Abstract
Aim
To describe neuronal networks underlying commonly reported migraine premonitory symptoms and to discuss how these might precipitate migraine pain.
Background
Migraine headache is frequently preceded by a distinct and well characterized premonitory phase including symptoms like yawning, sleep disturbances, alterations in appetite and food intake and hypersensitivity to certain external stimuli. Recent neuroimaging studies strongly suggest the hypothalamus as the key mediator of the premonitory phase and also suggested alterations in hypothalamic networks as a mechanism of migraine attack generation. When looking at the vast evidence from basic research within the last decades, hypothalamic and thalamic networks are most likely to integrate peripheral influences with central mechanisms, facilitating the precipitation of migraine headaches. These networks include sleep, feeding and stress modulating centers within the hypothalamus, thalamic pathways and brainstem centers closely involved in trigeminal pain processing such as the spinal trigeminal nucleus and the rostral ventromedial medulla, all of which are closely interconnected.
Conclusion
Taken together, these networks represent the pathophysiological basis for migraine premonitory symptoms as well as a possible integration site of peripheral so-called “triggers” with central attack facilitating processes.
Introduction
Among primary headache disorders, migraine is the only one that is preceded by a well characterized premonitory phase with a duration of hours to days before headache onset in a huge percentage of patients (1–7). Typical prodromal symptoms include variances in sleeping patterns like extreme tiredness or frequent yawning, alterations in appetite and food preference like craving of certain foods, changes in attention and concentration, mood swings and multiple others. These symptoms occur in about 30 to 90% of patients, as shown by multiple retrospective and a few prospective studies (1–7). On the other hand, complementary to these typical premonitory symptoms, various migraine triggers have been described, such as changes in weather or sleep regularity, skipping meals or stress in general (7–10). Some of these so-called triggers can be regarded as part of the premonitory symptoms. We have previously demonstrated that certain triggers, premonitory symptoms, and headache-associated symptoms correlate with each other. That is, patients who reported bright light as a trigger are more likely to experience photophobia not only in the premonitory phase, but also during the ictal phase (7). This interrelationship points towards a shared pathophysiology: Intrinsic factors might lower sensory thresholds and mediate appetite and other homeostatic signals. This might lead to an increased perception and occurrence of certain symptoms and possible misunderstanding as triggers. On the other hand, in a certain physiological situation; for instance, in the case of lowered light sensitivity, additional external influences such as bright light might actually be able to trigger a migraine attack.
The clinical observation and characterization of premonitory symptoms has led to various hypotheses of the involvement of certain brain centres and networks in the generation of a migraine attack. Most of the major rhythms in the human brain, such as sleeping, eating, hormonal cycles and various others, are controlled by the hypothalamus. The hypothalamus in turn derives input from many other parts of the brain and from the periphery and might thus be a major site of integrating attack triggering influences from the environment and attack promoting factors from inside the human brain and body (11,12). This review will first illustrate evidence of premonitory networks deriving from human studies to then further explore the anatomical and functional networks underlying premonitory symptoms in migraine.
Premonitory networks in migraine: Evidence from neuroimaging
Functional neuroimaging methods like positron emission tomography (PET) and functional magnetic resonance image (fMRI) make it possible to study activity changes of certain brain structures and also – in the case of fMRI – connectivity changes of these structures in the living human brain. In recent years, neuroimaging studies have begun to focus on pathophysiological mechanisms underlying the premonitory phase of migraine in particular: Triggering migraine attacks via the administration of nitric oxide (NO) is a widely used experimental model for studying migraine in humans and has been shown to also trigger the occurrence of typical migraine premonitory symptoms in the headache-free interval between NO-administration and development of migraine-like headaches (13–15). Using this experimental model with H215O-PET in eight migraine patients, distinct activation within the hypothalamus could be shown in the premonitory phase even before headache onset (14). In a study investigating one migraine patient daily using fMRI of trigemino-nociceptive stimulation over a whole month, the patient had three spontaneous and untreated migraine attacks. Hypothalamic activation was observed within the last 24 hours preceding headache onset (16). This study thus also provided insight into premonitory networks in migraine: Hypothalamic functional connectivity could be shown to alter during different stages of the migraine cycle. Whereas in the premonitory phase this area was more strongly coupled to the spinal trigeminal nuclei, functional connectivity to the dorsal rostral pons was increased during the headache phase. The connectivity changes of the hypothalamus and the brainstem, specifically the pain processing and modulating areas, provide a mechanism for how the premonitory symptoms might lead to headache attacks. The hypothalamus thus seems to change its connectivity to pain processing and modulating areas within the brainstem and might, via this mechanism, enable the generation of migraine pain. Although providing us with the unique opportunity to study migraine attacks in the living human brain, the spatial resolution of functional magnetic resonance imaging techniques is usually not high enough to attribute the activation to a subgroup of nuclei. Therefore, neuroanatomical and electrophysiological studies are of vast importance in further exploring the involvement of distinct hypothalamic nuclei and their structural connectivity in the pathophysiology of migraine premonitory symptoms.
