waveform registration and pulse pressure calculation
Pressure waveforms of the common carotid, brachial and femoral artery were recorded non-invasively using applanation tonometry. The applanation tonometer is a pencil-like probe incorporating a small pressure-sensitive transducer which is placed in contact with the skin at the site of the artery of interest and permits continuous and accurate recording of the local pressure wave contour (15). After calibrating these pressure wave contours, the local pulse pressures (ΔP) of the common carotid (ΔPCCA), brachial (ΔPBA) and femoral artery (ΔPFA) were calculated as previously reported (16). Briefly, by definition ΔPBA as measured with applanation tonometry = the pulse pressure (= SBP-DBP) as measured with the oscillometric device. Thus at the brachial artery the nadir and peak of the pressure wave as recorded with applanation tonometry were equalled to DBP and SBP, respectively. Subsequently, by integrating the area under the pressure–time curve of the brachial artery and dividing it by the duration of the cardiac cycle, mean arterial pressure (MAP) was calculated. Based on the observation that MAP and DBP hardly change throughout the arterial tree (17), the nadir and mean (assessed by integration) of the pressure waves of the common carotid artery and femoral artery were equalled to DBP and MAP, respectively. Finally, the corresponding SBP, ΔPCCA and ΔPFA were calculated.
Vessel wall movements and vessel wall properties
Arterial end diastolic diameter (D) and diameter changes (ΔD) were measured using an echo-tracking system as described by Hoeks et al. (18). This system consists of an ultrasound device equipped with a 7.5-MHz linear-array transducer connected to a data acquisition and processing unit. The system has been validated and has shown acceptable reproducibility (19). Both D and ΔD of the common carotid, brachial and femoral artery were measured at least three times. At the temporal artery only D was measured (at least five times). Each measurement lasted about 5 s (± 5 heart beats). For each artery the mean of these recordings was taken as the subject's reading. At the common carotid artery vessel wall movements and pressure waveforms were recorded ipsilateral and consecutively. At the brachial artery and femoral artery vessel wall movements (right) and pressure waveforms (left) were recorded contralateral and simultaneously.
Diameter–time recordings (ultrasound) and pressure–time recordings (applanation tonometry) were analysed off-line using commercially available software (Matlab®) and an analytical program developed by the biophysics department of the university as previously reported (16). By constructing diameter–pressure curves, vessel wall properties were calculated over the operating pressure ΔP according to the following equations: distensibility coefficient, DC = (ΔA/A)/ΔP and cross-sectional compliance, CC = ΔA/ΔP, where A is the end diastolic cross-sectional area (A = π(D/2)2) and ΔA the change in cross-sectional area during the cardiac cycle (ΔA = π(((D + ΔD)/2)2-(D/2)2)). In addition, isobaric vessel wall properties were calculated within the predefined pressure interval of MAP ± 5 mmHg (DCMAP and CCMAP) of each subject.
Blood flow and local vascular resistance
Velocity profiles of the arterial blood flow of the common carotid and temporal arteries were measured with a pulsed-Doppler signal (7.5 MHz) at a 70° angle to the vessel and a sampling volume in the centre of the artery. For each cardiac cycle the mean flow velocity was calculated electronically by multiplying the area under the velocity–time curve (corrected for angle) by the heart rate. At least eight velocity profiles of each artery were recorded; the mean of these recordings was taken as the subject's reading. Blood flow was calculated by multiplying the mean flow velocity by A. Local vascular resistance was defined as MAP divided by local blood flow.
Intima-media thickness
Using an established ultrasound method (20) posterior wall intima-media thickness (IMT) of the right common carotid artery (1–2 cm proximal of the bulb) and brachial artery (at the level of the elbow) was assessed. In short, the raw radio frequency (RF) signal, from which the B-mode (i.e. two-dimensional) and M-mode (i.e. moving image as a function of time) images on the ultrasound monitor are derived, was displayed on the computer monitor. By manually placing sample volumes at the blood–intima and media–adventitia interface of the amplitude signal of the end-diastolic RF-line, the value of IMT was given with an accuracy of two decimals (in mm).
