Abstract
Background and aims
Calcitonin gene-related peptide (CGRP) and glyceryl trinitrate (GTN) infusion in migraineurs provokes headache resembling spontaneous migraine, and CGRP receptor antagonists are effective in the treatment of acute migraine. We hypothesized that CGRP infusion would increase molecular markers of neuronal activation in migraine-relevant tissues of the rat.
Methods
CGRP was infused intravenously (i.v.) in freely moving rats to circumvent factors like anesthesia, acute surgery and severe hypotension, the three confounding factors for c-Fos expression. The trigeminal nucleus caudalis (TNC) was isolated at different time points after CGRP infusion. The level of c-Fos mRNA and protein expression in TNC were analyzed by qPCR and immunohistochemistry. c-Fos-stained nuclei were also counted in the nucleus tractus solitarius (NTS) and caudal ventrolateral medulla (CVLM), integrative sites in the brain stem for processing cardiovascular signals. We also investigated Zif268 protein expression (another immediate early gene) in TNC. The protein expression of p-ERK, p-CREB and c-Fos was analyzed in dura mater, trigeminal ganglion (TG) and TNC samples using Western blot.
Results
CGRP infusion caused a significant dose-dependent fall in mean arterial blood pressure. No significant activation of c-Fos in the TNC at mRNA and protein levels was observed after CGRP infusion. A significant increase in c-Fos protein was observed in the NTS and CVLM in the brain stem. Zif268 expression in the TNC was also not changed after CGRP infusion. p-ERK was increased in the dura mater 30 minutes after CGRP infusion.
Conclusion
CGRP infusion increased the early expression of p-ERK in the dura mater but did not increase c-Fos and Zif268 expression in the TNC. The rats may, thus, differ from migraine patients, in whom infusion of CGRP caused headache and a delayed migraine attack. The rat CGRP infusion model with c-Fos or Zif268 as neuronal pain markers in TNC is unsuitable for antimigraine drug testing.
Introduction
Calcitonin gene-related peptide (CGRP) is a potent vasodilator (1). CGRP receptor antagonists olcegepant (2) and telcagepant (3,4) are effective against acute migraine, demonstrating a key role of CGRP in migraine pathogenesis. CGRP infusion in migraine patients caused an immediate headache during and shortly after the infusion, and a delayed headache resembling spontaneous migraine attacks (5,6). As CGRP has a half-life of approximately 10 minutes, the direct effects of CGRP cannot account for the delayed headaches. CGRP therefore seems to trigger a cascade of events probably involving gene activation that results in delayed headache in humans. CGRP and CGRP receptor components are present in the dura mater, trigeminal ganglion (TG), trigeminal nucleus caudalis (TNC) and other pain-processing structures inside the rat brain (7–9).
The immediate early genes (IEGs) c-fos and zif268 have been used extensively to study pain pathways in different parts of the brain in a number of disease models (10–12). In the few studies conducted in animal models of migraine using other migraine-provoking agents, the increase in c-Fos expression has been ambiguous (13,14). These studies were performed in anesthetized rats subjected to acute surgery, which is known to up-regulate c-Fos expression (15). These interfering factors can be minimized in unanesthetized freely moving rats, as shown recently in a glyceryl trinitrate (GTN) infusion model from our lab (16,17). We therefore hypothesize that CGRP infusion in unanesthetized freely moving rats will induce migraine-like pain reflected by increase in c-Fos and Zif268 in the TNC. We also studied expression of p-extracellular signal-regulated kinase (p-ERK), p-cyclic AMP-response element binding protein (p-CREB) and c-Fos using Western blot in the dura mater, TG and TNC samples.
Methods
One hundred and four male Sprague-Dawley (SD) rats weighing 300–360 g (Taconic M&B, Denmark) were used in this study. The rats were maintained in cages with a 12-hour light/dark cycle and free access to food and water. All the experimental protocols were approved by the Danish committee for experiments with animals (2009/561-1664). Rats were weighed pre-surgery (331 ± 5 g, n = 76) and after the recovery period of eight days (354 ± 3 g, n = 76). Rats recovered well after surgery as indicated by body gain and there were no visible signs of distress or infection.
Drugs
CGRP (NeoMPS, Strasbourg, France) stock solution 0.5 mg ml−1 was prepared in water and aliquots were stored at −20℃. CGRP stock solution was further diluted in saline to their final concentrations just prior to administration. All the dilutions required for the study were freshly prepared.
