Abstract
Background
A detailed evaluation of migraine aura symptoms is crucial for classification issues and pathophysiological discussion. Few studies have focused on the detailed clinical aspects of migraine aura.
Methods
We conducted a prospective diary-based study of migraine aura features including presence, quality, laterality, duration of each aura symptom, their temporal succession; presence of headache and its temporal succession with aura.
Results
Seventy-two patients completed the study recording the characteristics of three consecutive auras (n = 216 auras). Visual symptoms occurred in 212 (98%), sensory symptoms in 77 (36%) and dysphasic symptoms in 22 (10%). Most auras had more than one visual symptom (median 2, IQR 1–3, range 1–4). The majority of patients (56%) did not report a stereotyped aura on the three attacks with respect to visual features, the combination and/or temporal succession of the three aura symptoms. Fifty-seven percent of patients also reported a different scenario of temporal succession between aura and headache in the three attacks. Five per cent of aura symptoms were longer than four hours.
Conclusion
These findings show a high inter- and intravariability of migraine with aura attacks. Furthermore, they provide reliable data to enrich and clarify the spectrum of the aura phenotype.
Introduction
Migraine is the third most common disorder in the world (1), with 30% of patients having migraine with aura (2). As for migraine without aura (3), the clinical presentation of attacks of migraine with aura varies considerably, both among patients and within each patient (4). While this variability is obvious in routine clinical practice it has not been extensively characterized prospectively, particularly for migraine with aura. Most reports of the clinical characteristics of migraine with aura are retrospective (5–10). Only a few prospective studies are available (11–13), and they are based on a limited number of patients.
A detailed description of the phenotype of the attacks and their inter- and intra-patient variability is important for achieving a more evidence-based nosographic framing of migraine with aura. This is a prospective diary-based study specifically focused on the thorough recording of the phenotypical characteristic of migraine with aura as regards both the aura symptoms and the associated headache. A part of this study, specifically dedicated to the temporal aspect of migraine with aura and based on the first 54 patients, has been previously published (14).
Methods
We recruited 224 consecutive patients affected by migraine aura attending the Headache Centers of Pavia and Trondheim (respectively 198 and 26).
The study received approval from the local Ethics Committees (‘C. Mondino’ National Neurological Institute, Pavia and REC-Central, Trondheim) and all patients signed an informed consent form. Enrollment started in October 2012 and ended in July 2014. Follow-up ended in May 2015. Inclusion criteria were: (i) patients affected for at least one year by migraine with aura fulfilling International Classification of Headache Disorders, 2nd Edition (ICHD-2) criteria (15) for 1.2.1 [G43.10] typical aura with migraine headache,1.2.2 [G43.10] typical aura with non-migraine headache, 1.2.3 [G43.104] typical aura without headache, excluding point 3 of C criteria (‘each individual aura symptom lasts 5–60 minutes’) and where only one of point C1 or C2 had to be verified to fulfill C criteria; (ii) age between 16 and 65 years. Exclusion criteria were: (i) hemiplegic migraine; (ii) brainstem aura; (iii) pregnancy; (iv) variation of the characteristics of aura and/or headache in the last 6 months; (v) patients with more than two vascular risk factors; (vi) history of myocardial infarction and/or transitory ischemic attack (TIA) and/or stroke and/or other thrombophilic disturbances; (vii) patients with episodes that are not clearly differentiated from other disturbances (TIA, seizures) (14). After diagnosing migraine with aura, a neurologist expert in headache verified the inclusion and exclusion criteria and instructed the patients to record prospectively the characteristics of three consecutive attacks in an ad hoc aura diary.