Neuronal networks of premonitory symptoms: The hypothalamus as a hub of integration
Appetite and food intake
Changes in appetite and food intake such as craving of and binging on certain foods are often reported as premonitory symptoms of migraine (1–3,6,7). Complementary skipping meals, irregular eating and fasting are associated with the development of migraine attacks (7–10,17). Feeding and fasting are controlled by a complex neuronal network involving the hypothalamus, the nucleus tractus solitarii (NTS), the ventral tegmental area (VTA) and other brainstem nuclei. The arcuate nucleus in the mediobasal hypothalamus receives signal from peripheral hormones such as leptin (18–20) and modulates energy balance and appetite via two groups of neurons residing within this region (21–24): Neuropeptide Y (NPY) and agouti-related peptide (AgRP) neurons stimulate appetite, whereas proopiomelanocortin (POMC) neurons inhibit feeding (25–27). These neurons project to many other hypothalamic and brainstem nuclei, among these the ventromedial, paraventricular, lateral and dorsal hypothalamus, thus promoting or inhibiting the release of appetite-promoting orexins (28). Of note, the key hormone involved in appetite and food intake regulation, leptin, also shows a diurnal rhythmicity (29) and this rhythmicity is further influenced by the alternation in sleep-waking rhythms and meal timing (30).
In addition to hypothalamic nuclei, brainstem centres are important in regulating appetite and food seeking behaviour: High serum glucose and leptin levels, along with other peptides from the gastrointestinal tract, lead to an activation of NTS, which subsequently suppresses the appetite (31). VTA promotes appetite and food craving but receives negative regulation from leptin. All of these areas are interconnected and represent a complex network regulating food intake, appetite, food seeking behaviour, and energy homeostasis. Changes within this network can explain the often-observed changes in appetite in the premonitory phase of a migraine attack, whereas skipping meals or fasting can also induce activity and connectivity alterations. But how are changes within feeding and fasting regulating networks able to precipitate migraine pain? These above-mentioned nuclei communicate directly or indirectly with various other nuclei of the brain and brainstem involving pain processing and pain modulating areas, such as the spinal trigeminal nucleus, the periaqueductal grey (PAG), and the locus coeruleus (LC). Thus, they are able to influence activity within many areas associated with the pain phase of migraine and integrate trigeminal pain processing and feeding behaviour. The crosstalk between two networks, the food seeking/appetite networks and the trigeminal pain network, provides a possible link to how the prodromal symptoms evolve into headache pain.
Circadian rhythm and sleep regulation
Clinically, migraine attacks somewhat follow a circadian rhythm, though not so strictly as those seen in cluster headaches. Nocturnal attacks are rare in episodic migraineurs, but the diurnal rhythmicity is less prominent in chronic migraine patients (32). A great percentage of migraine patients complain about excessive tiredness hours before headache onset, whereas others also describe sleep disturbances, concentration difficulties and changes in alertness preceding migraine pain (1–3,5–7). It is thus likely that brain structures and networks controlling sleep-waking rhythms and alertness are involved in the premonitory phase of migraine and the generation of migraine pain. Sleep-waking cycles show a strong circadian rhythmicity. This cycling is most likely generated by cells in the suprachiasmatic nucleus (SCN) in the hypothalamus (33–35). The intrinsically photosensitive retinal ganglion cells (ipRGCs) in the retina release pituitary adenylyl cyclase-activating protein (PACAP) and glutamate onto the SCN via a monosynaptic connection (36–39) and thus derive input directly from the retina with information about ambient light, which synchronises the SCN with the diurnal cycle.