Measurement of mean arterial blood pressure (MABP)
For assessment of MABP in unanesthetized rats, six rats underwent carotid artery and femoral vein cannulation. Rats were allowed to recover for one to three days and infused with CGRP (0.1, 0.25, 0.5 and 1 µg kg−1 minute−1 for 20 minutes at 30-minute intervals between two doses) through the femoral vein, and the MABP was measured via the carotid artery throughout the infusion.
Intravenous (i.v.) CGRP administration
Rats were first anesthetized by intraperitonial injection of a premixed solution of ketamine (Ketaminol®Vet, Intervet, Skovlunde, Denmark) (100 mg kg−1) and xylazine (Rompun®Vet, Bayer Inc, Germany) (7.5 mg kg−1). Prior to surgery the animals were injected with enrofloxacin (Baytril®Vet, Bayer Inc, Germany) (10 mg kg−1subcutaneously (s.c.)), an antimicrobial, and caprofen (Rimadyl®Vet, Pfizer Oy Animal Health, Helsinki, Finland) (5 mg kg−1 s.c.), a nonsteroidal anti-inflammatory drug (NSAID). Surgery was performed aseptically. The incision was first made in the left inguinal area and the femoral vein was isolated. The catheter was then inserted from the neck into the trocar and pulled out at the other end of the trocar facing the femoral vein. The vein was cannulated using 2 cm polythene tubing (0.40 mm, ID, 0.80 mm OD, Portex®, Smiths Medical ASD, USA), attached with polyurethane tubing and checked for patency. The catheter was then sealed using a blocking solution (Haemaccel, Heparin 500 IU ml−1, Voluven®). The animal was monitored until fully recovered from anesthesia as indicated by the ability to ambulate. The core body temperature was maintained using an automatic regulated heating plate. Enrofloxacin and caprofen treatments were repeated once in a day for the next two days, while buprenorphin (Temgesic®, RB Pharmaceuticals Limited, Berkshire, UK) (0.03 mg kg−1), an analgesic, treatment was repeated once a day for the next day. After a recovery period of eight days, the patency of the cannula was checked by infusing 100 µl of five-times diluted ketamine and xylazine solution. Afterwards, the cannula was connected to external polyurethane tubing coming out from a tether at the nape of the neck and the rats were allowed to move freely in AccuSampler cages (Dilab, Lund AB, Sweden) and were acclimatized for two days. On the third day, CGRP (0.25 µg kg−1 min−1 or 1 µg kg−1 min−1, for 20 minutes) was administered i.v. and the rats were sacrificed by pentobarbitone (50 mg kg−1, i.v.) at the required time points. Control rats were sacrificed and perfused after two hours of saline infusion.
Effect of anesthesia on c-Fos expression
To assess the effect of anesthesia on c-Fos expression, six rats (femoral vein cannulated rats, on day 10) were infused i.v. with 200 µl of diluted ketamine and xylazine anesthesia. This gives 15 to 25 minutes of anesthesia to rats; during that time period saline was infused in these rats, after which the rats recovered from anesthesia in the cage. These rats were deeply anesthetized with pentobarbitone and perfused two hours after the saline infusion.
mRNA expression studies
The rats were anesthetized with pentobarbital and perfused transcardially with cold 400 ml phosphate-buffered saline (PBS). TNCs were carefully dissected out and stored at −80℃ for later use. The tissues were homogenized thoroughly in RLT buffer (Qiagen® GmbH, Hilden, Germany) with a rotor-stator homogenizer. Total RNA was purified using an RNeasy Mini Kit (Qiagen® GmbH, Hilden, Germany) and its yield and purity were assessed spectrophotometrically (Nanodrop 200C, Thermo Fisher Scientific Inc). The cDNA synthesis and quantitative polymerase chain reaction (qPCR) analysis were performed as described previously (9). Briefly, cDNA was synthesized using the QuantiTect Reverse Transcription Kit (Qiagen), and all samples were subjected to reverse transcription-PCR (RT-PCR). qPCR was performed with LightCycler technology (Roche) and QuantiFast SYBR Green I dye (Qiagen). mRNA was quantified using a calibrator-normalized relative quantification approach. The detection ratios between each target gene and a non-regulated reference gene (β-actin) were used to calculate the relative abundance of mRNA transcripts in each experimental sample. A calibrator was included in every PCR run to provide a constant calibration point for all samples both within a LightCycler run and between LightCycler runs. Consequently, all final expression data were expressed as a target/reference ratio in experimental samples, normalized by the target/reference ratio of the calibrator. The TNC of the capsaicin-treated rat was used as the calibrator for all the target genes.
