Abstract
Background/Aim
Migraine is a risk factor for the formation of silent brain white matter lesions (WMLs) that are possibly ischemic in nature. Although dysfunction of the L-arginine/nitric oxide (NO) pathway has been associated with oxidative stress and endothelial dysfunction in migraine, its role in WML development has not been specifically investigated. Thus, this prospective study aimed to measure the serum concentrations of the NO substrate L-arginine, the NO synthase inhibitor asymmetric dimethylarginine (ADMA), and the L-arginine transport regulator symmetric dimethylarginine (SDMA) in migraine patients in a headache-free period.
Methods
All participants underwent MR imaging to assess for the presence of WMLs on fluid-attenuated inversion recovery imaging. Altogether 109 migraine patients (43 with lesions, 66 without lesions) and 46 control individuals were studied. High-performance liquid chromatography was used to quantify L-arginine, ADMA and SDMA serum concentrations. Migraine characteristics were investigated, and participants were screened for risk factors that can lead to elevated serum ADMA levels independent of migraine.
Results
Migraine patients and controls did not differ in regard to vascular risk factors. Migraineurs with WMLs had a longer disease duration (p < 0.001) and a higher number of lifetime headache attacks (p = 0.005) than lesion-free patients. Higher L-arginine serum levels were found in both migraine subgroups compared to controls (p < 0.001). Migraine patients with WMLs showed higher ADMA concentrations than lesion-free patients and controls (p < 0.001, for both). In migraineurs, the presence of WMLs, aura and increasing age proved to be significant predictors of increased ADMA levels (p = 0.008, 0.047 and 0.012, respectively). SDMA serum levels of lesional migraineurs were higher than in nonlesional patients (p < 0.001). The presence of lesions and increasing age indicated an increased SDMA level (p = 0.017 and 0.001, respectively). Binary logistic regression analysis showed that ADMA level (p = 0.006), increasing age (p = 0.017) and the total number of lifetime migraine attacks (p = 0.026) were associated with an increased likelihood of exhibiting WMLs. There was no significant effect of age on ADMA and SDMA concentrations in controls.
Conclusions
Elevated ADMA levels may impact the pathogenesis of migraine-related WMLs by influencing cerebrovascular autoregulation and vasomotor reactivity. Higher SDMA concentrations may indirectly influence NO synthesis by reducing substrate availability. Elevated L-arginine serum levels might reflect an increased demand for NO synthesis.
Keywords
Introduction
Migraine is a primary headache disorder with recurrent headache attacks (1). Migraine is an independent risk factor for the development of silent brain white matter lesions (WMLs) and infarcts (2–8). As such, WMLs are more prevalent in migraine patients than in the general population (2,3), and they can develop at any age during the active migraine years (3,9). Migraine, especially with aura, carries an increased risk for cerebro- and cardiovascular diseases that cannot be explained by traditional vascular risk factors (10–12). Although histopathological data are lacking in migraine, the microvascular ischemic injury theory (5,13) was supported by our previous quantitative magnetic resonance imaging (MRI) data demonstrating intralesional tissue damage (14) consistent with previous studies of WMLs of ischemic origin (15,16). In addition, longitudinal assessment of the same migraine patient group showed intralesional and intracerebral progression of WMLs over time (17).
A vascular etiology, such as impairment of the L-arginine/nitric oxide (NO) pathway with vascular endothelial dysfunction, could explain the development and progression of WMLs, and could also provide a link between migraine and ischemic stroke or coronary heart disease. Endothelial NO-synthase (eNOS) uses L-arginine to generate the strong vasodilator, anti-atherogenic NO (18). Methylated analogs of L-arginine, symmetric and asymmetric dimethylarginine (SDMA and ADMA), are modulators of the L-arginine/NO pathway (19,20). Both ADMA and SDMA levels are associated with an increased cardiovascular risk and mortality (19,21). ADMA has also been found to be a marker of oxidative stress and endothelial dysfunction in migraine (21,22).