Cardiac output and systemic vascular resistance
Cardiac measurements were performed in the left lateral position. Aortic diameter was measured using pulsed ultrasound (2.5 MHz) from a standard two-dimensional long-axis parasternal view at the site of the aortic annulus. Aortic blood velocity profiles were obtained with continuous ultrasound using an apical window. Stroke volume was calculated from aortic cross-sectional area multiplied by the mean blood velocity across the aortic valve during systole. Cardiac output was calculated electronically by multiplying stroke volume and heart rate, as determined from the duration of the cardiac cycle (21). Systemic vascular resistance (SVR) was defined as MAP divided by cardiac output.
Statistical analysis
It was calculated that with a significance level of 0.05 and a power of 80%, 40 subjects per group were required to detect between-group differences of 20% in distensibility of the femoral artery, which is the artery with the worst reproducibility. Comparison of clinical characteristics and cardiovascular parameters between both groups was done with the unpaired Student's t-test. Except for clinical data, which are presented as mean ± SD, data are presented as mean ± SEM. P < 0.05 was considered statistically significant.
Results
Subject characteristics
Clinical characteristics of migraine patients (aged 19–58 years) and control subjects (aged 21–59 years) did not differ (Table 1). Migraine patients had been suffering from migraine for on average 15 ± 1 years. Eleven patients had a history of recurrent migraine with aura (classic migraine), 39 without aura (common migraine). Migraine attacks recurred with a frequency of 2.6 ± 0.4 per month and lasted on average 1.4 ± 0.1 days. Headache was one-sided in 43 patients: either left-sided (n = 15), right-sided (n = 16) or alternating (n = 12). Nausea or vomiting (n = 39) and photophobia or phonophobia (n = 46) were frequently associated with it. As an abortive drug triptans (n = 21), analgesics (n = 20) and anti-inflammatory drugs (n = 15) were most frequently used. Only three patients mentioned the use of ergot alkaloids. Oral contraceptives or hormonal replacement therapy were used by a comparable number of migraine patients (n = 13) and control subjects (n = 14).
Subject characteristics at screening (mean ± SD)

P-values by unpaired Student's t-test.
Haemodynamic data
Mean arterial pressure did not differ (P = 0.856) between control subjects (88 ± 1 mmHg) and migraine patients (88 ± 1 mmHg). Pulse pressures of the common carotid, brachial and femoral artery of control subjects (41 ± 1, 44 ± 1 and 47 ± 1 mmHg, respectively) were comparable with those of migraine patients (39 ± 1, 43 ± 1 and 46 ± 1 mmHg, respectively); the corresponding P-values were 0.482, 0.195 and 0.447, respectively.
Vascular measurements
At the common carotid artery (Table 2 and Fig. 1) vascular parameters did not differ between migraine patients and control subjects. In both groups distension, DCMAP and CCMAP was smaller at the left common carotid artery compared with the right one. In a subgroup analysis of 31 migraine patients who consistently reported unilateral migraine on the same side of the head, the vascular parameters of the common carotid artery at the ‘diseased’ and ‘healthy’ side did not differ.
Common carotid artery: comparison of vascular parameters of migraine patients (n = 50) vs. control subjects (n = 50)

P-values by unpaired Student's t-test with the corresponding mean difference between groups and 95% confidence intervals (95%CI). DCMAP and CCMAP, distensibility coefficient and cross-sectional compliance at mean arterial pressure, respectively; IMT, intima-media thickness; ND, not determined.
∗ P < 0.05 by paired Student's t-test for comparison of left vs. right common carotid artery within each group.

Comparison of the vascular parameters of the right common carotid (□), brachial ( ) and common femoral artery (▪) of migraine patients with those of control subjects. Differences are expressed as percentage difference (± SEM) vs. control subjects. ∗P < 0.05 by unpaired Student's t-test vs. control subjects.
) and common femoral artery (▪) of migraine patients with those of control subjects. Differences are expressed as percentage difference (± SEM) vs. control subjects. ∗P < 0.05 by unpaired Student's t-test vs. control subjects.
Temporal artery diameter (Table 3 and Fig. 2) was larger (P = 0.033, right-sided) in migraine patients compared with control subjects. Temporal artery blood flow and local vascular resistance did not differ between both groups. A subgroup analysis comparing the temporal artery parameters of the ‘diseased’ and ‘healthy’ side of the head of migraine patients showed no differences.