The patient was asked to describe each aura symptom (visual, sensory and dysphasic) with his/her own words (in the diary there was a free-text box for each symptom), the main characteristics of headache attacks, and the time of onset/end of aura symptoms and headache. Once the patient had recorded three attacks, he/she returned to the Headache Center for a follow-up visit and the diaries were discussed with the neurologist. In particular, the neurologist verified that the symptoms described as auras: (i) have been present in the past during other typical auras experienced by the same patients; and (ii) were not premonitory symptoms (i.e. photophobia, difficulty with concentration/speech). In the analysis, the authors dissected every description of visual phenomena into elementary visual symptoms as reported by Queiroz and colleagues (7), adding also the features ‘visual snow’ (dynamic, continuous, tiny dots usually black/gray on white background and gray/white on black background) (15) and ‘deformed images’ (alteration of the shape of objects). In the evaluation of the succession of individual aura symptoms where two symptoms started simultaneously, we designated the first completing symptom as the first symptom (FS).
Statistical analysis
Categorical variables are presented as percentages or means with standard deviation, while continuous variables are presented as medians with interquartile ranges (IQR). Statistical comparisons between groups were based on the chi-squared test or Kruskall–Wallis test for categorical variables or continuous variables, respectively. Owing to the exploratory nature of the study, we reported