The primers used in the experiment were designed by Qiagen from NCBI GenBank extracted sequences (acc. no.).

NCBI: National Center for Biotechnology Information; TNC: trigeminal nucleus caudalis. Amplicon length denotes the expected size of amplified PCR products, as provided by Qiagen.
Antibody characterization
A list of primary and secondary antibodies used for Western blot (WB) and immunohistochemistry (IHC).

Ms: mouse; Rb: rabbit; HRP: horseradish peroxidase; Ig: immunoglobulin; p-ERK: p-extracellular signal-regulated kinase; p-CREB: p-cyclic AMP-response element binding protein; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
c-Fos and Zif268 IHC
At two and four hours after the end of the infusion, the animals were sacrificed with a high dose of pentobarbitone injection and perfused with PBS (7.4 pH) and paraformaldehyde (PFA) (4% in 7.4 pH PBS). TNCs were removed and post-fixed overnight in 4% PFA. The tissues were then stored in 30% sucrose solution in PBS for four days. The brain stems and upper cervical spinal cords were sectioned serially with a freezing microtome (Leica CM 3050S, Leica Biosystems Nussloch GmbH, Germany) from 0.5 mm rostral to obex to the C2 level (150 ± 5 sections), and every seventh 40-µm section (23 selected sections) was saved and processed for IHC. The immunolabeling procedures were performed according to the Avidin-Biotin-peroxidase Complex (ABC) method as described elsewhere (22). Sections were incubated overnight with polyclonal rabbit anti-c-Fos or anti-Zif268 antibody. Biotinylated donkey anti-rabbit immunoglobulin (Ig)G used as the secondary antibody. Buffer controls omitted either primary or secondary antibodies. The sections were subsequently mounted onto the glass slides and coverslipped, and observed through a Leica DMR HCS microscope using a 20× objective lens. Counting of c-Fos- and Zif268-positive cells in the region of interest was performed by an observer (c-Fos counting by ST and Zif268 counting by DKB) blinded to the treatment. The total numbers of c-Fos-positive cells were counted in lamina I–II of the TNC including the medullary and upper cervical dorsal horn. The distribution was counted from 0.5 mm rostral to obex to the C2 level. c-Fos counting was also performed in the nucleus tractus solitarius (NTS) and caudal ventrolateral medulla (CVLM), integrative sites for processing cardiovascular signals in the brain stem. The representative images of the treated and control sections were captured using a Nikon DS-Fi2 digital camera attached to a Nikon Eclipse (Ni) microscope.
Western blot
The rats were anesthetized with pentobarbital at 30 minutes and two hours post-infusion and perfused transcardially with ice-cold PBS. Dura mater, TG and TNC were isolated and immediately frozen in dry ice. Samples were grounded using a mortar and pestle kept in dry ice and transferred to a micro-centrifuge tube containing 300 µl lysis buffer. Tubes were left on ice for a minimum of half an hour. Afterwards, samples were briefly sonicated and were centrifuged at 12,000 g for 20 minutes at 4℃. Supernatant were collected and stored at −80℃ until further processing. Bio-Rad DC protein assay kit (Bio-Rad, CA, USA) was used to measure protein content. Protein samples were diluted with sample buffer and heated at 95℃ for five minutes, immediately kept on ice and centrifuged. Proteins were separated by sodium dodecyl sulfate polyacrylamide gel (12%) electrophoresis; 25 µg protein was loaded into the wells. The separated proteins were transferred to a polyvinylidene difluoride (PVDF) membrane. The PVDF blots were blocked in 5% bovine serum albumin (BSA) in tris-buffered saline containing 1% Tween-20 (TBS-T) for one hour at room temperature. The primary antibodies were made in 5% BSA dissolved in 1% TBS-T. PVDF blots were kept in different primary antibodies overnight at 4℃. The next day, PVDF blots were washed three times with TBS-T for 10 minutes each wash. The respective horseradish peroxidase-conjugated secondary antibodies were diluted in TBS-T and PVDF blots were incubated in this for one hour at room temperature. The PVDF blots were washed three times with PBS-T for 10 minutes each wash and detection was performed using the Amersham™ ECL™ Select Western Blotting Detection Reagent kit. The PVDF blots were scanned by an electronically cooled CCD camera system (Fujifilm LAS-1000) and chemifluorescent images were captured and stored digitally. Densitometric analysis was conducted for each band by Multi Gauge V3.2 and expressed as ratio to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) signal (loading control). Densitometric data were graphically visualized. For the expression of different proteins of interest in the same PVDF blot, the blots were stripped using stripping buffer followed by several washes and treated as mentioned above.