Temporal artery: comparison of vascular parameters of migraine patients (n = 50) vs. control subjects (n = 50)

P-values by unpaired Student's t-test with the corresponding mean difference between groups and 95% confidence intervals (95%CI).

Comparison of the vascular parameters of the left (□) and right ( ) temporal artery of migraine patients with those of control subjects. Differences are expressed as percentage difference (± SEM) vs. control subjects. ∗P < 0.05 by unpaired Student's t-test vs. control subjects.
) temporal artery of migraine patients with those of control subjects. Differences are expressed as percentage difference (± SEM) vs. control subjects. ∗P < 0.05 by unpaired Student's t-test vs. control subjects.
At the brachial artery (Table 4 and Fig. 1) distension and CCMAP were smaller in migraine patients compared with control subjects (P = 0.021 and 0.024, respectively). The increased IMT in migraine patients (5.3% vs. control subjects) did not reach statistical significance (P = 0.07). At the femoral artery (Table 4 and Fig. 1) no differences were demonstrated between both groups. Still, compared with control subjects in migraine patients distension (P = 0.20), CCMAP (P = 0.18) and DCMAP (P = 0.10) of the femoral artery were on average 12%, 14% and 17% smaller, respectively.
Brachial and common femoral artery: comparison of vascular parameters of migraine patients (n = 50) vs. control subjects (n = 50)
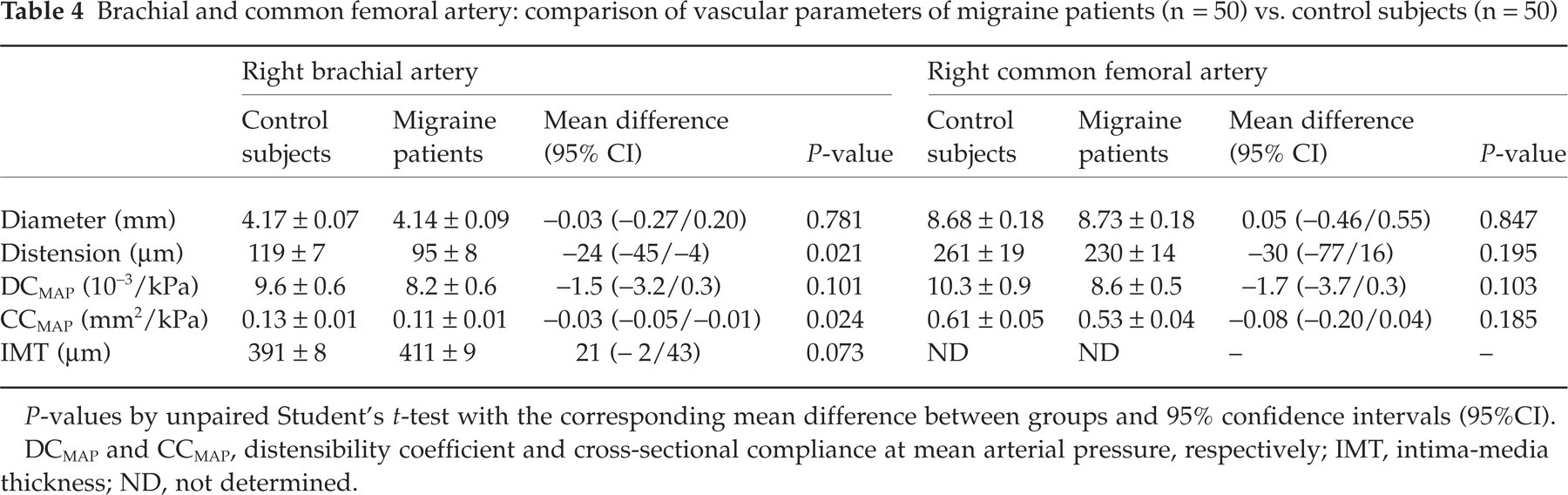
P-values by unpaired Student's t-test with the corresponding mean difference between groups and 95% confidence intervals (95%CI). DCMAP and CCMAP, distensibility coefficient and cross-sectional compliance at mean arterial pressure, respectively; IMT, intima-media thickness; ND, not determined.
