Abstract
Background
Migraine is much more common in females than in males, and occurrence is associated with changes in female sex hormones. Calcitonin gene-related peptide (CGRP) plays a key role in migraine, and variations in female sex hormones may affect CGRP sensitivity and/or production.
Objectives
Investigate repeatability, gender differences, influence of the menstrual cycle and of migraine on CGRP-dependent changes in dermal blood flow (DBF).
Methods
CGRP-dependent increases in DBF were assessed using laser Doppler perfusion imaging after topical application of 300 or 1000 µg capsaicin on the forearm of healthy subjects and migraine patients.
Results
In healthy males, DBF response did not vary over time and was comparable with DBF in male migraineurs. In healthy females, capsaicin-induced DBF responses to both doses of capsaicin were higher during menstruation compared to the late-secretory phase (p < 0.05); this menstrual cycle dependence was absent in female migraine patients. Compared to healthy subjects, female migraineurs displayed a higher DBF response both during menstruation and during the late-secretory phase (p < 0.05).
Conclusions
An increased capsaicin-induced, CGRP-mediated DBF response was observed during menstruation in healthy women, but in female migraine patients this increased response was not affected by the menstrual cycle.
Introduction
Migraine headache is a neurovascular disorder in which the trigeminovascular system is primarily involved. Migraine is much more prevalent in women than in men (1,2) and women with migraine also have higher rates of most migraine symptoms, more severe associated impairment, and higher healthcare resource utilization than males (2).
Migraine attacks seem to be associated with changes in female sex hormone levels (3) as peaks of migraine frequency occur when estrogen levels drop (4); i.e. around the menstruation period (5, 6), at the end of pregnancy (postpartum) and during the transition to menopause. In pre-adolescent boys and girls, migraine prevalence is similar. However after menarche, migraine prevalence becomes approximately three times higher in females than males during the reproductive years of life (7). Furthermore, in female migraine patients, a higher occurrence of attacks without aura is observed two days before onset of menses and on the first days of menses (8). The exact diagnostic criteria for pure menstrual migraine without aura and menstrually-related migraine without aura are defined by The International Classification of Headache Disorders (9). As mentioned above, a relationship between migraine and estrogen withdrawal has been suggested, while rising levels of estrogen appear to be protective against migraine (4). This theory is confirmed by studies reporting improvement of migraine during pregnancy (10, 11), particularly during the second and third trimester, when serum levels of estradiol and progesterone are much higher than peak levels during native menstrual cycles. Postpartum, migraine reoccurs, but less in lactating women. It has even been suggested that the improvement observed during pregnancy is maintained by breast-feeding, a period of anovulation (12). During the transition period to menopause, when estrogen levels vary, a higher migraine prevalence is observed (13). After the menopause, when female hormone levels are low, migraine in many patients disappears (14).
Sex steroids can exert effects on neurotransmitter systems and pain processing networks relevant to migraine headache (15), including CGRP expression (16), and at the level of downstream signaling (17). Estrogen affects growth factors and influences the function of neurons, glia cells and the vasculature. Therefore, estrogens may contribute to neurological disorders including migraine (18). Mechanisms by which estrogens exert vascular functional effects are nicely reviewed by Miller and Duckles (19).
Findings over the last two decades also clearly indicate a pivotal role for calcitonin-gene related peptide (CGRP) in the pathophysiology of migraine. CGRP is a very potent vasodilator and a neuropeptide involved in pain transmission in the trigeminovascular system. Blocking CGRP is efficacious in migraine treatment (20–22). To test receptor engagement of CGRP receptor antagonists in early clinical trials, a human exploratory biomarker model was developed (23). This capsaicin-induced dermal blood flow (DBF) model was validated (24) as a reproducible human neurogenic inflammation model to elicit endogenous CGRP release via transient receptor potential vanilloid 1 (TRPV1) activation at peripheral nerve endings (25). The resulting vasodilatation, as measured with laser Doppler perfusion imaging, was clearly shown to depend on CGRP (26).
After validating the long-term repeatability of the DBF model in healthy male volunteers, this study investigated the influence of the female menstrual cycle and of migraine on changes in skin perfusion resulting from the endogenous release of CGRP induced by the local application of capsaicin. We hypothesized that the CGRP response is fortified by migraine and/or dependent on the female hormonal cycle.