Data analysis
All values are presented as mean ± SEM. For blood pressure experiments, one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison test was used to determine the significant difference from the saline-treated group. For comparing c-fos mRNA and c-Fos-immunopositive cell count per section between different time points and treatments, Kruskal-Wallis test followed by Dunnett’s post hoc test were used. Mann-Whitney test or unpaired t-test was used for analysis of non-paired data. Differences were considered significant at p < 0.05. GraphPad Prism 5 (GraphPad Prism Software Inc, San Diego, CA, USA) was used for statistical analysis.
Results
Effect of CGRP infusion on MABP in unanesthetized rats
The 0.25, 0.5 and 1.0 µg kg−1 minute−1 infusion of CGRP for 20 minutes caused a significant fall in MABP of awake rats (N = 6) (Figure 1), whereas the infusion of 0.1 µg kg−1 minute−1 of CGRP for 20 minutes did not change the MABP significantly. For further studies of c-Fos expression, we chose to infuse 0.25 and 1 µg kg−1 minute−1 (for 20 minutes) of CGRP.
Percentage change in mean arterial blood pressure after increasing doses of CGRP infusion in conscious normotensive rats. One-way ANOVA followed by Dunnett’s multiple comparison test was used to determine the significant difference from the saline-treated group. *p < 0.05; **p < 0.01; ***p < 0.001 (n = 5–8).
c-fos mRNA expression
We studied the c-fos expression in the TNC as a marker of neuronal activation. After CGRP infusion (0.25 µg kg−1 minute−1 for 20 minutes), c-fos mRNA levels were unchanged at all time points (20 minutes, 30 minutes, one hour, two hours, four hours, six hours) in the TNC, compared to the saline-treated group (Figure 2(a)). However, at the higher (1 µg kg−1 minute−1 for 20 minutes) dose of CGRP, a non-significant increase in c-fos mRNA was observed in the TNC samples at the two-hour time point (Figure 2(b)).
Normalized ratio of c-fos mRNA in TNC at different time points after CGRP (a) (0.25 µg kg−1 minutes−1, 20 minutes) and (b) (1.0 µg kg−1 minutes−1, 20 minutes) intravenous infusion in unanesthetized rats. Kruskal-Wallis test followed by Dunnett’s post hoc test were used to determine the significant difference from the saline-treated group.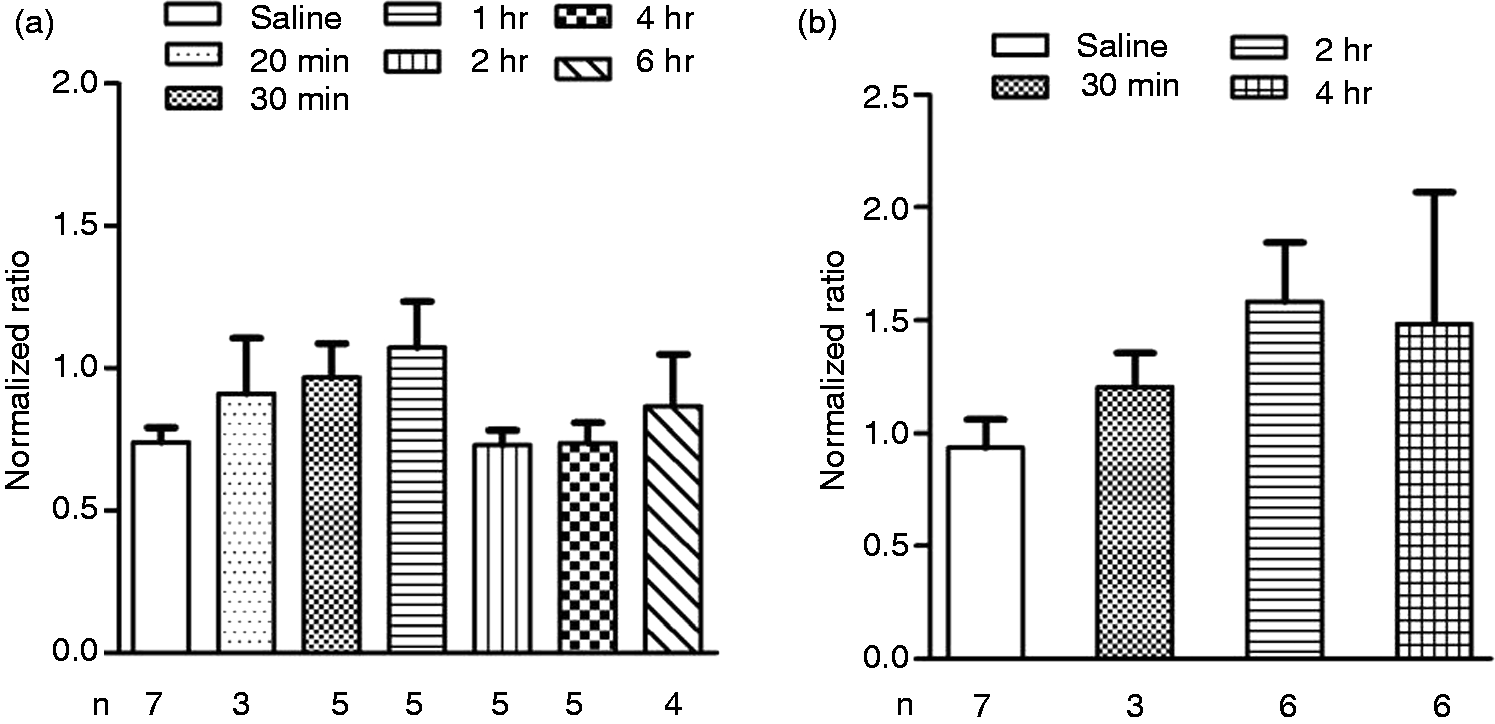
c-Fos and Zif268 protein expression
