Abstract
Introduction
We investigated whether interictal thalamic dysfunction in migraine without aura (MO) patients is a primary determinant or the expression of its functional disconnection from proximal or distal areas along the somatosensory pathway.
Methods
Twenty MO patients and twenty healthy volunteers (HVs) underwent an electroencephalographic (EEG) recording during electrical stimulation of the median nerve at the wrist. We used the functional source separation algorithm to extract four functionally constrained nodes (brainstem, thalamus, primary sensory radial, and primary sensory motor tangential parietal sources) along the somatosensory pathway. Two digital filters (1–400 Hz and 450–750 Hz) were applied in order to extract low- (LFO) and high- frequency (HFO) oscillatory activity from the broadband signal.
Results
Compared to HVs, patients presented significantly lower brainstem (BS) and thalamic (Th) HFO activation bilaterally. No difference between the two cortical HFO as well as in LFO peak activations between the two groups was seen. The age of onset of the headache was positively correlated with HFO power in the right brainstem and thalamus.
Conclusions
This study provides evidence for complex dysfunction of brainstem and thalamocortical networks under the control of genetic factors that might act by modulating the severity of migraine phenotype.
Keywords
Introduction
In recent years the thalamus has received growing attention as a key brain structure in migraine pathophysiology, not just as a relay station but also as a site of multisensory integration, being profoundly involved in its clinical and neurophysiological expression. Evidence from animal models have indicated that a temporary sensitisation of third-order thalamic neurons receiving convergent input from the dura, the periorbital skin, and from more distal cutaneous regions play a critical role in the clinical manifestation of central sensitisation – allodynia – beyond the original referred ictal headache region (1). Abnormal modulatory activity of the lateral geniculate complex, the thalamic relays of the visual system, might be involved in the process responsible for migraine-associated symptomology, such as photophobia (2). The anatomical correlates of this thalamic dysfunction are only recently beginning to be understood (3,4). However, as the thalamus is part of interconnected cortical and subcortical networks, neurophysiological measures offer a unique opportunity to investigate whether patients with migraine present a primary thalamic dysfunction between attacks.
Scalp responses to peripheral electrical stimulation (somatosensory evoked responses or SSEP) have been used to measure the influence of arousal systems on cortical somatosensory input processing. Studies have identified that SSEP are characterised by low-frequency oscillatory brain activity (LFOs), and by high-frequency bursts of wavelets (high-frequency oscillations, HFOs) that are more evident in the frontal-parietal region contralateral to the stimulated side (5). SSEP multichannel dipole source analysis has demonstrated four levels of sequential activation of HFOs: the first located in the brainstem (BS), followed by the thalamus (Th), the third located tangentially over the parietal area 3 b, and the fourth radially oriented over the sensorimotor associative area (5–7). This geometry of somatosensory pathway sources was recently confirmed in studies that used a novel approach called functional source separation (FSS) (8). This method identifies cerebral sources on the basis of their functional behavior instead of their anatomical position, allowing the functional characterisation of the oscillatory patterns of four nodes of the primary somatosensory pathway, localised respectively in the BS, the ventral posterior lateral nucleus of the Th, and two within the primary sensorimotor cortex (9,10).