Cardiac measurements
Cardiac output and systemic vascular resistance of control subjects (4.8 ± 0.1 l/min and 18.6 ± 0.4 mmHg/l per min, respectively) and migraine patients (4.9 ± 0.1 l/min and 18.5 ± 0.5 mmHg/l per min, respectively) were comparable.
Discussion
The present study set out to investigate whether migraine patients display different cardiovascular properties compared with matched control subjects during the interictal period. The most striking functional and structural vascular differences between both populations were observed at the medium-sized brachial artery displaying an increased arterial stiffness and a tendency towards an increased IMT in migraine patients. In addition, migraineurs showed a larger temporal artery diameter and tended to have an increased temporal artery blood flow compared with control subjects. Because of the importance of these findings, we suggest they be confirmed in a study where the observer is blinded with respect to the diagnosis of migraine in order to exclude observer bias. Neither at the left nor at the right common carotid artery were any differences observed between both groups. Likewise, cardiac output and systemic vascular resistance did not differ.
Vascular changes have frequently been investigated in migraine patients (5, 6). However, most studies only report on vascular changes observed during a migraine attack and focus on those vascular territories involved in the pathogenesis of migraine headache. In particular, research has focused on the external carotid artery and its branches, including the temporal artery (5), which largely perfuse meningeal blood vessels and are considered the pain-producing structures in migraine (2, 22, 23). Iversen et al. (5) have reported that in migraine patients the temporal artery diameters of both sides do not differ during the interictal period, while migraine headache is associated with an increased arterial diameter at the affected side. Accordingly, in the present study the temporal artery diameters of both sides did not differ; neither in migraine patients (between attacks) nor in control subjects. However, even between attacks, temporal artery diameter was larger in migraine patients compared with control subjects. In addition, the trend towards an increased temporal artery blood flow in migraine patients points to differences at the level of resistance vessels as well. Taken together, these data indicate that irrespective of migraine headache, patients display changes both of conduit arteries and resistance vessels in the vascular territory which is affected during an attack. A subgroup analysis of those patients who consistently reported unilateral headache did not reveal any interictal differences between the temporal artery of the affected and unaffected sides, arguing against localized vascular changes.
At the common carotid and brachial artery both functional (vessel wall properties) and structural (IMT) vascular parameters were investigated. The functional parameters distensibility (a measure of vascular elasticity) and cross-sectional compliance (a measure of buffering capacity) refer to mechanical vessel wall properties and decrease with increasing arterial stiffness (24). Distensibility and cross-sectional compliance of migraine patients and control subjects were compared at mean arterial pressure which did not differ between both groups. The present study demonstrates that the distension and buffering capacity of the brachial artery are decreased in migraine patients. Thus, in addition to changes at the temporal artery, migraine patients are also characterized by changes at the peripheral brachial artery. However, in agreement with Barenbrock and co-workers (25), at the common carotid artery no vascular changes were observed. The different behaviour of the common carotid artery compared with the brachial artery is probably related to differences in vessel wall structure. In general, the visco-elastic properties of the vessel wall depend on a passive component due to elastic and collagenous connective tissue and on an active component due to smooth muscle activity (26, 27). The common carotid artery is an elastic artery which has a higher proportion of elastic fibres and a smaller quantity of vascular smooth muscle cells compared with the muscular brachial artery (26). Thus, changes in vascular smooth muscle tone are likely to result in smaller changes of arterial stiffness in ‘elastic arteries’ compared with ‘muscular arteries’. At the femoral artery migraine patients display the same tendency towards an increased arterial stiffness as the one observed at the brachial artery.
