Abstract
Background
Autonomic dysfunction and light sensitivity are core features of the migraine attack. Growing evidence also suggests changes in these parameters between attacks. Though sensory and autonomic responses likely interact, they have not been studied together across the spectrum of disease in migraine.
Methods
We performed digital infrared pupillometry while collecting interictal photophobia thresholds (PPT) in 36 migraineurs (14 episodic; 12 chronic; 10 probable) and 24 age and sex-matched non-headache controls. Quantitative pupillary light reflexes (PLR) were assessed in a subset of subjects, allowing distinction of sympathetic vs parasympathetic pupillary function. A structured questionnaire was used to ascertain migraine diagnosis, headache severity, and affective symptoms.
Results
Photophobia thresholds were significantly lower in migraineurs than controls, and were lowest in chronic migraine, consistent with a disease-related gradient. Lower PPT correlated with smaller dark-adapted pupil size and larger end pupil size at PPT, which corresponded to a reduced diameter change. On PLR testing, measures of both parasympathetic constriction and sympathetic re-dilation were reduced in migraineurs with clinically severe migraine.
Conclusions
In summary, we show that severity of photophobia in migraine scales with disease severity, in association with shifts in pupillary light responses. These alterations suggest centrally mediated autonomic adaptations to chronic light sensitivity.
Keywords
Introduction
Migraine is characterized by paroxysms of head pain accompanied by sensory amplifications and autonomic dysfunction. Diagnosis is currently based on clinical criteria, including throbbing pain that is often unilateral, worsened by activity, and associated with nausea, sensitivity to light (photophobia) and/or sound (phonophobia) (1). Among these hallmark features, photophobia is a common and often disabling component of the migraine attack, serving as a key diagnostic criterion. Some definitions use the term photophobia to refer specifically to triggering and/or exacerbation of headache by light, though light sensitivity may also be present between attacks, as is supported by findings of lower interictal light sensitivity thresholds in migraineurs compared to non-headache controls (2,3). For the purposes of this report, we use the term to refer to discomfort or pain induced by light (light sensitivity), but do not specifically limit this definition to the context of the ocular or head pain.
Efforts to characterize the neural substrates of migrainous photophobia have implicated a number of potential circuit mechanisms, including cortical hypersensitivity to light (4), altered ocular parasympathetic outflow (5), and intrinsically photosensitive retinal ganglion cell pathways acting on dura-sensitive thalamocortical neurons (6), the latter of which may in turn influence adaptation of pupillary responses to light (pupillary light reflex; PLR) (7). These findings, and a robust literature on sensory/autonomic interaction in the trigeminovascular system (8), suggest that the sensory disruptions of photophobia may be linked to craniofacial autonomic dysfunction. Previous studies have focused on the effect of painful stimuli on light sensitivity and craniofacial trigeminovascular function in episodic migraineurs (9,10). To our knowledge, the relationship between photophobia and pupillary function has not yet been directly examined, particularly across the disease spectrum of migraine.
We used clinical assessment, psychophysical and physiologic measures to assess both photophobia and pupillary responses to light in migraine and non-headache subjects. We hypothesized that photosensitivity would be associated with alterations in autonomically-mediated pupil function, which could have important mechanistic and therapeutic implications.
Methods
Participants
In total, 60 subjects (18 male and 42 female) aged 18–75 years were recruited into migraine and age and sex-matched non-headache (NH) control groups. Subjects were recruited between June 2013 and December 2015 from the local community, community clinics and University of Utah headache and outpatient neurology clinics, via word of mouth, internet, and flyer advertisements. Institutional Review Board approval and informed consent were obtained.
Headache diagnosis was based on International Classification of Headache Disorders II criteria (2005) (11). Upon completion of a structured clinical questionnaire, modified from Samaan et. al. 2010 (12), the migraine group was further divided into episodic (EM), chronic (CM), and probable (PM) migraine for a total of 36 migraine subjects (18 migraine with aura, 18 migraine without aura). Subjects were excluded if they had comorbid medical, ocular, or neurological disorders known to directly affect autonomic function, pupillary control, or light sensitivity (e.g. prior eye injury, idiopathic blepharospasm, optic nerve disorder). Subjects were instructed not to consume alcohol, caffeine or nicotine for at least four hours prior to testing. A subset of subjects (n = 30; 11 non-headache (NH) and 19 migraine – all, consisting of 10 EM, four CM, and five PM) returned for PLR testing; age and sex-matching were again ensured for this selected group.