In the last decade, several HFO studies have assessed thalamic/thalamocortical activity in patients with migraine using the classical single-channel approach, and allowed measurement of the activity of thalamic and primary cortical nodes only, with some limitations in the degree of spatial accuracy. These studies have reported that the activity of the HFO somatosensory thalamocortical connections is reduced interictally in migraine (11,12), especially when associated with a worsening in the clinical course of the disease (13,14). To the best of our knowledge, there are no somatosensory HFO studies using multichannel electroencephalography (EEG) associated with FSS procedures to extract the functional activation of the signal generated at each of the sub-cortical (BS and Th) and cortical (early primary parietal area and late sensorimotor area) neuronal nodes. Biochemical (15) and neuroimaging (16) studies have indicated that BS monoaminergic transmission is altered in migraine, and that this may abnormally modulate preactivation levels and signal-to-noise ratio in cortical and thalamocortical neurons. Moreover, maladaptive responses of cortical neurones have been described in patients with migraine during direct cortical repetitive transcranial magnetic stimulation (rTMS). This finding led some authors to speculate that a primary cortical dysfunction could down-regulate thalamic activity via cortico-subcortical feedback loops (17). This body of evidence led us to explore in interictal migraine if the thalamus is the primary site of the dysfunction or whether the thalamic dysfunction occurs secondary to its functional disconnection from brain areas located proximally or distally along the somatosensory pathway and whether the severity of abnormal thalamic function correlated with clinical features of migraine.
Material and methods
Participants
We recruited 20 consecutive patients attending the headache clinic of ‘Sapienza’ University of Rome Polo Pontino, Latina (Italy) diagnosed with episodic migraine without aura (MO) (International Classification of Headache Disorders, third edition beta (ICHD IIIbeta)). Potential participants were identified during their first visit, and were asked if they consented to participate in the study, which was approved by the University Ethics Committee. The study complied with the principles set out in the World Medical Association (WMA) Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Subjects.
Inclusion criteria were as follows: no history of other neurological diseases, systemic hypertension, diabetes or other metabolic disorders, connective or autoimmune diseases or any other type of primary or secondary headache. Patients had uni/bilateral migraine headaches, but not fixed pain on the same side. In order to avoid confounding effects of pharmacologic treatment on the EEG signal, no preventive antimigraine drugs were allowed during the three months preceding the brain electrical activity recording. Patients were recorded during the interictal period, defined as absence of migraine attacks for at least three days before and after the recording session.
We also recruited as controls 20 healthy volunteers (HVs) from medical school students and health care professionals of comparable age and gender distribution; they had no personal or familial history (first- or second-degree relatives) of migraine and no detectable medical condition. All recruited individuals were right-handed. To avoid variability due to hormonal changes, women were recorded outside their pre-menstrual or menstrual periods. All recording sessions were performed in the afternoon between 3:00 and 6:00 p.m. None of the recruited individuals had sleep deprivation or alcohol consumption the day preceding the scans. Caffeinated beverages were not allowed on the day of the recording.
Data acquisition
All recordings were made at the Laboratory of Psychophysiology of the Psychiatric Clinic, Department of Systems Medicine of University of Rome ‘Tor Vergata’, Rome (Italy). Participants were comfortably seated on a chair in an illuminated room and asked to stay with their eyes open, orienting their gaze to a fixed point and their attention to the stimulated hand. Stable levels of arousal were further monitored using the on-line EEG signal. SSEPs were elicited after electrical stimulation of the median nerve at the wrist using a Digitimer DS7A device (Digitimer Ltd, UK) with constant-current square wave pulses (0.2 ms width, cathode proximal), stimulus intensity set at 1.5 times the motor threshold, and at a repetition rate of 4.4/s. For each patient, two series of 1000 sweeps were collected, one for each side. The stimulus side sequence was chosen randomly.
EEG was acquired from 39 electrodes using a pre-cabled electrode cap (Bionen, Florence, Italy) from the positions FP1, FPz, FP2, AF4, AF3, F7, F3, Fz, F4, F8, FC5, FC3, FC1, FCz, FC2, FC4, FC6, T3, C3, Cz, C4, T4, CP5, CP3, CP1, CPz, CP3, CP4, CP6, T5, P3, Pz, P4, T6, PO3, PO4, O1, Oz and O2. The montage was referenced to auricular electrodes, which were linked after placing a resistor in the series with each lead to compensate for any imbalance in impedance between the two auricular electrodes; the ground electrode was placed between Fz and Cz. Electrode impedance was monitored at the beginning and the end of each measurement and remained below 5 KΩ. The signal was amplified by a 40-channel EEG device (Galileo MIZAR-sirius, EBNeuro, Florence, Italy). Data were collected with a sampling rate of 4096 Hz and hardware EEG filters were set at 0.099 Hz high-pass and 0.45*SR (0.45 ×4096 Hz = 1843.2 Hz) low-pass; band-pass digital filtering between 1 and 900 Hz was performed before the off-line analysis.