nominal statistical associations (p-value < 0.05). Adjusted p-values based on the Bonferroni correction were also considered to avoid chance findings due to multiple testing for a total of 21 comparisons, and the significance level was lowered to p < 0.00238 (p = 0.05/21). Statistical analysis was performed with MedCalc Version 13.3.3 (MedCalc Software, Mariakerke, Belgium) software.
Results
Patients
Of the 224 patients recruited, 72 completed the diaries during three consecutive auras for a cumulative number of 216 auras recorded. Of the remaining 152 patients, 37 dropped out and 115 did not complete three aura attacks and were therefore not included in the analysis.
Characteristics of patients (n = 72).
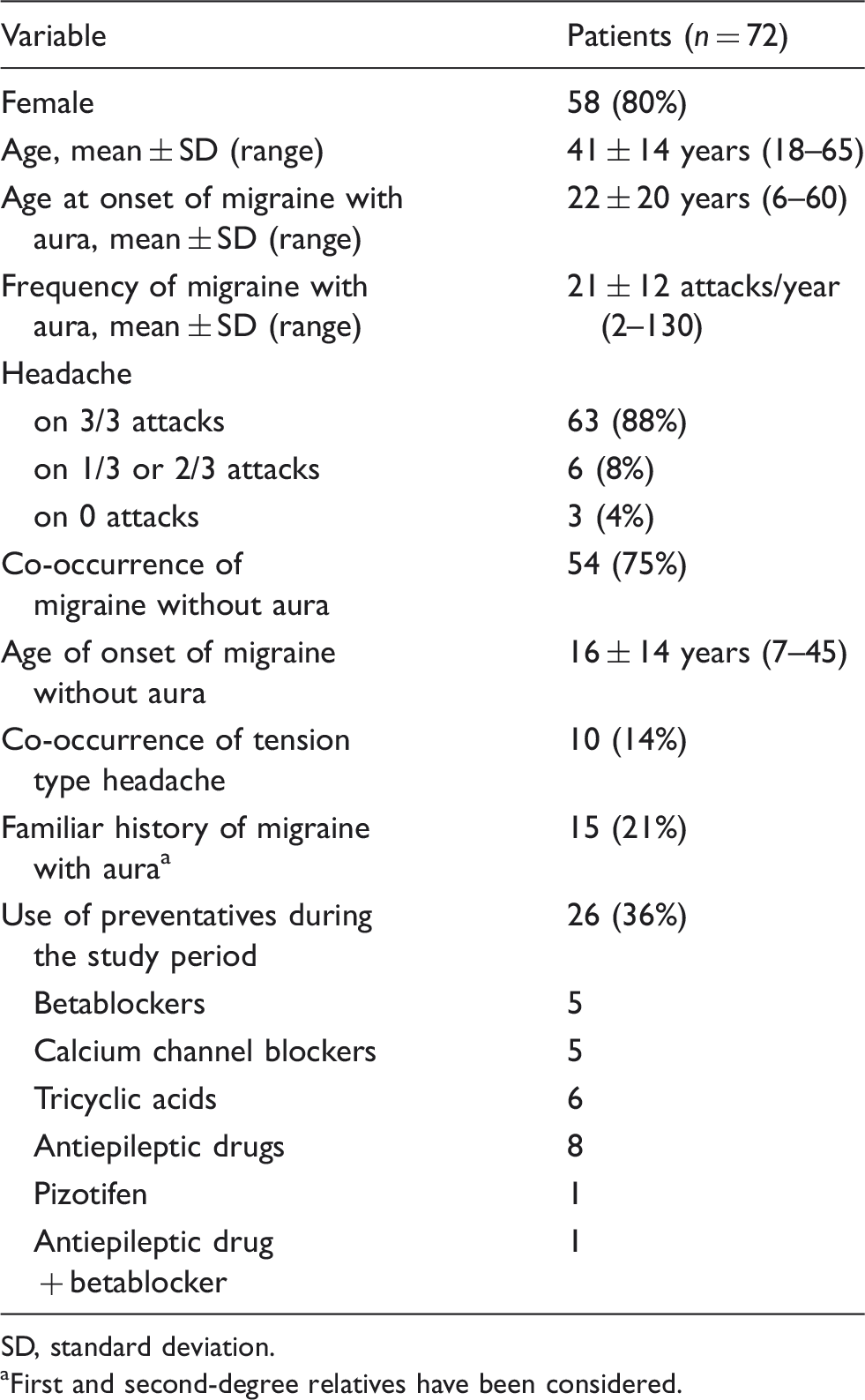
SD, standard deviation.
First and second-degree relatives have been considered.
Features of auras
Out of 216 auras evaluated, visual symptoms occurred in 212 (98%), sensory symptoms in 77 (36%) and dysphasic symptoms in 22 (10%). The distribution of aura symptoms is shown in Figure 1. A total of 135 auras out of 216 (62%) had one symptom, 67 (31%) had two symptoms and 14 (6%) had three symptoms. Sixty-six percent of patients (48/72) presented the same combinations of symptoms during the three attacks (i.e. just visual or visual and sensory or just visual, sensory and dysphasic), while in 34% (24/72) of patients the combination of aura symptoms varied across attacks. The latter group of patients has a statistically significantly higher number of symptoms per attack with respect to patients with the constant combination of symptoms across the three attacks (1.69 ± 0.67 vs 1.21 ± 0.50; p < 0.001).