The increased arterial stiffness in migraine patients might be related to an increased arterial tone. The regulation of arterial tone is complex and involves autonomic, humoral and endothelial mechanisms (23). Thus an increased arterial stiffness in migraineurs could result from a higher sympathetic tone. However, the conflicting evidence of the currently available data on autonomic nervous system function in migraine patients (28, 29) does not allow definite conclusions to be drawn. As far as humoral factors are concerned, serotonin (5-HT) is the most likely candidate to be involved. It probably plays an important role in the pathogenesis of migraine which is associated with low levels of 5-HT between attacks and increased levels during attacks (30, 31). However, given the biphasic vascular response to 5-HT depending on its plasma concentration (32), it may either increase or decrease vascular tone. Still, as within the physiological range the vasodilator effects of 5-HT predominate (33), the lower plasma concentrations in migraineurs might translate into less vasodilatation and be responsible for an increased arterial stiffness compared with control subjects. Finally, as far as endothelial mechanisms are concerned, Joannides et al. (34) recently reported that nitric oxide (NO) contributes to the regulation of mechanical properties of peripheral conduit arteries in humans. They showed that decreased NO production results in an unexpected increase in arterial compliance. Consequently, as in the present study migraine patients displayed a decreased compliance of the brachial artery, this might be related to an increased production of NO. Moreover, as previous reports suggest that the NO-pathway is activated during the headache-free period (35), this hypothesis fits well with the current opinion of NO playing a pivotal role in the pathophysiology of migraine (36). In contrast with the effects of NO on conduit arteries, at the level of resistance vessels an increased basal production of NO results in a decreased vascular tone (37), which could explain the tendency towards an increased flow at the temporal artery.
Apart from arterial tone, arterial stiffness is also determined by vessel wall structure. As the present study suggests that the IMT of the brachial artery is increased in migraine patients, this might contribute to the observed increased arterial stiffness. Furthermore, the recently reported elevated serum elastase activity in migraine (38) might also contribute by degrading elastic fibres. The resulting production of elastin peptides can induce a NO-dependent vasodilation, supporting the hypothesis of an increased interictal NO-production in migraine. However, the concept of an increased interictal NO-production remains to be established.
As calcitonin gene related peptide (CGRP) has previously been demonstrated to be involved in the pathogenesis of migraine headache (39), it is tempting to speculate about its putative contribution to interictal vascular changes. Could CGRP explain both functional and structural vascular changes which are apparently present in migraine patients? CGRP indeed has a physiological role in the regulation of vascular tone and has previously been shown to inhibit proliferation of cultured smooth muscle cells (40, 41). Consequently, a deficient basal release of CGRP could explain an increased interictal arterial stiffness related to an increased vascular tone as well as intima-media thickening due to smooth muscle cell proliferation. However, this hypothesis does not fit with current evidence suggesting either normal (39) or increased (42) interictal plasma levels of CGRP in migraine patients. An increased interictal plasma level of CGRP would, however, fit with an increased interictal temporal artery diameter.
The acute intake of both 5-HT1B/1D-receptor agonists (43) and ergot alkaloids (25, 44) results in a transient increase in arterial stiffness. Thus, it could be argued that the vascular changes observed in the present study might be related to the preceding use of anti-migraine drugs. However, with the exception of ergot alkaloids, most drugs used to abort an acute migraine attack are short-acting. Even for ergot alkaloids, a drug-free period of at least 3 days should be sufficiently long for functional vascular effects to subside (44, 45). Although it cannot be excluded that the increased arterial stiffness is related to structural vessel wall changes due to the repeated exposure to anti-migraine drugs, this is very unlikely. In addition, the repeated use of ergotamine has previously been shown not to affect IMT (25).
What are the consequences of the present findings? First, as vascular changes are present during the interictal period, it is tempting to speculate that drugs normalizing these changes might be useful in the prophylactic treatment of migraine. Interestingly, many of the currently available prophylactic anti-migraine drugs, including calcium antagonists, selective α-blockers and, very recently, angiotensin converting enzyme inhibitors (46), have previously been shown to decrease arterial stiffness (47, 48) and might thus normalize the vascular changes which render migraine patients at increased risk for a new attack. Second, as the interictal vascular characteristics of migraine patients differ from those of non-migraineurs, migraine patients may display different cardiovascular responses to the administration of vasoactive substances in general and of anti-migraine drugs in particular. Thus, the cardiovascular effects of potential anti-migraine drugs should be carefully looked at both in healthy subjects and in migraine patients. Finally, although it remains to be established whether the observed vascular changes are a primary cause of migraine or secondary to it, they do support the contention that migraine is associated with vascular abnormalities.
In conclusion, the present study suggests that even during the interictal period migraine patients display both cranial and peripheral vascular changes, indicating an important role of the vasculature in the pathogenesis of migraine. Although the contribution of the trigeminovascular system in migraine headache is generally accepted, migraine should not be regarded as an isolated neurovascular problem, but might be part of a more generalized vascular disorder.