Data analysis
All data were re-referenced to the Fz electrode to allow comparison with the majority of HFO literature (10,12).
EEG data preprocessing
A semi-automatic independent component analysis (ICA)-based procedure (18) was applied to identify and remove biological (cardiac, ocular and muscular) and non-biological (power line, instrumental noise) artefacts from the data without rejecting contaminated epochs. After artefactual IC identification, the ‘cleaned’ data were obtained by retro-projecting all the ICs except the artefactual ones.
FSS
The FSS (8) technique was used to identify and extract the cerebral sources active in the somatosensory pathways. The aim of FSS is to enhance the separation of signals of interest by exploiting some a priori knowledge on their physical properties. FSS, analogous to ICA, models the set of EEG-recorded signals X as a linear combination of an equal number of sources S via a mixing matrix A (i.e. X = AS). FSS expands ICA by incorporating information available about the specific brain area or neuronal pool under study into the algorithm. A functional requirement (R), is included with a proper weight (λ) into the contrast function (F), in formula F = J + λR, where J is the statistical constraint typical of ICA. FSS identifies a single functional source (FS) at a time, building a contrast function for that source that exploits fingerprint information associated to the neuronal pool to be identified (8). In general, FSS starts from the original EEG data matrix X for each source, and returns one FS with the required functional property. This scheme gives us the possibility to extract the FS that maximises the functional behavior in agreement with the functional constraint (9,10). Similarly to previous works (9,10) we used four ad-hoc functional constraints to extract the brain activity of the two subcortical and two cortical nodes along the somatosensory pathways (FS_BS: brainstem, FS_Th: thalamus, FS_S1: primary sensory (3b) and FS_SM: primary sensory-motor (SM)) from the broad-range frequency band (1–900 Hz).
FS positions
The spatial distribution of the field generated by the sources identified by FSS was obtained by retro-projecting the source activity into the sensors space and used as an input for inverse-problem computation. We used an equivalent current dipole (ECD) with four concentric conductive spheres model (see routine DIPFIT2 of EEGLAB v11.0, available at http://www.sccn.ucsd.edu/eeglab) to obtain ECD positions in Talairach space and projected them on the template brain of the Montreal Neurological Institute (MNI).
LFO-SSEP – peak latency and amplitude analysis
LFO analysis on the SSEP was obtained by applying a 1–400 Hz band-pass filter to the averaged FSs. The P14, P16, N20 and P22 peak latencies and the peak amplitudes (using the P14–0, P16–0, 0–N20 and P22–0 amplitude differences) were selected participant by participant in both groups and used for the following statistical analysis.
LFO and HFO – time frequency analysis
All time-frequency analyses were performed using a Morlet wavelet, with a constant parameter equal to seven that offered the best compromise between time and frequency resolution. Statistical significance of power changes was evaluated with a resampling bootstrap technique, and thresholded at p = 0.01. Non-significant changes were set to zero. LFO activities and HFO activities of each source were extracted by applying, respectively, a 1–400 Hz and a 450–750 Hz (10,12) band-pass filter to the averaged FSs.
All recordings were numbered anonymously and analysed blindly off-line by one investigator (CP), who was not involved in recruitment and inclusion of participants nor in the recording of the EEG.
Statistical analysis
Preliminary descriptive analysis showed that LFO-SSEP peak latency and amplitude of the four sources had a normal distribution, whereas LFO and HFO power amplitude of the four sources had a non-normal distribution. To obtain a better approximation to a Gaussian curve, the latter were logarithmically transformed before the analysis and achieved an appropriate equivalence to a normal distribution (Kolmogorov-Smirnov test, p > 0.2).