Methods
Subjects
This study was approved by the ethics committee of the University Hospitals of Leuven (Leuven, Belgium). Subjects were recruited via public advertisements at the university campus and in student magazines. Additionally, a volunteer database was used for subject recruitment. During a telephone call and email communication, a first eligibility check was performed. During a subsequent screening visit, all inclusion and exclusion criteria were further checked. Written informed consent was obtained from all subjects during the screening visit. All subjects were adult, white, non-smoking, healthy subjects (apart from migraine) based on medical history. No hormonal contraceptive method was allowed. As part of the health and demographic information questionnaire, women reported the first day and duration of their most recent menstruation and confirmed having a regular menstrual cycle. Migraine patients suffering from moderate to severe migraine headache with or without aura were included as diagnosed according to IHS criteria (International Committee on the Classification of Headache Disorders, 2004). Subjects using cardiovascular medication, or medication affecting the nervous system, were excluded. In case of acute anti-migraine medication use, a time window of five half-lives between medication intake and the experiment was taken into account. Subjects taking prophylactic migraine medication within one month of the screening visit or taking medication for acute headache on more than 10 days per month were excluded.
Study design
During a screening visit, medical history, inclusion and exclusion criteria were checked and basic demographic data collected. During each study visit, supine systolic blood pressure, diastolic blood pressure, and heart rate were measured at the dominant arm with a semi-automated oscillometric device (Omron HEM-705CP; Omron Healthcare, Hamburg, Germany) after an acclimatization period of at least 20 min. Subsequently, baseline DBF was determined as described previously (23), using a laser Doppler perfusion imager (HR-LDPI system, Periscan PIMII). Only responders, defined as subjects with a capsaicin-induced increase in DBF of ≥ 100% in both proximal sites of both forearms were included after the screening visit. All measurements were performed while the subjects rested in a supine position on a comfortable bed in a quiet, temperature-controlled room (ambient temperature 23 ± 1℃). Subjects were instructed to abstain from beverages and food containing chocolate, alcohol, and caffeine during the 12 h period preceding each study visit. General fasting was demanded for three hours before the experiment. Blood samples were taken from participating women to assess female hormone levels (i.e. estrogen and progesterone).
Repeatability of capsaicin-induced DBF over time
Healthy male subjects were included in a 4-period, single-blind study. Study periods with capsaicin-induced DBF assessments were separated by a wash-out period of 7 (±1) days.
Menstrual cycle effects in healthy females
For the part of the study assessing menstrual cycle effects in healthy females, capsaicin-induced DBF was evaluated weekly up to two menstrual cycles. Women were included at random periods of their menstrual cycle to avoid confounding effects over time. Measurements from women with an irregular menstrual cycle were excluded from the analysis.
Migraine patients
In the part of the study with migraine patients, male subjects were assessed at two study visits apart from the screening visit. Average capsaicin-induced DBF was calculated from these two visits. After screening, female migraineurs underwent the DBF experiment one week before expected menstruation, and once during their menstruation period. All experiments in migraine patients were performed during the interictal period.
Capsaicin-induced DBF response
Capsaicin powder (Sigma-Aldrich N.V., Bornem, Belgium) was dissolved in a 3:3:4 mixture of ethanol 100%, Tween-20 and distilled water to obtain 300 µg/20µl and 1000 µg/20µl solutions. In each subject, three equally spaced (minimum distance of 4 cm) 10 mm rubber O-rings (8 mm inner diameter) were placed on the volar surface of each forearm. In the two most proximal rings, subjects received a topical dose of 300 or 1000 µg/20 µL capsaicin (single-blind, randomized). In the distal ring of both arms, a placebo (i.e. vehicle) was applied. The application of the capsaicin/placebo started on the dominant arm (t = 0), with a one-minute interval between each ring. The applications at the non-dominant arm started five minutes after the first application at the dominant arm. DBF was assessed using laser Doppler perfusion imaging just before and at 10, 20, 30 and 40 minutes after capsaicin/vehicle application.
Estradiol and progesterone plasma levels
Serum estradiol and progesterone levels were determined with the Coat-A-Count Estradiol and the Coat-A-Count Progesterone kits (Siemens Medical Solutions, Netherlands). These are no-extraction, solid-phase 125I radioimmunoassay kits designed for measurement of estradiol and progesterone in serum.