Venn diagram illustrating the distribution of the various aura symptoms in 216 auras.
Visual symptoms
Frequency of occurrence of single visual disturbances.
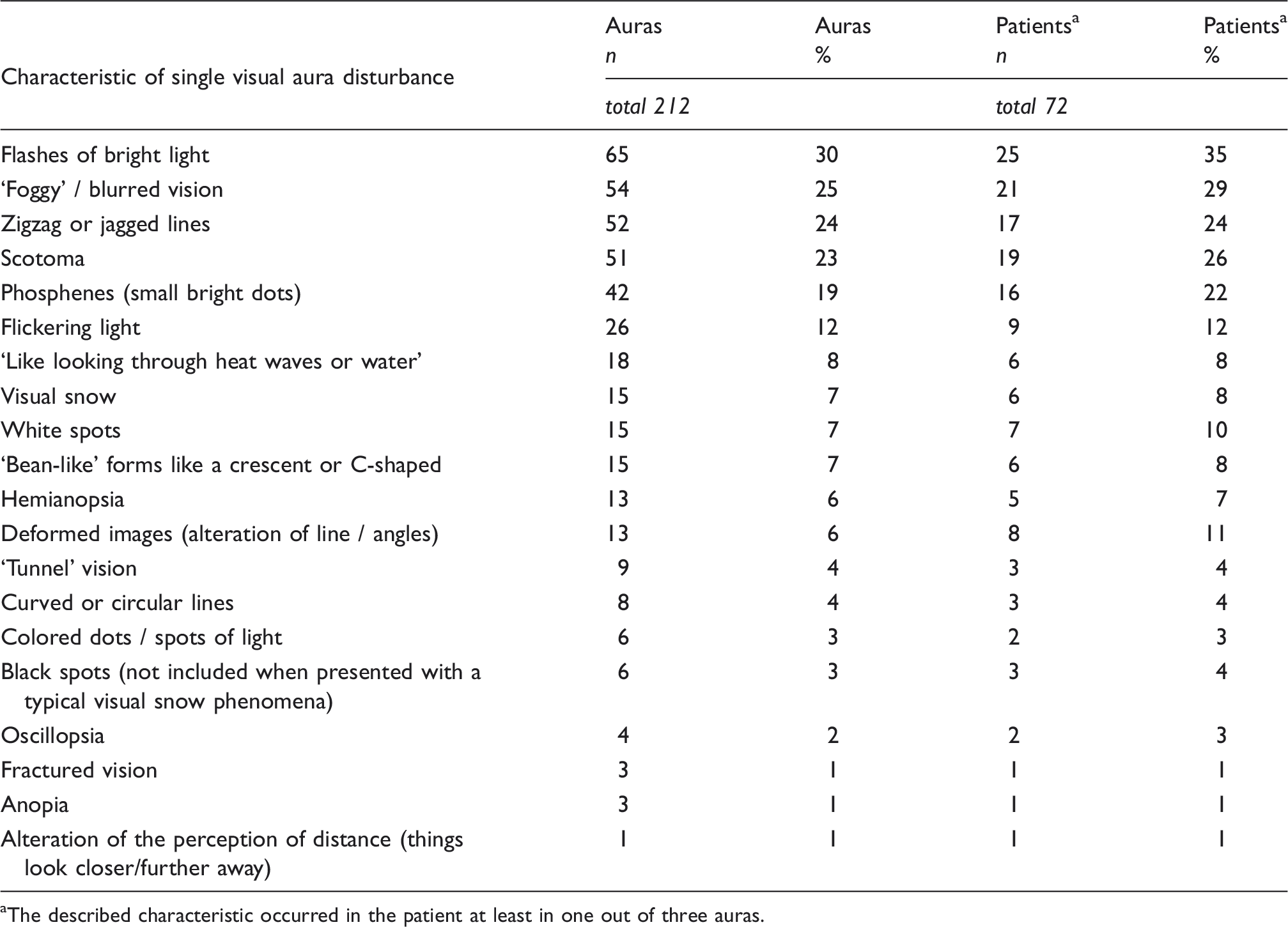
The described characteristic occurred in the patient at least in one out of three auras.
Frequency of occurrence of the overall visual phenomena.

The described phenotype occurred in the patient at least in one out of three auras.
Thirty percent of patients (22 out of 71) who presented at least two auras with visual symptoms, reported different combinations of the elementary visual symptoms (reported in Table 2) during the three auras, whereas 70% (49 out of 71) presented a stereotype set of visual phenomena.
We divided the simple visual symptoms into positive (phosphenes (small bright dots/stars), zigzag or jagged lines, flashes of bright light, flickering light, white spots, colored dots / spots of light, curved or circular lines, ‘bean-like’ forms like a crescent or C-shaped round forms), negative (scotoma, black spots, small black dots, hemianopia, tunnel vision, anopia), or disturbances of visual perception (DVP) (blurred/foggy vision, ‘like looking through heat waves or water’, deformed images (alteration of the shape of objects), visual snow, fractured vision, oscillopsia, corona phenomena, alteration of the perception of distance (things look close or far away)). Sixty-seven percent of visual aura (n = 142) included at least one positive phenomena, 38% (n = 82) at least one negative phenomena and 45% (n = 96) at least one DVP.
Distribution of auras/patients according to some of the visual aura characteristics.