A general linear model approach was used to analyse the ‘between-factor’ × ‘two within-factor’ interaction effect for LFO-SSEP peak latencies and amplitudes and LFO and HFO power amplitudes. The between-subject factor was diagnostic group (HV vs. MS); the two within-subject factors were: hemisphere (left vs. right) and source (FS_BS vs. FS_Th vs. FS_S1 vs. FS_SM). Two models of repeated-measures analysis of variance (ANOVA) (rm-ANOVA) followed by univariate ANOVAs were used to investigate the interaction effect on LFO-SSEP peak latency and amplitude and LFO and HFO power amplitude. Univariate results were analysed only if Wilks’ Lambda multivariate significance criterion was satisfied. The sphericity of the covariance matrix was verified with the Mauchly Sphericity Test. In case of violation of the sphericity assumption, Greenhouse-Geisser (G-G) epsilon (ɛ) adjustment was used. Statistical significance was set at p < 0.05. To define which comparison(s) contributed to the major effects, post hoc tests were performed with Fisher’s least significant difference (LSD). We adopted the Bonferroni adjustment for multiple comparisons (0.05/16; value of significance threshold divided by the number of tests presented in the interaction effect) and considered a Fisher’s LSD test significant at p < 0.003125. Finally, Cohen’s d, and its 95% confidence intervals (CI95), was calculated as a measure of effect size for significant post hoc test(s). Correlation analysis was carried out to search for relations among HFO power amplitudes and clinical variables (onset of migraine history; attack frequency; headache severity, measured on a 0–10 visual analogue scale; attack duration, and days elapsed from the last migraine attack).
Results
Descriptive statistics between healthy volunteers (HVs) and migraine without aura patients (MO). Data are frequencies and mean and standard deviation.

FS behavior
The localisation of FSs was consistent with the recruitment of neuronal pools at BS and Th levels (Figure 1, first row) for the subcortical generators. These were maximally activated between 14 ms and 16 ms after the stimulus in the LFO range (Figure 2 for the left hemisphere (LH) and Figure 3 for the right hemisphere (RH), SSEP column). Primary sensory (BA 3b or S1) and primary sensorimotor (BA4 or SM) areas (Figure 1, second row), were maximally activated between 20 and 22 ms after the stimulus in the LFO range (Figure 2 for the LH and Figure 3 for the RH, SSEP column).
Source positions. Grand average position and orientation of the ECD for healthy volunteers (left column) and migraine patients (right column) for the four sources (FS_BS, green; FS_Th, blue; FS_S1, red and FS_SM, Magenta) superimposed on the MNI brain template in axial, coronal, and sagittal views for the left and right hemisphere. For each source in both groups Talairach (x, y, z) coordinates are also shown. Source behaviours, left hemisphere. For the grand average on the left hemisphere, topographic map (topography), somatosensory evoked potential (SSEP) and high-frequency oscillation spectral analysis (time frequency) for the four functional sources (FS_BS, green, first row; FS_Th, blue, second row; FS_S1, red, third row and FS_SM, magenta, fourth row), respectively. BS: brainstem; Th: thalamus; S1: primary sensory; SM: primary sensory motor. Source behaviours, right hemisphere. For the grand average on the right hemisphere, topographic map (topography), somatosensory evoked potential (SSEP) and high-frequency oscillation spectral analysis (time frequency) for the four functional sources (FS_BS, green, first row; FS_Th, blue, second row; FS_S1, red, third row and FS_SM, magenta, fourth row), respectively. BS: brainstem; Th: thalamus; S1: primary sensory; SM: primary sensory motor.