Data analysis and statistics
Based on the within-subject standard deviation observed in the reproducibility study and given a type I error probability (α) of 0.05, a sample size of 11 subjects provides 80% power for detecting a difference in DBF increase of 20% (23). To assess the repeatability of DBF after capsaicin/vehicle application, the percentage change in DBF from the baseline to 40 minutes after capsaicin/vehicle application was calculated by ANOVA including dose, time point and week in the model. For comparison between healthy and migraine subjects, the percentage change in DBF (corrected for the vehicle response) was calculated at each time point (pre-capsaicin, and 10, 20, 30 and 40 minutes post-capsaicin). The area under the curve of DBF response from 0 up to 40 minutes after capsaicin application (AUC0-40, % · min) was calculated as a summary measure using DBF ratios. Unless stated otherwise, the mean and standard error of the mean (SEM) are presented.
SPSS version 21 (SPSS inc, IBM,Chicago, IL, USA) was used for statistical analysis. Repeated-measures analysis of variance (ANOVA) with a Bonferroni post-hoc test was used to assess reproducibility in healthy males, menstrual cycle effects in healthy females, and for comparison of differences between healthy subjects and migraine patients. To compare the effects of female hormones on capsaicin-induced DBF with those recently reported in literature (27), we performed paired t-tests comparing the response during menses and the late-secretory phase of the menstrual cycle. Kruskal-Wallis ANOVA was used to assess hormonal changes during the four-week menstrual cycle of healthy subjects and migraine patients. With a Dunn’s post hoc test we compared the hormone levels within and between groups. A two-tailed p-value < 0.05 was considered statistically significant.
Results
Subjects
Five healthy subjects and two migraineurs were excluded at screening, being non-responders to capsaicin. Data for five subjects were excluded from analysis because of medication use; four others were excluded because of irregular menstrual cycles. Due to technical breakdown of the LDI scanner, data from seven out of 26 healthy females were exclusively used for analysis of menstrual cycle effects within the group of healthy females and excluded from comparison with migraine women, as they were scanned with a differently calibrated scanner. Likewise, data from two out of 16 healthy men were only used for weekly repeatability analysis within healthy males, and not for inter-group comparison.
Repeatability in males
Figure 1 describes DBF response 10, 20, 30 and 40 min after application of vehicle, 300 or 1000 µg capsaicin solution, assessed on four consecutive weeks in 16 healthy men (average age 23.6 years, range: 20–28). Significant differences were found between doses (placebo, 300 µg and 1000 µg capsaicin; p < 0.001) and between time points (10, 20, 30 and 40 minutes post-capsaicin; p < 0.001). Over a period of four weeks the capsaicin-induced increase in DBF did not change (p = 0.220).
Repeatability of dermal blood flow over four consecutive weeks in healthy men (n = 16).
Menstrual cycle effects in healthy females
Twenty-six healthy females, not taking hormonal contraceptives, were enrolled (average age 21.6 years, range 18–26). DBF was expressed as (i) the percentage increase in DBF 40 minutes post-capsaicin application compared to the placebo, as well as (ii) area-under-the-curve of percentage change in DBF response, expressed as a percentage increase compared to the placebo, from 0 up to 40 minutes (AUC0-40). Repeated measures ANOVA did not reveal a significant effect of the menstrual cycle on the 40 minute DBF response to 300 µg capsaicin (Figure 2a), nor on the 40 minute DBF response (Figure 2b) or on the AUC of the DBF response to 1000 µg (Figure 2d). We did, however, find a trend for significance of the menstrual cycle effect on the AUC of the DBF response to 300 µg capsaicin (Figure 2c, p = 0.053). When specifically comparing the menses with the late secretory phase, in most comparisons we observed increased DBF responses to both doses of capsaicin during menstruation (Figure 2a: p = 0.049, Figure 2b: p = 0.004, Figure 2c: p = 0.023, Figure 2d: p = 0.065).
DBF during each week of the menstrual cycle in healthy females (n = 26).
Migraine patients
Inter-group comparison of demographics of healthy subjects and migraine patients.