Episodic and probable migraine participants were studied after being headache-free for at least 48 hours, and subjects were excluded from analysis if a migraine occurred within 24 hours of testing. CM subjects were assessed when migraine attack-free for at least 48 hours, though testing during daily or non-migrainous headaches was permitted. Subjects had not used opiate medication or migraine-specific abortive medications during the 48 hours prior to testing, and did not take any medications that could affect autonomic or pupillary function, including eye drops (other than artificial tears), psychotropics, antihistamines, and benzodiazepines or their derivatives. Headache diaries were used to assess attack frequency for one week prior to testing and for subsequent attacks occurring within the two weeks of testing, as well as medication use. The group of age and sex-matched NH control subjects reported no history of recurrent or disabling headaches and were studied in their usual state of health. Five subjects were excluded from photophobia testing due to frequent blinking or intolerance to testing (one NH, one EM, and three CM), for a total of 55 of 60 subjects completing photophobia testing for analysis; two subjects (one PM and one EM) were excluded from PLR testing on account of persistent blink artifact, for a total of 28 of 30 subjects completing PLR testing for analysis.
Measurements
Questionnaire
Subjects completed a modified written Structured Migraine Interview (SMI) (12) along with a headache diary to characterize migraine diagnosis and headache frequency; additional questions were included to ascertain how many days per month the participant experienced their “typical migraine” type headache, how often they experienced “any other” type of headache, and whether their headaches began following a head injury. For expanded symptom characterization, we included questionnaire items to assess photophobia (Short Form Photophobia Questionnaire (2)) and affective symptoms (Patient Health Questionnaire-9 (PHQ-9) (13); Generalized Anxiety Disorder-7 (GAD-7) (14)). Migraine Disability Assessment (MIDAS) (15) and Headache Impact (HIT-6) (16) were also collected as well-validated clinical measures of disease burden. A MIDAS score ≥6 is used to define the presence of migraine with related disability (grade II or greater) (15); similarly, a HIT-6 score ≥50 was used to define significant headache related functional impairment (17).
Photophobia threshold testing
Psychophysical assessment of light sensitivity thresholds was adapted from Adams et al. (18). Subjects were positioned in a comfortable seated position facing two white-light 500 W halogen lamps (SL-1002; Bayco, Wylie, TX). A 12 × 12 in double-pane sheet of infrared-filtering glass (Solarban 60, ¼ in Clear; Oldcastle, Atlanta, GA) was mounted 20 cm from lamps and 35 cm from the subject’s eyes to reduce heat exposure. Subjects underwent dark-adaption for three minutes prior to testing. Using a calibrated rheostat (LX1010B Luxmeter), light intensity was increased in a fixed stepwise fashion at two-second intervals from 0.1 lux to photophobia threshold, or a maximum of 16,450 Lux (0.1, 0.2, 2.5, 10, 30, 73, 146, 242, 395, 591, 883, 1203, 1663, 2220, 2810, 3590, 4490, 5420, 6550, 7830, 9180, 1064, 12220, 13800, 15520, 16450). Subjects were asked to look at a fixed point between the two light sources and to say “stop” when the intensity of light became “uncomfortable” (defined as the level of stimulus that made the subject want to blink or turn away from the light source, and physiologically confirmed by video review of grimace). Testing was repeated three times with a three-minute dark adaptation between each trial; the light intensity of the final position on the rheostat was considered the photophobia threshold for that trial and was reported in log(lux).
Pupil responses during photophobia threshold testing were recorded using an infrared camera positioned to include a standard 5 mm reference in the field of view from which pupil size could be measured. Recordings were saved for offline analysis in ImageJ version 1.48u4. Baseline dark-adapted and maximally constricted pupil size at photophobia threshold were measured and reported in millimeters. If maximally constricted pupil size at photophobia threshold could not be visualized due to obstruction from eyelids, the recording was excluded from further analysis.
Quantitative pupillary light reflex (PLR) testing
To independently assess parasympathetic and sympathetic contributions to the PLR arc, we utilized a standardized PLR protocol adapted from Muppidi et al (19) and a binocular pupillometer (DP-2000, Neuroptics Inc, Irvine, CA; image acquisition 30 Hz, pixel resolution 0.05 mm) to precisely quantify pupillary diameter changes to a standardized stimulus. Following one minute of dark adaptation, a light stimulus was presented to one eye at a time using a circumferential array of white light-emitting diodes. Three trials separated by one minute of dark adaptation were completed in each eye, and then repeated in the opposite eye. The only ambient light in the exam room (from the computer screen of the pupillometry device) was shielded and directed away from the subject. Dynamic recordings of pupil diameter were saved for offline analysis.