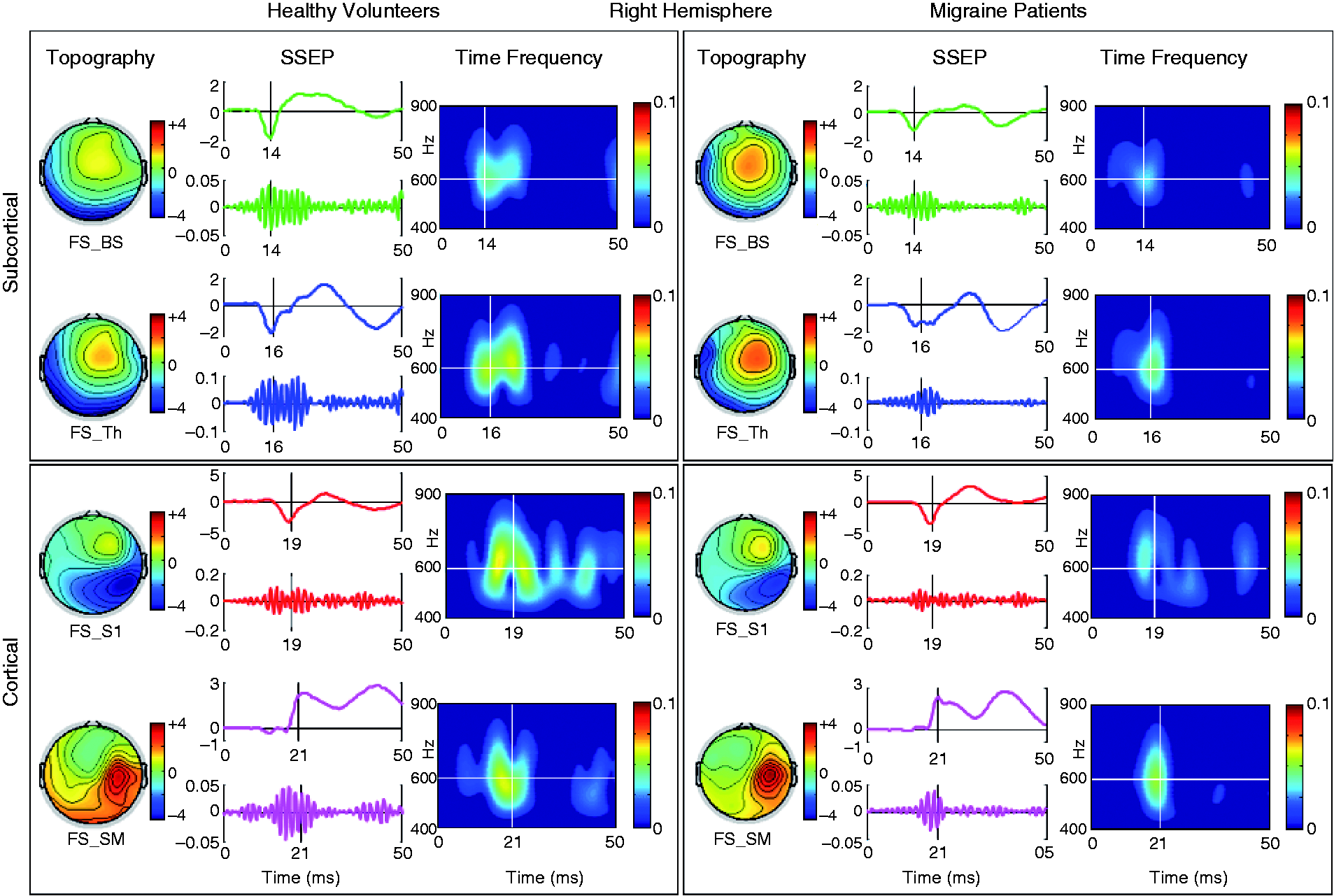
The LFO-SSEP time-series of all sources are shown in the time domain in Figures 2 and 3 (SSEP column). At LFO-SSEP peak latencies in both hemispheres clear bursts of high-frequency activity were presented as time-series revealed in the time domain (SSEP column) and spectral properties revealed in the time frequency plots (Time Frequency column).