The described characteristic occurred in the patient in at least one out of three auras.
Sensory symptoms
Distribution of auras/patients according to some of the sensory aura characteristics.

The described characteristic occurred in the patient at least in one out of three auras.
Other than paresthesias, in some moments of aura these patients experience a sensory reduction and in other moments a complete sensory loss.
Number of sensory auras = 77; number of patients with at least one sensory aura = 35.
Dysphasic symptoms
Paraphasic errors (substitution of words/letters) were reported in 15% cases (3/20), aphasia nominum in 20% (4/20), while in other cases the disturbances were described as generically impaired language production.
Temporal features
This aspect has been already described in a previous paper (14), where we reported the results of the first 54 patients. In this paragraph we report aspects that were not yet investigated, while in Supplementary Tables 1 and 2 we updated previously reported results.
Twenty-two patients had at least two migraine auras with multiple aura symptoms: 14 of them (64%) reported a different pattern of succession between the first symptom (FS) and the second symptom (SS) or the SS and the third symptom (TS) (i.e. in one aura the FS and SS started simultaneously, in one aura the SS started when the FS stopped, and in one case the SS started after a free interval of time after the end of the FS) on two/three attacks, while in eight patients (36%) the pattern was the same.
The median duration of the whole aura was 30 minutes (min) (IQR 25–60 min, range 5–10,860 min). Auras with one symptom (visual in 98% of cases) were significantly shorter than auras with two or three symptoms (median 30 min (IQR 20.75) vs 60 min (IQR 106.25); p < 0.0001).
The total number of aura symptoms recorded was 311 (135 auras with one symptom + 67 auras with two symptoms (134 symptoms) + 14 auras with three symptoms (42 symptoms)). With respect to the duration of aura symptoms, when considering them as a whole (n = 297; for 14 of the 311 we do not have data of duration), 46 symptoms lasted for longer than one hour (15%). Their duration fell in the following ranges: more than one hour to two hours (n = 21), more than two hours to four hours (n = 10), more than four hours to eight hours (n = 5), more than eight hours to 24 hours (n = 4), > 24 hours (n = 6). Based on these data, only 5% of aura symptoms lasted for more than four hours.
Intravariability of the aura phenotype
If we consider together the visual features, the combination of aura symptoms, and the succession of subsequent symptoms (where available), 40 out of 71 patients (56%) did not report a stereotyped aura on the three attacks. The variability was due to either a different combination of visual phenomena (in terms of quality) (n = 12), a different combination of visual and/or sensory and/or dysphasic symptoms (in terms of presence/absence of the three symptoms and/or temporal succession among them) (n = 18) or both (n = 10).
Headache
In 197 out of the total 216 (91%) auras there was associated headache. Headache characteristics will be the subject of a future paper. For 40 out of the 197 migraine auras with headache, data about headache onset were lacking. Of the remaining 157, in 15 (12%) headache started before aura; in 19 (10%) headache started simultaneously with aura; in 45 (28%) headache started during aura; in 19 (12%) headache started when aura stopped; and in 59 (37%) headache started after a free interval of time after the end of aura. Fifty-seven patients had at least two auras where data about headache onset were present: 54% (n = 31) did not report a stereotyped scenario of succession aura-headache, while 46% (n = 26) did.
Visual auras with positive symptoms versus negative symptoms
We subdivided visual aura attacks into two groups: a first one in which there was just positive symptoms, with or without DVP and a second one in which there was just negative symptoms, with or without DVP. We performed an analysis to evaluate if there was any clinical variable associated to one group (see Supplementary Table 3). In univariate analyses, the only clinical parameter significantly associated with one group after multiple comparison was the duration of sensory auras, which was longer in the group with positive symptoms (median 30 min (IQR 117.5) vs 15 min (IQR 9.75); p = 0.002).