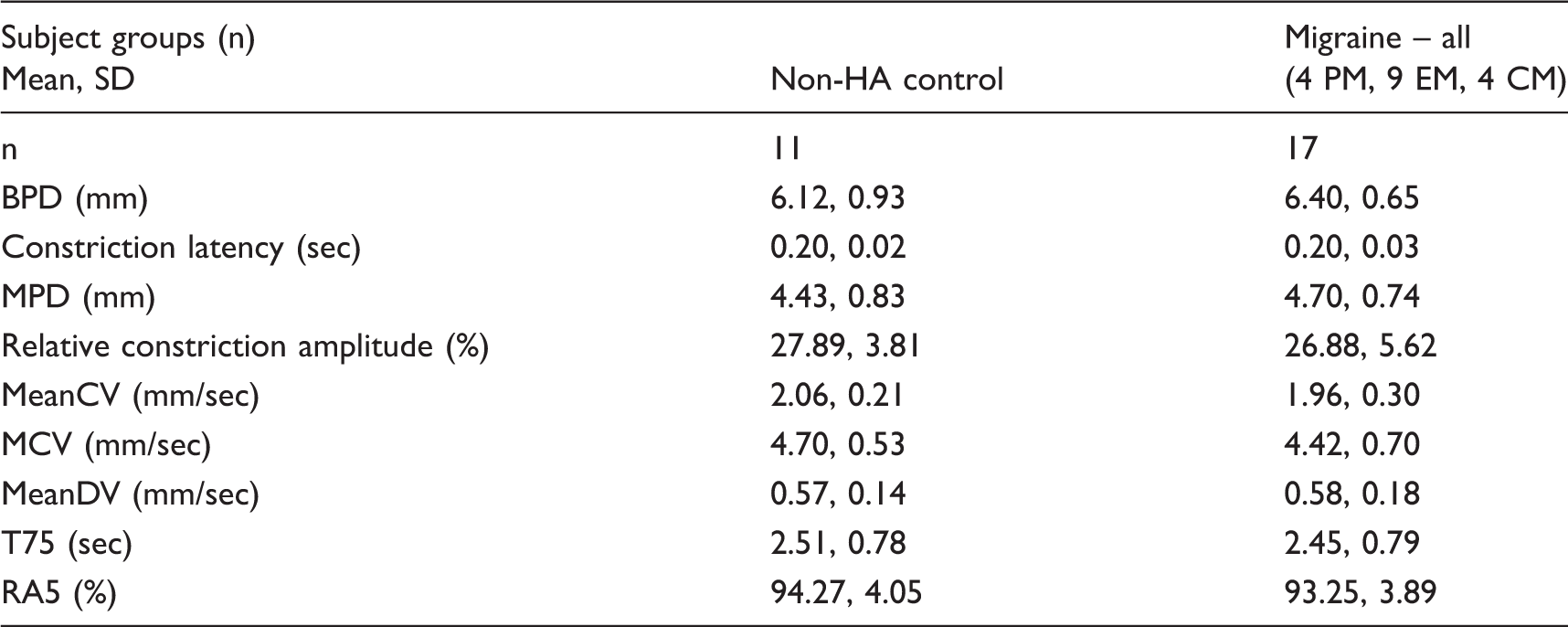
In pilot experiments in four healthy subjects, the effects of variation in intensity, duration of light stimulus, and recording length (during which time subjects are asked to avoid blinking) were examined. Based on these results, a standard brief light stimulus (40 ms duration, 2.30 Lux) and recording duration (five seconds) was used in all further studies. The following standardized indices of pupillary constriction and dilation in response to each light stimulus were assessed: baseline pupil diameter (BPD), latency to constriction onset, minimal pupil diameter measured at the end of constriction (MPD), constriction amplitude (CA), maximum constriction velocity (MCV), mean constriction velocity (meanCV), mean dilation velocity (meanDV), time to reach re-dilation to 75% of BPD (T75), and five-second re-dilation amplitude relative to BPD (RA5) (19) (Figure 1). For analysis of all PLR parameters, the three trials performed in each eye were averaged to provide a total of two measurements for each subject, one for the right and one for the left. Because no significant side-to-side differences in PLR measures were found, measures from both eyes were pooled for analysis.
Normal pupillograph with light reflex indices. Baseline pupil diameter (BPD), constriction latency, maximum constriction velocity (MCV), mean constriction velocity (MeanCV), minimum pupil diameter (MPD), constriction amplitude (CA), mean dilation velocity (MeanDV), time to 75% re-dilation (T75) and re-dilation amplitude at five seconds (RA5).
Pupillary measures were visualized and analyzed using NeurOptics software and MATLAB (Version 2015b; Mathworks, Natick, MA). BPD was calculated as the average pupil size during the interval from stimulus onset (time zero) to onset of the constriction. CA was determined and expressed as a percentage (BPD-MPD/BPD*100%). MeanCV was calculated by averaging the constriction velocities that occurred after the light stimulus was delivered until the minimum pupil diameter was reached. MCV was calculated as the maximum of the first derivative during the constriction. MeanDV was determined by averaging the dilation velocities from the minimum pupil diameter to the end of the trial. T75 was calculated by first determining the 75% recovery diameter ((BPD-MPD*0.75) + MPD) and recording the time at which the 75% recovery diameter was achieved; T75 was recorded as the time elapsed between MPD and 75% recovery. RA5 was calculated using the formula (pupil diameter at 5 sec/BPD*100%) and expressed as a percentage.