Despite the growing evidence of oxidative stress and vascular endothelial activation in migraine (23–27), the abovementioned biomarkers of endothelial dysfunction have not been comprehensively analyzed in relation to WMLs. Therefore, our objective in the present study was to quantify the L-arginine, ADMA and SDMA serum concentrations of migraine patients with or without cerebral WMLs in a headache-free period, to detect group differences between lesional and non-lesional migraineurs, to investigate the effect of migraine characteristics, and to determine the differences between migraine patients and controls.
Patients and methods
Participants
Clinical data of migraine patients and healthy controls and differences among groups.
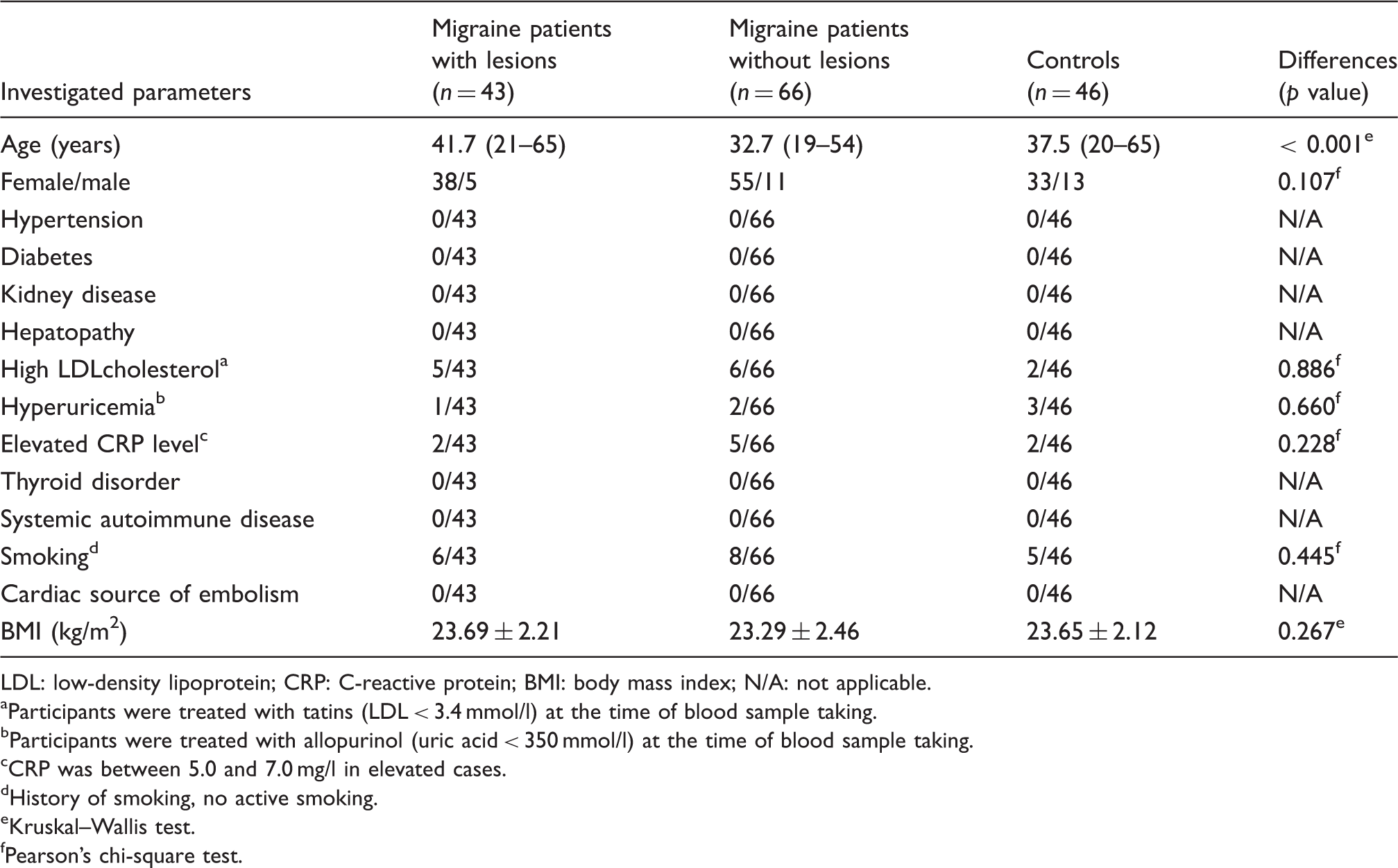
LDL: low-density lipoprotein; CRP: C-reactive protein; BMI: body mass index; N/A: not applicable. aParticipants were treated with tatins (LDL < 3.4 mmol/l) at the time of blood sample taking.