The SCN subsequently synchronizes with other hypothalamic nuclei and the pineal gland and hence controls the diurnal rhythm of the release of certain hormones, including melatonin. Additionally, the SCN also modulates arousal via output into the sympathetic nervous system and the hypothalamic–pituitary–adrenal (HPA) axis (40). The sleep/arousal network is strongly intertwined with the feeding/fasting network: The SCN projects to the dorsomedial hypothalamus, which serves as a leptin sensor and modulates the appetite. Efferents from the dorsomedial hypothalamus project to the paraventricular hypothalamic nucleus (41–44), which subsequently controls the activity of the spinal trigeminal nucleus and hence represents a major site of integration between sleep/arousal, feeding/fasting, and trigeminal pain processing networks (45). Neurons in the lateral hypothalamus sense blood glucose levels and release orexins to increase appetite when blood glucose levels are low (46,47). Orexins – in addition to appetite – also promote alertness via effects on the cerebral cortex, the thalamus, other hypothalamic nuclei and brainstem areas: Orexinergic projections from the hypothalamus directly activate arousal and alertness promoting neurons within the LC, a key nucleus also involved in pain processing, specifically trigeminal pain processing (48–51). Furthermore, altered connectivity between the hypothalamus and the LC has been reported in migraine (52). The sleep/arousal network further modulates certain “pain modulators” in the brainstem including the rostroventral medulla (RVM) and PAG. Within RVM and PAG, certain neurons act as the ON-and-OFF switch. The ON-cells facilitate sensory input from the periphery, and OFF-cells inhibit it (53). During sleep, OFF-cell activity is enhanced and subsequently inhibits sensation of peripheral stimuli (including pain), whereas ON-cells are more active within the arousal state and facilitate nociception. In the end, the lateral hypothalamus serves as another hub to connect the sleep/arousal network and the trigeminal pain network (54–56).
Alterations in circadian rhythmicity, sleep, waking and alertness might thus influence feeding and fasting cycles, just as changes in energy homeostasis and the orexinergic networks can lead to sleep disruption and changes in waking rhythms. Via the direct and indirect links to many sites of trigeminal pain processing within the brain and brainstem, distinct changes within the sleep/waking network can account for many frequently reported premonitory symptoms in migraineurs and present a possible mechanism for the transition from the premonitory to the pain phase of a migraine attack. Furthermore, they can account for the often-observed circadian rhythmicity of migraine attacks.
Mood change and stress response
Stress, and paradoxically relief of stress, are often reported as migraine triggers and premonitory symptoms of migraine often involve feeling distressed, moody or dysphoric. This is another example that shows certain triggers and premonitory symptoms may share the same origin, in this case the stress and emotion modulating networks. Stress leads to an upregulation of the hypothalamic-pituitary-adrenocortical (HPA) axis and subsequently increases the release of corticotropin-releasing hormone (CRH), adrenocorticotropin (ACTH) and cortisol (57). CRH is produced by the paraventricular nucleus in the hypothalamus. The release of CRH is modulated under the following circumstances: Fasting and states of high arousal and stress all lead to an increased release of CRH, and this effect might – at least partly – be mediated by orexins (58–60). Orexins are, as mentioned previously, important neurotransmitters both in the feeding/fasting network and the sleep/arousal network and can in addition influence trigeminonociceptive processing (48–51). CRH might reciprocally also trigger orexin release from hypothalamic neurons projecting to areas involved in reward and aversive processing such as the amygdala and the nucleus accumbens (61). Neuropeptides typically involved in the feeding/fasting network are also important regulators of the HPA-axis: Leptin counteracts the stress-induced elevation of cortisol levels. On the other hand, NPY might induce CRH release and subsequently an increase in cortisol levels (62–64). The release of CRH follows a circadian rhythmicity with the highest levels in the early morning hours. This releasing cycle is synchronised with ambient light/darkness via direct retinal input to the SCN of the hypothalamus (35,65).
Functional units in the hypothalamus pertinent to the prodrome of migraine.
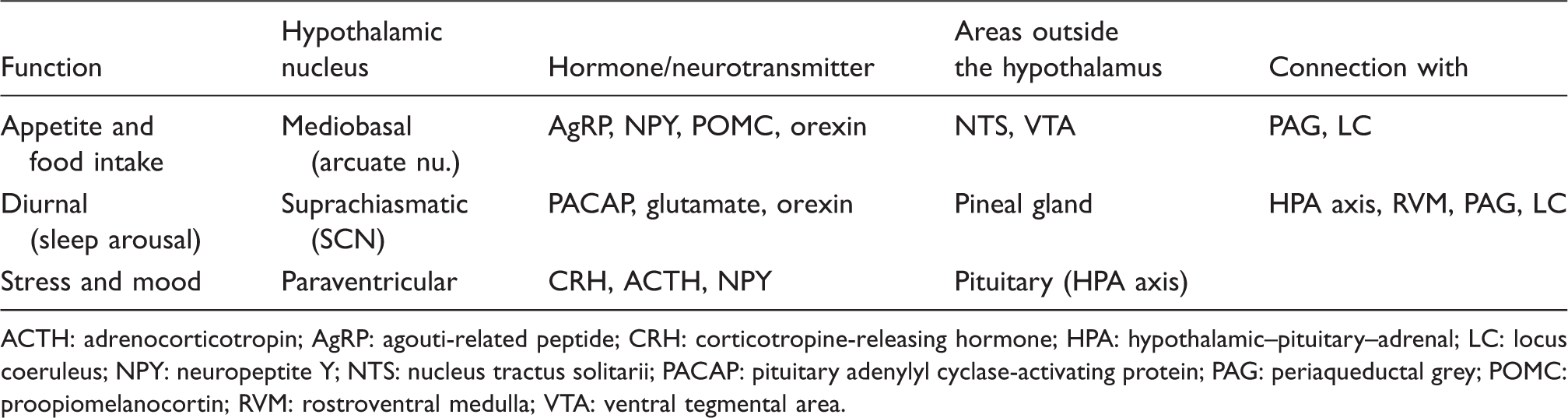
ACTH: adrenocorticotropin; AgRP: agouti-related peptide; CRH: corticotropine-releasing hormone; HPA: hypothalamic–pituitary–adrenal; LC: locus coeruleus; NPY: neuropeptite Y; NTS: nucleus tractus solitarii; PACAP: pituitary adenylyl cyclase-activating protein; PAG: periaqueductal grey; POMC: proopiomelanocortin; RVM: rostroventral medulla; VTA: ventral tegmental area.