Auras with and without DVP
We subdivided all visual auras into two groups: the first one (n = 96) where at least one DVP was present and a second group (n = 116) where no DVP were included. We performed an analysis to evaluate if there was any clinical variable associated to one group. In univariate analyses (Supplementary Table 4) after multiple comparisons, we found that auras with DVP had more positive and more negative phenomena.
In the DVP group, 36 auras out of 96 (37%) where just characterized by DVP symptoms, 52 auras (54%) had also either one positive or one negative symptom, and eight auras (9%) had both one positive and one negative symptoms. In 27 out of 36 visual auras where only DVP were present, data about localization in the visual field or lateralization were reported: in 23 cases (85%) the disturbances were localized to a part of a visual field or hemifield, while in four cases (15%) the disturbances involved the whole visual field bilaterally.
Discussion
We have studied prospectively, using an ad hoc diary, several characteristics of migraine with aura attacks. Visual symptoms are almost universal, and among the visual symptoms patients most often had more than one form. The majority of patients did not have stereotyped attacks either in terms of features, their combination or the succession of headache. Since 95% of aura symptoms lasted less than four hours, it could be suggested that this would be a more logical upper limit for aura symptom aura than one hour.
We confirmed that almost all migraine auras (98% of 216) had visual symptoms. This is in agreement with two retrospective studies where visual symptoms occurred in 99% of auras (6,10). We found that the most common symptoms were flashes of bright light, foggy/blurred vision, zigzag or jagged lines, scotoma and phosphenes. Those symptoms were also reported as the most frequent in other studies, with the exception of foggy/blurred vision. The latter symptom has a high frequency of occurrence in studies in which it was specifically investigated (7,8,16). On the other hand, in other studies, DVP have not been included among aura symptoms at all. The latter circumstance is probably due to the fact that DVP are not considered part of aura by some authors. Here we show that DVPs share much with typical aura symptoms: (i) our patients did not experience DVP outside auras but just during migraine with aura attack (92% of which were auras with headache, the same frequency of occurrence as visual auras without DVP); (ii) in the majority of cases, DVP occur together with positive or negative aura visual disturbances; and (iii) auras with DVP were similar to auras without DVP with respect to the majority of aura features investigated (nine out of 11, see Supplementary Table 4). The only differences we found after multiple comparisons were that auras with DVP had a higher prevalence of positive and negative phenomena. Whether the pathophysiology of typical aura and DVPs is the same is an entirely unresolved question. Many DVPs, such as blurred or foggy vision or visual snow (17), have no obvious link to cortical spreading depression as the latter's physiology is now understood (18). Moreover, since typical aura has provided the epidemiological underpinning to comorbidity observations, such as those with stroke (19), any change in terminology or widening of the use of the term ‘aura’ would be complex.
Our data show also for the first time that positive visual phenomena are associated with longer duration of sensory disturbances. This finding needs to be reconfirmed in future studies before being interpreted.
We analyzed the descriptions of visual aura not just ‘as a whole’ but dissecting it into a wide variety of simple visual disturbances (n = 25) and analyzing the interaction among them. We confirm the retrospective data recently published where the median number of visual aura symptoms was the same as we found (n = 2), with a similar range (1–5) (4). This analysis showed us that some simple visual phenomena can occur alone but can also occur in multiple associations with other ones. In some cases the association included up to four different visual phenomena. Other studies evaluated the prevalence of multiple symptoms just in ‘preconstructed’ single association (with a maximum of two symptoms) such as ‘scintillating scotomas’ (9,16). These evidences of auras with multiple elementary visual symptoms should be discussed together with (i) the results recently published by the authors of this paper (14) showing that two aura symptoms (such as visual and sensory) may start, even in the same patient, in some cases simultaneously and in other cases in succession (one starts after the previous one ends); and (ii) the data we report here that a patient can present visual and sensory disturbances in one attack and visual and dysphasic disturbances in another attack. At present, all the pathophysiologic mechanisms underlying migraine aura are still not understood but it seems very likely that cortical spreading depression (CSD), a primarily neuronal event, is an intrinsic property of the migrainous cerebral cortex and is responsible for the aura symptoms (20). Indeed, there are no available direct data demonstrating a precise correlation between the phenomenology of CSD, such as site of origin, direction of spreading, brain areas involved, and the different phenomenology of aura symptoms. Many questions related to the mechanism underlying the multifaceted phenomenology of aura need answers. For instance, why do two different aura symptoms, such as visual and sensory, start in some cases simultaneously and in other cases in succession (14)? If, in the latter case, which is most typical, the textbook explanation is that CSD spreads gradually from visual to sensory areas, we should infer either that multifocal CSD exists or that a single CSD originates in a silent area of the brain that later involves simultaneously two eloquent areas. The latter is plausible given the documentation of what seems to be silent aura that should be visual in a study with more than 1000 observations (21).