Participants were treated with allopurinol (uric acid < 350 mmol/l) at the time of blood sample taking.
CRP was between 5.0 and 7.0 mg/l in elevated cases.
History of smoking, no active smoking.
Kruskal–Wallis test.
Pearson’s chi-square test.
Results of measured variables and statistical differences between migraine subgroups and controls.
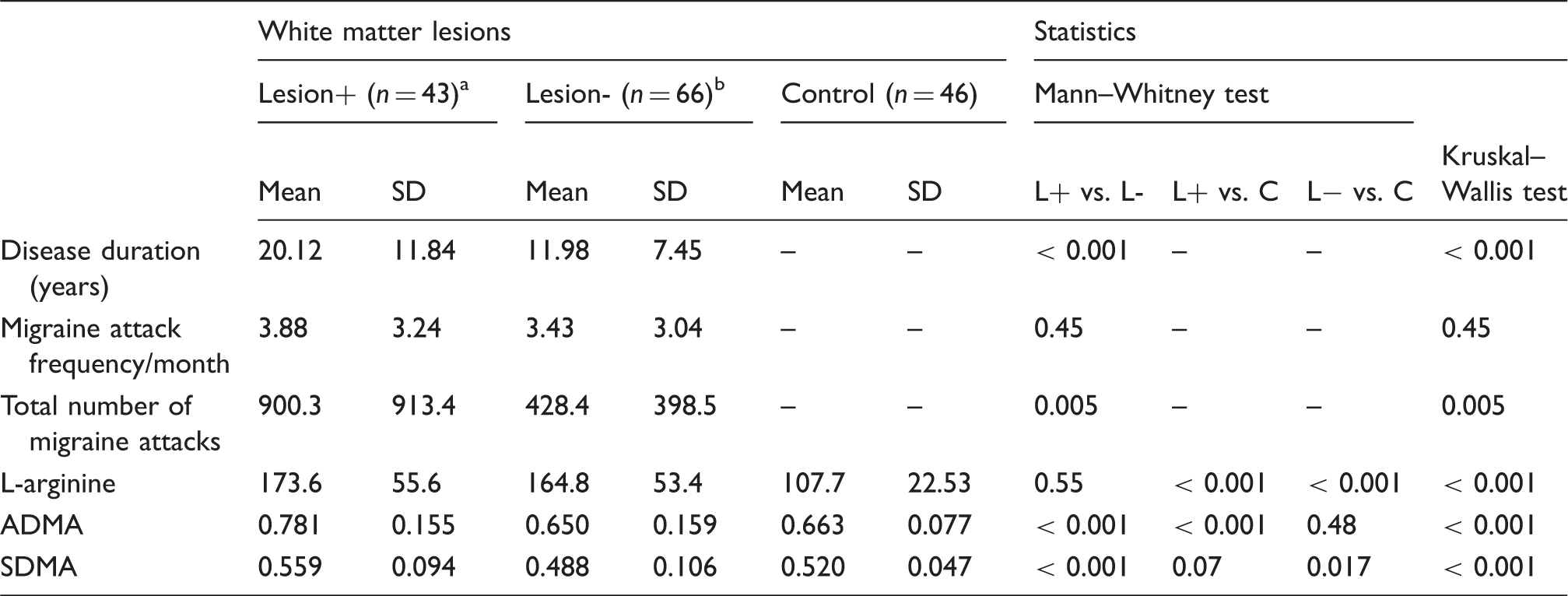
ADMA: asymmetric dimethylarginine; SDMA: symmetric dimethylarginine.
Thirteen migraine patients with aura.
Fourteen migraine patients with aura.