Representative images of c-Fos-immunoreactivity in the NTS and CVLM two hours after CGRP infusion is shown in Figure 3. Representative images of c-Fos and Zif268 immunoreactivity in the spinal trigeminal nucleus is shown in Figure 4. There was no significant difference observed in c-Fos-immunopositive cells per section in the lamina I–II of the TNC comparing CGRP (1 µg kg−1 minute−1, 20-minute infusion, two hours and four hours post-infusion) and saline-treated rats (Figure 5(a)). c-Fos expression in the NTS (Figure 5(c)) and CVLM (Figure 5(e)) was significantly increased two hours after the CGRP infusion. Four hours after the CGRP infusion, c-Fos expression in the NTS (Figure 5(c)) and in the CVLM (Figure 5(e)) was not significantly different from saline-infused rats. A lower dose of CGRP (0.25 µg kg−1 minute−1, 20-minute infusion, two hours post-infusion) also increased c-Fos expression in NTS (4 ± 1.7 (saline treated) to 10 ± 1.5, p = 0.004, n = 7) and in CVLM (2 ± 0.3 (saline treated) to 10 ± 1.3, p < 0.0001, n = 7) significantly, but it did not increase c-Fos count in TNC (8 ± 1.5 (saline treated) to 4 ± 0.8, p = 0.08, n = 7).
Representative image of c-Fos-immunoreactivity in the nucleus tractus solitarius (NTS) and caudal ventrolateral medulla (CVLM). Cross-sections through the medulla with the NTS and CVLM stained for the activation marker c-Fos in neuronal nuclei. Representative specimen fixed two hours after a 20-minute intravenous (i.v.) CGRP (1 µg kg−1 minute−1) infusion in unanesthetized rats. Magnification bars 100 µm. Representative images of c-Fos (a) and Zif268 (b) immunoreactivity in the spinal trigeminal nucleus. Cross-sections through the medulla with the trigeminal nucleus caudalis stained for the activation marker c-Fos and Zif268 in the neuronal nuclei. Representative specimen fixed two hours after a 20-minute intravenous (i.v.) CGRP (1.0 µg kg−1 minute−1) infusion in unanesthetized rats and stained for c-Fos and Zif268. (a)1 and (b)1 are areas showing mediolateral trigeminal nucleus caudalis at higher magnification. Magnification bars 100 µm. Changes in c-Fos-immunoreactive cell count per section at two hours and four hours after intravenous (i.v.) CGRP (1 µg kg−1 minute−1, 20 minute) infusion in unanesthetized rats, compared to the saline-infused rats. (a) TNC; (c) NTS and (e) CVLM. ***p < 0.001 and **p < 0.01 (Kruskal-Wallis test followed by Dunnett’s post hoc test). Changes in c-Fos-immunoreactive cell count per section after saline (20 minutes) infusion in anesthetized rats, compared to the saline infusion (20 minutes) in unanesthetized rats. (b) TNC; (d) NTS, and (f) CVLM. ***p < 0.001 and **p < 0.01 (Mann-Whitney test, compared to control rats).