LFO-SEP – peak latencies and amplitudes
Mean and standard deviation for the LFO-SSEP peak latencies and amplitudes of the four sources in both hemispheres in both groups.

HVs: healthy volunteers; MO: migraine without aura patients. LH: left hemisphere; RH: right hemisphere; BS: brainstem; Th: thalamus; S1: primary sensory; SM: sensory motor; LFO: low-frequency oscillation; SSEP: somatosensory evoked potential.
LFO and HFO power
In the rm-ANOVA model with LFO power as dependent variable multivariate test was not significant for the ‘diagnostic group’ × ‘hemisphere’ × ‘source’ interaction effect (Wilks’ Lambda = 0.873, F3,36 = 1.755, p = 0.173). Figure 4 shows raw data of LFO power amplitudes in all the conditions.
LFO power amplitude. The means and CI95 of LFO power amplitudes of the four sources in both hemispheres in both groups.
On the contrary, the rm-ANCOVA model with HFO power amplitude as dependent variable multivariate test showed a significant effect for the ‘diagnostic group’ × ‘hemisphere’ × ‘source’ interaction (Wilks’ Lambda = 0.620, F3,36 = 7.344, p < 0.001). After G-G correction was applied for univariate tests due to a violation in sphericity, univariate rm-ANOVAs for HFO power confirmed the difference between groups (F3,114 = 3.931, ɛ = 0.743, p = 0.020). Post hoc tests revealed that the main contributors to the difference between MO and HVs in HFO power were BS (left: p < 0.0001; Cohen’s d (CI95) = –1.16 (–1.36 to –0.94); right: p < 0.0001; Cohen’s d (CI95) = –1.48 (–1.70 to –1.36)) and Th sources (left: p < 0.0003; Cohen’s d (CI95) = –1.44 (–1.63 to –1.22); right: p = 0.003; Cohen’s d (CI95) = –0.95 (–1.15 to –0.79)). According to conventional values of Cohen’s d, the effect size could be considered as large. As shown in Figure 5 (raw data), MO had significant lower power values than HVs for left and right BS and Th sources, suggesting a dysfunction of these subcortical structures. The absence of significant between groups power difference in cortical sources is not supportive of a primary dysfunction at cortical level in the somatosensory region in migraine.
HFO power amplitude. The means and CI95 of HFO power amplitudes of the four sources in both hemispheres in both groups.
Correlation analysis showed that the age of headache onset was positively correlated with HFO power amplitudes of the right BS (r = 0.602, p = 0.005; Figure 6(a)) and Th sources (r = 0.640, p = 0.002; Figure 6(b)). No other correlations were significant.
Migraine onset and the HFO power activities correlation. Graphs depict the correlation between migraine onset and the HFO activities in the right BS and Th. In the graphs, the continuous line is the fitting line and dotted lines show the regression bands of CI95 for mean.
Discussion
The most relevant finding of this study is that evoked BS and Th HFO power is bilaterally reduced in patients with MO after median nerve stimulation at the wrist. The severity of this impairment correlated with the age of onset of migraine manifestations. This spectral abnormality was not present in the two cortical sources, or in the LFO activity both for subcortical and cortical sources.