All included participants underwent brain MRI in a headache-free period using the same scanner and acquisition protocol to evaluate the presence of white matter hyperintensities. Given the previous findings of focal tissue damage (14,17), we will refer to these as WMLs later in the text. WMLs were detected in 43 migraineurs, whereas no WMLs or other structural abnormalities were found in the rest of the studied population including controls.
Migraine patients were divided into two subgroups based on presence of WMLs (L+) or absence of those (L−). Presence of aura, disease duration in years and monthly attack frequency were investigated, followed by the estimation of lifetime migraine attack number (average monthly attack number × 12 × number of migraine disease years to date) in all migraineurs.
Serum levels of L-arginine, ADMA and SDMA were quantified from fasting blood samples taken between 8 and 9 a.m. by venipuncture from the antecubital vein in all enrolled participants. Patients were headache free for at least three days before blood sampling. The MR scanning and the taking of blood samples were performed during the same week.
MR scanning protocol and identification of WMLs
A 3.0-Tesla clinical MRI scanner (Magnetom TIM Trio, Siemens Medical Solutions, Erlangen, Germany) with a 12-channel phased-array head coil was used for MR image acquisitions. Routine T1-, T2-weighted and fluid-attenuated inversion recovery (FLAIR) imaging were performed. For standard and accurate axial slice positioning the anterior and posterior commissural line (AC-PC line) was used as a reference for T2-weighted and FLAIR images. Sagittal T1-weighted images were obtained using a fast low angle shot (FLASH) two-dimensional (2D) sequence: repetition time (TR)/echo time (TE) = 300/2.46 ms; flip angle = 88 degrees; 27 slices; slice thickness = 4 mm; distance factor = 30%; field of view (FOV) = 220 × 220 mm2; matrix size = 256 × 320; receiver bandwidth = 330 Hz/pixel. For T2-weighted images a turbo spin echo sequence was used: TR/TE = 6000/93 ms, 30 slices, slice thickness = 4 mm, distance factor = 20%, FOV = 193 × 220 mm2, matrix size = 280 × 320, bandwidth = 220 Hz/pixel, number of echo trains = 18. A turbo spin echo sequence was also used for the FLAIR images: TR/inversion time (TI)/TE = 15710/2750/105 ms, 100 slices, slice thickness = 1.5 mm, distance factor = 0% (no gap), interleaved slice readout with two concatenation, FOV = 220 × 220 mm2, matrix size = 192 × 192, bandwidth = 400 Hz/pixel, number of echo trains = 14.
Two qualified neuroradiologists who were blinded to migraine diagnosis and clinical data rated the WMLs. WMLs were considered if visible as hyperintense areas on T2-weighted and FLAIR images without hypointensity on T1-weighted scans and larger than 3 mm appearing in at least two consecutive slices (2) (Figure 1).
Axial fluid-attenuated inversion recovery brain magnetic resonance imaging (MRI) images of a migraine patient with aura. The figure shows bifrontal white matter hyperintensities in two different slices ((a) and (b)).
Measuring serum concentrations of dimethylarginines and L-arginine
The amino acids were extracted from the blood serum via solid-phase extraction (SPE) according to the method of Nonaka et al. (28): a mixture of 250 μl serum sample and 700 μl borate buffer (pH = 9.00) was mixed with the internal standard of L-homoarginine hydrochloride (Aldrich, Germany) and 50 μl of 1000 µmol/l solution was applied. Then the mixture with the internal standard was passed through SPE cartridges (OASIS® MCX 3 cc SPE) at 750 mbar in a 12-column manifold (J.T. Baker). The elution was performed by concentrated aqueous ammonia (Reanal, Hungary), water and methanol with a volume ratio of 10/40/50. The solvent was evaporated beginning under a nitrogen atmosphere and finished in a vacuum at 60℃. The dry residue was dissolved in 200 µl deionized water (Millipore, Milli-Q) and derivatized according to Molnar-Perl and Vasanits (29); at room temperature the reaction time was 10 minutes. The high-performance liquid chromatography (HPLC) analysis was performed with a Waters 2695 Separations Module equipped with a thermostable autosampler (5℃) and column module (35℃). Separation was achieved with a Waters Symmetry SB C18 (4.6 × 150 mm, 3.5 µm) column and detected by a Waters 2475 fluorescence detector (Waters, Milford, MA, USA) (30). For the measurements, 10 µl was injected from the samples and the gradient elution was applied during the analysis with two mobile phases: (a) (20 mM (NH4)2CO3 in water, pH = 7.50 ± 0.05) and (b) (acetonitrile). The gradient program was as follows: 0–16 min: 91% (a) and 9% (b), 16–17 min: linear change to 70% (a) and 30% (b) and hold this for five minutes, 22–23 min: linear change to 91% (a), 9% (b) and hold this for 12 minutes. The flow rate was constant (1 ml/min). Arginine and homoarginine were detected at λex = 337 nm, λem = 520 nm, but λem = 454 nm was used for ADMA and SDMA.