Hypersensitivity to external stimuli within the premonitory phase of migraine: Evidence for thalamic involvement
Photophobia and phonophobia are typical accompanying symptoms of the headache phase in migraineurs (66) but have also been described to precede headache onset (1–7). In both cases, external stimuli (light or sound) are perceived as unpleasant or abnormally intense. Furthermore, bright light or loud noises are frequently named as migraine triggers (7–10). Patients with photophobia were more likely to report bright light as a migraine trigger. Similar stands true with phonophobia. The neural basis for photophobia in the premonitory phase of migraine is not entirely understood but emerging evidence suggests the role of a complex network interaction involving direct retinal afferents, the spinal trigeminal nuclei, the hypothalamus, the thalamus and the visual cortex: Bright light induces enhanced trigeminonociceptive activity within the spinal trigeminal nucleus, most likely via direct activation of intraocular trigeminal neurons (67–70), hence leading to enhanced activity in nociceptive thalamic neurons. Trigeminonociceptive neurons within the posterior thalamus additionally derive input from retinal ganglion cells (71) and are the origin of broad cortical projections to the visual cortex, the somatosensory cortex and various association cortices. Thus, discomfort and pain coding pathways are directly activated by bright light and one can hypothesize that transmission within those pathways might be altered in photophobic migraineurs. This is corroborated by the fact that lower light luminance selectively activated the visual cortex in migraineurs but not in controls (72). Similar crosslinks exist between the trigeminal and the auditory system: The auditory cortex has fibre connections to other cortical areas like the visual and the somatosensory cortex, and auditory cortex activation has been found in functional imaging studies as a response to somatosensory stimulation (73–77). There are at least two possible mechanisms behind the reciprocal crosslink: Enhanced trigeminonociceptive activity might facilitate auditory processing, thus leading to a pronounced and even unpleasant perception of auditory stimuli. Alternatively, co-activation of areas involved in pain processing might be sensitized during the premonitory phase and thus lead to the named perception.
Inter-relationship of the main hypothalamic and brainstem networks likely involved in the generation of premonitory symptoms in migraine.
As migraineurs are not usually photophobic all the time but only during certain phases of the migraine cycle, these changes are not likely to be present all the time but to occur periodically. One possible mediator responsible for the cyclic rhythmicity could be the hypothalamus, as the key integrator of light information with intrinsic rhythms.
Conclusion
Multiple evidence suggests a central role of hypothalamic networks during the premonitory phase of migraine. The interconnected nuclei in the hypothalamus serve as hubs of modulation and integration between external factors such as information about energy homeostasis and circadian rhythmicity and internal factors such as hormonal cycles. It thus makes this small area of the brain a key candidate for integrating so-called migraine triggers and internal bodily signals. This small area of the brain may also play an important role in a threshold model of migraine pathophysiology: The baseline hypothalamic activity in migraineurs may already be altered through certain physiological conditions (78). When an external factor further stresses the system; for example, skipping a meal, a critical level of activation may be reached in facilitating trigeminal pain processing and thus ultimately leading to a migraine attack. The understanding of the premonitory phase cannot be narrowed down to a single network: It involves cortical, thalamic and brainstem areas, in the centre of which is the hypothalamus.
Footnotes
Article highlights
Multiple homeostatic networks are involved in the pathophysiology of the premonitory phase of migraine. The interconnected nuclei within the hypothalamus serve as hubs of integration between external and internal factors and thus possibly between so-called migraine triggers and premonitory symptoms. The understanding of the premonitory phase of migraine can not be narrowed down to a single network: It involves cortical, thalamic and brainstem areas, in the center of which is the hypothalamus.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