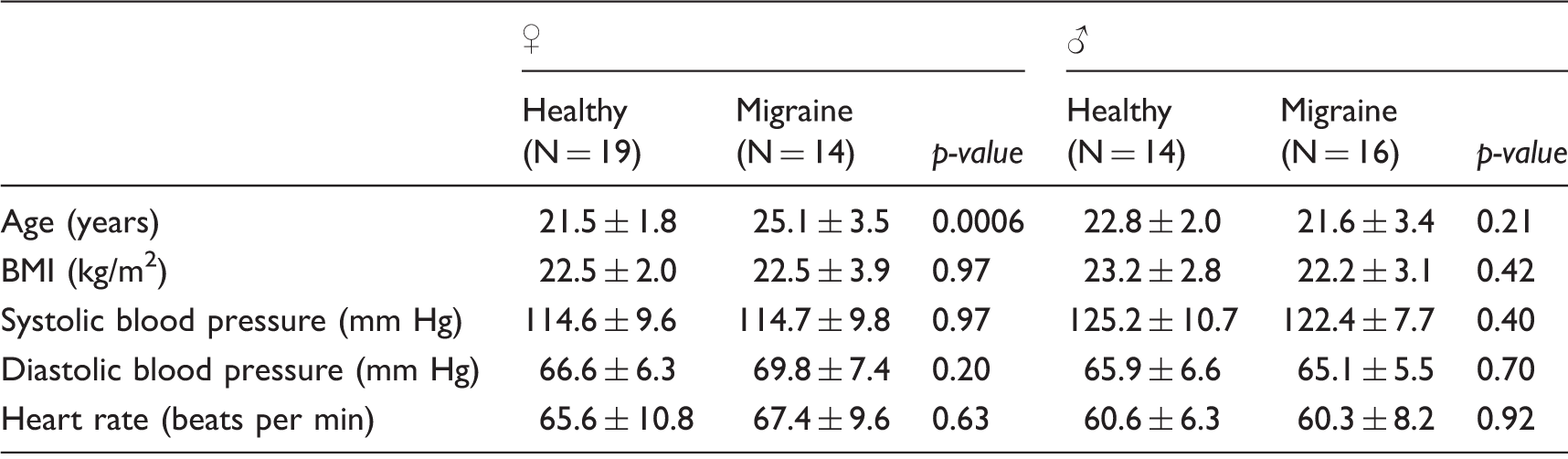
Data presented as mean ± SD.
Repeated measures ANOVA did not reveal a significant effect of the menstrual cycle phase within the migraine patients on the 40 minute response or on the AUC of the capsaicin-induced DBF, neither for 300 nor for 1000 µg capsaicin (Figure 3a – Figure 3d). When comparing the DBF response at 40 minutes between migraine patients and healthy women together in one analysis, the effect of disease state (healthy vs. migraine, p = 0.052) appeared to influence the response to the low dose of capsaicin (300 µg), but not (p = 0.071) when studying the high dose (1000 µg) of capsaicin. In contrast, the AUC was significantly affected by disease state for both doses of capsaicin (p = 0.017 and 0.006, respectively). These differences in AUC values are in line with the observation that migraine patients tended to reach their maximal response to capsaicin at an earlier time point than the healthy women (supplementary figure 1). No difference in DBF was found between healthy men and men suffering from migraine after both 300 and 1000 µg capsaicin (Figure 3).
DBF in migraine women (n = 14) compared to healthy women (n = 19). No difference between healthy men (n = 14) and migraine men (n = 16) after 300 and 1000 µg capsaicin.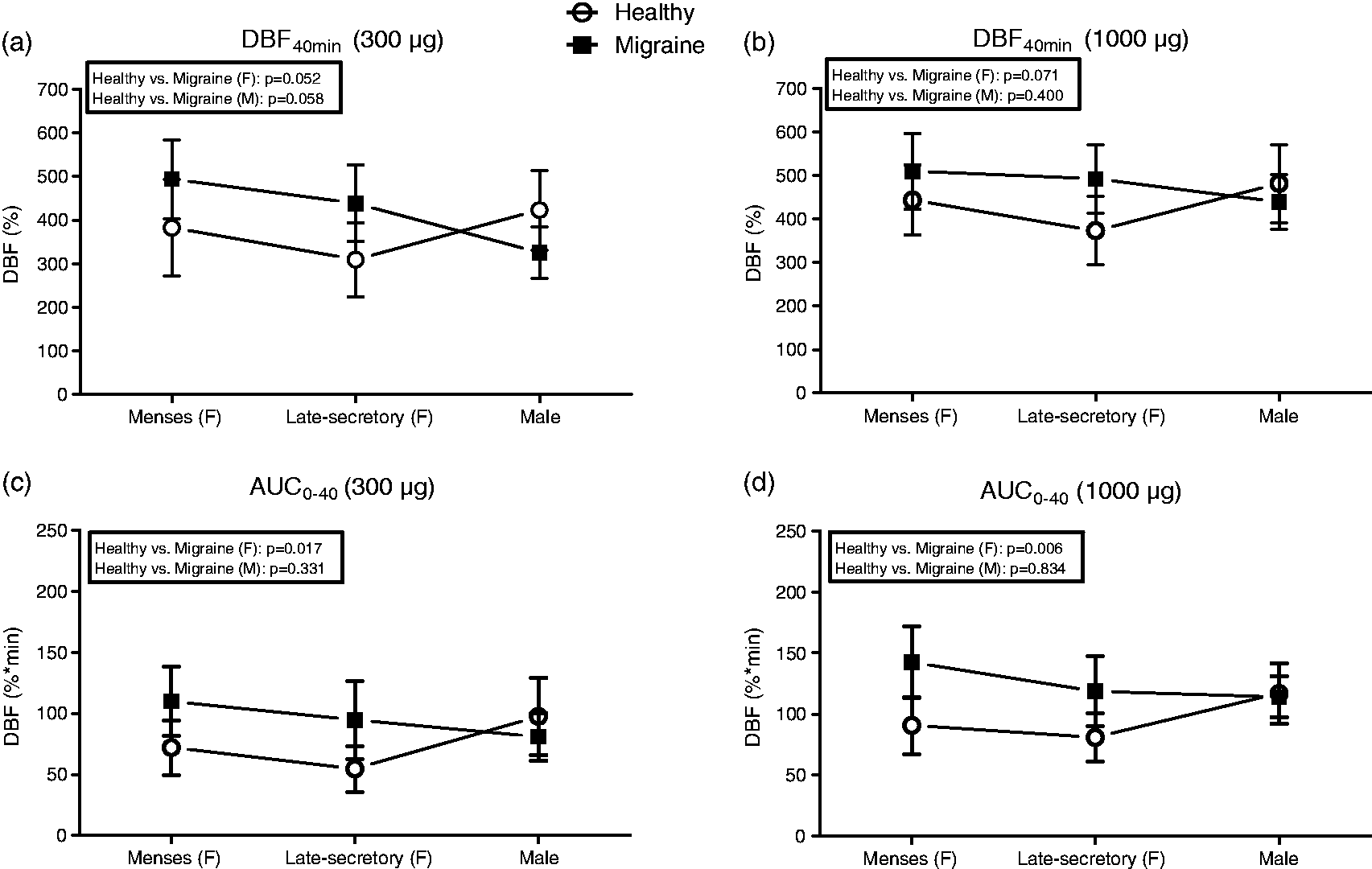
Hormone levels
During the menstrual cycle within the healthy women (73 ± 12 pg/ml to 25 ± 3 pg/ml, p < 0.05) and within migraine patients (75 ± 12 pg/ml to 28 ± 3 pg/ml, p < 0.001), a significant drop in estrogen before menstruation was observed. No differences in estrogen levels were found between both groups during menstruation and the late-secretory week.
Similarly, within both the healthy women (10 ± 2 ng/ml to 0.6 ± 0.1 ng/ml, p < 0.001) as well as migraine patients (5 ± 1 ng/ml to 0.7 ± 0.2 ng/ml, p < 0.05), a significant drop in progesterone before menstruation was observed. No differences in progesterone levels were found between groups during either the late-secretory week or menstruation.
Discussion