While the constriction phase of the PLR is primarily a parasympathetic function, re-dilation following a brief light stimulus occurs in two phases: 1) an early rapid phase influenced by combined parasympathetic withdrawal and sympathetic activation, and 2) a later, slower phase predominantly controlled by sympathetic innervation (20). Based on this, meanDV likely reflects mixed parasympathetic and sympathetic influences. For the purposes of interpretation in this study, constriction latency, MCV and meanCV were considered reflective of parasympathetic function, whereas T75 and RA5 were considered most reflective of sympathetic function (20) (Figure 1).
Statistical methods
Data distributions were visually inspected and tested for normality using the Shapiro-Wilk test; overall, the PLR and photophobia threshold were found to be normally distributed whereas most clinical parameters, including GAD-7, PHQ-9, headache days per month, MIDAS, HIT-6 and age, were not. Normally distributed data was analyzed using one-way ANOVA and post-hoc two-sided independent samples t-tests with Bonferroni correction. For non-parametric data, the Kruskal-Wallis rank sum test was utilized for across-group comparisons, with post-hoc pair-wise comparisons using two-sided Wilcoxon rank sum test with Bonferroni correction. Correlations were made initially using two-sided Spearman’s correlation as a conservative method, and confirmed with age-adjusted partial correlations with Holm’s correction for multiple comparisons. Results were considered significant for p values < 0.05. Statistical analyses were performed with R for Windows (Version 3.1.0; R Core Team, Vienna, Austria.)
Results
Clinical characteristics
Clinical characteristics.

Kruskal-Wallis rank sum test across subgroups, NH vs PM vs EM vs CM:
Non-significant – years of age: p = 0.72,
Abbreviations: CM, chronic migraine; EM, episodic migraine; F, female; GAD-7, generalized anxiety disorder-7; IQR, interquartile range; M, male; NH, non-headache; PHQ-9, patient health questionnaire; PM, probable migraine; SD, standard deviation.
Headache sample clinical characteristics.

Kruskal-Wallis rank sum test across subgroups, PM vs EM vs CM:
HA days/month –
MIDAS –
HIT-6 – p = 0.22.
Abbreviations: CM, chronic migraine; EM, episodic migraine; HA, headache; HIT-6, headache impact test; IQR, interquartile range; MIDAS, migraine disability assessment; PM, probable migraine; SD, standard deviation. Bold text is used to emphasize the groups comparisons and their corresponding p-value (stat of significance).
Photophobia thresholds