The diverging behaviour of HFO and LFO is not unexpected given that multichannel source localisation data and pharmacological manipulation studies have shown that LFO and HFO activities are functionally independent, and different generators within the same or nearby cortical areas have been hypothesised (19). Low-frequency activity along the somatosensory pathway represents slow postsynaptic responses, sequentially evoked during passage of each somatosensory node, and clearly confined in their corresponding anatomical location (19). HFOs on the contrary reflect more direct and very fast neuronal spike-activity with a continuous bottom-up outflow of synchronised activity (19). In animal models, dorsal column BS nuclei possess an intrinsic capability to generate HFO activity that synchronise with the contralateral ventroposterolateral (VPL) nucleus (20), while in humans, HFO superimposed on the dorsal column nuclei (DCN) potential were detected in intra-operatory SSEP recordings (21) or by nasopharyngeal electrode (22). The HFO extracted at the BS level might reflect activity of cuneothalamic projection neurons from the DCN in the pons (medial lemniscal pathway in the BS) (21). These nuclei are constituted by a shell and a core region; the latter is the site of the great majority of the cells projected to the VPL nucleus of the contralateral thalamus (20). It is of particular interest for migraine that in experimental and animal models the spinothalamic projecting neurons are reported to have branches ending both in the VPL and nucleus ventralis posteromedialis (VPM) (23–25), where the trigeminothalamic tract converges, and that in turn these thalamic nuclei send inputs to the region of primary somatosensory cortex processing information from the head (26), where the migraine pain is located. HFO activity within or close to the Th and thalamocortical fibres has been recorded in experimental models (27) and in humans during deep brain electrode implantation (21), and is suggested to reflect near-field activity from the somatosensory relay thalamic nucleus. Magnetoencephalographic and EEG studies have identified two generators of HFOs, one in somatosensory area 3b and the other in neighbouring sensorimotor areas with an orthogonal orientation (see Curio (5) for review). These cortical generators may reflect presynaptic repetitive discharges conducted in the terminal segments of thalamocortical axons and/or postsynaptic contributions from specialised, intracortical, very fast-spiking neurons that closely approximated the periodicity of the surface-recorded HFOs as showed in animals (6) and in humans with implanted electrodes (28).
The role of HFO as an intermediate phenotype in migraine patients has been explored in the last decade using the classic single-channel approach. This methodology lacks the spatial accuracy necessary to resolve the contribution of subcortical and cortical regions to this phenomenon. Decreased power of early HFOs in migraine between attacks was independently reported by two groups (12,29) and explained as reflecting generic activity in thalamocortical afferents. Moreover, low thalamocortical drive between attacks was associated with the most pronounced cortical neurophysiological abnormalities and a worsening in the severity of migraine clinical course, such as increase attack frequency (13), intensity and duration of the headache phase (14).
The importance of the integrity of subcortical sources in modulating the burden of migraine is supported by our finding that right BS and Th power amplitude values correlate positively with the age of headache onset, i.e. the earlier the age of first migraine manifestation, the higher the malfunction of subcortical nodes along the somatosensory pathway. The different outcome in correlation analysis between the two sides may result from BS/Th structural asymmetries that have been previously reported by magnetic resonance (MR) imaging analyses (30,31). Migraine is a functional brain disorder in which genetics play a relevant role in setting the individual ‘threshold’ for developing migraine attacks (32). The body of evidence from population-based family studies and twin studies indicates that genetic factors account for approximately 50% in liability to MO (33) and that first-degree family members of patients affected by MO have twice the risk to develop migraine compared with the general population (34). These findings and the evidence emerging from cohort studies suggest that a genetic predisposition to migraine makes probands more susceptible to migraine earlier in life (35). Interestingly, neurophysiological studies in asymptomatic individuals with a family history of migraine reported subcortico/cortical neurophysiological abnormalities similar to those seen in migraineurs (36). The prevalence of these correlated significantly with the number of migraine sufferers among first- and second-degree relatives. Collectively, these observations in conjunction with the positive correlation between age of migraine onset and severity of subcortical BS and Th impairment in HFO activity strongly suggest a link between the underlying genetic load and the interictal abnormal sensory processing in migraineurs.