The present study confirms the repeatability of DBF changes induced by weekly applications of capsaicin in healthy males without evidence for major desensitization. Unlike men, healthy females displayed fluctuations in the CGRP-dependent dermal blood flow response, which was increased during the menstruation period. DBF responses were elevated in female migraine patients compared to healthy subjects.
Since the capsaicin-induced DBF response is repeatable over time in healthy male volunteers, the “capsaicin model” can also be used as a target-engagement biomarker for the prospective follow-up of long-acting biologicals including monoclonal antibodies targeting CGRP (28,29), next to short acting small molecule CGRP-receptor antagonists in early clinical drug development (26).
As there was no significant desensitization to capsaicin observed in healthy males and, moreover, because women were included at random weeks during their menstrual cycle, the changing DBF response in healthy women during the menstrual cycle can most likely be attributed to hormonal changes related to the menstrual cycle. This was previously suggested by the findings of Ibrahimi et al. and Gazerani et al. (27,30). After intradermal capsaicin injection to the forehead, they found an increased flare area in women during the menstrual phase compared to the luteal phase. Accordingly, after topical application of capsaicin on the forearm, we found an increased DBF in healthy females during their menstrual phase compared to the luteal phase. In contrast, we did not find an effect of the menstrual cycle on the capsaicin-induced DBF in migraine patients. Interestingly, upon comparison of corresponding phases of the menstrual cycle, female migraine patients responded with larger increases in DBF to capsaicin application than healthy women did. This effect may have been augmented by an earlier response to capsaicin in patients (Supplemental Figure 1) and suggests an increased availability of CGRP or an increased sensitivity to capsaicin and/or CGRP. The lack of menstrual cycle-dependent variation in migraine has previously been reported in our study where we assessed the influence of the menstrual cycle on capsaicin-induced forehead DBF, thus comparing trigeminovascular activation in healthy women to that in women with menstrually-related migraine (27).