Statistical analyses
Statistical analyses were performed using the IBM SPSS Software (version 21, SPSS Inc, Chicago, IL, USA). First, the distribution of each studied variable was assessed using the Kolmogorov–Smirnov and Shapiro–Wilk tests. Differences in age between the whole migraine group and healthy controls were assessed by the Mann–Whitney U-test.
Univariate analyses
Differences of continuous variables (e.g. age, body mass index (BMI), disease duration, migraine attack frequency, total number of migraine attacks, L-arginine/ADMA/SDMA levels) among groups (L+, L−, control) were compared using the Kruskal–Wallis test followed by the Mann–Whitney U-test. Differences in binary population characteristics (e.g. gender, smoking, hypercholesterolemia, hyperuricemia, elevated C-reactive protein (CRP) level) among the same groups were assessed using the Pearson’s chi-square test.
Multivariate analyses
First, we tested predictors of L-arginine/ADMA/SDMA levels using stepwise multiple linear regression analyses. Age, gender, disease duration, migraine attack frequency, total number of migraine attacks and presence of aura were included as independent variables along with the grouping variable (L+ versus L−), and L-arginine/ADMA/SDMA level as the dependent variable. Similar analyses were performed to assess the L-arginine/ADMA/SDMA level differences between patient and control groups (L+ versus control and L− versus control), excluding migraine characteristics (not present in the control group) from the list of independent variables. In the control group, multiple linear regression analyses were performed to assess the potential effects of age and gender on L-arginine, ADMA, and SDMA concentrations. Serum concentrations served as dependent, while age and gender as independent, variables. The assumptions of multiple linear regression were satisfied, as judged by testing for linearity and normality. In addition, in the patient group, binary logistic regression was performed including the covariates of age, gender, L-arginine/ADMA/SDMA levels, disease duration, frequency of migraine attacks, total number of migraine attacks and presence of aura to assess the likelihood of WML presence. The final covariates were selected using a stepwise method (forward conditional).
Finally, Spearman’s correlations were performed to assess the internal dependencies between the three reported lab variables (L-arginine, ADMA, SDMA) in all participants, and to investigate the correlation between age and disease duration in the migraine group. The level of statistical significance was set as <0.05.
Results
Age
Since the age of controls was matched to the whole migraine group, there was no significant difference in age between them. The Kruskal–Wallis test revealed significant age differences among migraine subgroups and controls (p < 0.001) (Table 1). Post-hoc testing indicated significant differences between all possible pairs of the three groups (p < 0.05).
Control group
Since the study subgroups were not age-, and gender-matched, the healthy control group was investigated next by multiple linear regression. There were no significant effects of either age or gender on L-arginine, ADMA, or SDMA concentrations; there were no interactions between age and gender (i.e. age*gender).
Categorical data
Regarding the gender of the participants, and the presence of vascular risk factors, no significant differences were found among the groups (Table 1).
Migraine characteristics
Compared to lesion-free migraineurs, patients with WMLs had a longer disease duration (p < 0.001; Table 2), and a higher number of lifetime headache attacks (p = 0.005; Table 2). The attack frequency did not show a significant difference between the L+ and L− groups (Table 2).