Two hours after saline infusion in the anesthetized rats, the c-Fos expression (per section) in the lamina I and II of the TNC (23 ± 6) (Figure 5(b)), in the NTS (63 ± 12) (Figure 5(d)) and in the CVLM (29 ± 5) (Figure 5(f)) was significantly increased, compared to c-Fos expression in lamina I and II of the TNC (8 ± 1), in the NTS (4 ± 0.4) and in the CVLM (4 ± 1) of saline-treated unanesthetized rats. There was no difference observed in Zif268-immunopositive cells per section in the lamina I–II of the TNC between CGRP- (176 ± 18, n = 12, two hours post-infusion) and saline- (150 ± 27, n = 12, two hours post-infusion) treated rats.
Western blot
A characteristic band of the p-ERK protein was identified at 42 and 44 KDa. A significant increase in the intensity of the p-ERK band was observed in the dura mater samples 30 minutes after CGRP (1 µg kg−1 minute−1, 20 minutes) infusion, compared to vehicle control (Figure 6(a)). Phospho-CREB (Ser133) antibody detects endogenous levels of CREB only when phosphorylated at serine 133 and shows a band at 43 KDa. Thirty minutes after the CGRP infusion, p-CREB expression did not change in the dura mater (Figure 6(a)). Two hours after the CGRP infusion, no significant changes in the levels of p-ERK and p-CREB were observed in the dura mater (Figure 6(b)). Thirty minutes (Figure 7(a)) and two hours (Figure 7(b)) after the CGRP infusion, p-ERK levels were not changed in the TG. Furthermore, there was no significant change in the expression of p-ERK (p = 0.06), p-CREB and c-Fos levels observed in the TNC at 30 minutes (Figure 8(a)) and two hours (Figure 8(b)) after the CGRP infusion.
Western blot analysis of p-ERK and p-CREB in the dura mater 30 minutes (a) and two hours (b) after a 20-minute intravenous (i.v.) CGRP (1.0 µg kg−1 minute−1) infusion in unanesthetized rats. *p < 0.05, unpaired t-test. Western blot analysis of p-ERK and p-CREB in the TG 30 minutes (a) and two hours (b) after a 20-minute intravenous (i.v.) CGRP (1.0 µg kg−1 minute−1) infusion in unanesthetized rats. Western blot analysis of p-ERK, p-CREB and c-Fos in the TNC 30 minutes (a) and two hours (b) after a 20-minute intravenous (i.v.) CGRP (1.0 µg kg−1 minute−1) infusion in unanesthetized rats. *p < 0.05, unpaired t-test.


Discussion
Effect of CGRP infusion on MABP
CGRP infusion decreased blood pressure dose dependently, which is in line with a study showing a blood pressure-lowering effect (–24 ± 4 mmHg) after i.v. bolus injection of CGRP (0.1–5 µg kg−1) in unanesthetized rats (23). As we could not detect an increased expression of c-Fos-positive nuclei in the TNC after infusion of the lower dose (0.25 µg kg−1 minute−1, 20 minutes) of CGRP, we decided to infuse a higher dose (1.0 µg kg−1 minute−1, 20 minutes) of CGRP. At this concentration the fall in MABP was distinct; therefore we also investigated c-Fos expression in the NTS and in the CVLM, which together with the rostral ventrolateral medulla (RVLM) are integrative sites for processing cardiovascular signals in the brain stem (24,25). Two hours after infusion of CGRP, both at the lower dose and the higher dose, c-Fos-positive cells in NTS and CVLM were significantly upregulated compared to saline-infused rats. Infusion of CGRP causes a decrease in MABP and an increase in c-Fos-positive cells in NTS and CVLM, which confirms the involvement of the baroreceptor reflex after CGRP infusion.