Photophobia thresholds differed significantly across groups, generating an apparent gradient of disease severity with the lowest thresholds in CM, followed by EM and PM. Pairwise comparisons between migraine subgroups and NH showed that this gradient was driven by CM (Figure 2, Table 3). When stratifying disease severity based on clinical measures of headache impact (HIT-6) and headache related disability (MIDAS) (21), photophobia thresholds were again found to be significantly lower in those with higher headache severity scores (Figure 2b and c, mean photophobia threshold 2.2 in HIT-6 ≥50 vs. 2.5 in HIT-6 score <50, p = 0.03; mean threshold 2.1 in MIDAS ≥6 vs. 2.5 in MIDAS < 6, p = 0.002, two-sample t-test.
Interictal photophobia thresholds differ significantly in (a) ICHD-II defined migraine subgroups compared to non-headache controls (single-asterisk, indicates significant difference across all groups; p = 0.003, one-way ANOVA. Double-asterisk indicates significant pair-wise difference from NH; p = 0.001, post-hoc pair-wise independent t-test). Photophobia thresholds are also lower in those with the most clinically severe migraine (b) HIT-6 ≥50 vs <50 (p = 0.03, 2-sample t-test) and (c) MIDAS ≥6 vs <6 (p = 0.002, 2-sample t-test). Note the effect of both disease and disease severity, with the lowest thresholds in chronic migraine and those with a high disease burden. There were no statistically significant differences between probable, episodic, and chronic migraine when stratified by HIT-6 or MIDAS (not shown). Photophobia threshold parameters. One-way ANOVA test across subgroups, NH vs PM vs EM vs CM: Photophobia threshold – Non-significant – dark-adapted pupil size: p = 0.22, pupil size at PPT: p = 0.57, relative constriction amplitude: p = 0.42 Abbreviations: CM, chronic migraine; EM, episodic migraine; mm, millimeter; NH, non-headache; PM, probable migraine; PPT, photophobia threshold; SD, standard deviation. Bold text is used to emphasize the groups comparisons and their corresponding p-value (stat of significance).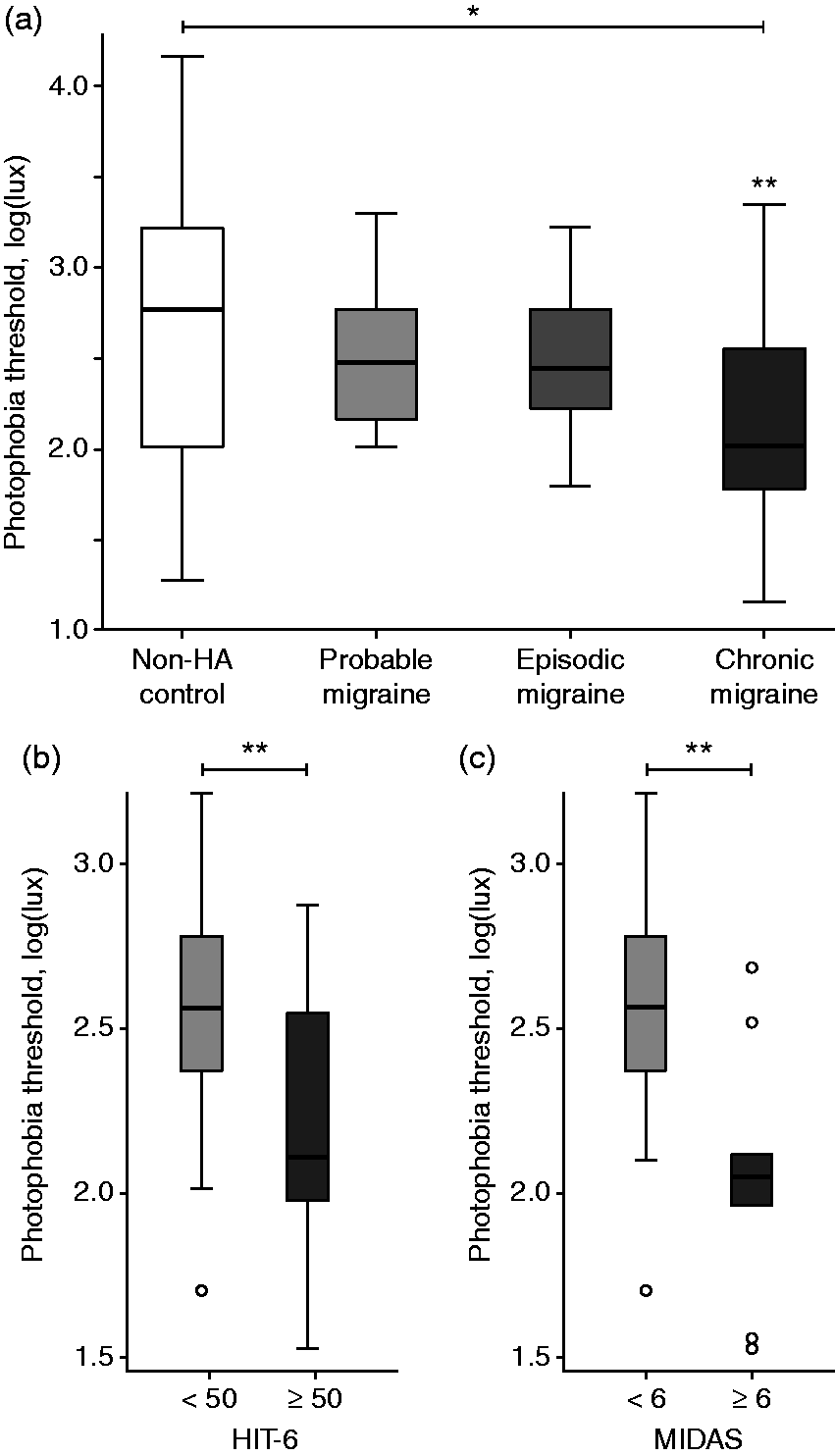

Dark-adapted pupil size, size at photophobia threshold, and relative amplitude change during photophobia testing did not significantly differ between NH and migraine subgroups (Table 3). However, among migraine subjects there was a significant correlation between photophobia thresholds and dark-adapted pupil size at baseline (Figure 3a), and pupil size at photophobia threshold (Figure 3b), which was confirmed by a positive correlation between photophobia thresholds and overall amplitude of constriction at photophobia threshold (Figure 3c). The lowest photophobia thresholds were associated with the smallest dark-adapted pupil sizes, largest end pupil size at photophobia threshold, and lowest overall constriction amplitudes. Interestingly, the latter relationship showed a gradient across migraine subgroups, with the strongest correlation in CM > EM > PM (Figure 3d). Importantly, we did not observe this same pattern of correlations in non-headache controls.