L-arginine levels
Markedly higher blood serum concentrations were measured in the migraine groups (L+ 173.6 ± 55.6, L− 164.8 ± 53.4) than in the control group (107.7 ± 22.53) (p < 0.001, in all comparisons; Table 2, Figure 2). Statistically significant differences were not found between the migraine groups (Table 2, Figure 2). Since the multiple linear regression analyses revealed no additional significant predictors (e.g. age, gender), the pattern of significance remained unchanged.
L-arginine, asymmetric and symmetric dimethylarginine (ADMA and SDMA) serum concentrations in migraineurs and controls. The figure demonstrates the differences between the lesion+, and the lesion– migraine groups and the control group. Whiskers are set at minimum and maximum, and the horizontal line marks the median, whereas the box indicates the interquartile range (25%–75%). The statistically significant differences between the groups are indicated by * on the figures (*p < 0.05, ***p < 0.001).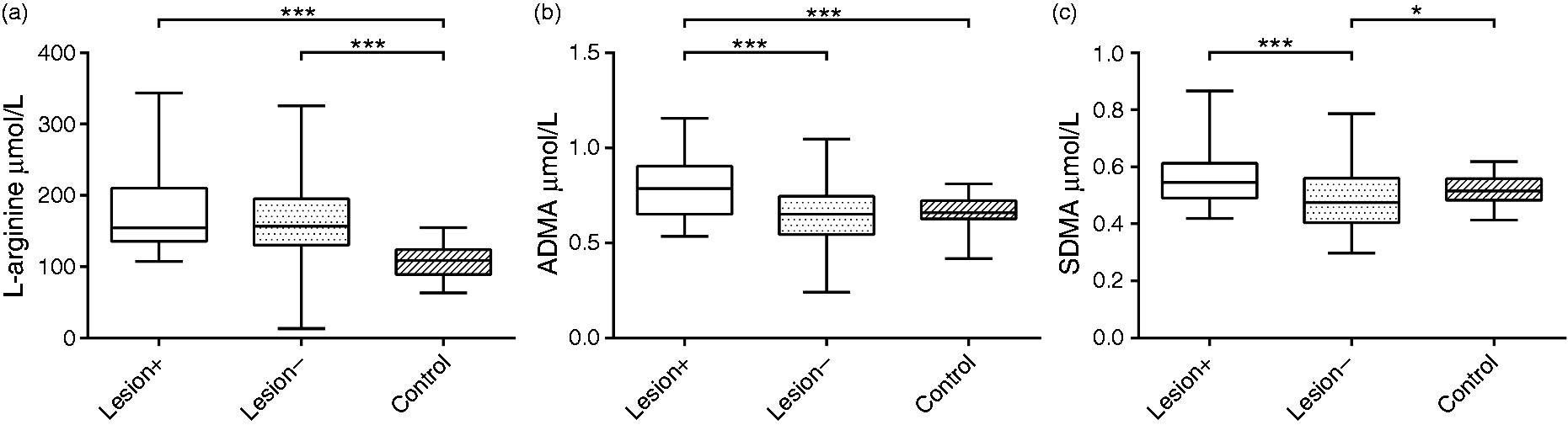
ADMA levels
L+ migraine patients (0.781 ± 0.155) showed higher serum concentrations than L− patients (0.650 ± 0.159) and controls (0.663 ± 0.077) (p < 0.001, in both comparisons) (Table 2, Figure 2). Controlling for the relevant covariates using multiple linear regression analyses, there was no change in the pattern of significance. When comparing the L+ group to the control group no additional significant predictors (e.g. age, gender) were found. In the comparison of the L+ and L− groups, aura and age proved to be additional significant predictors; the presence of WMLs, aura and increasing age indicated an increased ADMA level (p = 0.008, 0.047 and 0.012, respectively). There were no interactions between the independent variables.
SDMA levels
SDMA levels of L+ migraineurs (0.559 ± 0.094) were higher than L− patients (0.488 ± 0.106) (p < 0.001) and controls (0.520 ± 0.047), but the latter comparison was not statistically significant (p = 0.07) (Table 2, Figure 2). SDMA levels of controls were higher than L− patients (p = 0.017). When comparing L+ and L− groups using a multiple linear regression model, age proved to be an additional significant predictor; the presence of lesions and increasing age indicated an increased SDMA level (p = 0.017 and 0.001, respectively). Comparing the L- and control groups, age proved to be a significant predictor (increasing age associated with increased SDMA level, p = 0.011), but the group difference (i.e. L− vs. control) was no longer significant (p = 0.145). A significant interaction was found between the grouping variable and age (p = 0.037).