Effect of CGRP infusion on c-Fos expression in the TNC
CGRP infusion in migraineurs caused an immediate headache during infusion and shortly after a delayed headache resembling spontaneous migraine attacks (6). We assumed that changes in c-Fos protein expression at the two-hour and four-hour time points will match the immediate and delayed phase of human migraine provocation studies, respectively. We have used a validated model in which infusion of GTN in unanesthetized freely moving rats caused an increase of c-Fos in TNC, and pretreatment with sumatriptan (16) and olcegepant (17) attenuated c-Fos expression. Mechanical stimulation of dural blood vessels produced c-Fos-positive labeling primarily in lamina I, whereas facial stimulation produced c-Fos-positive labeling both in lamina I and in the outer part of lamina II (26). Capsaicin and GTN infusion in rats also increased c-Fos-positive labeling only in lamina I and the outer part of lamina II (16,27). The number of c-Fos-positive nuclei were not increased significantly in lamina III–IV in all three previous studies. In the present study, we have counted c-Fos-positive nuclei only in lamina I and the outer part of lamina II. Neither a lower nor higher dose of CGRP activated c-Fos in TNC both at the mRNA level and at the protein level. This finding was supported by two studies: In the first study, both topical (on dura surface) and systemic infusion of CGRP in rats were not able to activate or sensitize mechanosensitive meningeal nociceptors in the TG (28). More recently in the second study, it has been shown that the activity of the spinal trigeminal neurons with meningeal afferent input is normally not controlled by CGRP receptor activation or inhibition in the TG (29), emphasizing that CGRP-induced vasodilation and CGRP application on TG may not be an effective stimulus for the activation of pain. Both studies suggested a more central site of action to explain the facilitatory effect of CGRP on central neuron discharge. In contrast to the previous two studies, it has been reported that a bolus injection of CGRP produced facilitation of responses to innocuous vibrissal stimulation in non-nociceptive brain stem trigeminal neurons (30).
Our findings are in agreement with a number of studies that have failed to detect a hyperalgesic effect of peripherally administered CGRP (31,32). Anti-nociceptive effects of CGRP administered intracerebroventricularly (i.c.v.) and into the nucleus raphe magnus were reported in rats by tail-flick and hot plate tests, respectively (33,34). Furthermore, it has been shown that CGRP receptors are not present on sensory axons in cranial dura mater (7). Activation of meningeal nociceptors resulted in activation of trigeminal neurons and i.v. administration of the CGRP receptor antagonist olcegepant at a very high (0.9 mg kg−1) dose inhibited neuronal activity, whereas the local meningeal application of olcegepant had no effect (35). Pre-treatment with olcegepant (0.9 mg kg−1) inhibited capsaicin-induced c-Fos expression throughout the spinal trigeminal nucleus (18). These results suggest a more central site of action of olcegepant in inhibiting migraine. However, a peripheral mode of action of CGRP cannot be excluded as human positron-emission tomography (PET) studies revealed that central antagonism of CGRP receptors is not necessary for therapeutic efficacy in migraine pain relief (36,37).
Effect of CGRP infusion on Zif268 expression in the TNC
We also tested another IEG, Zif268, which follows a similar expression time profile as c-Fos. IHC for Zif268 was conducted only in limited samples to verify c-Fos data. It has been shown that Zif268 (early growth response protein 1 (EGR-1)) and c-Fos levels were increased in TNC after formalin injection in the whisker pad of the rat (12). Compared to the c-Fos-positive cell, we have seen more Zif268-positive cells in saline-infused rats, which is in accordance with previous studies (12,38). Zif268-positive cells per section were also not increased significantly in TNC two hours after CGRP (1.0 µg kg−1 min−1, 20 minutes) infusion. Both IEGs gave similar results, which further confirmed that CGRP infusion did not activate second-order neurons in TNC.