Relevance to migraine pathophysiology
In this study we found that low thalamic/thalamocortical drive is accompanied by equally low BS activation. The notion of altered BS monoaminergic neurotransmission in migraine has been supported by biochemical (15), neurophysiological (37), and neuroimaging (16) studies. Brain imaging studies have identified increased blood flow in the dorso-lateral part of the BS during attacks of MO (16). A number of studies in migraine have shown dysfunctional endogenous pain modulation at BS level (38–40). Altered turnover of monoaminergic neurotransmitters (serotonin, dopamine, acetylcholine, etc.) released by BS nuclei may also underlie the aberrant BS pain modulation system in migraine (see for a review Hamel (15)). This was confirmed with neuroimaging focused on receptor populations expressed in the anatomical structures involved in migraine pathophysiology, including the BS, following the migraine cycle (16). Reduced monoaminergic, especially serotonergic, availability in the migraineur brain was claimed as a possible culprit of the electrocortical abnormalities frequently observed in migraine interictally. In fact, lack of habituation to repetitive stimuli detected with BS auditory evoked potentials (41) and event-related cognitive potentials (42) was found to be related to platelet serotonin content during the migraine cycle. The intensity dependence of auditory evoked potentials (IDAP) – known to be an inversely related marker of synaptically released serotonin in the central nervous system (CNS) – was found to be stronger interictally in migraine patients compared with to healthy controls (43).
The body of evidence reviewed here seems to suggest that the pathophysiology of migraine could be driven by a complex dysfunction of BS and thalamocortical connectivity. A simultaneous dysfunction of thalamocortical activity and of BS monoaminergic nuclei is the hallmark of various functional brain disorders grouped under the name ‘thalamocortical dysrhythmia’ (TCD) syndromes. The TCD theory proposes that a functional and anatomical disconnection of the Th from the BS monoaminergic subcortical areas induces a change of rhythmic thalamocortical activity favouring cortical rhythms of lower frequency. This in turn is known to reduce the excitation of pyramidal cells at the beginning of the stimulus and of fast-spiking inhibitory interneurons during stimulus repetition (44). Whether this abnormal oscillatory pattern may be responsible for the symptoms that accompany migraine both ictally and, to a minor extent, interictally such as photo/phonophobia and vertigo, remains to be determined.
A limitation of this study is that we are not in a position to evaluate whether our findings are specific to migraine or shared by other headache syndromes. To achieve this, comparative studies with other primary headache disorders are necessary.
Conclusions
In conclusion, the results from this study confirm and extend those previously obtained with the classic single-channel approach. The novelty of our findings is in providing evidence that low interictal thalamic/thalamocortical drive in migraine can be due to low BS activation, and not to a primary cortical dysfunction. The evidence of greater impairment in subcortical HFOs in patients with earlier onset of the disease suggests a role of predisposing genetic factors in the pathophysiology of this electrophysiological intermediate phenotype. Finally, although neurophysiological procedures have not been recognised as useful tools for the diagnosis of non-acute primary headache disorders (45), a more systematic application of the proposed evoked EEG analysis techniques may provide relevant indications about subcortical-cortical activity in migraine. These data can be acquired in a routine clinical setting and the procedure requires minimal patient collaboration. A prospective study could determine the value of this diagnostic procedure in supporting the decision on appropriate pharmacological and non-pharmacological treatment (46).
Article highlights
Morpho-functional evidence suggests that the thalamus plays an important role in migraine. We report that reduced interictal thalamocortical drive in migraine is due to low brainstem activation, not to primary cortical dysfunction. The age of onset of the headache is positively correlated with high-frequency oscillation power in the right brainstem and thalamus. We propose that functional and anatomical thalamic disconnection from the brainstem can contribute to migraine attack recurrence.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The contribution of the G.B. Bietti Foundation to this paper was supported by the Ministry of Health and Fondazione Roma. The contribution of the LET'S-ISTC-CNR was partly supported by FISM – Fondazione Italiana Sclerosi Multipla – Prot. N. 13/15/F14 and PNR-CNR Aging Program 2012–2018.