Photophobia thresholds correlate with pupillary responses to light in migraineurs. Lower photophobia threshold correlates with (a) smaller dark-adapted/baseline pupil size and (b) larger end-pupil size at photophobia threshold in migraineurs (age-adjusted partial correlation rho 0.52, p < 0.0001 and rho −0.30, p = 0.04 respectively; n = 28, 73 measurements). (c) This pattern is associated with reduced amplitude of pupil diameter change in those with lower photophobia threshold (age-adjusted partial correlation rho 0.52, p < 0.0001). This overall pattern in baseline and end pupil diameter was not observed in non-headache controls. (d) These adaptations to light sensitivity appear to scale with disease severity, showing a gradient of correlation across migraine subgroups, with the strongest correlation in CM (black line/circles; age-adjusted partial correlation rho = 0.81, p < 0.0001; n = 8, 22 measurements) > EM (dotted line/triangles; rho = 0.37, p = 0.04; n = 11, 31 measurements) > PM (dashed line/squares; rho = 0.22, p = 0.75; n = 9, 20 measurements).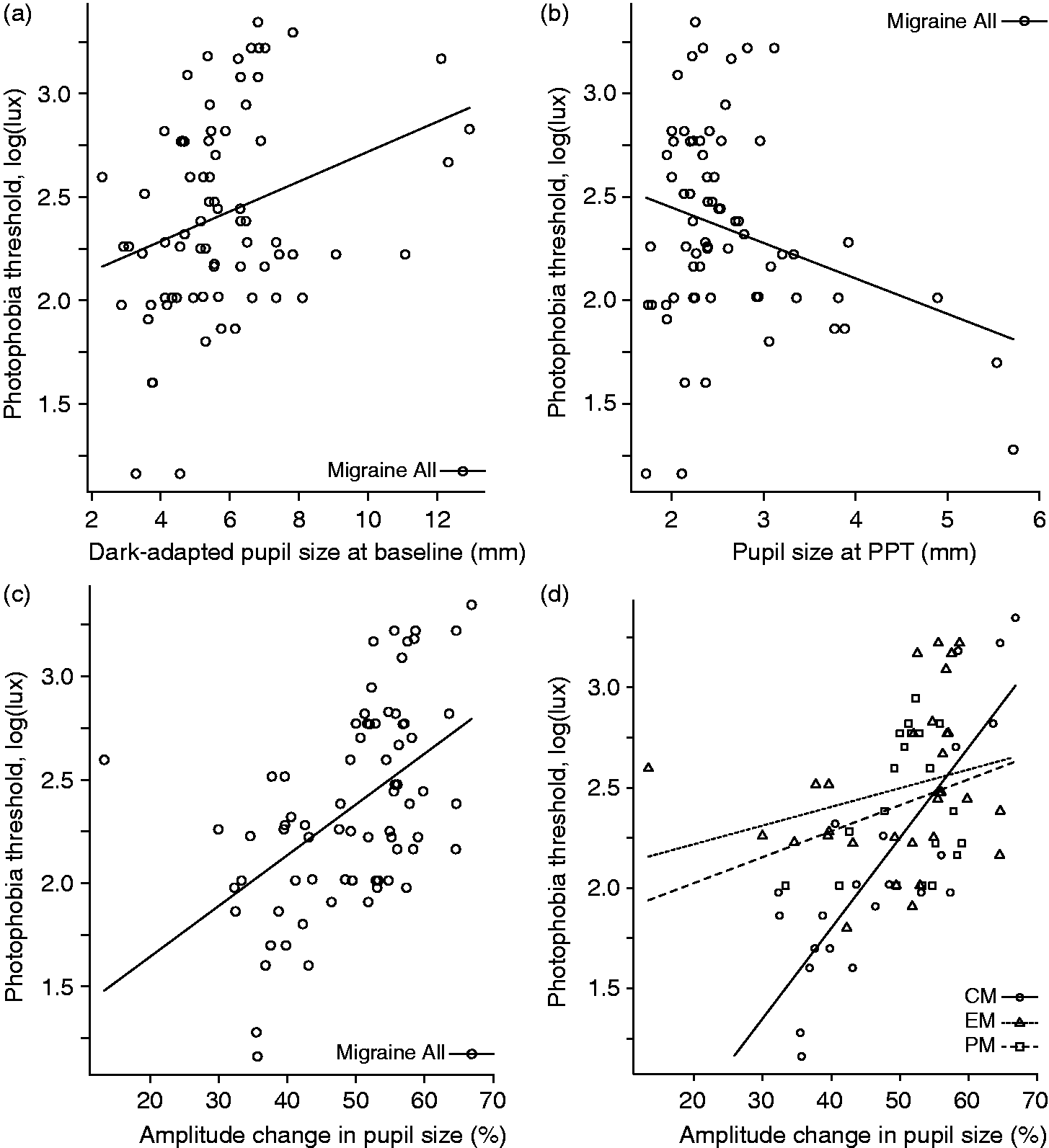
Altered PLR in migraine subjects
In order to assess possible autonomic mechanisms, we collected quantitative PLR data in a subset of 17 out of the 26 migraine (migraine – all; four of 10 PM, nine of 14 EM, four of 12 CM) and 11 of the 24 NH subjects. Due to a lower total number of subjects, ICHD-II subgroups were analyzed together. In order to maintain a measure of disease severity across the migraine group, we stratified based on migraine severity, with HIT-6 score ≥50 and MIDAS ≥6 (21) representing the most clinically affected migraine subjects.