Our results were not supported by a recent study showing an increase in c-Fos expression in TNC 60 minutes after a single i.v. injection of CGRP (0.6 µg kg−1); however, this study was performed in pentobarbitone-anesthetized rats. Increase in transient receptor potential cation channel subfamily V member 1 (TRPV1), CGRP, phosphorylated protein kinase C and phosphorylated CREB protein was also reported in TG (39). In our study, infusion of saline in ketamine- and xylazine-anesthetized rats caused a significant increase in c-Fos expression in the lamina I–II of the TNC, in the NTS and in the CVLM, compared to saline-infused rats in unanesthetized rats. It is already known that anesthesia and acute surgery by themselves affect expression of c-Fos (15,40,41). The study by Chatchaisak and colleagues (39) raised a few points. First, the authors have shown that after 45 minutes and 60 minutes of CGRP infusion TRPV1 content is increased in TG; even in the chronic pain models a change in TRPV1 expression takes days (42). Second, they have shown an increase in CGRP content in TG after 60 minutes of CGRP infusion; even in TG culture it takes a minimum of four-hours’ incubation of CGRP to promote CGRP mRNA synthesis (43). Furthermore, in isoflurane-anesthetized rats four hours after i.v. infusion of GTN (250 µg kg−1 over a period of two hours), an increase in CGRP and neuronal nitric oxide synthase (nNOS) immunoreactivity was reported in the TG (44). After one short bolus infusion of CGRP in rats, the 60-minute time window is very short to expect increased TRPV1 and CGRP protein expression. In the rat closed-cranial window (CCW) model, a 20-minute i.v. infusion of CGRP caused a robust increase in middle meningeal artery (MMA) diameter and decrease in blood pressure; these effects returned to normal baseline within 10 minutes after the infusion was stopped (45). We have used the same cannulas (polyurethane) for CCW and infusion studies. A robust increase in MMA diameter by CGRP infusion excludes the possibility of any absorption of CGRP by the polyurethane cannula.
Western blot
We have seen a significant activation of p-ERK in the dura mater 30 minutes after CGRP infusion. The mitogen-activated protein kinase (MAPK) cascades represent major downstream effectors of CGRP (46). CGRP receptor activation produced protein kinase A (PKA) and protein kinase C-dependent mechanical hyperalgesia and central sensitization (47). ERK signaling pathway plays an important role in peripheral sensitization after noxious stimulation to the peripheral tissues (48). Both c-Fos and p-ERK are used as a marker of neuronal activation and central sensitization after noxious stimulation tissue injury (49). Compared to c-Fos, pERK can be induced within a minute, which can be correlated well with the induction of immediate pain after noxious stimulus. c-Fos is localized in the nuclei of neurons but pERK can be induced in different subcellular structures of neurons such as nuclei, cytoplasm, axons and dendrites (49). Recently, it was reported that infusion of GTN evoked a delayed meningeal nociceptor sensitization that was associated with a robust ERK phosphorylation in the meningeal arteries (50).
We did not see any delayed activation of p-ERK two hours after CGRP infusion in the dura mater and in the TNC. Capsaicin-stimulated p-ERK activation was reported in the TG within 10 minutes, and the p-ERK activation was not affected by pre-treatment with the CGRP receptor antagonist, olcegepant (27). ERK is the principal mediator of Ca2+-dependent c-Fos induction via two different modules, (i) ERK/Elk-1 (on serum response element, SRE site) and (ii) ERK/? (probably via an indirect mechanism involving the family of ribosomal S6 kinase, RSKs)/CREB (on cAMP-response element, CRE site), a pathway positively controlled by calcium-calmodulin kinases (CaM-Ks) (20,51). Previously, CREB expression was reported to be upregulated by CGRP in cultured spinal neurons and in a primary culture of TG enriched for glia (52,53). However, we did not see activation of p-ERK and p-CREB in TG 30 minutes and two hours after CGRP infusion.
Clinical implications
Calcitonin gene-related peptide (CGRP) infusion activates blood pressure-regulating centers in the brain stem. CGRP infusion increased the early expression of p-extracellular signal-regulated kinase (p-ERK) in the dura mater. CGRP-induced vasodilation of dural arteries is not enough to activate neurons in the trigeminal nucleus caudalis. The rat CGRP infusion model with c-Fos or Zif268 as neuronal pain markers in TNC is unsuitable for antimigraine drug testing.
Footnotes
Funding
This work was supported by an International Mobility Grant (Faculty of Health and Medical Sciences, University of Copenhagen), Candy’s Foundation, Lundbeck Foundation and the A.P. Møller Foundation for the Advancement of Medical Science.
Conflict of interest
None declared.