The importance of CGRP in the pathophysiology of migraine and a correlation between migraine incidence, menstrual cycle and decreasing estrogens, as described in literature, are in line with our findings. A substantial period with high estrogen exposure seems to be necessary for migraine attacks to result from estrogen withdrawal (31–33). Many studies have described an association between the occurrence of migraine attacks and menstrual cycle (4), pregnancy (34,35), partus (36) and menopausal transition (13). However, effective hormonal treatment of migraine associated with menstruation and the menopause is lacking (3,37–39). Also, association studies focusing on the role of sex hormone receptor gene polymorphisms in migraine remain inconclusive (40). Nevertheless, changes in levels of female sex hormones like estrogen and progesterone clearly modulate neurotransmitter systems (18), and influence brain function (15) and vascular function (19) in diverse ways.
In contrast to females, comparison of healthy men with male migraine patients showed no difference in DBF response. Gender differences (due to hormonal fluctuations) are suggested to have implications on both migraine susceptibility and migraine mechanism between males and females (41). Gender differences further illustrate the complexity of migraine, and may support the hypothesis that clinically-observed migraine features are modulated by different mechanistic pathways that trigger migraine.
Migraine subjects included in this study met the criteria for episodic migraine as classified in the International Classification for Headache (ICHD-II). No distinction was made between menstrual, menstrually-related or non-menstrual migraine subjects due to small subgroup under powering. As there are subtle differences between these groups, the hormonal effects observed in our study should represent a more general migraine population, but might be more pronounced in purely menstrual migraine without aura patients. Hormone levels between healthy female subjects and migraine patients in our study did not differ, suggesting that in this group of migraine patients higher sensitivity to female hormones is more important than absolute plasma levels.
The exclusion of non-responders to capsaicin could be a limitation of this study, as this may bias our results. However, we excluded five healthy subjects and only two migraineurs because of this criterion, implying that inclusion of non-responders would even fortify our findings. The reason why some subjects do not respond to topical applied capsaicin is unknown, but has been observed in other studies as well (24,42). Genetic analysis of non-responders to detect TRPV1 polymorphisms or other mutations could provide an explanation. However, it has been suggested that the absence of a DBF response may be purely related to insufficient skin penetration rather than to the absence of (functional) TRPV1 channels. This was supported by the observation that subjects with an apparently absent response to capsaicin applied on the forearm do respond after application on the forehead skin (43).
Molecular and physiological mechanisms for increased DBF response after capsaicin application could be the result of several mechanisms, including increased neuronal sensitivity to capsaicin, perhaps due to increased TRPV1 expression, increased release of CGRP, or increased sensitivity to CGRP. Female hormones influence neurotransmitter systems (18), vasculature (19) and thus migraine (15), but further research is needed to clarify which mechanisms are involved (44).
In summary, we first validated the long-term repeatability of the CGRP-dependent DBF response in men. Secondly, we showed an increased DBF response during the menstruation period in healthy women. Finally, a higher and seemingly faster CGRP-dependent DBF response that was not influenced by the menstrual cycle was confirmed in women suffering from migraine. Our data demonstrate the influence of the female menstrual cycle and migraine on CGRP-mediated vascular responsiveness, which further supports hormonal effects and increased female susceptibility to migraine.
Clinical implications
The capsaicin-induced DBF model is weekly repeatable in males without showing evidence for significant desensitization. Therefore, this model can be incorporated as a pharmacodynamic target-engagement biomarker in clinical trials with long-acting antimigraine drugs including monoclonal antibodies targeting CGRP for migraine prophylaxis. Capsaicin-induced DBF increases in healthy women depend on the menstrual cycle and are highest during the menses. Response to this CGRP-mediated human in vivo model is increased in migraine women and during menstruation in particular, confirming a hormonal influence on the increased susceptibility to migraine observed in women. More knowledge of the physiological and molecular modulating mechanisms involved in peripheral sensitization will improve our understanding of how female hormones increase susceptibility to migraine.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Amgen 10.13039/100002429 Unrestricted grant.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