Bilateral localization of aura symptoms was not uncommon. Forty percent of 42 patients reporting data during three auras had either visual aura always on both sides of vision (35%) or sometimes on one side, and sometimes on both sides (5%). Similar data have been reported in retrospective studies where 18% to 53% of patients have experienced visual symptoms on both sides of vision (6,7,10,13). With respect to sensory symptoms, about 15% of patients had a bilateral localization, which is in line with previous findings (6,10).
In our sample, dysphasic aura occurred in 10% of cases, while in other studies it was 9–32% (5,6,10,12). Our frequency is therefore in the lower range of these studies. A diary to record in a prospective way helped us to exclude some symptoms that patients addressed as dysphasic aura, which were actually premonitory symptoms such as general difficulty in concentration and/or reading and/or speaking that can happen overall in up to one-third of patients (22).
We prospectively confirm that aura attacks presenting exclusively with visual symptoms have a shorter duration than those with visual symptoms in association with other aura symptoms (6,23).
In this study we also evaluated the prevalence of the intravariability of aura phenotype. During the three auras, about one-third of patients presented a different combination of visual disturbances while two-thirds have a stereotyped constellation of phenomena. This aspect has been reported already in two retrospective studies with similar results: 72% to 73.8% of patients have stereotyped overall visual phenomena (7,8). Moreover, our results show that more than half of the patients did not report a stereotype aura either for a different combination of simple visual phenomena, a different combination of visual and/or sensory and/or dysphasic symptoms, or both. In this analysis we did not include the quality, lateralization and localization of sensory disturbances, the quality of dysphasic auras (as these were present for few cases) and the duration of each aura symptom (as it would be difficult to define in which range two symptoms should be considered of the same duration). In addition, we did not include, as they were not available, other characteristics of visual symptoms such as: (i) the temporal features of each single visual phenomena (i.e. a curvilinear positive wavefront lasting for 7 minutes, followed after 5 minutes by a circular scotoma lasting for 6 minutes, after which the flickering wavefront resumed) (21); (ii) the color of visual disturbances; (iii) the frequency of flickering (for intermittent phenomena); (iv) the shape of the visual phenomena; (v) the site of origin in the visual field (VF); (vi) the direction of spreading in the VF; (vii) the laterality of whole visual disturbances (unilateral or bilateral); (viii) the duration of gradual developing; and (ix) preserved central vision. Although we do not have data for these variables, our impression is that whether included in the intravariability analysis, the proportion of patients with a stereotype aura will even decrease. Collection of these data in new studies will also help to better describe the aura spectrum and probably to shed more light on its pathophysiological mechanism.
With respect to the headache phase, we confirmed our previous data: only in one-half of patients there is the textbook progression of aura followed by headache. Our data are in line with those of a previous prospective study in which a different diary/questionnaire was used on a smaller number of auras (13) and two retrospective studies on a larger number of auras (6,10). These findings suggest that the pathophysiological explanation for which CSD initiates pain needs revision in a substantial proportion of cases (14).