Pupillary light reflex results.
Independent samples t-test, NH vs migraine – all:
Non-significant – BPD: p = 0.24, constriction latency: p = 0.59, MPD: p = 0.34, Relative constriction amplitude: p = 0.40, MeanCV: p = 0.22, MCV: p = 0.97, MeanDV: p = 0.64, T75: p = 0.84, RA5: p = 0.39. NH, n = 11 subjects, 22 measurements; PM, n = 4 subjects, 8 measurements; EM, n = 9 subjects, 18 measurements; CM, n = 4 subjects, 8 measurements.
Abbreviations: BPD, baseline pupil diameter; CM, chronic migraine; EM, episodic migraine; HA, headache; MCV, maximum constriction velocity; MeanDV, mean dilation velocity; MeanCV, mean constriction velocity; MPD, minimum pupil diameter; NH, non-headache; PM, probable migraine; RA5, re-dilation amplitude at five seconds; T75, time to 75% of BPD; SD, standard deviation.

Pupillary light reflex kinetics are altered in the most severe migraineurs. (a) Representative traces of pupillary light reflex responses in mild (HIT-6 <50 and MIDAS <6; thin black line) and severe (HIT-6 ≥50 and MIDAS ≥6; thick grey line) migraine subjects, illustrating differences in parasympathetically and sympathetically mediated responses to light. (b and c) Constriction latency (parasympathetic) was prolonged in those with MIDAS ≥6 (p = 0.03, two-sided independent samples t-test); latency was also longer in those with HIT-6 ≥50, though this was non-significant (p = 0.59, two-sided independent samples t-test). (d–g) Sympathetic re-dilation measures were also altered in those with the most severe headaches, with a longer time to reach 75% of baseline pupil size (T75) (p = 0.009, two-sided independent samples t-test) and reduced relative re-dilation amplitude at five seconds (RA5) (HIT-6 p = 0.003 and MIDAS p = 0.006, two-sided independent samples t-tests). T75 was also longer in those with MIDAS ≥6, though this was non-significant (p = 0.36, two-sided independent samples t-test).
Discussion
Photophobia is a hallmark feature of migraine headache, and has been proposed as a potential contributor to central sensitization in chronic migraine (22,23). Yet the underlying physiological mechanisms of migrainous photophobia remain poorly understood. Using clinical, psychophysical and physiologic measures, we assessed pupillary function relative to interictal photophobia. Our principal finding was that of mixed parasympathetic and sympathetic pupillary dysfunction associated with lower interictal photophobia thresholds in migraine subjects. Importantly, we also observed a gradient of disease, with the lowest photophobia thresholds in chronic migraineurs (Figure 2). Low photophobia thresholds were associated with altered dark-adapted size and pupillary constriction amplitudes at photophobia threshold (Figure 3), and greater impairment of both parasympathetically mediated constriction latency and sympathetically mediated pupillary re-dilation were observed in those with the most clinical impairment based on measures of disease severity (Figure 4).
While prior studies have reported either isolated parasympathetically or sympathetically mediated pupillary deficits in migraineurs (24–26), more recent work using contemporary techniques and diagnostic definitions has shown mixed parasympathetic and sympathetic dysfunction (27,28). Importantly, few studies have formally assessed disease severity within their subject sample. Based on the observation that only selected subsets of migraine subjects demonstrated alterations in craniofacial sympathetic function (29), Drummond postulated that these changes may only arise in those with the most severe headache. In agreement with this hypothesis, we found altered pupillary light responses predominantly in those with clinically more severe migraine (Figure 4). However, these differences were not limited to sympathetic dysfunction, and included both parasympathetic and sympathetically mediated pupillary alterations. Moreover, we observed lower photophobia thresholds in chronic migraine (Figure 2), which were associated with lower amplitudes of pupillary constriction at photophobia threshold (Figure 3c and d). Our findings support the overall hypothesis of mixed interictal pupillary dysfunction in migraine, and link it to both light sensitivity and disease severity.
Possible disease-based circuit alterations
The correlation between photophobia and altered pupillary responses to light suggests the possibility of a causal relationship, though of course the direction of causality cannot be definitively determined here, and indeed the observed relationship may be due to an independent, unmeasured cause. An intuitive hypothesis is that sensitivity to light leads to an alteration in circuit function that favors reduced light entry through a smaller pupil (relative sympathetic hypofunction). Larger pupil size at time of photophobia threshold may simply result from a greater sensitivity to lower light levels (i.e., the psychophysical sensitivity threshold is met before full pupillary constriction is achieved), or could be due to relative parasympathetic pupillary hypofunction, preventing adequate constriction in response to light. Increased light sensitivity with more severe disease could drive larger adaptive (allostatic) changes in circuit function, driving photophobic migraineurs towards a smaller dark-adapted pupil, while mixed interictal pupillary dysfunction further contributes to alteration of pupillary responses to light in those with the most severe migraines.