Binary logistic regression analysis
The logistic regression model explained 37.4% (Nagelkerke R2) of the variance in WMLs and correctly classified 71.6% of WMLs. ADMA levels were the most significant independent predictors of WML presence, with more than an 85-fold higher likelihood of exhibiting WMLs in those with a 1 µmol/l elevation in ADMA (Exp(B) = 85.33, p = 0.006). Increasing age and total number of migraine attacks were also associated with an increased likelihood of exhibiting WMLs (Exp(B) = 1.06, p = 0.017 and Exp(B) = 1.001, p = 0.026, respectively). There were no interactions between the independent predictors. None of the other examined variables added significantly to the model.
Spearman’s correlations
The ADMA level was positively correlated with both L-arginine and SDMA levels (ρ = 0.158, p < 0.05 and ρ = 0.403, p < 0.001, respectively). No significant correlation was found between the L-arginine and SDMA levels. A significant positive correlation was found between age and disease duration in the whole migraine group (ρ = 0.628, p < 0.001) and in both subgroups (L+, ρ = 0.703, p < 0.001; L−, ρ = 0.479, p < 0.001), as well.
Discussion
The main finding of the present study is the elevated serum ADMA concentration in migraine patients with WMLs outside the attack period. ADMA levels were the best predictor of WMLs, independent of other significant factors such as age and estimated lifetime headache attack number. Higher SDMA serum levels distinguished lesional migraine patients from lesion-free patients, while the elevated L-arginine concentrations differentiated migraineurs from controls.
Age, vascular risk factors, migraine characteristics
Increasing age was associated with an increased likelihood of exhibiting WMLs, and older age proved to be a predictor of higher ADMA and SDMA in migraineurs with lesions. Since positive correlation was found between age and disease duration, and there were no significant effects of age on ADMA and SDMA concentrations in controls, long duration of migraine, rather than older age, may explain the elevation of dimethylarginines.
Increased plasma ADMA concentrations have been associated with the presence of numerous vascular risk factors and chronic diseases such as obesity, hypertension, diabetes, dyslipidemia, hyperhomocysteinemia, ischemic heart disease, transient ischemic attack, silent brain infarcts, ischemic stroke, renal and liver failure, smoking and physical inactivity (19,31). Vascular risk factors were rare both in patients and controls in our study, and all of them were treated during the study period. It is known that lipid-lowering therapy and smoking cessation reverse the endothelial dysfunction, if present (32). Therefore, a significant influence of vascular risk factors on the findings is unlikely.
In the present study, migraine patients with WMLs had a longer disease duration than nonlesional patients, and it was associated with a higher number of lifetime headache attacks. The number of lifetime headache attacks proved to be an independent predictor of WMLs, while the presence of aura indicated higher ADMA levels in migraineurs. Beyond the investigated migraine parameters, there are other factors such as attack intensity and duration, frequency and severity of the cortical spreading depression, which may have an influence on lesion formation. In concordance with previous studies (22,27), the interictal elevation of ADMA in lesional patients indicates that oxidative stress can be present both in the ictal and interictal phases in migraine.
Effects of migraine on the biosynthesis and metabolism of dimethylarginines and L-arginine
NO is produced by vascular endothelial cells, and plays a crucial role in the regulation of blood pressure, cerebral blood flow and neuroprotection (18,33). The amino acid L-arginine is the main precursor of NO (Figure 3) (20,34). Posttranslational methylation of arginine residues on nuclear proteins leads to the generation of ADMA, SDMA and N-monomethyl-L-arginine (L-NMMA) that are released into the cytosol upon proteolysis, then migrate into the extracellular space and thence into the blood plasma (35). Both ADMA and the less potent L-NMMA are competitive inhibitors of all three isoforms of NOS, thus lowering NO levels (35). The concentration of ADMA and L-NMMA is regulated mainly by degradation in the endothelial cells by the dimethylarginine dimethylaminohydrolase-2 isoform (DDAH-2), and to a lesser degree by urinary excretion (35,36). SDMA is the structural counterpart of ADMA, and like ADMA, blocks cellular L-arginine uptake competitively, thus indirectly influencing NO bioavailability by reducing substrate availability (37–39).