Limitations of the study
This study has some methodological limitations. First, the patients were recruited from tertiary referral centers. One could then argue that in such a setting more difficult cases are seen or unusual aura is referred for evaluation. This would account for the longer or more complicated aura presentations. We cannot, therefore, be clear what the population proportion of these aura phenomena are with these data. This does not negate the accuracy of the observations, since all patients were reviewed by experts who retook the history of secondary and aura-like presentations. Practically speaking, the use of a detailed aura diary in a population-based study has its challenges, and population-based studies always have the issue of how detailed one can make the clinical evaluation. Second, unless these are prospective, some data were reported by patients in a voluntary rather then driven way. Therefore, some items not specifically requested (such as the unilaterality of visual disturbances) have been reported by just some of the patients. This could have biased the data toward one particular condition (i.e. patients with a unilateral visual disturbance were more prone to report the aspect of uni/bilaterality of visual disturbance). Indeed, other features were specifically required, such as time of onset of headache and each aura symptom.
Conclusion
In conclusion, we have demonstrated that the clinical features of migraine aura are heterogeneous and variable. This variability is particularly apparent with prospective recording of multiple attacks in the same patient. The growing use of electronic diaries provides an opportunity for more reliable prospective recording both within and outside of clinical trials (4,24). One thing that should be decided is which features should be recorded in the diary. Of course it is preferable to include a large number of questions for a complete description of the aura. Yet we must take into account that ‘the more you ask, the less you get’ and in order to increase compliance and reliability the amount of information requested must be limited. Therefore, it is important to discuss which clinical aspects of migraine with aura need to be better evaluated with a proper clinical tool and methodology. This will be the ultimate chance to have the best and most comprehensive vision of the spectrum of migraine aura phenotype. This exercise could help us in shedding light into pathophysiological mechanisms of the disorder, which in turn may lead to improved patient care.
Clinical implications
This prospective diary-aided study on migraine aura results shows a high variability of presentation between patients and in the same patient. The variability includes the characteristics of visual, sensory and dysphasic symptoms (in terms of quality and duration), presence of associated headache, pattern of temporal succession between two subsequent aura symptoms, pattern of temporal succession between aura and headache. Visual symptoms are almost universal, and patients most often had more than one visual disturbance in the same aura. The most common symptoms were flashes of bright light, foggy/blurred vision, zigzag or jagged lines, scotoma and phosphenes. Disturbances of visual perception, such as blurred/foggy vision, ‘like looking through heat waves or water’ and deformed images, are 45% of visual manifestations and have similar characteristics to the 55% of more typical visual aura. Fifteen percent of aura symptoms last for more than one hour, whereas only 5% last for more than four hours.
Footnotes
Acknowledgements
This study was carried out in collaboration with UCADH (University Consortium for Adaptive Disorders and Head Pain), University of Pavia, Italy.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MV, GS, ML, NG, EG, MA, ST, GN, CT have no conflict of interest. PJG reports personal fees from Ajinomoto Pharmaceuticals Co., Ltd, personal fees from Akita Biomedical, personal fees from Alder Biopharmaceuticals, grants and personal fees from Allergan, grants and personal fees from Amgen, personal fees from Avanir Pharma, personal fees from Cipla Ltd, personal fees from Colucid Pharmaceuticals Ltd, personal fees from Dr Reddy’s Laboratories, personal fees from Electrocore LLC, grants and personal fees from Eli Lilly and Company, grants and personal fees from eNeura Inc., personal fees from Ethicon US, personal fees from WL Gore & Associates, personal fees from Heptares Therapeutics, personal fees from NuPathe, Inc., personal fees from Pfizer, Inc., personal fees from Promius Pharma, personal fees from Scion, personal fees from Teva Pharmaceuticals, other from Trigemina Inc., personal fees from medico-legal work, personal fees from Journal Watch, personal fees from UpToDate, personal fees from Oxford University Press (outside the submitted work); in addition, PJG has a patent for magnetic stimulation for headache pending.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants of the Italian Ministry of Health to RC 2013-2015.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