An opposite hypothesis of mixed autonomic changes could also be entertained, with the disease-based ‘driving force’ being chronic systemic sympathetic activation in response to chronic pain, with relative parasympathetic hyperfunction due to increasing interictal trigeminovascular activation with disease severity. However our measures do not show evidence of sympathetic or parasympathetic hyperfunction relative to either NH controls or to those with the lowest migraine burden (e.g. PM, HIT-6 < 50, MIDAS < 6). Ultimately these hypotheses need to be tested within experimental paradigms that can determine direction of causality, e.g. with demonstration (likely in animal model systems) that induced photophobia results in reduced sympathetic and/or parasympathetic tone, and not vice versa.
Potential limitations
It is important to realize the potential limitations of pupillometric studies. Dark-adapted pupil size can potentially be influenced by age, sex, medication effect and level of alertness. Because a variety of parameters affect pupillary function, we ensured age and sex matching for all comparisons, and tested under standardized conditions, with exclusion of potentially confounding medications. In our study, we also instructed our study subjects to abstain from alcohol, caffeine and nicotine for a period of at least four hours prior to testing. Minimal data is available on which to base potential duration of effect of these agents on pupil function, though there is data to suggest that caffeine effects on pupil function begin to resolve by four hours (30,31), nicotine effects within 20 minutes (32), and alcohol with effects on pupil size that resolve with signs of intoxication (33). In alert subjects, dark-adapted pupil size has been shown to have minimal day-to-day variability, without the need to control for sleep, diet or activity levels (34), and remains stable for up to six months in individuals up to 47 years of age (35,36). While these data are not generalizable to our methods, and do not account for theoretical influence of lingering alcohol, caffeine and nicotine on PLR, we found that a majority of reports describing PLR do not carefully control for these agents; thus, we selected a practical time frame in which the acute effects of such agents could be expected to have worn off, and one at which our study subjects could reliably comply. However, the timing of testing parameters relative to migraine attacks is important. We required that EM subjects be headache free at least 24 hours after the interictal measurement to avoid testing in the premonitory phase of the migraine attack, and ascertained by patient report the date of the last headache. Importantly, in our sample, we did not find any significant relationship between time since last headache and pupil parameters or photophobia thresholds.
Alterations in pupillary diameter and kinetics of the PLR due to excitement or mental stress are recognized (37) and are used in experimental models of affective disorders, cognitive tasks, and stress (38,39). Pupillary constriction in response to light is inhibited in the excited subject, and is associated with a relative reduction in the amplitude of contraction (20). Along these lines, it is possible that the reduced amplitude of pupil constriction during photophobia threshold testing in our study is a result of heightened sympathetic drive, though this mechanism would not be consistent with our other findings suggesting sympathetic hypofunction in the same subjects. Additionally, we found no significant relationships between affective symptoms (as measured by the GAD-7 and PHQ-9) and pupil parameters or photophobia threshold.
Conclusions
In summary, we show that photophobia and pupillary autonomic function are correlated in migraineurs, and that both vary on an apparent gradient of disease severity. Our findings implicate potential mechanistic links between sensory sensitization and autonomic allostasis, and help further validate both photophobia and pupillary function as biomarkers of disease in migraine.
Article highlights
We combined clinical, psychophysical and physiologic measures to assess disease state, photophobia and pupillary responses to light in migraine and non-migraine subjects. Our key finding was that of mixed pupillary sympathetic and parasympathetic dysfunction associated with lower interictal photophobia thresholds in migraine subjects, and in clinically more severe migraine. We found a gradient of disease, with the lowest photophobia thresholds in chronic migraineurs, and greater impairment of both parasympathetically mediated constriction and sympathetically mediated pupillary re-dilation in those with the most clinical impairment. Our findings are hypothesis-generating for possible causal links between sensory sensitization and autonomic allostasis, and aid in validating both photophobia and pupillary function as biomarkers of disease in migraine.
Footnotes
Acknowledgements
The authors would like to acknowledge Jeremy Theriot for his expertise in protocol design and implementation, data analysis and graphics preparation; Jonas Peterson for his assistance in data collection and recruitment; and Ben Caler for his assistance in protocol design and implementation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NIH NIMHD LRP (MMC); American Academy of Neurology Medical Student Summer Research Program (NAR, LAH); Moran Eye Center, University of Utah (KBD); NIH NINDS R01 085413, DoD CDMRP PR 130373 (KCB).