Effects of migraine headache on the biosynthesis and metabolism of dimethylarginines and L-arginine. SAM: S-adenosyl-methionine; SAH: S-adenosyl-homocysteine; PRMTs: protein arginine methyltransferases; ADMA: asymmetric dimethylarginine; L-NMMA: N-monomethyl-L-arginine; SDMA: symmetric dimethylarginine; NO: nitric oxide; DDAH-2: dimethylarginine dimethylaminohydrolase-2; eNOS: endothelial nitric oxide synthase.
Since DDAH-2 is sensitive to oxidative stress triggered by the excessive generation of NO, under pathophysiological conditions, for example, cardiovascular diseases and migraine, reactive O2 species (ROS) inhibit DDAH-2 activity with corresponding accumulation of ADMA (35). It is conceivable that the NO-caused vasodilatation is overbalanced by the increased amount of ADMA during migraine headache, which may lead to a reduced bioavailability of NO, vasoconstriction with cerebral hypoperfusion and endothelial dysfunction (22). Since elevation of NO and ADMA concentrations are not restricted to the ictal state (22), inhibition of eNOS can also be present in a headache-free period. Chronic endothelial dysfunction has a role in mediating impaired cerebral autoregulation, and in the small perforating cerebral blood vessels it could result in poor white matter blood supply and the accumulation of white matter injury (40,41).
It is probable that migraine is a systemic factor of vascular endothelial injury and WML development during the active headache years. Our findings support the importance of migraine headache management to avoid vascular complications of migraine. This includes not only acute and prophylactic headache treatments, but also the pharmacotherapy of comorbid cerebro- and cardiovascular diseases, as well as lifestyle changes.
Comparability of serum and plasma L-arginine and dimethylarginine measurements
The samples were measured with the internal standard method using L-homoarginine as the internal standard in the present study. L-homoarginine with its similar structure and chemical properties to ADMA, SDMA and L-arginine provides a chance to eliminate the matrix effects and the loss of components during sample preparation. Furthermore, the calibrants were prepared the same way as the samples. Thus, it can be assumed that there is no significant influence on the comparability of the results because the matrix effect can be eliminated.
Study limitations
The lack of age- and sex-matched study subgroups may limit the comparability of groups. Although hyperhomocysteinemia is a cardiovascular risk factor, blood homocysteine levels were not measured in the present study.
Conclusion
The elevated ADMA levels in lesional migraineurs may indicate a role of migraine-related vascular endothelial dysfunction in the development of WMLs, supporting the ischemic injury theory of WML formation. These findings indicate that elevated ADMA concentrations may be a risk factor for clinically silent brain WMLs and point out the necessity of therapeutic interventions. SDMA may also have a role in lesion formation by indirect inhibition of NO bioavailability. The higher L-arginine serum concentrations might reflect an increased demand for NO synthesis in migraine.
Clinical implications
Elevated asymmetric dimethylarginine (ADMA) concentrations may be a risk factor for clinically silent brain white matter lesions (WMLs). Detection of ADMA levels in the clinic may help to estimate the risk of vascular endothelium impairment in migraineurs. The elevation of ADMA in migraine patients with lesions points to the importance of the clinical management of migraine headache and the pharmacotherapy of comorbid cerebral and cardiovascular diseases.
Footnotes
Acknowledgement
All participants provided informed consent. Studies were performed in accordance with the approval (4022) of the Regional Research Ethics Committee of the Medical Center, Pécs.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from EEA/Norwegian Financial Mechanism HU 0114—“Save what can be saved”—applied neurological research using high-field magnetic resonance imaging and by TÁMOP-4.2.1/B. N.K. was supported by the Bolyai Scholarship of the Hungarian Academy of Science and by OTKA PD 103964.
